Abstract
Background:
Early detection and removal of adenomas through colonoscopy significantly reduces colorectal cancer (CRC) incidence and mortality. However, there is a lack of summarized evidence evaluating the necessity of colonoscopy in patients with upper gastrointestinal (UGI) adenomas.
Objectives:
We aimed to assess the risk of colorectal neoplasms in individuals with UGI adenomas to provide evidence on whether patients with UGI adenomas should undergo colonoscopy examination.
Design:
Systematic review and meta-analysis.
Data sources and methods:
A search of PubMed and Embase was performed up to November 30, 2024. The studies comparing the risk of colorectal neoplasms in patients with and without UGI adenomas were included. Pooled odds ratios (ORs) of colorectal adenomas were estimated using random-effects models. Trial sequential analysis (TSA) was performed to control for random errors.
Results:
A total of 15 studies with 210,508 participants were included. Our analysis revealed a significant association between UGI adenomas and an increased risk of colorectal adenomas (pooled OR 2.36, 95% confidence interval (CI), 1.82–3.06). TSA confirmed the reliability of these findings. In addition, the presence of duodenal adenomas was linked to an increased risk of advanced colorectal adenomas (pooled OR 2.95, 95% CI, 1.80–4.84) and CRC (pooled OR 2.15, 95% CI, 1.51–3.06). Sensitivity analyses supported the robustness of these associations. Subgroup analysis suggested that patients with either gastric or duodenal adenomas had a higher risk of colorectal adenomas.
Conclusion:
This is the first meta-analysis suggesting that patients with UGI adenomas are at increased risk of colorectal adenomas and CRC. These findings highlight the need for colonoscopy in this patient population and supplement colonoscopy screening guidelines.
Plain language summary
Introduction
Colorectal cancer (CRC) was ranked as the third most common cancer worldwide in 2022. 1 The adenoma–carcinoma pathways originating from conventional adenomas are the major pathways of colorectal carcinogenesis in sporadic CRC. 2 Early detection and removal of colorectal adenomas and other precancerous lesions through colonoscopy can effectively reduce the incidence and mortality rates of CRC.3,4 Therefore, for patients at high risk of colorectal adenomas, colonoscopy screening is recommended, such as those over 50 years old, with inflammatory bowel disease, or with a family history of CRC.3,5
Some studies have shown that upper gastrointestinal (UGI) adenomas are associated with an increased risk of colorectal adenomas, but the conclusions across different studies have been inconsistent. A previous study involving 787 patients demonstrated that patients with gastric adenomas have an increased risk of colorectal adenomas compared to healthy controls without gastric polyps (odds ratio (OR) 4.03, 95% confidence interval (CI), 1.21–13.40). 6 Similarly, research by Genta et al. 7 found that duodenal adenomas are associated with an increased risk of colonic adenomas (OR 2.65, 95% CI, 2.16–3.25). However, other studies have not found a significant link between UGI adenomas and colorectal adenoma risk.8,9 These discrepancies across studies, coupled with the fact that most investigations on this topic have been limited by small sample sizes, highlight the lack of pooled evidence to definitively determine whether UGI adenomas are associated with an increased risk of colorectal adenomas.
Therefore, given that UGI adenomas share common risk factors with colorectal adenomas, such as smoking, alcohol consumption, and other adverse lifestyle factors,10–13 we conducted this meta-analysis to summarize the risk of concurrent or subsequent colorectal adenomas and CRC in patients with UGI adenomas, and aimed to evaluate the need for colonoscopy for patients in whom UGI adenomas have been identified. Trial sequential analysis (TSA) was employed to determine the required sample size and confirm the reliability of the conclusions, 14 providing evidence-based guidance on the indications for colonoscopy screening in patients with UGI adenomas.
Methods
The study adhered to the Meta-Analysis of Observational Studies in Epidemiology (MOOSE; Supplemental Table 1) 15 and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA; Supplemental Table 2) 16 guidelines and was registered in advance on PROSPERO (CRD42024623610).
Literature search
A comprehensive search of the electronic databases PubMed and EMBASE was conducted up to November 30, 2024, to identify pertinent literature. The search strategy employed a combination of Medical Subject Headings and relevant text words pertaining to UGI adenoma and colorectal neoplasms. The search terms included concepts such as duodenal adenoma, gastric adenoma, colorectal tumor, and prevalence. There were no restrictions regarding study design, publication date, or language. Furthermore, a manual examination of the reference lists of potentially relevant articles was undertaken. The detailed search strategy is provided in Supplemental Table 3.
Selection criteria
Two independent reviewers (H.X. and K.S.) carefully screened study titles and abstracts based on predefined inclusion criteria. They thoroughly assessed full-text articles for eligibility, resolving any disagreements through discussion or with a third author (Y.Z.) if needed. For studies with multiple reports, only the most recent and comprehensive one with the largest sample size was included.
We incorporated studies that satisfied the following criteria: (1) participants aged over 18 years; (2) investigations assessing the prevalence of colorectal neoplasms in patients with adenomas in the UGI tract, compared to control groups; (3) diagnoses confirmed through pathological findings; (4) colonoscopy performed concurrently with, after, or within 1 year prior to gastroscopy; (5) participants with no history of hereditary nonpolyposis colorectal cancer or familial adenomatous polyposis (FAP), and (6) providing ORs or relative risks (RRs) with corresponding 95% CIs as measures of association, or that allowed for the computation of these measures based on count data reported within the article. Studies were excluded if they (1) lacked control groups; (2) were narrative or systematic reviews, case reports, commentaries, editorials, or conference abstracts; (3) did not provide data for patients with UGI adenomas; (4) investigated the impact of intestinal lesions on the risk of UGI lesions; or (5) involved patients who underwent colonoscopy more than 1 year prior to UGI endoscopy.
Data extraction
Data extraction for each study was independently conducted by two reviewers (W.C. and C.L.). The extracted data included the following: (1) study characteristics (first author’s name, publication year, country, study type); (2) total participants; (3) participants’ age and sex distribution; (4) lesion characteristics (location and pathological types) in UGI and colorectal neoplasms; (5) association measures (ORs or RRs; since all the included studies were case–control studies and used ORs to measure risk, we only extracted ORs) with 95% CIs; and (6) confounding factor adjustments, if applicable.
Statistical analysis
The analyses were performed utilizing R software version 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria). The primary outcome measure was the pooled OR of colorectal adenomas in individuals with UGI adenomas compared to those without, based on count data, since the articles reporting the adjusted OR values had substantial heterogeneity in their primary research endpoints. Secondary outcome measures included the pooled ORs for colorectal advanced adenoma and CRC. A meta-analysis was conducted for the exposure variable (UGI adenomas) and each outcome variable (colorectal neoplasms) using random-effects models.
Grading of evidence followed the Grading of Recommendations Assessment, Development and Evaluation (GRADE) recommendations using the GRADEpro software (McMaster University and Evidence Prime, 2024). 17 Appraisal was limited to primary outcomes.
The heterogeneity among the studies was evaluated using Cochran’s Q test and I2 value.18,19 For the Cochran Q test, p-value <0.05 represented significant heterogeneity. For the I2 value, it reflected the amount of true heterogeneity between studies, with values of 25% or lower corresponding to low degrees of heterogeneity, 26%–50% to moderate degrees of heterogeneity, and greater than 50% to high degrees of heterogeneity. To identify the specific study contributing to the heterogeneity in the primary outcome, a Galbraith plot was utilized. In this plot, each study is depicted as a single point, with a central regression line traversing the plot. The quality of the studies was assessed using the Newcastle–Ottawa Scale (NOS) in three criteria: patient selection, comparability of control groups, and outcome ascertainment. 20 To evaluate publication bias, funnel plots and Egger’s regression test were employed. 21
Sensitivity analysis and subgroup analysis
Sensitivity analysis included (1) excluding studies that contributed significantly to heterogeneity, (2) conducting a meta-analysis on articles reporting adjusted OR values, (3) summarizing results only for multicenter studies, and (4) performing a meta-analysis after excluding studies with sample sizes much larger than the others. Subgroup analysis included examining (1) the risk of colorectal adenomas associated with UGI adenomas in different anatomical locations, (2) the risk of colorectal serrated lesions associated with UGI adenomas, and (3) the risk of colorectal adenomas in different anatomical locations associated with UGI adenomas.
Trial sequential analysis
We used TSA to control for the risk of random errors caused by small sample size and repeated significance testing of the included studies, adjusting the thresholds for statistical significance in the meta-analysis. 14 The analysis was performed using TSA software version 0.9 beta (Copenhagen Trial Unit, Centre for Clinical Intervention Research, Copenhagen, Denmark). We set the model to a random-effects model (DerSimonian–Laird) to calculate the effect measures. Based on previous studies in this field and considering the reported ORs and 95% CIs, we adopted a conservative estimate of a 20% increased risk of colorectal adenomas associated with UGI adenomas.22–24 This conservative effect size was used in the TSA to provide a more stringent estimate of the required information size. If the true risk increase is greater than the assumed value, the TSA results are expected to remain valid, potentially offering even stronger statistical power. The analysis was conducted with a power (1−β) of 80% and a type I error (α) of 5%. The software automatically generated heterogeneity correction based on model variance. The statistical significance of the meta-analysis was assessed based on the position of the cumulative Z curve relative to the conventional boundary, trial sequential monitoring boundary (TSMB), and futility boundary. A stable and definitive conclusion was drawn if the cumulative Z curve crossed the TSMB or entered the futility area below the futility boundary, whereas more studies are required to confirm the results if the sample size is not reached or if the cumulative Z curve does not cross the relevant boundary and area mentioned above. 25
Results
Study selection
The initially systematic literature search yielded 1451 results. After removing duplicates and excluding records based on title and abstract screening, 56 full-text articles were reviewed. Full-text screening identified 15 eligible publications, including 210,508 participants, and all of which were case–control studies.6–9,22–24,26–33 The study selection process is shown in Figure 1. The studies that were excluded, along with the reasons for their exclusion, are presented in Supplemental Table 4. In studies examining the age distribution of patients, the majority reported a mean age exceeding 50 years, aligning with the current guidelines for recommended colonoscopy screening. In terms of the classification of UGI adenomas, nine studies focus specifically on gastric adenomas,6,8,9,22,24,27,29,30,32 while the remaining studies concentrate on duodenal adenomas. Concerning the geographic distribution of the studies, seven were conducted in South Korea,8,9,23,27,29,30,32 three originated from China,6,22,24 three were based in Europe (The Netherlands, France, and Germany),26,31,33 one was conducted in Australia, 28 and one was from the United States. 7 The characteristics of these studies are detailed in Table 1. The quality of the studies, as assessed using the NOS, is shown in Supplemental Table 5. Nine of 15 articles were of good quality.

Flow diagram on search and article inclusion.
The characteristics of the included studies.

AA, advanced adenoma; CI, confidence interval; H. pylori, Helicobacter pylori; NA, not available; OR, odds ratio; SD, standard deviation.
Risk of colorectal adenomas in patients with UGI adenomas
A total of 13 articles reported the association between UGI adenomas and colorectal adenomas, encompassing a total of 208,337 individuals.6–9,22,24,26–32 Our analysis indicates a significant association between the presence of UGI adenomas and an increased risk of developing colorectal adenomas (pooled random-effects OR 2.36, 95% CI, 1.82–3.06; I2 = 60%, p-value of Cochran’s Q test <0.01; Figure 2(a)). The summarized findings demonstrate a substantial level of heterogeneity.

Forest plot of results for the risk of colorectal adenomas in patients with upper gastrointestinal (UGI) adenomas. (a) Forest plot of results for the risk of colorectal adenomas in patients with UGI adenomas. (b) Forest plot of results for the risk of colorectal adenomas in patients with UGI adenomas after excluding studies contributing to high heterogeneity. (c) Forest plot of results for the risk of colorectal adenomas in patients with gastric adenomas. (d) Forest plot of results for the risk of colorectal adenomas in patients with duodenal adenomas.
TSA revealed that the required information size was 7336. The cumulative Z curve has reached the required information size and has crossed the TSMB and conventional boundary, suggesting the positive result was reliable (Figure 2(b)).
Risk of advanced colorectal adenoma and CRC in patients with UGI adenomas
Nine articles reported the association between UGI adenomas and the risk of advanced colorectal adenomas.6,7,9,23,28–31,33 The results similarly show that UGI adenomas significantly increase the risk of advanced colorectal adenomas (pooled random-effects OR 2.95, 95% CI, 1.80–4.84; I2 = 62%, p-value of Cochran’s Q test <0.01; Figure 3(a)).
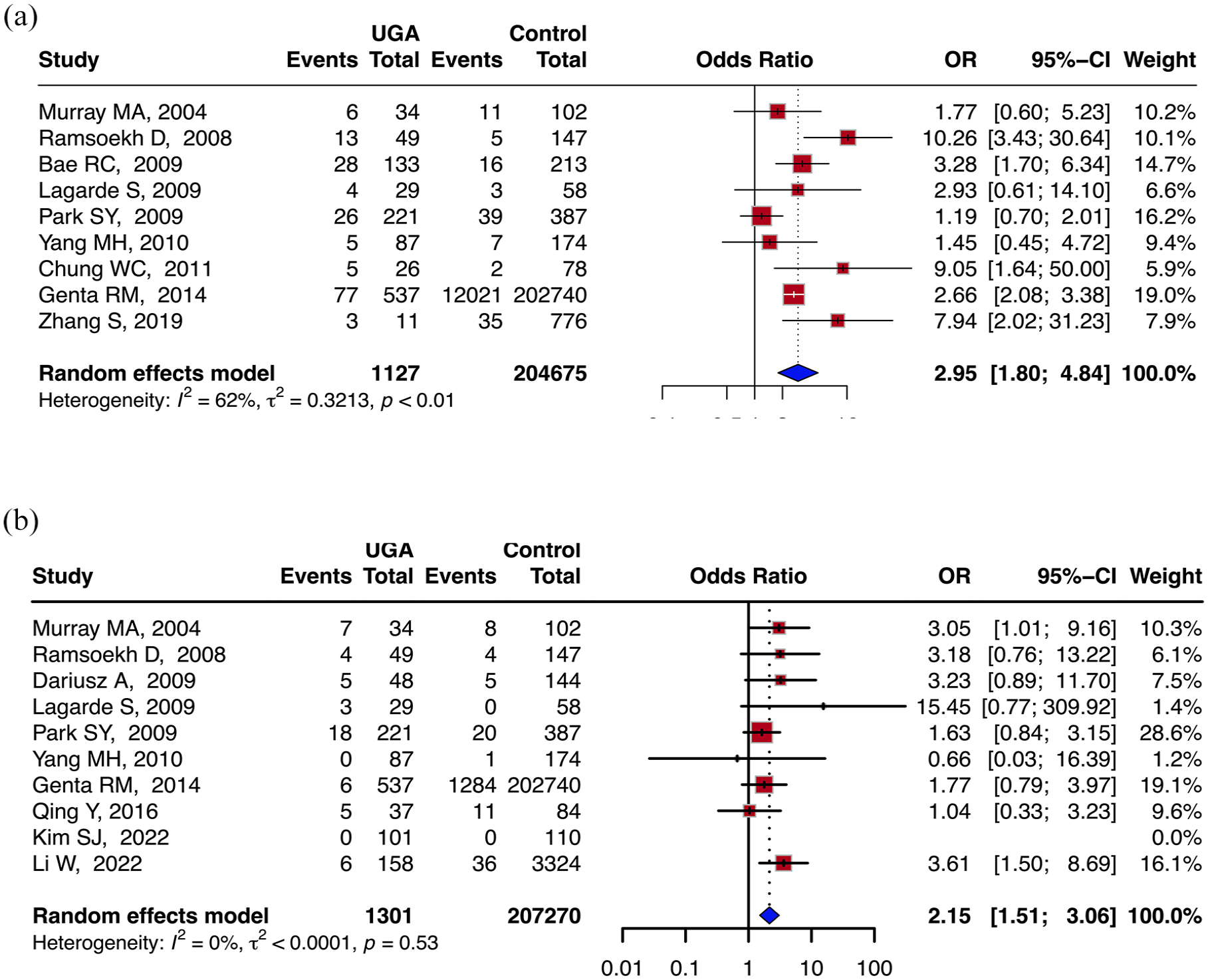
Forest plot of results for the risk of colorectal advanced adenomas and colorectal cancer (CRC) in patients with upper gastrointestinal (UGI) adenomas. (a) Forest plot of results for the risk of colorectal advanced adenomas in patients with UGI adenomas. (b) Forest plot of results for the risk of CRC in patients with UGI adenomas.
Ten articles reported the association between UGI adenomas and the risk of CRC.7,22,24,26–31,33 UGI adenomas notably increased CRC risk (pooled OR 2.15, 95% CI, 1.51–3.06; I2 = 0%, p-value of Cochran’s Q test = 0.53; Figure 3(b)).
Sensitivity analysis
Considering the moderate heterogeneity in the pooled results of the primary endpoint, heterogeneity testing revealed that the studies conducted by Bae et al. 9 and Dariusz and Jochen 26 significantly contributed to the variability (Supplemental Figure 1). After the exclusion of these studies, the pooled analysis continued to indicate a significant association between UGI adenomas and an elevated risk of colorectal adenomas (pooled OR 2.36, 95% CI, 1.82–3.06; I2 = 0%, p-value of Cochran’s Q test = 0.74; Supplemental Figure 2(A)). In addition, we conducted a meta-analysis for the adjusted OR values from five articles reporting on the primary endpoint of colorectal adenomas.6,8,9,29,30 The pooled results similarly support that UGI adenomas increase the risk of colorectal adenomas (pooled OR 1.45, 95% CI, 1.08–1.95; I2 = 53%, p-value of Cochran’s Q test = 0.08; Supplemental Figure 2(B)). When confined to multi-center studies,6,7,23,31 the findings indicated that UGI adenomas are associated with an elevated risk of colorectal adenomas (Supplemental Figure 2(C)). Furthermore, after excluding the study by Genta et al., which had a sample size much larger than the other studies, the conclusion remained unchanged (Supplemental Figure 2(D)).
Subgroup analysis
Due to limited data provided by the included studies here, it is hard to conduct a pooled analysis on variables such as patient age, Helicobacter pylori (H. pylori) infection status, the anatomical location of UGI adenomas, and the site or size of colorectal tumors.
In the subgroup analysis of gastric or duodenal adenomas, the results similarly demonstrated an increased risk of colorectal adenomas (pooled OR 2.03, 95% CI, 1.44–2.86; I2 = 65%, p-value of Cochran’s Q test <0.01; pooled OR 2.87, 95% CI, 2.01–4.09; I2 = 44%, p-value of Cochran’s Q test = 0.11; Supplemental Figure 3(A) and (B)). We further conducted a test for subgroup differences using a random-effects model. The result showed no statistically significant heterogeneity between subgroups (Q = 1.88, p = 0.17), indicating that the increased risk of colorectal adenoma associated with UGI adenomas at different anatomical sites was consistent. Regarding another significant precancerous lesion in CRC, namely, serrated lesions, only a single study provided relevant results. In a case–control study of 537 patients with duodenal adenomas and 202,740 controls, Genta et al. 7 found 12 cases of concurrent serrated adenomas (2.2%) in the duodenal adenoma group, compared to 5326 cases (2.6%) in the control group, with no statistically significant difference observed (OR 1.51, 95% CI, 0.84–2.73).
Regarding the location of colorectal adenomas, Yang et al. 30 analyzed the distribution of colorectal adenomas in patients with/without concomitant gastric adenomas. They found that in patients with gastric adenomas, colorectal adenomas were more commonly located in the distal colon (14/42 vs 10/58) or simultaneously present in both sides of the colon (19/42 vs 13/58). By contrast, for patients without gastric adenomas, colorectal adenomas were more often found in the proximal colon (35/58 vs 9/42). Meanwhile, Park et al. 29 did not observe any significant difference in the distribution of colorectal adenomas despite the presence of gastric adenomas. Genta et al. 7 analyzed all colorectal polyps and found that duodenal adenomas were associated with an increased risk of polyps in both the left and right sides of the colon (Left colon: OR 2.16, 95% CI, 1.74–2.67; Right colon: OR 1.88, 95% CI, 1.54–2.30). As for the size of colorectal adenomas, duodenal adenomas were associated with an increased risk of colorectal adenomas sized 0–19 mm (0–9 mm: OR 2.09, 95% CI, 1.66–2.63; 10–19 mm: OR 3.07, 95% CI, 1.76–5.33). No increased risk was observed for colorectal adenomas larger than 20 mm, possibly due to the limited number of positive events in this category. 7
Assessment of the certainty of evidence and publication bias
All pooled results of primary outcomes were rated as low-certainty evidence (Table 2). Supplemental Figure 4 presents a funnel plot that illustrates the potential for publication bias. The distribution of studies around the pooled effect size, both within and beyond the funnel’s contours, indicates a diminished likelihood of substantial bias in smaller studies, which are more prone to producing nonsignificant results and, consequently, may be overlooked. This observation is corroborated by Egger’s test (p = 0.567).
Summary of primary findings table and certainty of evidence.

CI, confidence interval; OR, odds ratio; UGI, upper gastrointestinal.
Discussion
To the best of our knowledge, this is the first systematic review and meta-analysis to investigate the association between UGI adenomas and the risk of colorectal adenomas and CRC. Our findings demonstrate that UGI adenomas significantly increase the risk of colorectal adenomas, advanced adenomas, and CRC. By conducting subgroup analyses, we found that the increased risk of colorectal adenomas was consistent across different anatomical sites of UGI. Given that we included only studies in which colonoscopy examination was conducted within 1 year before or after a UGI endoscopy, our results reflect the risk of concomitant or subsequent colorectal adenomas and CRC in patients with UGI adenomas. These findings suggest that individuals with UGI adenomas should be considered for colonoscopy examination.
Genetic factors, lifestyle factors, and other common risk factors may contribute to the association between UGI adenomas and colorectal neoplasms.12,34–36 The shared pathogenic role of upper and lower gastrointestinal tumors is increasingly recognized. A study by Li et al. 35 indicated that patients with BRCA1 pathogenic variants had an elevated risk of both gastric cancer (RR = 2.17; 95% CI, 1.25–3.77) and CRC (RR = 1.48; 95% CI, 1.01–2.16). Although the association between BRCA1 pathogenic variants and CRC risk disappeared in gender-stratified analyses, the results still suggest a potential genetic factor in the development of both upper and lower gastrointestinal tumors. A study by Sasaki et al. 37 on Japanese patients with FAP, an autosomal dominant condition caused by pathogenic variants in the APC gene, demonstrated that the cumulative incidence rates of gastric adenoma and gastric cancer in 50-year-old patients with FAP were 22.8% and 7.6%, respectively. Moreover, regarding the incidence of gastric cancer, the hazard rates increased moderately up to the age of 40 years, but the increase accelerated from the age of 50 years. Similarly, a meta-analysis by Singh et al. 38 elucidated that for patients with juvenile polyposis syndrome (autosomal dominant, hereditary hamartomatous GI cancer predisposition syndrome) which is prone to progressing to colorectal polyps, the risk of gastric cancer was higher in SMAD4 pathogenic germline variant carriers (OR = 11.6; 95% CI, 4.6–29.4; I2 = 18.3%) compared with patients whom without the variant.
Helicobacter pylori infection, a risk factor for gastric tumors, has also been shown to increase the risk of colorectal carcinoma and colorectal adenomatous polyps. Furthermore, after treatment for H. pylori, the risk of CRC is correspondingly reduced.39–42 The immune microenvironment of colorectal tissue induced by H. pylori, characterized by a reduction in regulatory T cells and pro-inflammatory T cells, along with the activation of pro-carcinogenic STAT3 signaling in the colonic epithelium and a loss of goblet cells, may be one of the mechanisms promoting colorectal tumorigenesis.40,43 In addition, shared risk factors for both UGI tumors and colorectal tumors include obesity,11,36,44 cigarette smoking,10,11 and alcohol consumption.11,12 Meanwhile, certain disease states can simultaneously increase the risk of both upper and lower gastrointestinal tumors. A study by Mantovani et al. 45 found that non-alcoholic fatty liver disease was significantly associated with a nearly 1.5-fold to 2-fold increased risk of developing gastrointestinal cancers (including stomach, colorectal, esophageal, and pancreatic cancers). This association remained significant after adjusting for confounding factors such as age and gender.
An important consideration for clinical translation of our findings is the cost–benefit and risk assessment associated with recommending colonoscopy following the diagnosis of UGI adenomas. While our pooled analysis suggests a higher prevalence of colorectal adenomas and advanced neoplasia among patients with gastric or duodenal adenomas, it remains important to evaluate the clinical implications in terms of health economics. 46 Specifically, the Number Needed to Treat to identify one additional colorectal adenoma or advanced lesion, and the potential Number Needed to Harm due to colonoscopy-related complications, should be estimated. 47 These metrics can help contextualize whether the diagnostic yield justifies the additional burden on patients and healthcare systems. Future studies should prospectively assess these outcomes to guide evidence-based recommendations and improve screening guidelines.
Although our study holds clinical significance for guiding endoscopy, it still has certain limitations. First, we did not conduct further subgroup analyses, so we are unable to assess the impact of factors such as H. pylori infection status, patient age, and gender on the risk of colorectal tumors. Second, our study was cross-sectional, which means it cannot reflect the long-term risk of colorectal tumors following UGI adenomas. For patients with UGI adenomas but negative findings on colorectal endoscopy, the long-term risk of colorectal tumors remains a subject for further investigation. In addition, some studies explicitly included serrated adenomas as part of colorectal adenomas,7,22,27 making it difficult to exclude them during data extraction in two studies.22,27 As a result, the pooled results might be slightly influenced.
Conclusion
We have summarized the current research evidence and clarified that patients with UGI adenomas are at increased risk of developing colorectal adenomas and CRC and should be considered for thorough colonoscopy screening. Our findings provide an important complement to the guidelines for colorectal tumor screening, which could help reduce the incidence and mortality of CRC.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251351522 – Supplemental material for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848251351522 for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis by Hanyuan Xu, Kai Song, Yuelun Zhang, Chengzhen Lyu, Kun He, Wangyang Chen, Wei Chen and Dong Wu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-2-tag-10.1177_17562848251351522 – Supplemental material for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis
Supplemental material, sj-jpg-2-tag-10.1177_17562848251351522 for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis by Hanyuan Xu, Kai Song, Yuelun Zhang, Chengzhen Lyu, Kun He, Wangyang Chen, Wei Chen and Dong Wu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-3-tag-10.1177_17562848251351522 – Supplemental material for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis
Supplemental material, sj-jpg-3-tag-10.1177_17562848251351522 for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis by Hanyuan Xu, Kai Song, Yuelun Zhang, Chengzhen Lyu, Kun He, Wangyang Chen, Wei Chen and Dong Wu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-4-tag-10.1177_17562848251351522 – Supplemental material for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis
Supplemental material, sj-jpg-4-tag-10.1177_17562848251351522 for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis by Hanyuan Xu, Kai Song, Yuelun Zhang, Chengzhen Lyu, Kun He, Wangyang Chen, Wei Chen and Dong Wu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-5-tag-10.1177_17562848251351522 – Supplemental material for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis
Supplemental material, sj-jpg-5-tag-10.1177_17562848251351522 for Follow your “gut”—calling for colonoscopy in patients with upper gastrointestinal adenomas: a systematic review and meta-analysis by Hanyuan Xu, Kai Song, Yuelun Zhang, Chengzhen Lyu, Kun He, Wangyang Chen, Wei Chen and Dong Wu in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
