Abstract
Background:
The harmonic scalpel (HS) and cavitron ultrasonic surgical aspirator (CUSA) are two common techniques for liver parenchymal transection (LPT). The second International Consensus Conference on laparoscopic liver resection (LLR) recommended the utilization of the HS for superficial layer LPT and the CUSA for deep layer LPT. Some centers currently employ the HS for deep-layer LPT. However, the potential effect of HS during deep layer transection in LLR remains unclear.
Objectives:
The study aims to investigate the feasibility, safety, and efficiency of HS in LLR compared with CUSA.
Design:
This is a study protocol for a randomized controlled trial (RCT).
Methods and analysis:
This RCT will be carried out at West China Hospital of Sichuan University from January 2024 to December 2025. Consecutive patients who underwent LLRs during this period will be recruited based on the specified inclusion and exclusion criteria. The participants will be randomly allocated to the HS group or the CUSA group. The primary outcome is intraoperative blood loss. Secondary outcomes include parenchymal transection velocity, operative time, conversion rate, postoperative hospital stays, hospital cost, morbidity, and mortality. Subgroup analysis will be performed according to liver cirrhosis. The corresponding statistical approach will be used for statistical analysis.
Ethics:
The trial has been reviewed and approved by the Biomedical Ethics Review Committee of West China Hospital, Sichuan University, on January 19, 2024.
Discussion:
This study will clarify the feasibility, safety, and efficiency of HS for LPT in LLRs. The results will provide more reliable clinical evidence for the selection of LPT devices in LLRs.
Trial registration:
The study protocol was registered in the Chinese Clinical Trial Registry (http://www.chictr.org.cn) on April 26, 2024, ChiCTR2400083493. The protocol version is V2.0.
Keywords
Background
The liver features with abundant and intricate vascular network, which poses a significant risk of intraoperative bleeding. 1 Intraoperative blood loss (IBL) during liver resection ranged from 200 ml to 2000 ml per patient. 2 The IBL was identified as the main factor affecting the prognosis of patients who underwent liver resection. 3 According to previous studies, excessive IBL was associated with an increased risk of postoperative complications, higher mortality, and elevated healthcare costs.4,5 In addition, significant blood loss served as a negative prognostic factor for overall survival and disease-free survival in patients with liver malignancies. 6 Although laparoscopic liver resection (LLR) has been demonstrated to be advantageous in reducing IBL over open liver resection,7 –9 the control of intraoperative bleeding is still the main focus in LLR.
During LLR, the main source of bleeding arises during liver parenchymal transection (LPT). Currently, a range of advanced instruments are available to enhance the safety and efficacy of LPT, including cavitron ultrasonic surgical aspirator (CUSA), harmonic scalpel (HS), LigaSure, water jet and sealing device, etc. 10 According to the second International Consensus Conference on LLR, 11 superficial parenchymal dissection could be safely performed using HS, while CUSA, water jet, and LigaSure were recommended for deeper parenchymal dissection (>2 cm). However, the selection of instruments for LLR typically relies on experience and performance of surgeons in most centers.10,11
A previous study 12 showed that the CUSA was the most commonly used surgical instrument for LPT in LLR. Nevertheless, CUSA may be unavailable due to high prices in underdeveloped areas. In recent years, HS technology has been increasingly used during the LPT phase of LLR, recognized for its effectiveness in sealing smaller vessels. Previous studies13 –15 have indicated that the use of HS was associated with reduced operative time, less blood loss, decreased costs, and shorter hospital stays compared to CUSA. Furthermore, perioperative safety was comparable between the two devices with similar rates of postoperative morbidity and mortality.13 –15
However, due to the retrospective and non-randomized design of the above studies, there is currently a lack of high-quality medical evidence to support the utilization of HS for LPT. Herein, we conduct this randomized controlled trial (RCT) to investigate the feasibility, safety, and efficiency of HS in LLR by comparing it with CUSA.
Methods and analysis
Trial design
This is a single-center, parallel, single-blinded, RCT. This study aims to investigate the feasibility, safety, and efficiency of HS in LLR compared with CUSA. This study will be conducted in accordance with the principles of the Declaration of Helsinki. The reporting of this study conforms to the SPIRIT statement (Supplemental Material). 16 The protocol is written based on “Standard Protocol Items: Recommendations for Interventional Trials” checklist. 17
Study setting
This study will be conducted at West China Hospital of Sichuan University, China. The Liver Surgery Department of China West Hospital performs more than 2000 cases of liver resection per year. The main investigators in the trial have overcome the learning curve with over 10-year experience in LLR.
Recruitment
A dedicated researcher is responsible for patient recruitment. After preoperative assessment is finished, patients will be screened for eligibility according to the inclusion and exclusion criteria by the researcher. Then the eligible patients will receive a comprehensive introduction to the surgical protocol, including surgical-related benefits and risks, to both the patients and their families. They will be included or excluded from this study according to their decisions. All participants will sign the written informed consent for entering the trial and for authorizing the perioperative data for recording and publishing. Table 1 and Figure 1 present the study procedure and the participant timeline.
Participant timeline.

CUSA, cavitron ultrasonic surgical aspirator; HS, harmonic scalpel; IBL, intraoperative blood loss.
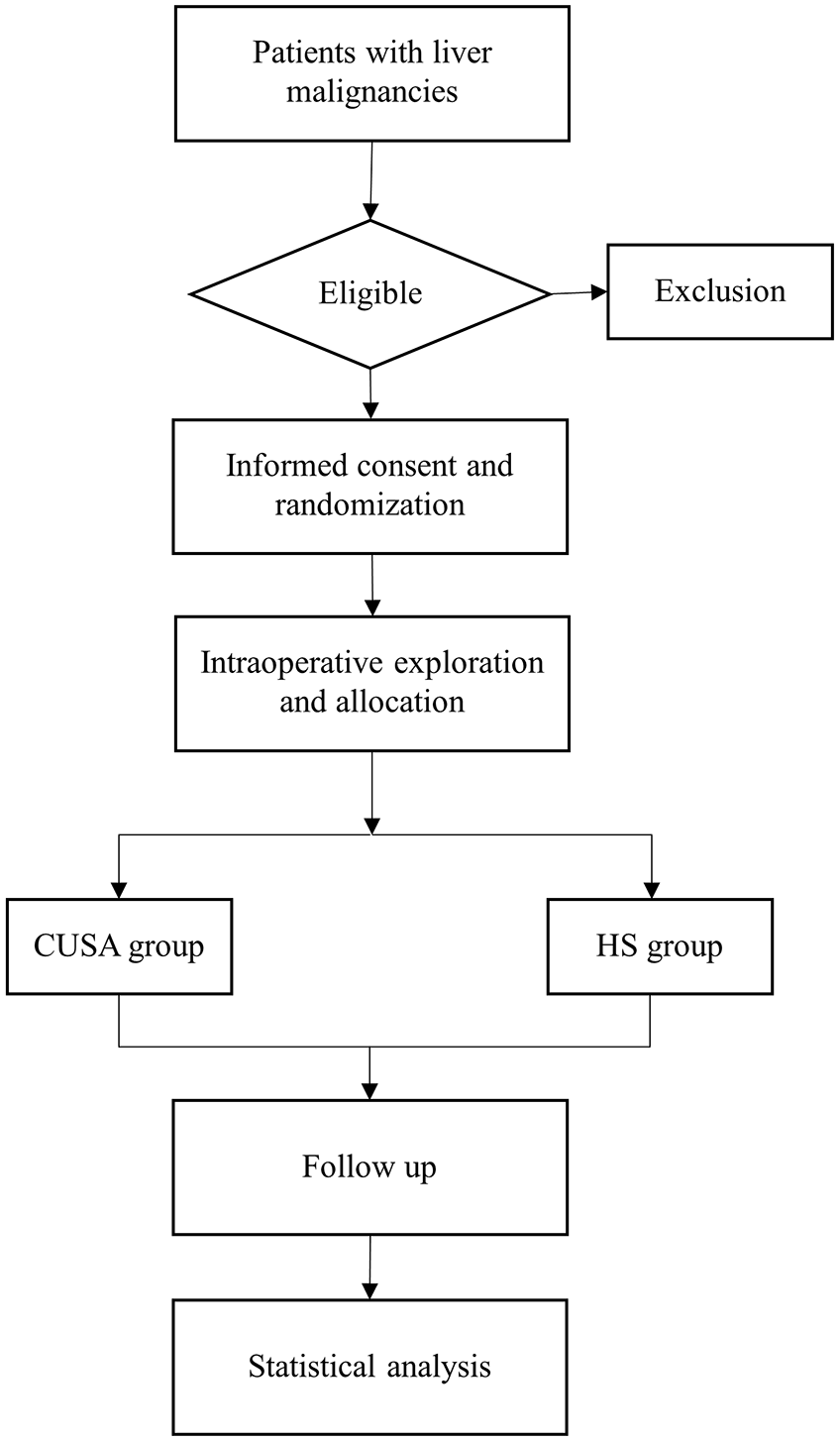
Flowchart illustrating the randomization and flow of study participants.
Inclusion criteria
Patients aged from 18 to 80 years;
No contraindication to general anesthesia and LLR;
Liver function is evaluated as Child-Pugh class A;
Laparoscopic anatomical liver resection;
The surgical indications are liver malignancies, including hepatocellular carcinoma, colorectal liver metastasis, intrahepatic cholangiocarcinoma, etc.;
Understanding and readiness to provide written informed consent.
Exclusion criteria
Combined with resection or radiofrequency ablation of another liver segment;
Multi-visceral resection planned except cholecystectomy;
Reconstructive surgery after LPT (including bile ducts, portal veins, arteries);
Previous history of major upper abdominal surgery.
Intervention description
In the HS group, HS (Ethicon Endo-Surgery, OH, USA, Harmonic ACE®+7) will be used for LPT throughout the hepatectomy by the method that we described in a previous study. 15 In the CUSA group, HS will be only used for dissecting for capsule of liver, while LPT will be dissected by CUSA (Integra LifeSciences, NJ, USA, CUSA Excel+™). For vascular and biliary structures ⩽ 3 mm in diameter will be clipped with titanium clips. While the larger ones will be clipped by Hem-o-lok and divided. The corresponding Glissonean pedicles and large hepatic veins will be transected by linear endoscopic staplers. Bipolar coagulation will be used for hemostasis.
The surgical technique for LLR has been described previously.18,19 Patients will be positioned in the anti-Trendelenburg position and rotated 30° to the left. Five trocars will be strategically placed on the upper abdomen. Controlled low central venous pressure (CVP) and Pringle maneuver will be routinely performed during LPT. Intraoperative ultrasound will be utilized to identify tumor situations and boundaries. After completion of the resection, one or two silicone drainage tubes will be placed in the resection area.
Criteria for discontinuing or modifying allocated interventions
The surgery will be suspended in the presence of extensive abdominal metastasis or invasion of the inferior vena cava and other large blood vessels. It will also be suspended in case of other medical emergencies. If patients choose to withdraw from this treatment, it will be allowed, and they will be considered dropouts.
Provisions for post-trial care and follow‑up management
All the patients will receive the same standardized postoperative care pathway for liver resection, without concomitant care. Perioperative antibiotics will be used prophylactically for 48 h. ECG monitoring and nasal cannula oxygenation will be maintained for 24 h. Routine blood tests, liver and renal function tests, and coagulation evaluation will be performed routinely on postoperative days 1, 3, 5, and 7.
Patients will undergo follow-up appointments at the outpatient clinic monthly for postoperative 90 days. Tumor check-ups will be scheduled every 3 months following surgery, including AFP, CEA, CA199, PIVKA-II, ultrasonography, and dynamic contrast-enhanced CT when necessary. The contact information for participants will be collected by the follow-up team members to ensure regular communication during subsequent follow-up visits. Any necessary assistance required by trial participants will be readily provided at these follow-up appointments.
Outcomes
The primary outcome is the IBL, which will be calculated by subtracting the volume of flushing fluid from the total aspiration volume plus the weights of soaked gauze minus dry gauze. 20
Secondary outcomes encompass parenchymal transection velocity, IBL during the LPT, transaction surface evaluation, operative time, conversion rate, postoperative hospital stays, hospital cost, 30-day morbidity, and 90-day mortality. Parenchymal transection velocity is calculated as the ratio of transection area to transection time. 21 The transection area is estimated using a grid paper with squares measuring 0.5 cm * 0.5 cm. IBL during the LPT will be recorded as IBL before LPT minus IBL upon completion of LPT. The transection surface will be evaluated based on the following three aspects: smoothness of cut surface, extent of localized thermal injury, and presence of significant vascular damage. Operative time is measured from skin incision to skin closure. Postoperative morbidity is categorized according to the Clavien–Dindo classification and documented for up to 30 days or during the same hospitalization, including readmissions within 30 days. 22
Sample size
The calculation of sample size is based on the primary outcome according to a retrospective study of our center. 15 In the previous study, the mean IBL was 198.18 ml with a standard deviation (SD) of 237.15 ml in the HS group and 389.09 ml with an SD of 482.39 ml in the CUSA group. Significance level was set at 5% with a power of 80%. The sample size was calculated by SPSS Statistics Version 27 (IBM, NY, USA) with a dropout rate of 10%. Consequently, this study needs to enroll 69 participants in each group.
Randomization and blinding
A dedicated researcher is responsible for sequence generation and enrolling participants. Random sequences were generated by a computer random number method using SPSS Statistics Version 27 (IBM, NY, USA) before the study began. The random number and corresponding intervention plan were sealed into an opaque envelope. The researcher will choose envelopes according to the order of patient enrollment. After intraoperative exploration, the researcher will open the envelope to determine the instrument for LPT.
The researcher for sequence generation and patient enrollment will be blinded to subsequent experimental procedures. An independent researcher will conduct follow-ups with patients and collect data. Both the data analyst and data collector will be blinded to the group allocation. Patients and the nursing team will be blinded to the group allocation. It is impossible to blind the investigators since the intervention is conducted by surgeons. There is no procedure for unblinding since the researchers who conduct the intervention are unblinded.
Data collection and management
Baseline characteristics of patients will be assessed before allocation. Perioperative data will be recorded before discharge. The outcomes will be collected by an independent, blinded researcher in accordance with the pre-established case record form (CRF) prior to the commencement of the study. Identification information of all patients will be linked to a unique number.
The paper CRF will be stored in a locked closet, and access will only be granted with proper authorization. Following the completion of enrollment, two researchers will meticulously verify the accuracy of the CRF before transferring it into a local database for thorough data analysis. Both the paper CRF and the local database are only available to research members. The data will be kept confidential before the final publication of the study; the full protocol and data will be available in the submitted manuscript. It is also available to individuals with suitable requests.
Statistical analysis
Data analysis is performed using SPSS Statistics Version 27 (IBM, NY, USA) based on the intention-to-treat principle. Continuous variables will be presented as mean ± standard deviation or median with an interquartile range in terms of distribution. Categorical variables will be expressed as numbers and percentages. The Mann–Whitney U test is analyzed for continuous variables, whereas the Pearson’s chi-square test or Fisher’s exact test are applied for comparisons of categorical variables. p Value < 0.05 will be considered statistically significant in this study.
Given that liver cirrhosis negatively affects the outcomes of LLR, 23 a subgroup analysis will be conducted based on the presence of cirrhosis. Patients will be stratified into two subgroups: with and without liver cirrhosis. We will then compare the primary and secondary outcomes between the HS and CUSA groups across these subgroups to evaluate the impact of cirrhosis on the effectiveness of the two surgical techniques.
Oversight and monitoring
This study will be supervised by the Biomedical Ethics Review Committee of West China Hospital. Any amendments will be reviewed by the Biomedical Ethics Review Committee of West China Hospital again. And it will be submitted to the trial registry. Self-auditing will be conducted every month between the researchers and members of the committee. The coordinating center is the Division of Liver Surgery, Department of General Surgery of West China Hospital. The trial steering committee is composed of three investigators and three physicians.
Any adverse events will be monitored and managed by the attending physicians within a 7-day timeframe. In the case of serious adverse events, they will also be promptly reported to the Biomedical Ethics Review Committee of West China Hospital. Since the HS and CUSA are two mature and safe instruments in liver resection, this study will not introduce any additional surgical risks.
Dissemination plans
The findings will be submitted for publication in peer-reviewed journals and may also be presented at medical conferences.
Discussion
Typically, liver resection involves surgical field exposure, parenchyma fracturing, blood and parenchyma suction, stanching, and vessel handling. The CUSA is capable of performing three out of the five aforementioned operations (fracturing, suctioning, and stanching). 24 Deep layer parenchyma can be dissected safely by CUSA with or without bipolar cautery.10,11 After fracturing and aspirating liver parenchyma, the CUSA provides a clear visual field for vascular protection. Thus, the second International Consensus Conference on LLR 11 reported that CUSA was recommended for deeper parenchymal dissection (>2 cm). However, Gurusamy et al. 25 reported that the CUSA could not reduce blood loss compared to radiofrequency dissecting sealer, hydrojet, and clamp crushing techniques. In addition, a previous study conducted at our center 19 found that the CUSA group had a longer operative time and higher hospitalization costs compared to the LigaSure group.
Compared to CUSA, the HS was always used for superficial layer LPT and sealing vessels smaller than 5 mm.10,11,26 The coagulating effect through protein denaturation is attributed to the disruption of hydrogen bonds within proteins and the generation of heat in vibrating tissue. Simultaneously, the high-speed vibration of the blade produces a cutting effect. 27 Hence, surgeons could directly deal with bleeding vessels using HS without the need for equipment replacement. 15 Thus, the application of HS for LPT may pose the potential benefits in timing saving during LLR. Moreover, the prolonged operative time was identified as an independent risk factor for postoperative complications.28,29 Therefore, the HS is expected to enhance both the efficiency and safety of LLR by minimizing IBL and reducing operative time.
A previous study 14 examined the efficacy of the HS/TissueLink combination and CUSA/TissueLink combination in liver resection. This retrospective analysis revealed that the HS/TissueLink combination group exhibited significantly reduced blood loss (250 vs 1035 ml) compared to the CUSA group. Another prospective study focused on hemi-right hepatectomy 13 found that the HS group also presented a tendency toward less blood loss compared to the CUSA group. Furthermore, in a retrospective study conducted at our center, 15 the HS group demonstrated significantly less blood loss, shorter operative times, and reduced hospitalization costs compared to the CUSA group. And the postoperative morbidity and mortality were comparable in the above studies between the HS and CUSA groups. However, most studies comparing CUSA and HS are retrospective, have small sample sizes, and lack related high-quality medical evidence. Therefore, we design this RCT to evaluate the feasibility, safety, and efficiency of HS for LPT in LLR.
Intraoperative hemorrhage is still a tough problem in LLR because of the inherent vascularity of the organ, 30 which could affect perioperative morbidity and mortality.4,5 In addition, uncontrollable bleeding during LLR was also a main reason for conversion to open surgery. 31 In addition, excessive blood loss has been identified as a negative prognostic factor for both overall survival and disease-free survival in patients with liver malignancies. 6 Thus, we considered the IBL as the primary outcome in this study.
The present study is a single-blinded, RCT. The random sequence was generated by SPSS Statistics Version 27 (IBM, NY, USA), and the corresponding intervention plan was sealed into an opaque envelope. Patients will be recruited according to their order of admission. The randomization could minimize the risk of confounding by unmeasured factors. 32 Moreover, blinding researchers apart from the surgical team will help to eliminate unconscious information bias. 32
Meanwhile, the Pringle maneuver and controlled low CVP will be routinely performed during LPT to control blood loss, which is facilitated to mitigate potential confounding factors that may impact intraoperative bleeding. Moreover, to decrease the bias caused by cut surface and enhance the accuracy of transection area measurements, we propose a method similar to the cell counting plate for calculating the transaction area. The included patients will also be restricted to those undergoing laparoscopic anatomical liver resection according to the 2000 Brisbane classification. 33 Given that liver cirrhosis has a negative impact on the outcomes of LLR, 23 subgroup analysis will be performed according to the cirrhotic status of the background liver.
This protocol has several limitations that warrant acknowledgment. First, as a single-center study, the findings may not fully represent outcomes in hospitals with differing surgical workflows or device availability. Second, the 90-day follow-up period focuses on short-term complications; long-term outcomes should be evaluated in future studies. Third, the current inclusion criteria are confined to liver malignancies, it is necessary to explore the efficacy and safety profiles of these transection modalities in benign pathologies. Therefore, multicenter studies with diverse cohorts are needed to verify the results of this study.
Conclusion
In conclusion, to provide more high-quality evidence of the role of HS in the LPT, we conducted this RCT to verify the feasibility, safety, and efficiency of the HS in LLR compared to the commonly used CUSA. To the best of our knowledge, this is the first RCT on this topic.
Trial status
The trial has been reviewed and approved by the Biomedical Ethics Review Committee of West China Hospital, Sichuan University, on January 19, 2024. The study protocol was registered in the Chinese Clinical Trial Registry on April 26, 2024 (ChiCTR2400083493). The protocol version is V2.0. Participant recruitment started on May 1, 2024, and recruitment is anticipated to be completed on May 1, 2025.
Supplemental Material
sj-doc-1-tag-10.1177_17562848251348974 – Supplemental material for Laparoscopic liver parenchymal transection using CUSA versus harmonic scalpel: a protocol for a prospective randomized controlled trial
Supplemental material, sj-doc-1-tag-10.1177_17562848251348974 for Laparoscopic liver parenchymal transection using CUSA versus harmonic scalpel: a protocol for a prospective randomized controlled trial by Bin Liang, Yufu Peng, Wugui Yang, Yubo Yang, Bo Li, Yonggang Wei and Fei Liu in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
