Abstract
Background:
If severe acute pancreatitis (SAP) is complicated by disseminated intravascular coagulation (DIC) or walled-off necrosis (WON), the mortality rate may increase. Therefore, prevention of the development of WON and treatment of DIC are likely to play important roles in improving survival in SAP. Although recombinant human soluble thrombomodulin (rhTM) might play a useful role in treating DIC, the impact of rhTM on improving survival and resolution of DIC due to SAP is still unclear.
Objective:
This study aimed to evaluate the clinical impact of rhTM in patients with SAP and DIC.
Design:
A single-center retrospective study.
Method:
The patients were divided into two groups: the rhTM group and the non-rhTM group, in which rhTM was not administered. The primary outcome was the DIC resolution rate at 7 days after starting treatment. The mortality rate at 28 days was secondarily evaluated.
Results:
Among 321 SAP patients, 63 patients were complicated with DIC, and they were divided into rhTM (n = 28) and non-rhTM (n = 35) groups. The rate of development of WON was significantly higher in the non-rhTM group (51.4%, 18/35) compared with the rhTM group (25.0%, 7/28) (p = 0.033). The resolution rate of DIC within 7 days was significantly higher in the rhTM group (89.3%, 25/28) compared with the non-rhTM group (60.0%, n = 9/35) (p = 0.009). The mortality rate within 14 days, which might have been strongly influenced by the presence of DIC, was significantly higher in the non-rhTM group (20.0%, 7/35) compared with the rhTM group (0%, 0/28) (p = 0.01). According to multivariate analysis, rTM non-administration was an independent factor for failed DIC resolution or developing WON.
Conclusion:
In conclusion, rhTM may play a role not only in improving the resolution rate of DIC and improving the survival rate of SAP, but also in preventing the development of WON.
Plain language summary
Why was the study done?
When patients were complicated with severe inflammation of the pancreas (pancreatitis), it can sometimes lead to dangerous problems like blood not flowing properly or parts of the pancreas getting badly damaged. These problems can increase the risk of death. Therefore, stopping these issues early and treating them quickly is very important. There is a drug, called rhTM, that might help with the blood-related problems, but it's not clear yet if it really helps people survive or recover better. This study wanted to find out if using rhTM would help people with severe pancreatitis do better
What did the researchers do?
We tried to find out if using rhTM would help people with severe pancreatitis do better. The patients were split into two groups. One group received rhTM, and the other group didn’t. The researchers looked at following things: how many people got better from the blood problem within one week, and how many people were still alive after four weeks.
What did the researchers find?
Out of 321 patients with serious pancreatitis, 63 had blood problems. These 63 people were divided into two groups: 28 got the medicine (rhTM group), and 35 did not (non-rhTM group). More people in the group without the medicine had serious damage to their pancreas (51%) compared to those who got the medicine (25%). More people in the rhTM group got better from the blood problem within one week (89%) compared to the non-rhTM group (60%). In the first two weeks, none of the patients in the rhTM group died, while 20% of the non-rhTM group did. The study found that not using the medicine was linked to worse outcomes.
What do the findings mean?
This medicine (rhTM) may help people with severe pancreatitis by helping them recover from blood problems, preventing further damage, and possibly saving lives.
Keywords
Introduction
Acute pancreatitis (AP), which is caused by various factors, including acute inflammation of the pancreas, has the potential to affect other systemic organs. Clinically, AP is diagnosed based on the presence of symptoms, such as epigastric pain, elevation of pancreatic enzyme levels, and findings on imaging modalities, such as multidetector contrast-enhanced computed tomography (MDCT).1,2 Clinically, one-third of AP patients develop severe AP (SAP), with elevation of inflammatory markers and other organ dysfunction. The mortality rate of SAP was reported 20%–40%. 3 The main reason for mortality due to SAP is organ failure (OF), which can be divided into local and systemic. In case of local OF, pancreatic ischemia occurs first, followed by pancreatic necrosis.4,5 Systemic organ failure, such as respiratory, renal, or heart failure, is usually critical in patients. OF with AP occurs secondary to abnormal coagulability, which develops as a result of micro-circulatory failure due to endovascular injury of the pancreas.6,7 In case of AP due to endovascular injury, damage-associated molecular patterns or inflammatory cytokines, which produce tissue factors and thrombin, can lead to disseminated intravascular coagulation (DIC), which further increases the mortality rate.8,9 Walled-off necrosis (WON) following an acute necrotic collection (ANC) can also develop due to tissue micro-circulatory failure. WON increases the mortality rate of SAP from 1.3% to 6.1%. 10 Therefore, prevention of the development of WON and treatment of DIC are likely to play important roles in improving survival in SAP.
Thrombomodulin is an important molecule involved in blood coagulation that is mainly expressed in vascular endothelial cells. It is known to bind to thrombin, exerting anticoagulant effects by changing its activity. In addition, thrombomodulin suppresses fibrinolysis, promotes thrombolysis, and promotes the endogenous anticoagulant mechanism of DIC. 11 Several studies have shown that recombinant human soluble thrombomodulin (rhTM) might play a useful role in preventing the development of WON.12–15 However, the impact of rhTM on improving survival and resolution of DIC due to SAP is still unclear. Therefore, this study aimed to evaluate the clinical impact of rhTM in patients with SAP and DIC.
Patients and methods
This study was conducted between February 2012 and October 2023. This retrospective observational study included consecutive patients with complicated SAP aged >18 years, who had undergone treatment within 24 h of symptom onset. Exclusion criteria were traumatic pancreatitis, cancer-related pancreatitis, and/or those with immunosuppressive conditions. The study protocol was approved by the institutional review board of our hospital and conformed to the ethical guidelines of the 1975 Declaration of Helsinki. A priori approval for the conduct of this study was given by the human research committee of Osaka Medical and Pharmaceutical University (IRB No 2024-0199). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 16
Diagnosis and treatment of SAP
AP was diagnosed by the presence of two or three symptoms, including abdominal pain, abnormal elevation of amylase and/or lipase, and pancreatic inflammation on MDCT imaging.1,3 Several parameters were assessed to evaluate the severity of AP, such as C-reactive protein (CRP), blood urea nitrogen (BUN) level ⩾20 mg/dL, acute physiology and chronic health evaluation II (APACHE II) score, systemic inflammatory response syndrome (SIRS) score, sequential organ failure assessment (SOFA) score, and the Japanese severity score.17–20 Enrolled patients were treated according to the strategy recommended in the Japanese guidelines for the management of early-stage AP. Briefly, the treatment regimen included fasting, fluid therapy, and administration of a protease inhibitor (nafamostat mesilate: 0.06–0.20 mg/kg/h, continuously infused; or gabexate mesilate: 20–39 mg/kg/day). For patients with ischemic or necrotizing pancreatitis, continuous regional arterial infusion of the protease inhibitor (nafamostat mesilate) and prophylactic antibiotics (CRAI) was administered.20,21 Antibiotics were administered intravenously if sepsis was suspected, based on the presence of symptoms such as high fever or a shock-like state. In cases complicated with ANC or WON, endoscopic ultrasound (EUS)-guided drainage was first considered. Briefly, the ANC or WON was punctured using a 19-G needle, and contrast medium was injected. After guidewire deployment within the ANC or WON, tract dilation was performed using a dilator or balloon catheter, followed by stent deployment. If an adequate puncture route was not identified under EUS, the percutaneous approach was secondarily selected. In cases of WON, necrosectomy was performed. If clinical success was not obtained by these methods, surgical treatment was considered.
Diagnosis and treatment of DIC
DIC due to SAP was diagnosed based on the Japanese Association for Acute Medicine (JAMA)-DIC criteria. Briefly, 1 DIC point was assigned for each of the following: SIRS score ⩾3, 22 prolongation of the prothrombin time-international normalized ratio (⩾1.2), mild thrombocytopenia (platelet count (PLT) 8.0–12.0 × 1010/L, or a >30% decrease within 24 h from admission), and mild elevation of fibrin/fibrinogen degradation product (FDP) values (10–25 μg/mL). In addition, three DIC points were assigned for each of severe thrombocytopenia (PLT < 8.0 × 1010/L or a >50% decrease in platelet count within 24 h) and severe elevation of FDP values (⩾25 μg/mL). Patients with a DIC score of ⩾4 were diagnosed with complicating DIC. DIC with SAP was treated using rhTM (Asahi® Kasei Pharma Corporation, Tokyo, Japan) (380 or 130 U/kg/day), which was usually administered at a dose of 380 U/kg for six consecutive days, although treatment was continued based on the judgment of the attending physician if DIC resolution was not obtained. If patients developed renal failure as a complication, rhTM was administered at a dose of 130 U/kg/day. From 2015, because the clinical efficacy of recombinant thrombomodulin (rTM) for SAP has been reported, 13 we basically administered rTM for DIC; however, the judgment of rTM administration was performed by the attending physician.
Administration of other drugs to treat DIC, such as gabexate mesylate or antithrombin III, was based on the judgment of the attending physician.
Definitions and statistical analysis
The patients in the present study were divided into two groups: the rhTM group, in which rhTM was administered for at least 2 days, and the non-rhTM group, in which rhTM was not administered. The primary outcome was the DIC resolution rate at 7 days after starting treatment. The mortality rate at 28 days was secondarily evaluated.
Adverse events associated with rhTM, such as intestinal bleeding, were evaluated, as previously described. 23 The Mann–Whitney U-test and Fisher’s exact test were used to compare patients’ characteristics, DIC scores, and laboratory data between the two groups. The stratified log-rank test was used to compare survival curves, and censored data were taken into account. Univariate and multivariate logistic regression analyses were conducted to determine risk factors for failed DIC resolution and the development of WON, and evaluated as the hazard ratio (HR) with 95% confidence intervals (CI). Differences with a p-value less than 0.05 were considered significant. Finally, continuous variables were expressed as mean values. All analyses were performed with SPSS version 13.0 (SPSS, Chicago, IL, USA) statistical software.
Results
Baseline characteristics
During the study period, SAP was complicated in 342 patients. Among them, 21 patients were excluded based on exclusion criteria (Figure 1). Among the 321 patients, development of WON was observed in 75 patients (23.4%). In addition, DIC was observed in about one-fifth of the patients (19.6%, 63/321). The mortality rate at 28 days was 3.1% (27/321).
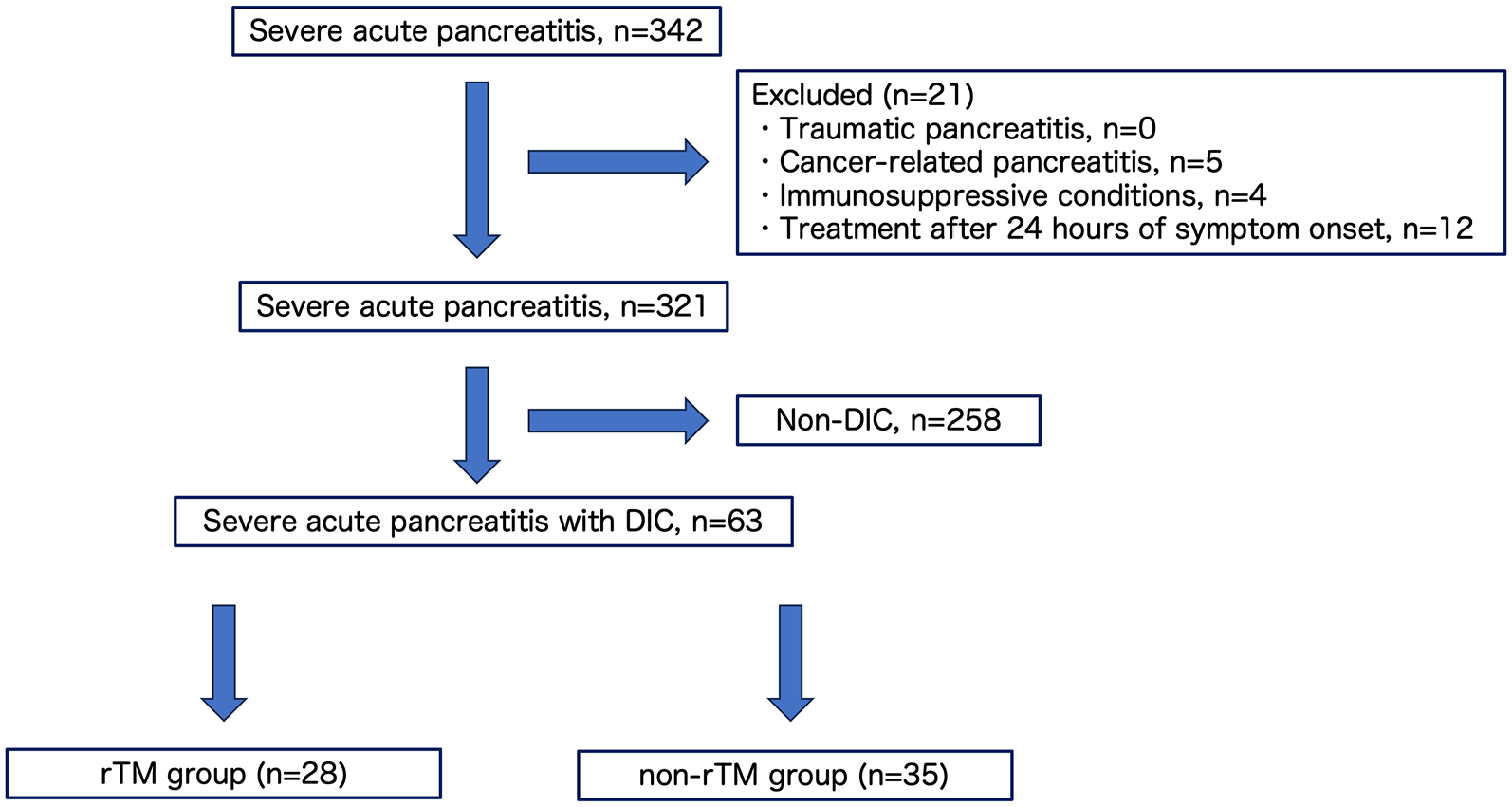
Patient’s flow chart.
Table 1 shows the characteristics of patients with DIC divided into rhTM (n = 28, mean age 64.2 years; 21 males) and non-rhTM (n = 35, mean age 64.7 years; 26 males) groups. Among the DIC patients, other anti-DIC drugs, such as antithrombin inhibitors, were not administered to any of the patients. The mean duration of rTM administration was 9.2 ± 2.4 days. The mean body mass index was 24.1 kg/m2 in the rhTM group and 22.7 kg/m2 in the non-rhTM group (p = 0.412). The main reason for SAP was alcohol in both the rhTM (67.9%, n = 19/28) and non-rhTM groups (77.1%, n = 27/35) (p = 0.500). The Charlson risk index score was not significantly different between the two groups (p = 0.458). Furthermore, 31.1% of patients in the rhTM group (9/28) and 25.7% of patients in the non-rhTM group (9/35) were treated in the ICU setting, indicating no significant difference (p = 0.575). Also, there were no significant differences between the two groups regarding antibiotics, somatostatin analogs, or gabexate mesylate administration, and undergoing continuous renal replacement treatment. Baseline mean DIC score was significantly higher in the rhTM group (4.43 ± 0.634) compared with the non-rhTM group (4.03 ± 0.169) (p = 0.001). Factors such as mean FDP and Prothrombin Time-International Normalized Ratio (PT-INR) were not significantly different between the two groups. Also, inflammation data, including White Blood Cell (WBC) and CRP, were not significantly different between the two groups. Mean APACHE II scores in the rhTM and non-rhTM groups were 23.1 and 21.6, respectively, with no significant difference (p = 0.335).
Comparison of characteristics of patients in the rhTM and non-rhTM groups.

APACHE, Acute Physiology and Chronic Health Evaluation; CRP, C-reactive protein; DIC, disseminated intravascular coagulation; FDP, fibrin/fibrinogen degradation product; rhTM, recombinant human soluble thrombomodulin.
Clinical results
Table 2 shows the clinical results in the rhTM and non-rhTM groups. ANC as a complication was observed in 53.6% (15/28) of rhTM and 57.1% (20/35) of non-rhTM group patients (p = 0.46). Among them, the rate of development of WON was significantly higher in the non-rhTM group (51.4%, 18/35) compared with the rhTM group (25.0%, 7/28) (p = 0.033). Although the mean duration of hospitalization was shorter in the rhTM group (31.9 days) compared with the non-rhTM group (50.6 days), the difference was not significant (p = 0.109).
Clinical outcomes in the rhTM and non-rhTM groups.

DIC, disseminated intravascular coagulation; rhTM, recombinant human soluble thrombomodulin; WON, walled-off necrosis.
As the primary outcome, the resolution rate of DIC within 7 days was significantly higher in the rhTM group (89.3%, 25/28) compared with the non-rhTM group (60.0%, n = 9/35) (p = 0.009). Mortality rate at 28 days, which was the secondary outcome, was not significantly different between rhTM (14.3%, 4/28) and non-rhTM groups (25.7%, 9/35) (p = 0.265), although the mortality rate within 14 days, which might have been strongly influenced by the presence of DIC, was significantly higher in the non-rhTM group (20.0%, 7/35) compared with the rhTM group (0%, 0/28) (p = 0.01). Finally, adverse events associated with bleeding after rhTM administration were not observed in any of the patients.
Risk factors associated with failed DIC resolution on day 7 and the development of WON
Risk factors associated with failed DIC resolution on day 7 were evaluated using logistic regression analysis (Table 3). According to univariate analysis, although age (>75 years, HR 1.15 95% CI: 0.626–2.11, p = 0.653), sex (male, HR 0.89, 95% CI: 0.468–1.69, p = 0.723), ICU admission (presence, HR 0.511, 95% CI: 0.468–1.692, p = 0.723), WBC count (>12,000/μL, HR 0.846 95% CI: 0.464–1.54, p = 0.585), CRP level (>10 mg/L, HR 0.984, 95% CI: 0.541–1.79, p = 0.959), DIC score (⩾5, HR 0.758, 95% CI: 0.338–1.70, p = 0.500), APACHE II score (⩾20, HR 0.993, 95% CI: 0.549–1.80, p = 0.982), and SOFA score (>3, HR 0.929, 95% CI: 0.432–1.99, p = 0.849) were not significant factors associated with failed DIC resolution on day 7, rhTM non-administration (HR 0.475, 95% CI: 0.26–0.867, p = 0.0153) was significantly related to DIC non-resolution. Furthermore, according to multivariate analysis as well, rhTM non-administration (HR 0.445, 95% CI: 0.239–0.830, p = 0.011) showed a significant association with failed DIC resolution on day 7.
Risk factors for failed DIC resolution on day 7.

APACHE, Acute Physiology and Chronic Health Evaluation; CRP, C-reactive protein; DIC, disseminated intravascular coagulation; rhTM, recombinant human soluble thrombomodulin; SOFA, sequential organ failure assessment.
Next, the risk factors associated with the development of WON were evaluated, as shown in Table 4. According to univariate analysis, although sex (male, HR 0.614, 95% CI: 0.184–2.05, p = 0.427), ICU admission (presence, HR 1.813, 95% CI: 0.599–5.49, p = 0.293), WBC count (>12,000/μL, HR 1.28, 95% CI: 0.451–3.64, p = 0.641), DIC score (⩾5, HR 0.511, 95% CI: 0.122–2.15, p = 0.360), APACHE II score (⩾20, HR 0.863, 95% CI: 0.304–2.45, p = 0.781), and SOFA score (>3, HR 0.511, 95% CI: 0.122–2.15, p = 0.360) were not significant factors associated with the development of WON, CRP level (>10, HR 4.67, 95% CI: 1.56–14.00, p = 0.0059) and rhTM non-administration (HR 3.177, 95% CI: 1.076–9.374, p = 0.0363) were significantly related to the development of WON. Furthermore, according to multivariate analysis, rhTM non-administration (HR 3.223, 95% CI: 1.077–9.648, p = 0.0365) was also significantly associated with the development of WON.
Risk factors for the development of WON.
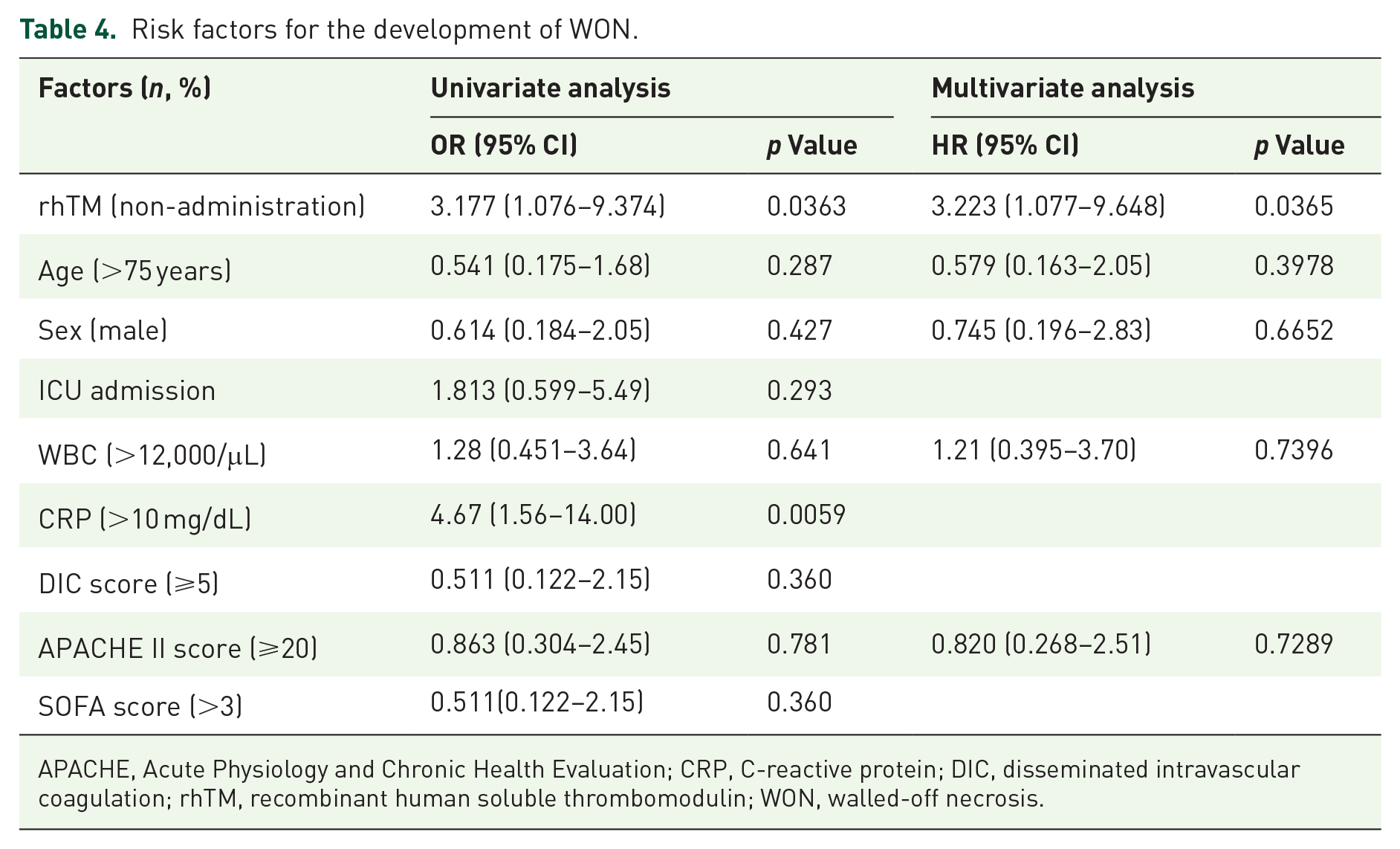
APACHE, Acute Physiology and Chronic Health Evaluation; CRP, C-reactive protein; DIC, disseminated intravascular coagulation; rhTM, recombinant human soluble thrombomodulin; WON, walled-off necrosis.
Discussion
SAP still has a high mortality rate, with the complication of WON further increasing the mortality rate. 10 The presence of DIC as a complication of SAP makes the situation even more critical. Hamada et al. performed a nationwide epidemiological survey in Japan to evaluate the influence of DIC on AP. 8 They compared mortality rates between AP (n = 256) and AP with DIC (n = 32), and observed that the mortality rate was significantly higher in AP with DIC (15.6%, 5/32) (p = 0.001). Therefore, to improve the mortality rate of SAP, DIC resolution and prevention of the development of WON are clinically important.
rhTM has been shown to exert anticoagulant effects by binding to thrombin and activating protein C. Recently, rhTM has been widely used as the key drug for treating DIC. According to a meta-analysis of the use of rhTM in DIC, including 17 studies and 2296 patients, 24 administration of rhTM significantly reduced mortality (OR 0.54, 95% CI: 0.42–0.71) and improved DIC resolution (OR 2.88, 95% CI: 1.83–4.52), with no significant differences in the incidence of bleeding complications between the rhTM and control groups (OR 0.92, 95% CI: 0.66–1.28). Yamakama et al also performed a nationwide observational study to research the time trends of outcome and treatment options for DIC in Japan from 2010 to 2021. 25 Their survey described the clinical status of the concomitant use of antithrombin and rhTM. During the observation period, 8% of DIC patients received antithrombin and rhTM concomitantly. Sequential use of the two anticoagulants, in which one drug was administered after the other was completed, was limited to only 3% of DIC patients. The upward trend indicates a preference for the concomitant use of antithrombin and rhTM. Also, rhTM possesses anti-inflammatory properties through its lectin-like domains. According to an experimental study using SAP rats, 26 20 rats were used for experiments in the SAP + rTM group, which were all alive at the end of the study, indicating a survival rate of 100%. By contrast, of 20 rats in the SAP group, only eight rats survived (survival rate: 40%). The survival rate was, thus, significantly better in the SAP + rhTM group than in the SAP group. The extent of edema was also significantly lower in the SAP + rhTM group than in the SAP group (mean (SD), 3.38 (0.30) vs 2.53 (0.20); p = 0.02). Histological examination in that study also showed that the extent of acinar necrosis was significantly milder in the SAP + rhTM group than in the SAP group (mean (SD), 2.75 (0.37) vs 0.90 (0.24); p = 0.0003), and microvessel counts of endothelial cells (mean (SD)) were 17 (5.12) in the normal group, eight (2.84) in the SAP group, and 13 (3.20) in the SAP + rhTM group. The differences in microvessel density between the normal and SAP group and between the SAP and SAP + rhTM groups were statistically significant. This suggests that rhTM might prevent the development of pancreatic necrosis by maintaining the microcirculation. Therefore, according to the above descriptions, rhTM administration might be both theoretically and practically reasonable for DIC resolution and for preventing the development of WON. Antithrombin III may also be key drug as another key drug for DIC treatment. According to recent meta-analysis regarding efficacy of antithrombin III for DIC including 95 patients (47 in the antithrombin III group, 48 in the control group), 27 the antithrombin III group’s DIC resolution rate was higher than the control group’s (OR = 5.21 (2.10, 12.90), p = 0.0004), while the 28-day mortality and bleeding-related adverse events did not differ significantly (OR = 0.45 (0.16, 1.31), p = 0.14; OR = 1.02 (0.22, 4.74), p = 0.98). In this meta-analysis, the clinical efficacy of antithrombin III was similar to rTM. However, because the clinical study of antithrombin III for DIC associated with SAP has not been sufficiently reported, further clinical evaluation is needed.
To date, only a few reports have evaluated the treatment of DIC secondary to SAP.12,13,28 Eguchi et al. evaluated whether rhTM might prevent the development of WON in SAP patients. 13 In their study, which included 54 AP patients, rhTM was administered to treat 24 AP patients with DIC, while the remaining 30 patients were included in the non-rhTM without DIC group. Although no significant differences in the rate of pancreatic necrosis/ischemia were observed between patients treated with rhTM on the admission day and controls (58.3% vs 63.3%, p = 0.71), the rate of development of WON was significantly lower in the rhTM group compared with the non-rhTM group {29.2% (7/24 patients) vs 56.7% (17/30 patients), p < 0.05}. Therefore, they concluded that rhTM might prevent progression from pancreatic necrosis/ischemia to WON. However, in their study, since they compared SAP patients with versus without DIC, the study population might have been inadequate. We believe that the efficacy of rhTM should be evaluated in a similar population setting, such as by including only DIC cases. In addition, their study did not describe the resolution rate of DIC. On the other hand, our comparative study only included SAP with DIC cases. In addition, our study showed that rhTM plays a role in SAP cases by not only preventing the development of WON but also improving the resolution rate of DIC. Yano et al. evaluated the effect of rhTM on prognosis in SAP patients with DIC. 28 In their study of 38 patients, rhTM was administered to 13 patients, and the mortality rate 60 days after ICU admission was significantly lower in the rhTM group (15%) compared with the control group (56%) (p = 0.036). Furthermore, the DIC resolution rate on day 7 was significantly higher in the rhTM group than in the control group (rhTM 62%, non-rhTM 24%, p = 0.035). In addition, logistic regression analysis revealed only rhTM as a contributory factor to survival on the 60th day (OR 12.5, 95% CI: 1.80–160, p = 0.009). However, although the results related to the DIC resolution rate were the same as those in our study, they found that the mortality rate was significantly improved in the rTM group at 60 days although this was not a significant difference at 28 days. Clinically, DIC might influence survival in the early phase during SAP treatment. Therefore, the role of rhTM for 60-day mortality might not be clear. On the other hand, in our study, although mortality rates at 28 days were not significantly different between the two study groups, the mortality rate within 14 days was significantly higher in the non-rhTM group compared with the rhTM group. Therefore, our results might suggest the utility of rhTM in clinical practice. However, our study has several limitations. First, it was a retrospective study. Second, our study involved a small patient cohort; therefore, our study should be evaluated using a larger patient cohort. In addition, because rTM administration was performed by the judgment of the attending physician, our study included selection bias in rTM administration. Finally, in our study, pancreatitis due to alcohol was mainly included. This rate might be higher compared with previous reports, because many patients were referred from hospitals where alcohol-dependent cases were treated. Therefore, the etiology of our study might be different for many centers.
Conclusion
In conclusion, rhTM may play a role not only in improving the resolution rate of DIC and the survival rate of SAP but also in preventing the development of WON. Further randomized controlled trials are needed to verify our results.
Supplemental Material
sj-doc-1-tag-10.1177_17562848251353626 – Supplemental material for Clinical impact of recombinant thrombomodulin administration on disseminated intravascular coagulation due to severe acute pancreatitis (Recover-AP study)
Supplemental material, sj-doc-1-tag-10.1177_17562848251353626 for Clinical impact of recombinant thrombomodulin administration on disseminated intravascular coagulation due to severe acute pancreatitis (Recover-AP study) by Takeshi Ogura, Saori Ueno, Akitoshi Hakoda, Atsushi Okuda, Nobu Nishioka, Jun Sakamoto, Jun Matsuno, Yuki Uba, Mitsuki Tomita, Nobuhiro Hattori, Junichi Nakamura, Kimi Bessho, Taro Iwatsubo, Ahmad F. Aboelezz and Hiroki Nishikawa in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
