Abstract
Background:
Liver disease is a leading cause of morbidity and mortality. Patients with end-stage liver disease (ESLD) experience multiple physical symptoms. Despite the poor prognosis and significant symptom burden, palliative care integration remains limited.
Objectives:
To assess the symptom burden in ESLD patients, the viability of applying a symptom scale in routine evaluations, and to assess the impact of palliative care on symptom management.
Design:
Observational, prospective cohort study.
Methods:
We prospectively included patients with chronic liver disease following their first episode of decompensation or diagnosis of hepatocarcinoma (HCC). Data collected included patient demographics, ESLD etiology, history of decompensation, and patient-reported symptom burden. Two-sided tests were used to identify factors of disease severity and evaluate the benefits of palliative care intervention.
Results:
Forty-four patients were assessed, divided into two cohorts: palliative care cohort (52.3%; n = 23) and hepatology care cohort (47.7%; n = 21). Patients in the palliative care cohort were older (69.35 ± 11.71 vs 59.86 ± 7.11 years; p = 0.002), had lower functional status (59.13 ± 2.51 vs 72.38 ± 2.92; p = 0.002), and higher prevalence of unstable decompensated cirrhosis (60.9% vs 28.6%; p = 0.043) and HCC (p < 0.001). This cohort reported a higher overall symptom burden, with rates of 82.6% for asthenia, 65.2% for pain, and 56.5% for anorexia. Palliative care interventions tended to reduce the prevalence of pain, anorexia, and dyspnea, with a significant decrease in pain intensity from 86.7% to 23.1% (p = 0.008) and asthenia intensity from 100% to 84.2% (p < 0.001).
Conclusion:
Significant differences in symptom prevalence were observed between the two cohorts, likely due to specific clinical characteristics of each group. The use of a symptom assessment scale proved to be simple and effective, revealing a high prevalence of symptoms. Palliative care was associated with a positive impact on symptom management.
Trial registration:
NCT06181474.
Plain language summary
Liver disease is a major health issue, with people with severe liver disease often facing many uncomfortable symptoms. This study aimed to understand the symptoms experienced by patients with severe liver disease, how these symptoms relate to the severity of their condition, and the way palliative care could help manage these symptoms. The researchers included patients who had recently experienced a worsening of their condition or were diagnosed with liver cancer. The study involved 44 patients, divided into two groups: one receiving palliative care (23 patients) and another receiving standard liver care (21 patients). The palliative care group had older patients with more severe symptoms. Palliative care was found to help reduce the pain and improve the management of other symptoms. The study found notable differences in symptoms between the two groups, likely because of the unique health conditions of each one of them. Overall, palliative care can have a positive effect on managing symptoms for those patients.
Introduction
Liver cirrhosis represents an advanced stage in the continuum of liver disease, initially characterized by an asymptomatic period known as compensated cirrhosis. This phase can progress to decompensated cirrhosis when complications associated with portal hypertension or liver dysfunction, such as ascites, hepatic encephalopathy, or hepatocarcinoma (HCC), arise.1,2 The transition to the end-stage liver disease (ESLD) phase, through the appearance of one or more complications, translates into a reduction in survival, which, on average, is estimated at 2 years. 1 In addition to these complications, some patients may develop acute-on-chronic liver failure (ACLF), a clinical syndrome characterized by multiple organ dysfunctions and high short-term mortality after an episode of liver decompensation.3,4 Recent evidence indicates that the clinical phenotype of each patient has a significant impact on prognosis.5,6 Patients may present with pre-ACLF, defined as acute decompensation of cirrhosis that progresses to ACLF within a 90-day follow-up period; unstable decompensated cirrhosis (UDC), characterized by rehospitalization due to acute decompensation within 3 months of the initial episode; or stable decompensated cirrhosis (SDC), referring to those who do not require rehospitalization for acute decompensation during the same timeframe.5,6
Liver disease is a leading global cause of morbidity and mortality, accounting for approximately 1–2 million deaths/year,7,8 primarily affecting the young adult population (25–64 years). 9 The SUPPORT study indicates that patients experiencing their first hospitalization episode present a 29% mortality rate, with another 29% patients dying within the first year. 10 This study demonstrated that patients with ESLD experience a marked decline in quality of life, characterized by a wide range of physical, cognitive, and social symptoms that are often challenging to manage. 10 Commonly reported symptoms include pain, dyspnea, fatigue, and mental confusion. 10 A 2019 meta-analysis conducted by Peng et al. 11 reported symptom prevalence ranging from 30% to 79% for pain, 20% to 88% for dyspnea, 56% to 68% for muscle cramps, 52% to 86% for asthenia, and 47% to 64% for pruritus. Notably, the majority of the studies included in the analysis reported the presence of only one or two symptoms per patient. 11
Palliative care is a specialized, multidisciplinary medical care that addresses the physical, psychological, social, and spiritual needs of patients with advanced illness and their caregivers, regardless of prognosis and survival.12,13 Furthermore, a pivotal study by Baumann et al. 14 demonstrated that referral to palliative care for patients on the liver transplant list resulted in symptomatic improvement. Moreover, a growing body of evidence highlights the benefits of implementing palliative care in ESLD patients. In addition to symptomatic control, palliative care teams facilitate timely discussion of the care plan, helps in reducing unnecessary hospitalizations and in better-coordinated care, also being important in supporting caregivers,10,15,16 which led to publication by the American Association for the Study of the Liver of the first recommendations on integrating palliative care and symptom management in patients with decompensated cirrhosis and incorporating palliative care elements into multidisciplinary decision-making for patients with HCC.17,18 Despite increasing recognition of the benefits of early palliative care in ESLD, multiple studies have identified significant barriers to its implementation by specialists.19,20 These include the widespread misconception among patients that palliative care is exclusively associated with end of life, the continued prioritization of curative treatment strategies by attending physicians, and the insufficient number of available palliative care specialists.19,20
One crucial question that emerged is when to refer ESLD patients to palliative care. In fact, traditional models, such as the Supportive & Palliative Care Indicators Tool (SPICT) and the NECPAL CCOMS-ICO Tool©, identify patients with signs of deterioration in their health status, but fail to present objective data on the severity of underlying liver disease.21,22 Specific models for ESLD patients have been developed, aiming to demonstrate the relationship between several variables, like the Model for End-Stage Liver Disease (MELD) score, associated mortality, and the implementation of palliative care.23–25 However, these models, which tend to be mathematical, may not be able to demonstrate patient frailty or symptomatic burden. Effectively, studies show that even with low MELD scores, up to 70% of these patients may present with a high symptom burden. 26
The primary objectives of this observational study are to assess the symptom burden experienced by ESLD patients, the viability of implementing a symptom scale in routine evaluations of ESLD patients, and to evaluate the intervention of palliative care on the symptom burden of ESLD patients. We also aim to explore the correlation of the symptom burden with MELD-sodium (MELD-Na) score and each patient’s clinical phenotype.
Materials and methods
Study design
This study is an observational, prospective cohort study of patients with chronic liver disease following their first episode of decompensation or diagnosis of stage C or D HCC, according to the Barcelona Clinic Liver Cancer (BCLC) criteria. 27 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Figure 1 and Supplemental Material). 28

STROBE flow chart.
Patients and setting
Between January and October 2024, all adult patients with chronic liver disease were consecutively enrolled following either their first episode of decompensation or a new diagnosis of HCC. Recruitment took place at a Portuguese secondary referral urban hospital equipped with comprehensive facilities, including integrated hepatology, oncology, and palliative care services. The study employed a convenience and purposive sampling strategy without randomization. Exclusion criteria included: (1) patients under 18 years old; (2) patients with chronic liver disease who died from causes unrelated to the disease progression; (3) presence of HCC without cirrhosis; (4) presence of non-hepatic cancer; and (5) persistent hepatic encephalopathy, psychiatric disorders, dementia, or other cognitive impairments preventing informed consent.
Eligible patients were approached by the study staff (which included a physician or a nurse) to obtain informed consent. Referrals to palliative care were carried out independently by the clinicians, without influence from the study staff. During the study period, a total of 49 patients were assessed for eligibility. Of these, five patients (10.2%) were excluded: two declined to participate, one was lost to follow-up, and two developed non-hepatic malignancies during the follow-up period. The final analysis included 44 patients, who were stratified into two cohorts: those referred to palliative care, comprising the palliative care cohort, and those managed exclusively by hepatology services, comprising the hepatology care cohort.
Data collection
Clinical characteristics were collected from electronic medical records at the time of enrollment and during subsequent follow-up chart reviews. These included the etiology of liver disease, classified as alcohol-related, metabolic dysfunction-associated steatotic liver disease, viral hepatitis B or C, or other causes. Additional data comprised cirrhosis-related complications such as ascites, hepatic encephalopathy, and esophageal variceal bleeding; disease severity measured by the MELD-Na and Child-Pugh scores; functional status assessed using the Karnofsky Performance Scale; and comorbidities evaluated through the Charlson Comorbidity Index.26,29–31
Patient-reported symptom burden was assessed resorting to the revised Edmonton Symptom Assessment System (ESAS-r) scale.32,33 The ESAS-r evaluates the severity of nine common symptoms experienced by patients with serious illnesses (pain, tiredness, nausea, depression, anxiety, drowsiness, appetite, well-being, and shortness of breath), with an option to include a 10th symptom. We specifically included cramps and pruritus, which are prevalent in patients with chronic liver disease. Symptoms were rated on a numerical scale from 0 to 10 (0 indicating absence and 10 indicating the worst severity). The choice for this scale was due not only to its recurrent use among palliative care teams but also to existing studies demonstrating its applicability in patients with ESLD. 34
All patients underwent a minimum of two assessments. In the palliative care cohort, symptoms were evaluated at the initial consultation and reassessed 5 days following the intervention. In the hepatology care cohort, assessments were conducted 1 month after the episode of decompensation and again 3 months later.
Statistical analysis
Statistical analysis was carried out using Statistical Package for the Social Sciences for Mac (version 26), IBM® SPSS®. Descriptive statistics were used to present continuous variables as mean and standard deviation, and categorical variables as frequency and percentage. For each item of the ESAS scale, we report the overall symptom prevalence and the percentage of patients with a moderate to severe symptom burden (defined as an individual item score ⩾4). The statistical significance of the differences between the two groups was assessed using the appropriate tests. Comparisons between categorical variables were performed with the Pearson’s χ2 test or Fisher’s exact test, where appropriate. Comparisons between continuous variables were carried out using Student’s t test for normally distributed variables. If it was not possible to apply the t test, we resorted to the nonparametric Mann-Whitney test. The nonparametric Wilcoxon test was used to compare related (paired) results. Statistical significance was determined by a two-sided p-value of less than 0.05, or a 95% confidence interval that did not include 1.
Ethical considerations
The study protocol complied with the ethical principles outlined in the 1975 Declaration of Helsinki and was approved in advance by the institutional Ethics Committee (reference number 131/CES/JAS). Informed consent was obtained from all participants, who received no financial compensation for their participation. To ensure anonymity, all personal identifiers were removed, and each participant was assigned an alphanumeric code, accessible only to the principal investigator. The study was registered on ClinicalTrials.gov under the identification number NCT06181474.
Results
General characterization of the sample
A total of 44 patients were evaluated, and all their data were complete. They were divided into two cohorts: those referred to palliative care (52.3%; n = 23—palliative care cohort) and those receiving standard hepatology care (47.7%; n = 21—hepatology care cohort). Most patients were male, with similar distributions in both groups (Table 1). The average age of participants was 64.82 ± 10.80 years (Table 2), with the palliative care cohort being older compared to the hepatology care cohort (69.35 ± 11.71 vs 59.86 ± 7.11 years; p = 0.002). Karnofsky functional status revealed that the palliative care group had a lower average score compared to the hepatology cohort (59.13 ± 2.51 vs 72.38 ± 2.92; p = 0.002). According to the Charlson Comorbidity Index, patients in the palliative care cohort had a higher score versus those in the hepatology care cohort (6.52 ± 1.81 vs 3.95 ± 1.63; p < 0.001).
Characterization of the population and underlying liver disease.

p, significance level.
ACLF, acute-on-chronic liver failure; n.d., not detectable.
Statistically significant findings are marked in bold.
Characterization of age, Charlson index, Karnofsky score, MELD-Na, number of hospitalizations, and number of decompensations.

Statistically significant findings are marked in bold.
IQR, interquartile range; MELD-Na, model for end-stage liver disease-sodium.
The main cause of ESLD was alcohol 72.7% (n = 32), with similar distributions in both groups. Main forms of decompensation included ascites (44.7%; n = 17) and hepatic encephalopathy (21.1%; n = 8), with similar distributions between cohorts (Table 1). The MELD-Na score documented in the population undergoing palliative care follow-up was 20.43 ± 1.43 versus 16.81 ± 1.31 (p = 0.071). Notably, there was a progressive improvement in the MELD-Na score over time for the hepatology cohort (from 16.81 ± 1.31 to 13.90 ± 1.15; p = 0.017).
Regarding the clinical phenotype, the palliative care group had a higher prevalence of UDC type (60.9% vs 28.6%; p = 0.043). Average hospitalization rates were similar for both cohorts (1.57 ± 0.95 vs 1.48 ± 1.08; p = 0.414), as were rates of decompensation (2.09 ± 1.38 vs 1.62 ± 1.07; p = 0.219). The prevalence of HCC in the study population was 38.6%, with 10 patients in BCLC stage C and 7 in stage D, all within the palliative care cohort (p < 0.001). During the follow-up period, 50% (n = 22) of the patients died, with 87% (n = 20) of those belonging to the palliative care cohort (p < 0.001).
Symptomatic burden in ESLD patients
In the studied population, four symptoms were reported with a prevalence exceeding 30%: asthenia (77.3%), pain (43.2%), anorexia (43.2%), and drowsiness (34.1%). In contrast, the prevalence of cramps and pruritus (specific symptoms of ESLD) was low, at 9.1% and 0%, respectively.
The palliative care cohort exhibited (Figure 2) a higher overall symptom burden, with rates of 82.6% for asthenia, 65.2% for pain, and 56.5% for anorexia at initial assessment. Notably, all patients with asthenia reported a moderate to high intensity. In terms of pain, 86.7% (n = 13) rated it as moderate to high intensity.
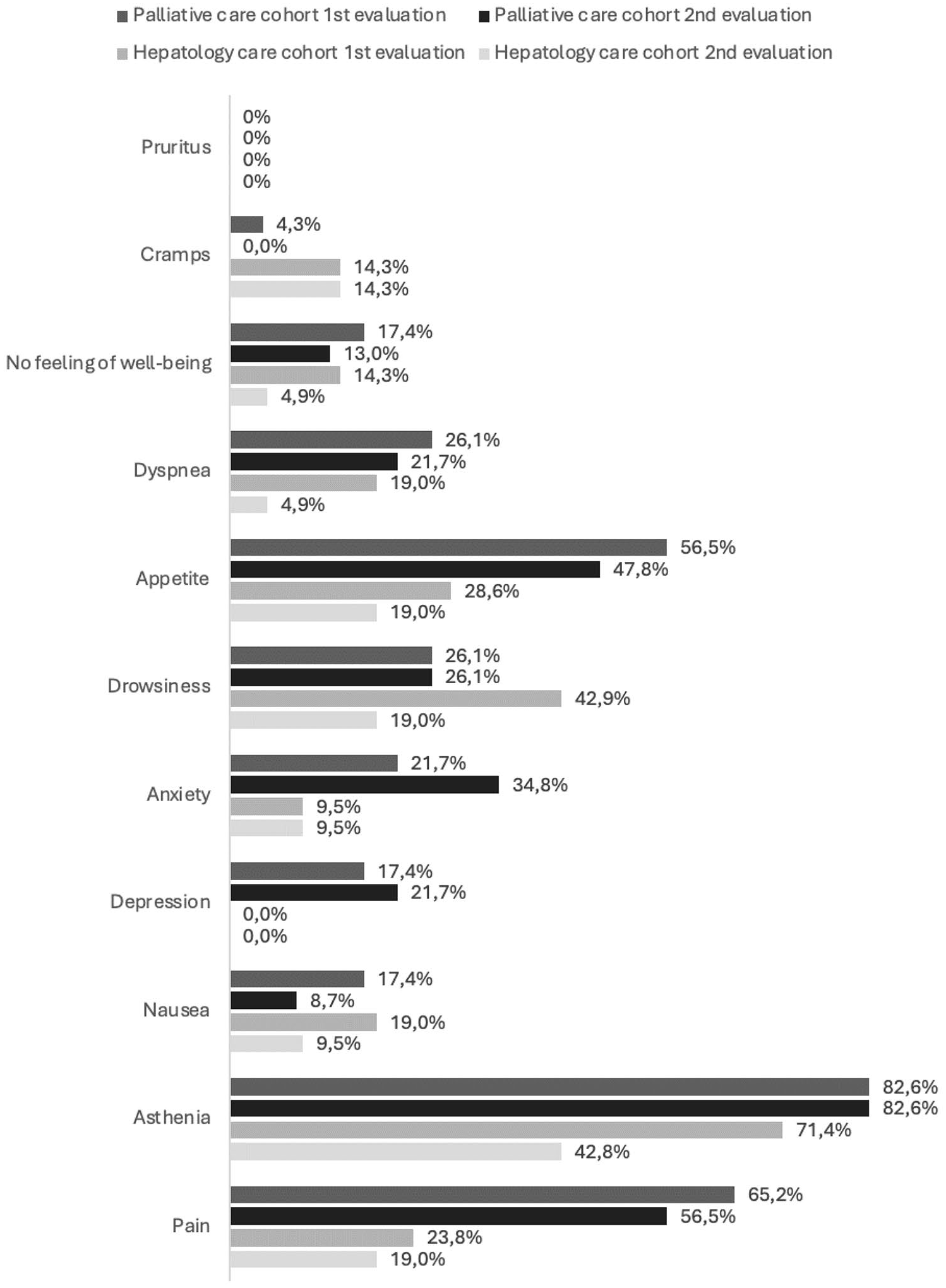
Prevalence of symptoms by moment of assessment in the populations under study.
In contrast, the hepatology care cohort exhibited a significantly higher proportion of asymptomatic patients and generally reported symptoms of lower intensity. At the initial assessment, the prevalence rates were 71.4% for asthenia, 42.9% for drowsiness, 28.6% for anorexia, and 23.8% for pain. Within this cohort, 46.7% (n = 7) of patients described asthenia as moderate to severe, while 44.4% (n = 4) rated drowsiness as of moderate intensity. By the 3-month reassessment following the initial decompensation event, the prevalence of asthenia and drowsiness had declined substantially to 42.8% and 19.0%, respectively.
Impact of palliative care on symptom burden
On average, there were 44.43 ± 40.06 days between the initial decompensation and the evaluation by the palliative care team. Five days following the initial intervention by the palliative care team, the prevalence of pain decreased (Table 3) to 56.5%, anorexia to 47.8%, and dyspnea to 21.7%. Moderate to high intensity pain showed a reduction from 86.7% to 23.1% (p = 0.008). The prevalence of asthenia remained unchanged at 82.6%, but there was a reduction of moderate to high intensity asthenia, from 100% to 84.2% (p < 0.001). Furthermore, anxiety and depression symptoms worsened between the two assessments (Figure 2).
Outcomes of symptoms by group and evaluation period.

Fisher’s exact independence test was applied to infer the possible association of symptoms with the group under study at each assessment.
McNemar test for each group considering the assessments performed (paired).
n.d., not detectable.
Statistically significant findings are marked in bold.
During the follow-up period, the palliative care cohort exhibited a mortality rate of 87% (n = 20). The mean duration from the initial palliative care assessment to death was 55.55 ± 59.42 days, with 50% of patients surviving for at least 90 days following the assessment.
Discussion
Our study shows the considerable symptom burden in patients with ESLD, with significant differences in symptom prevalence between the palliative care and hepatology care cohorts, possibly due to specific clinical characteristics of each group. We highlight that the palliative care group involved older patients, with a worse functional score and higher comorbidities. In this context, implementing a symptom assessment scale in routine evaluations proved to be simple and effective. Palliative care was associated with a reduction in symptom burden, spotlighting the need for its broader integration in ESLD care.
We documented a prevalence of asthenia, pain, anorexia, and drowsiness in this population, which closely aligns with the data reported by Peng et al., 11 corroborating findings from the SUPPORT study, which emphasizes the substantial symptom burden experienced by patients with ESLD. 10 However, with no explanation found for this difference, the prevalence of cramps and pruritus in our study was significantly lower. 11 We found considerable differences in symptom prevalence between patients referred to palliative care and those who were exclusively monitored by hepatology. Patients in the latter group exhibited a higher proportion of asymptomatic cases and generally reported symptoms of lower intensity.
Although the two cohorts shared similar characteristics, including the etiology of ESLD, type of decompensation, number of hospitalizations, and notably comparable MELD-Na scores, there may be no direct association between MELD score and symptomatic burden. This observation aligns with previous studies suggesting that the MELD scale does not reliably reflect either the need for palliative care or the intensity of symptomatology.26,34–39 We identified other specific differences between the populations. Patients in the palliative care cohort tended to be older, had lower functional status, and exhibited more comorbidities. These observations corroborate studies indicating that older patients are more likely to be referred to palliative care,40–43 in addition to support the classic models for identifying patients with deterioration in their health status, such as SPICT and NECPAL.21,22 In fact, the functional status appears to be the most consistent parameter between classical models and more specialized models for the ESLD population.23–25 In addition, some studies indicate that lower functional status is associated with a greater symptom burden,44,45 potentially explaining the higher symptom prevalence observed in the palliative care cohort. Although functional status may not be an ideal prognostic marker, it is a crucial indicator of frailty and palliative needs, reinforcing that referrals should be based on patient needs rather than on prognosis.
Our analysis revealed that the clinical phenotype of UDC was more prevalent in the palliative care cohort, which had a significantly higher mortality rate (87%). Conversely, the hepatology cohort frequently displayed the SDC phenotype, associated with a lower mortality rate. These findings reaffirm that patients with ESLD progressing to ACLF or UDC have a poorer prognosis, even in subclinical cases.5,6,46 It is not possible to establish a definitive association between clinical phenotype and symptom prevalence, as multiple interacting factors may influence this relationship. This notion is supported by findings from a study conducted by Hansen et al., 39 which had previously highlighted the complexity of these interactions. However, identifying these clinical phenotypes remains essential for optimizing the allocation of healthcare resources and ensuring more targeted and effective care strategies.
Furthermore, our study documented a high prevalence of HCC in the palliative care cohort, which demonstrates that referral to palliative care seems to be easier when ESLD patients are diagnosed with HCC,37,40,43,47–49 highlighting a potential direct association between cancer diagnosis and palliative care access.
The ESAS scale, which is adapted and validated in several languages, proved to be simple to implement, echoing findings from a recent study. 34 There was a need for initial training in its application, but its regular practice allows to create a routine without increasing consultation time. We believe that the routine use of this scale could facilitate the prompt identification of symptoms requiring intervention in patients with ESLD. Nevertheless, future research should explore feasibility in different clinical settings.
We demonstrated that intervention by the palliative care team led to a reduction in the prevalence of pain, anorexia, and dyspnea, with a significant reduction in pain intensity. In addition, while the prevalence of asthenia remained unchanged, its intensity also decreased. The benefit of implementing palliative care in the symptomatic improvement of patients with ESLD has already been referred to in the pioneering study carried out by Baumann et al. 14 However, published studies about this topic and the optimal pharmacological strategies for this population are limited. 50 An increasing prevalence of anxiety and depression has also been observed, potentially linked to heightened awareness of disease progression. This underscores the critical importance of integrating psychological support within palliative care teams to address the profound emotional response many patients experience, helping them coping with ESLD. 51
It is important to foreground that in the hepatology cohort, the stabilization and improvement of ESLD through medical management of ascites and hepatic encephalopathy, as well as nutritional support, contributed to symptom alleviation, strengthening what is mentioned in international recommendations about the importance of the best possible control of ESLD complications in symptom control. 17
Moreover, there are also other data that should be spotlighted. The referral rate for palliative care in our study was 52.3%, which is consistent with recent literature and higher than previously reported data in our country,35,48,52–54 which indicates a growing integration of palliative care in ESLD management. We also observed a marked increase in the average time between palliative care assessment and death, with 50% of patients presenting a period of at least 90 days, representing a significant improvement over previous reports.38,53
There are some limitations to our study, notably the unicentric nature that limits data generalization, as well as the sample size, which was smaller than our initial goal, potentially reducing statistical power and limiting detection of other significant differences, namely in the analysis of palliative care on symptom management. Nevertheless, the sample reflects our national scale and is close to the study published by Shinall et al. 55 We must also underline that the palliative care group patients had specific characteristics: they were older patients, had a worse functional score, and a high percentage of HCC, which may impact the symptoms described. The challenges in patient enrollment and follow-up required adjustments to our initial protocol, which aimed for quarterly evaluations or assessments following each decompensation episode in the hepatology cohort.
Despite these limitations, our study provides noteworthy insights: it applies a recognized symptomatic assessment scale to all ESLD patients, demonstrating a considerable prevalence of symptoms; it showed a growing a positive impact of palliative care on symptom management; and it emphasizes the critical role of the prompt identification of symptoms in promoting timely referrals to palliative care for ESLD patients. Furthermore, we can consider this as a pilot study with the aim of progressing toward a multicentric analysis.
Conclusion
Patients with ESLD carry a significant symptom burden. The routine use of symptom assessment scales is effective in facilitating the timely identification of symptoms requiring intervention. The positive impact of palliative care on patients with ESLD highlights the importance of its broader integration into standard ESLD management.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251353624 – Supplemental material for Symptom burden in end-stage liver disease: a prospective cohort study of the symptoms experienced by patients and the role of palliative care
Supplemental material, sj-docx-1-tag-10.1177_17562848251353624 for Symptom burden in end-stage liver disease: a prospective cohort study of the symptoms experienced by patients and the role of palliative care by Hugo M. Oliveira, Filipa Ribeiro, Graça Lopes, Eliana Frias, Filipe Andrade, Verónica Guiomar, Eduardo Eiras, Francisca Rego and Rui Nunes in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
