Abstract
Objective
To investigate the hepatic effects of high-dose intravenous (IV) iron, including those on liver function and the degree of fibrosis, in a rat model of cirrhosis.
Methods
We evenly allocated 25 Sprague–Dawley rats into five groups: normal rats (control group), cirrhotic rats receiving IV normal saline (liver cirrhosis [LC] group), and cirrhotic rats receiving 20, 40, or 80 mg/kg IV ferric carboxymaltose (LC-iron20, LC-iron40, and LC-iron80 group, respectively). Biochemical parameters were compared at 0, 7, 14, 21, and 28 days. The degrees of hepatic fibrosis and iron deposition were evaluated. Inflammatory and oxidative stress markers were also compared.
Results
There were no significant differences in the 28-day serum alanine aminotransferase levels among the LC-iron20, LC-iron40, and LC-iron80 groups (69 ± 7, 1003 ± 127, 1064 ± 309, 919 ± 346, and 820 ± 195 IU/L in the control, LC, LC-iron20, LC-iron40, and LC-iron80 groups, respectively). Hepatic iron accumulation increased in a dose-dependent manner, but the degree of hepatic fibrosis was comparable among the groups. The inflammatory and oxidative stress marker levels did not differ significantly according to the IV iron dose.
Conclusions
Administration of IV iron at various high doses appears safe in our rat model of cirrhosis.
Keywords
Introduction
Chronic liver disease often results in anemia, which affects approximately 75% of patients with advanced liver disease. 1 These patients’ underlying causes of anemia are multifactorial, including bone marrow suppression due to viruses or alcohol use, renal insufficiency, and variceal bleeding.2–4 Iron homeostasis is a complex process that can be significantly disrupted in patients with different etiologies and stages of chronic liver disease. Iron deficiency anemia is often documented in this population because of chronic blood loss. 5 Iron deficiency is a treatable cause of anemia, and several studies have demonstrated the benefits of iron treatment for patients in various surgical settings.6,7 However, preoperative iron therapy has not been established for patients undergoing hepatectomy or liver transplantation because of concerns about excessive iron overload and the potentially negative clinical outcomes of iron accumulation in the livers of patients with cirrhosis.8,9
Our previous animal study 10 showed that a single dose of intravenous (IV) iron (20 mg/kg) did not significantly change liver function test results or the degree of liver fibrosis in cirrhotic rats. Although these results provide preclinical evidence that IV iron injection might not impair liver function or aggravate the degree of fibrosis and that it might provide benefits in terms of inflammation and oxidative stress, we used only one dose of iron (20 mg/kg, which is the standard dose recommended by the manufacturer). Therefore, further preclinical experiments using higher doses of IV iron in a rat model of cirrhosis are needed to identify and confirm safe and effective dosing levels. In the present study, we further investigated the hepatic effects of various doses of ferric carboxymaltose, an IV iron supplement, in a rat model of cirrhosis.
Materials and methods
Study design and treatment
In total, 27 male Sprague–Dawley rats aged 5 weeks and weighing 140 to 160 g were housed in an experimental animal unit at 23°C ± 3°C, 55% ± 15% relative humidity, 10 to 20 cycles/hour ventilation frequency, 12-hour light/dark cycles, and 150 to 300 lux. The environmental conditions were regularly assessed during the experimental period, and no changes likely to affect the test results were observed. Dreambio standard rat chow (Cargill Agripurina Co., Ltd., Seoul, South Korea) and water from a polycarbonate drinking bottle were provided
Establishment of rat model of liver cirrhosis
Figure 1 shows the overall study design, including the pilot study and main experiment. Carbon tetrachloride (CCl4) was dissolved in purified olive oil to a concentration of 50% and administered twice weekly via intraperitoneal injection (2 mL/kg). 12 Blood samples were obtained at 8 weeks, and the aspartate aminotransferase (AST) and alanine aminotransferase (ALT) concentrations were measured to assess the state of hepatocellular injury. The blood samples were collected through the tail vein, and the serum was centrifuged at 3000 rpm for 10 minutes. The AST and ALT levels were measured using an automatic blood biochemical analyzer. Liver fibrosis was confirmed by gross histopathological examination and Sirius red staining of liver samples collected from two rats not included in the grouped analyses described below. Following administration of the experimental and control substances, CCl4 injection was continued twice weekly until euthanasia to maintain the cirrhotic state of the liver in all animals.
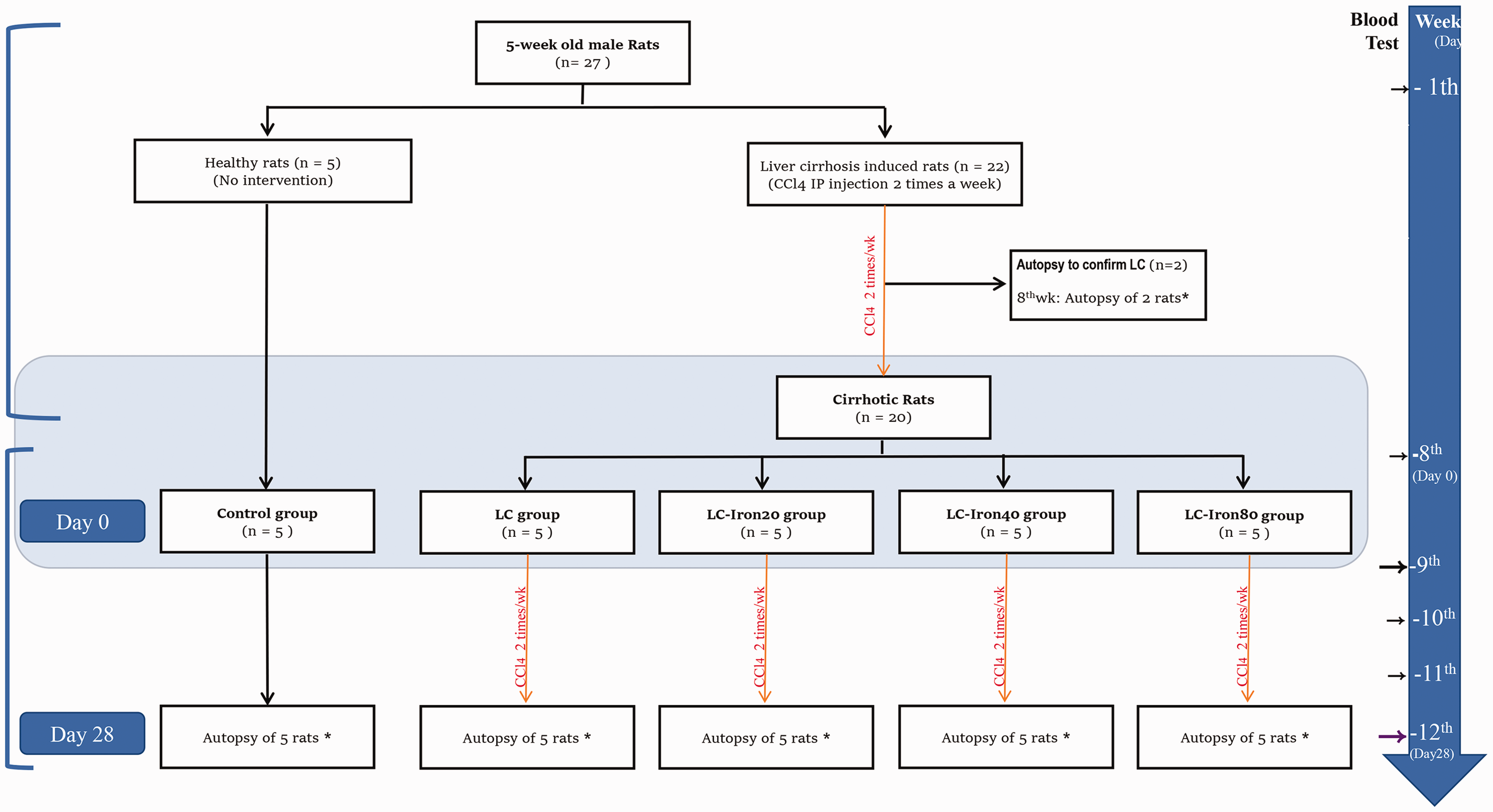
Diagram of experimental protocol. CCl4, carbon tetrachloride; LC, liver cirrhosis; Wk, week; Control group: normal, healthy rats without cirrhosis; LC group: rats with induced liver cirrhosis that received CCl4 twice weekly with intravenous saline; LC-iron20 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 20 mg/kg intravenous iron on day 0; LC-iron40 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 40 mg/kg intravenous iron on day 0; LC-iron80 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 80 mg/kg intravenous iron on day 0. Day 28 reflects the time after infusion of iron. The red arrow indicates CCl4 intraperitoneal injections two times a week. The black arrow indicates the timing of blood sample collection for liver function tests. The purple arrow indicates the timing of blood sample collection for liver function tests and measurement of iron-related parameters. *Autopsy for histopathologic examination.
Main experiment
Twenty-five rats were equally allocated into five groups. Five normal, healthy rats without cirrhosis were maintained as a baseline control group. Of the 20 remaining rats (all with cirrhosis), 5 continued to receive CCl4 twice weekly with IV saline (liver cirrhosis [LC] group), and 15 continued to receive CCl4 in combination with iron treatment (Ferinject; Vifor Pharma, Glattbrugg, Switzerland) at three doses: 20 mg/kg iron [LC-iron20 group], 40 mg/kg iron [LC-iron40 group], and 80 mg/kg iron [LC-iron80 group]. 13 The investigators were blinded to the animal treatments. Ferinject is a recently developed IV iron supplement that contains ferric carboxymaltose as its main component.14,15 In rat models, Ferinject has shown greater liver, heart, and kidney function benefits than those provided by other ferric carboxymaltose agents. 16
After confirmation of LC, the animals in the LC-iron groups were fixed in a calibrated frame. Iron was slowly injected through the tail vein at a rate of 1 mL/minute using a syringe with a 26-gauge needle. The dose contained 20, 40, or 80 mg iron/kg body weight of Ferinject diluted with normal saline and was adjusted for the body weight of each animal. This drug was administered only once. All experimental protocols performed in this study were approved by the institutional committee of KNOTUS Co., Ltd. and conducted at the KOTUC Co., Ltd. laboratory in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (Bethesda, MD, USA).
Necropsy
Twenty-eight days after iron administration, five animals from each group were anesthetized by 1.9% inhaled ether (0.08 mL/L of container volume). Blood was collected via posterior vena cava puncture, and the animals were euthanized by exsanguination. Liver tissues were harvested, and the right lobe was divided into two sections: one was frozen at −70°C, and the other was fixed in 10% neutral buffered formalin. The left lobe was fixed in a 10% neutral buffered formalin solution. During the surgery, portal vein pressure was measured by direct cannulation using a 26-gauge needle.
Biochemistry
Blood samples were collected before injection and at 7, 14, 21, and 28 days after iron administration. Complete blood count vials containing ethylenediaminetetraacetic acid dipotassium as an anticoagulant were used to collect samples for hemoglobin and mean corpuscular volume measurements using a hematology system (ADVIA 2120; Siemens Healthineers, Erlangen, Germany).
Portions of the blood collected before iron treatment and at 7, 14, 21, and 28 days after treatment were retained in a vacutainer tube containing a clot activator and incubated for 15 to 20 minutes at room temperature. Serum was centrifuged at 3000 rpm for 10 minutes, and the AST and ALT concentrations were measured using an automatic biochemical blood analyzer.
Histology and quantification of fibrosis
The extracted liver tissues were fixed, embedded in paraffin, sectioned, and stained using hematoxylin–eosin and Prussian blue to identify fibrosis and iron deposits, respectively. Histopathological changes were observed using an optical microscope (BX53; Olympus, Tokyo, Japan). Morphological examinations were performed at a magnification of 100×. In all instances, two independent observers performed blinded evaluations, and mean percentages were calculated. Histochemical localization in the liver tissue of each rat was performed using a computer and image analyzer and expressed as the percentage of the area per millimeter of positive iron deposit staining. Liver cell fibrosis was scored from 0 to 4 17 : 0 = no sign of fibrosis or cirrhosis; 1 = enlarged, fibrotic portal tracts; 2 = periportal or portal–portal septa but intact architecture; 3 = fibrosis with architectural distortion but no obvious cirrhosis; and 4 = probable or definite cirrhosis. Iron accumulation was scored from 1 to 2. 18
Chemicals and reagents
Stored blood and liver tissue samples collected on day 28 were further analyzed using enzyme-linked immunosorbent assays and other assay kits. Serum was isolated for analysis of the levels of the total iron binding capacity, serum iron (cat. no. ab239715; Abcam, Cambridge, UK), ferritin (cat. no. ab157732; Abcam), C-reactive protein (cat. no. ab260058; Abcam), and hepcidin (cat. no. CSB-EL0 101 24RA; Cusabio, Houston, TX, USA). Proteins were isolated from liver tissues to measure the levels of malondialdehyde (cat. no. ab238537; Abcam), tumor necrosis factor-α (cat. no. ab100785; Abcam), and interleukin-6 (cat. no. ab100772; Abcam). The absorbance was measured using a microplate spectrophotometer (Epoch; BioTek, Winooski, VT, USA).
Statistical analysis
The normality of the data distribution was assessed using the Kolmogorov–Smirnov test. Data with a normal distribution were compared using one-way analysis of variance with the Bonferroni post hoc test to assess differences among groups. Data without a normal distribution were compared using the Friedman test and Dunn post hoc test. Multiple groups were compared using the Kruskal–Wallis test. Quantitative variables are expressed as median (interquartile range) or mean ± standard deviation, and qualitative variables are expressed as proportion (%) unless otherwise noted. All comparisons were two-tailed, and a
Results
Confirmation of liver fibrosis
Eight weeks after initiation of CCl4 administration, the AST concentration was significantly higher in the LC (1201 ± 252 IU/L), LC-iron20 (1246 ± 102 IU/L), LC-iron40 (1236 ± 223 IU/L), and LC-iron80 (1215 ± 1135 IU/L) groups than in the control group (65 ± 7 IU/L,
Main experiment: Liver fibrosis and iron deposition
Histological examination indicated that CCl4 administration significantly decreased the degrees of freedom from fibrosis; the LC and LC-iron groups had significantly greater degrees of fibrosis than the control group (Figure 2(a)). The level of iron accumulation was significantly greater in the LC-iron groups than in the LC group at all measurement points, and the difference was dose-dependent (Figure 2(b)). The degrees of fibrosis were comparable among the LC, LC-iron20, LC-iron40, and LC-iron80 groups on day 28. The degree of hepatic iron accumulation on day 28 increased significantly from the LC group to the LC-iron80 group in a dose-dependent manner (
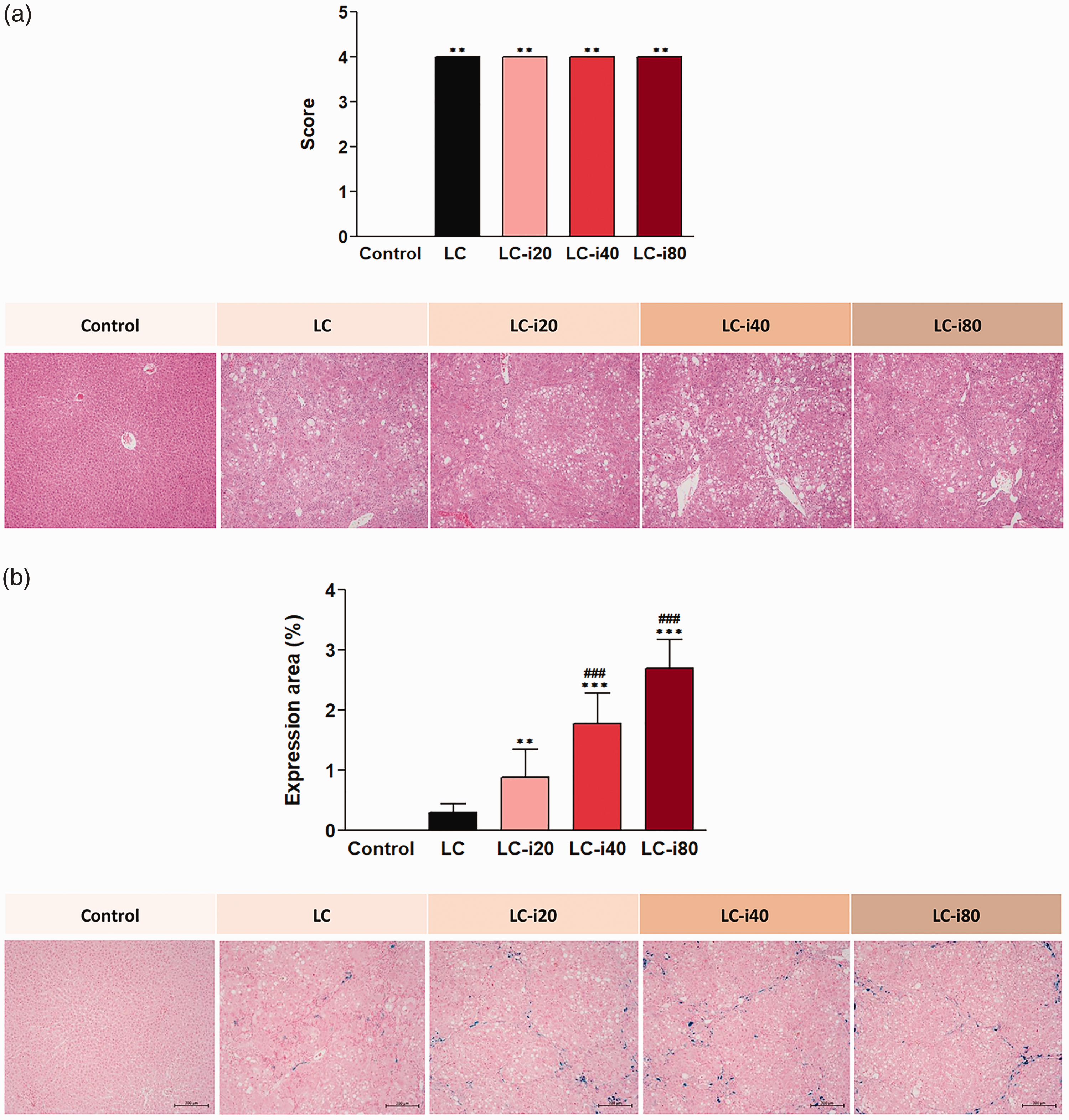
Histopathologic findings 4 weeks after intravenous iron injection. (a) Liver fibrosis (hematoxylin–eosin staining, ×100). (b) Iron deposition (Prussian blue staining, ×100). (a) Liver cell fibrosis was scored from 0 to 4
17
: 0 = no sign of fibrosis or cirrhosis; 1 = enlarged, fibrotic portal tracts; 2 = periportal or portal–portal septa but intact architecture; 3 = fibrosis with architectural distortion but no obvious cirrhosis; and 4 = probable or definite cirrhosis. (b) Histochemical localization in the liver tissue of each rat was calculated using a computer and image analyzer and is expressed as the percentage of the area per millimeter of positive iron deposit staining. Control group: normal, healthy rats without cirrhosis; LC group: rats with induced liver cirrhosis that received CCl4 twice weekly with intravenous saline; LC-iron20 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 20 mg/kg intravenous iron on day 0; LC-iron40 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 40 mg/kg intravenous iron on day 0; LC-iron80 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 80 mg/kg intravenous iron on day 0. ***/**Significant difference at
Portal vein pressure
The portal vein pressure was higher in the LC and LC-iron groups than in the control group (6.7 ± 2.1 mmHg in the control group,
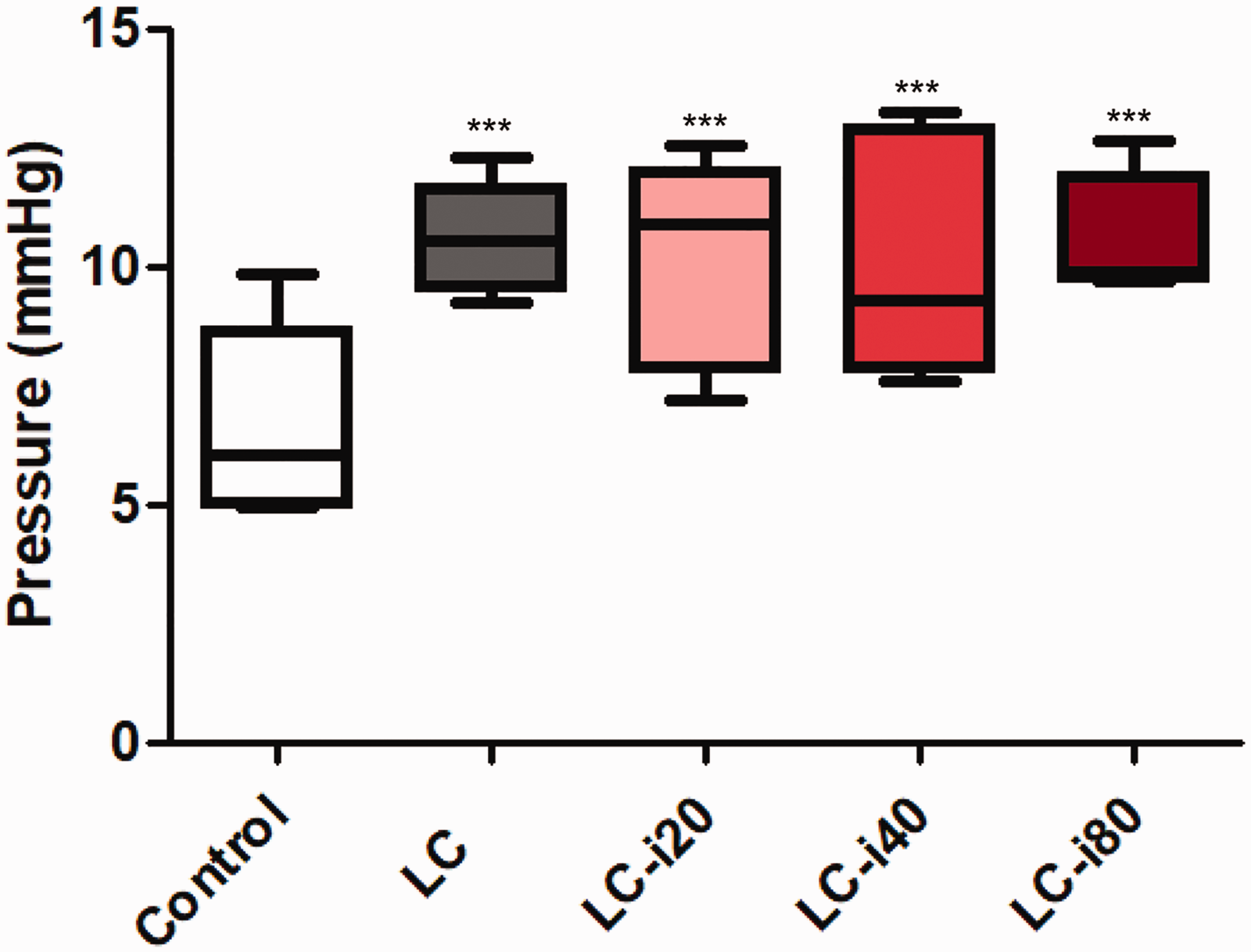
Measurement of portal vein pressure 4 weeks after intravenous iron injection. Data are shown as mean ± standard deviation. Control group: normal, healthy rats without cirrhosis; LC group: rats with induced liver cirrhosis that received CCl4 twice weekly with intravenous saline; LC-iron20 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 20 mg/kg intravenous iron on day 0; LC-iron40 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 40 mg/kg intravenous iron on day 0; LC-iron80 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 80 mg/kg intravenous iron on day 0. ***Significant difference at
Liver function tests
During the test period, the AST and ALT concentrations were significantly higher in the LC and LC-iron groups than in the control group (
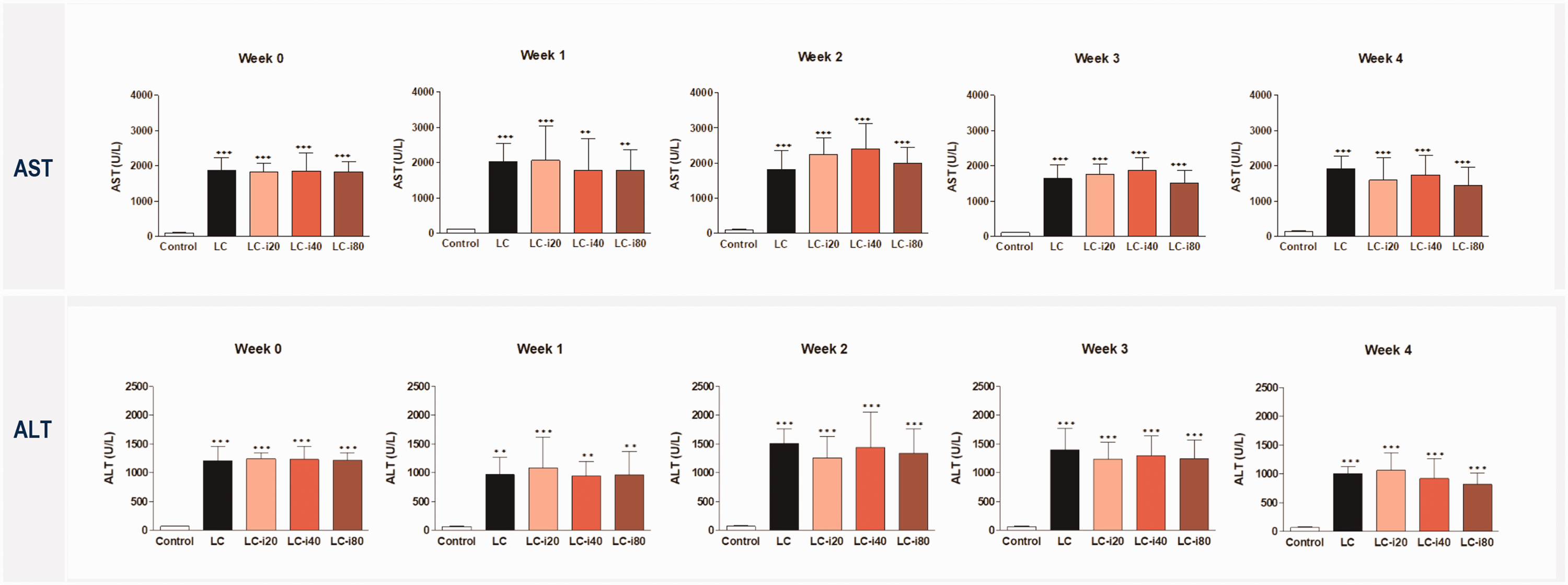
Measurement of liver enzymes. Data are shown as mean ± standard deviation. AST, aspartate transferase; ALT, alanine transferase; LC group: rats with induced liver cirrhosis that received CCl4 twice weekly with intravenous saline; LC-iron20 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 20 mg/kg intravenous iron on day 0; LC-iron40 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 40 mg/kg intravenous iron on day 0; LC-iron80 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 80 mg/kg intravenous iron on day 0. Weeks 1, 2, 3, and 4 reflect times after infusion of iron. ***Significant difference at
Iron deficiency-related parameters
Before IV iron administration, the hemoglobin concentration was comparable among the five groups. The LC and LC-iron groups did not significantly differ in other hematological measures. On day 28 after Ferinject administration, the hemoglobin concentration was significantly higher in the LC-iron40 and LC-iron80 groups than in the LC group (14.5 ± 1.0 g/dL in the control group, 10.9 ± 1.5 g/dL in the LC group, 11.8 ± 1.4 g/dL in the LC-iron20 group, 13.6 ± 1.5 g/dL in the LC-iron40 group, and 13.8 ± 1.0 g/dL in the LC-iron80 group) (Table 1). The LC and LC-iron groups did not significantly differ in ferritin, transferrin saturation, C-reactive protein, hepcidin, iron, or total iron binding capacity.
Anemia-related parameters at week 4.
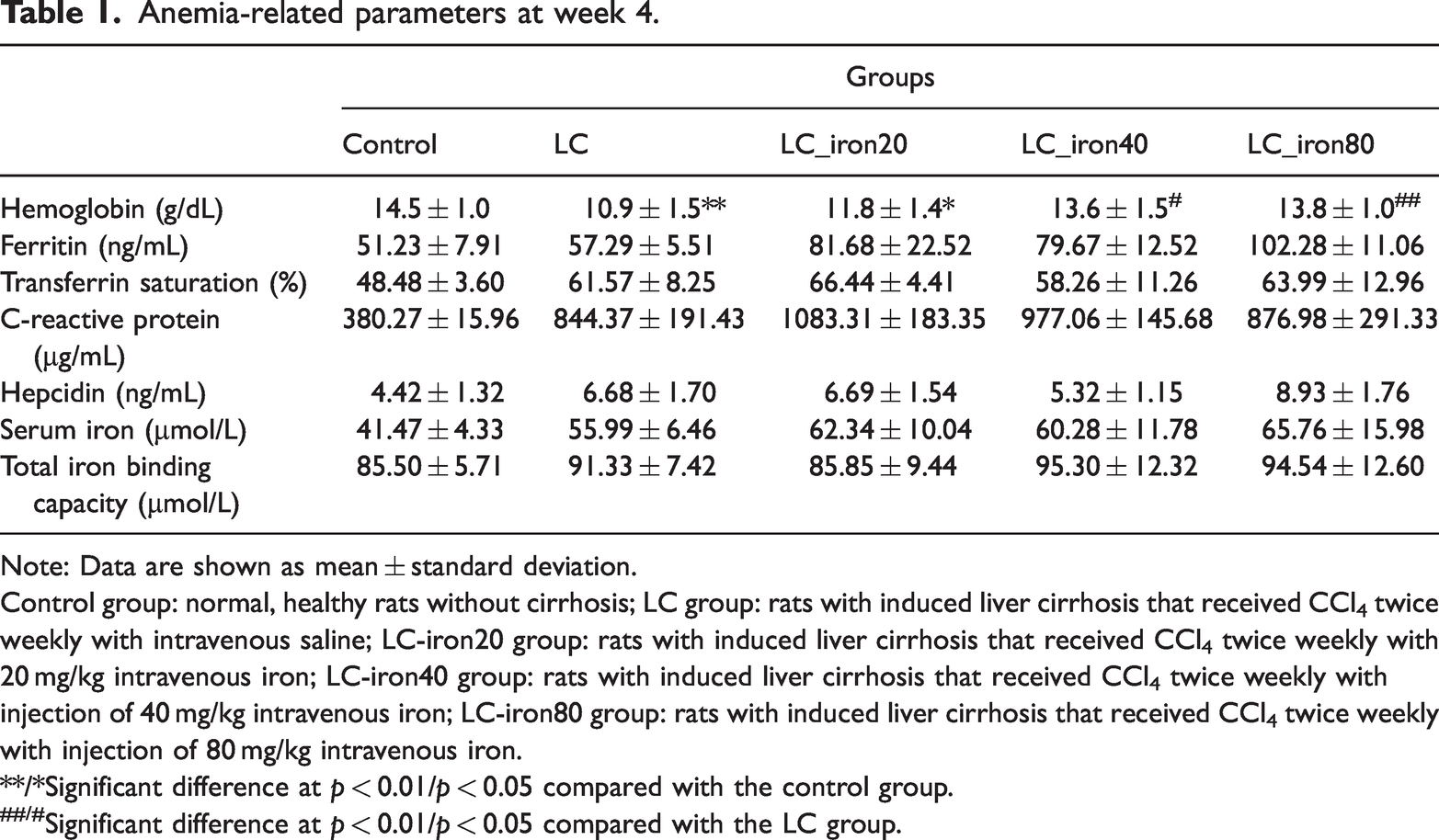
Note: Data are shown as mean ± standard deviation.
Control group: normal, healthy rats without cirrhosis; LC group: rats with induced liver cirrhosis that received CCl4 twice weekly with intravenous saline; LC-iron20 group: rats with induced liver cirrhosis that received CCl4 twice weekly with 20 mg/kg intravenous iron; LC-iron40 group: rats with induced liver cirrhosis that received CCl4 twice weekly with injection of 40 mg/kg intravenous iron; LC-iron80 group: rats with induced liver cirrhosis that received CCl4 twice weekly with injection of 80 mg/kg intravenous iron.
**/*Significant difference at
##/#Significant difference at
Proinflammatory and oxidative stress markers
On day 28, the tumor necrosis factor-α concentration was significantly higher in the LC, LC-iron20, LC-iron40, and LC-iron80 groups than in the control group. The interleukin-6 concentration was also significantly higher in the LC and LC-iron20 group than in the control group (
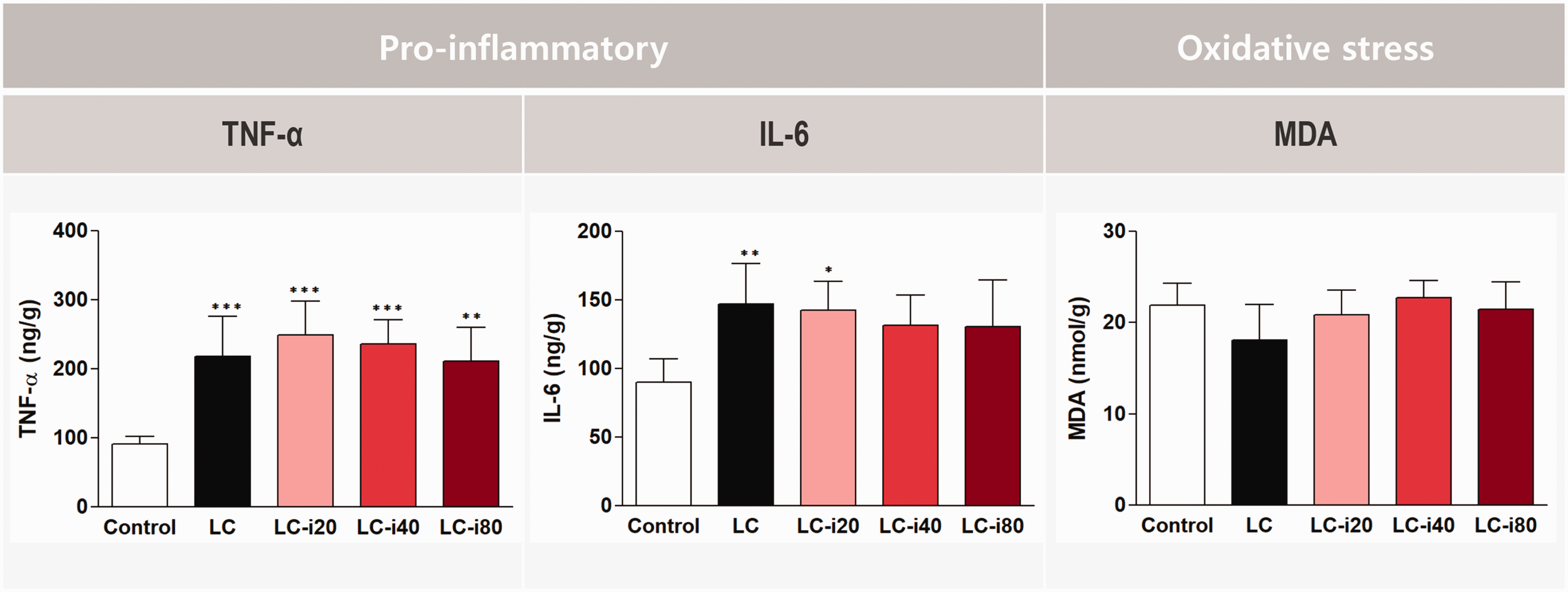
Measurement of proinflammatory and oxidative stress markers 4 weeks after intravenous iron injection. Data are shown as mean ± standard deviation. TNF-α, tumor necrosis factor-α; IL-6, interleukin-6; MDA, malondialdehyde; Control group: normal, healthy rats without cirrhosis; LC group: rats with induced liver cirrhosis that received CCl4 twice weekly with intravenous saline; LC-iron20 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 20 mg/kg intravenous iron on day 0; LC-iron40 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 40 mg/kg intravenous iron on day 0; LC-iron80 group: rats with induced liver cirrhosis that received CCl4 twice weekly after administration of 80 mg/kg intravenous iron on day 0. ***/**/*Significant difference at
Discussion
Interest in iron deficiency without anemia has recently grown, and the indications for iron supplementation are therefore also expanding.7,19,20 However, preoperative iron therapy to correct iron deficiency has not been implemented in patients with end-stage liver disease despite its benefits in other surgical settings. Although our previous animal study showed the potential safety of iron treatment in a rat model of cirrhosis, 10 concerns about supplementary iron in patients with liver cirrhosis remained with respect to the potentially toxic effects of a higher dose. In the present study, contrary to speculation that liver function would deteriorate at higher doses, we found that iron treatment, even at double and quadruple doses, did not induce significant changes in liver function, the degree of fibrosis, or portal vein pressure. However, the liver parenchyma showed substantial iron accumulation after treatment. The degrees of inflammation and oxidative stress in liver tissues did not differ between the LC-iron groups and the LC group (no iron treatment). These results in a rat model of cirrhosis provide preclinical evidence that a high dose of IV iron, exceeding the manufacturer’s recommended dose, might not impair liver function, aggravate the degree of fibrosis, or produce inflammation or oxidative stress.
Because the liver plays a major role in iron metabolism, patients with hepatic disease often have abnormalities in iron homeostasis. As liver disease progresses, the concentration of hepcidin, an iron regulatory hormone, can decrease, which causes iron overload and leads to iron deposition in the liver. Moreover, when the labile, non-transferrin-bound iron level in the bloodstream becomes high, transferrin iron, representing the normal form of circulating iron, is usually low. 21 Accordingly, the paradoxical coexistence of iron excess and iron deficiency is observed in patients with liver disease, hindering iron treatment in this population. Liver fibrosis is a pathological stage of chronic liver disease. 22 Excessive iron accumulation can accelerate the Fenton reaction, generating harmful reactive oxygen species. This process induces the expression of malondialdehyde and both profibrinogenic and proinflammatory mediators such as tumor necrosis factor-α and interleukin-6, leading to liver parenchymal damage and fibrosis. Thus, it is important to determine whether additional hepatic iron deposition after IV iron treatment can exacerbate inflammation and oxidative stress or accelerate the progression of liver fibrosis.
After administration of a supplementary iron injection, the iron distribution and storage pathway becomes implicated in hepatic toxicity. Iron storage within the reticuloendothelial system, particularly in Kupffer cells, signifies appropriate deposition, implying a reduced likelihood of oxidative damage. 23 However, detecting iron in non-reticuloendothelial hepatocytes within the liver, such as in parenchymal and sinusoidal epithelial cells, is considered abnormal and associated with oxidative stress. In this study, iron deposition (visualized using Prussian blue staining) was restricted to reticuloendothelial cells in the LC-iron groups, which implies that the injected IV iron was deposited in the appropriate locations. Previous studies in rat models showed that Ferinject has a favorable safety profile with respect to liver function, oxidative stress, and inflammatory responses, with appropriate iron deposition in reticuloendothelial system cells in the liver.24–26 Our findings suggest that Ferinject is a stable IV iron product that induces a lower level of labile iron. The safety profile of Ferinject might have contributed to our favorable findings in iron-treated rats. 16
The hemoglobin level was significantly higher in the LC-iron40 and LC-iron80 groups than in the LC group on day 28. However, as we found in our previous experiment, the LC-iron20 and LC groups did not differ significantly. This finding implies that higher doses can be more effective in treating anemia, and trends in other iron-related parameters support that possibility. Inflammatory liver diseases often result in a high serum ferritin concentration, but iron circulation and delivery to the bone marrow can be impaired, leading to iron-restricted erythropoiesis.27,28 Iron deficiency, even without anemia, has been identified as an important prognostic marker of postoperative mortality. 29 Recent evidence suggests that absolute and functional iron deficiency are important in predicting outcomes in both anemic and non-anemic patients, further highlighting the significance of iron homeostasis in clinical settings.
In patients with liver disease undergoing hepatectomy or liver transplantation, heavy blood loss is generally expected during and after surgery because of impaired coagulation, a rich portosystemic collateral system, and surgical procedures involving major vessels.30,31 In this population, blood transfusion can cause major organ dysfunction because of a greater circulating blood volume and dilutional coagulation disorders. 32 Accordingly, efforts to treat preoperative anemia focus on reducing the need for red blood cell transfusions. 33 In several case reports, preoperative IV iron administration to Jehovah’s Witnesses with end-stage liver disease enabled liver transplantation surgery without blood transfusion, suggesting that IV iron can be administered safely in that population.20,34–38 The findings of the present study provide preclinical evidence to support the adoption of preoperative iron therapy in patients with end-stage liver disease.
This study has two main limitations. First, because our goal was to evaluate the safety of IV iron in patients with liver cirrhosis, we used a rat model of cirrhosis. We did not assess the presence of anemia in this model. Although the groups of rats that received higher iron doses had better profiles for several iron-deficiency markers, our findings are not sufficient to suggest the efficacy of IV iron in treating anemia. Further studies using an animal model of anemia should be conducted to evaluate efficacy. Second, although we aimed to examine perioperative iron metabolism, our protocol did not include a surgical procedure. Therefore, an appropriate animal experiment involving a surgical procedure is warranted before clinical investigations are considered.
In conclusion, administering high-dose IV iron in a rat model of CCl4-induced cirrhosis did not significantly change liver function test results or the degree of liver fibrosis. Although IV iron administration appeared to be safe in our rat model of cirrhosis, further studies are warranted to confirm the safety and efficacy of IV iron in patients with liver cirrhosis or end-stage liver disease.
Footnotes
Author contributions
(I) Conception and design: J.-H. Kwon, R. Kang, and J.S. Ko; (II) Administrative support: S.M. Lee, T.S. Hahm, H.S. Cho, and G. Jin; (III) Provision of study materials or patients: R. Kang, S.M. Lee, T.S. Hahm, H.S. Cho, and G. Jin; (IV) Collection and assembly of data: All authors; (V) Data analysis and interpretation: J.-H. Kwon, R. Kang, and J.S. Ko; (VI) Manuscript writing: J.-H. Kwon and J.S. Ko; (VII) Final approval of manuscript: All authors.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was supported by a grant from the Korean Health Technology R&D Project through the Korean Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI22C1895); a grant provided by Samsung Medical Center (grant number: SMO1240161); and a grant provided by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science and Information & Communication Technology (grant number: 2019R1F1A1063201).
