Abstract
With the increasing prevalence of endoscopic procedures, peri-endoscopic management of antithrombotic agents remains challenging due to inconsistent guideline adherence. This review highlights barriers to suboptimal guideline adherence, which include inadequate guideline education, delayed regional guideline updates, ambiguous anticoagulation responsibility, drug cost disparities, and uneven international resource allocation. Proposed strategies to improve compliance include: (1) Enhanced education for physicians and patients to align practices with evidence-based recommendations; (2) Digital tools such as clinical decision aids to standardize risk stratification and medication management; (3) Unified antithrombotic management teams to clarify responsibilities and reduce inter-specialty discrepancies; and (4) International collaboration to harmonize guidelines and address regional disparities in healthcare access and practice patterns. Existing guidelines from societies such as the European Society of Gastrointestinal Endoscopy, the American Society for Gastrointestinal Endoscopy, and the British Society of Gastrointestinal Endoscopy provide risk-stratified frameworks for procedure-specific antithrombotic management, yet their adoption varies globally. Eastern practices often prioritize shorter warfarin cessation windows (e.g., 3 days for routine colonoscopies in China), contrasting with Western protocols that align more closely with international guidelines. Future directions emphasize integrating artificial intelligence into clinical workflows to personalize risk assessments and adopting telemedicine platforms for real-time decision support. Addressing these gaps is critical to reducing thromboembolic and hemorrhagic complications in high-risk patients undergoing endoscopic procedures.
Plain language summary
With the increasing prevalence of endoscopic procedures among aging patients worldwide, managing individuals with atrial fibrillation (AF) and thromboembolic disorders (TE) presents a significant challenge for physicians. Recent guidelines from major gastrointestinal endoscopy societies, including those from Europe, the USA, Asia, and the Pacific regions, provide detailed recommendations for managing antithrombotic agents based on varying risks. However, adherence to these guidelines in clinical practice remains suboptimal. To address this issue, we first summarized the varying recommendations across these guidelines and conducted a systematic analysis of current clinical compliance rates. We identified potential barriers to compliance, categorizing them into physician-related, patient-related, and cultural perspective barriers. Furthermore, we highlighted promising measures to overcome these challenges, including enhanced patient and physician education, telephone-based instructions, the formation of unified antithrombotic management departments, the use of digital clinical decision aid tools, and the promotion of international academic communication. By implementing these strategies to improve clinical compliance, the standardized use of antithrombotic agents can be achieved, effectively reducing coagulation-related adverse events and addressing the global health burden associated with these conditions.
Introduction
The use of antiplatelet and anticoagulant medications is growing, especially among patients with atrial fibrillation (AF) and thromboembolic disorders. 1 Driven by advanced endoscopic therapy, more patients under antiplatelet or anticoagulant medications were now facing the situation of taking the above therapy at the same time. Gastrointestinal bleeding (GIB) is a recognized risk of systemic antithrombotic therapy, which may be exacerbated during endoscopic activities, especially therapeutic ones. 2 Clinicians are thus faced with the critical task of deciding whether to withhold or continue antithrombotic therapy in patients undergoing endoscopy, balancing the risks of GIB against those of thromboembolic events. 3
In response to these trends, the American Society for Gastrointestinal Endoscopy (ASGE) developed the first international guideline in 1996. 4 This guideline, which was based on expert consensus, focused on managing patients on antithrombotic therapy during the peri-endoscopic period. Subsequently, numerous high-quality clinical trials have been undertaken, leading to the publication and updates of several position statements and guidelines by major gastroenterology societies across Europe, the United States, and Asia.5–7 These guidelines have played a crucial role in enhancing the quality of medical care and addressing unwarranted clinical variations, which are often attributed to inexplicable differences based on patient conditions or preferences, thereby potentially improving patient outcomes.
Despite more evidence-based data on using anticoagulants during the peri-endoscopic period, there remains considerable resistance among physicians to adhering to these guidelines in clinical practice. 8 Recognizing this gap, our review aims to identify the barriers to the global implementation of these guidelines and explore effective strategies to overcome these obstacles. Ultimately, our goal is to lay a new groundwork for guiding future research and shaping practical recommendations in this area, thus addressing these persistent challenges comprehensively.
Literature search
In this review, we employed specific search terms including “Endoscop*,” “Exp gastroscopy/,” “Exp endoscopy/,” “Gastroscop*,” “Periendoscop*,” “Esophagogastroduodenoscopy,” “Periendoscopic,” “Anticoagulant,” “Antithrombotic,” “Antiplateletand,” “Warfarin,” “Clopidogrel,” “Aspirin,” “Guideline,” “Compliance,” “Adherence,” “Current state,” “Implementation,” “Survey,” and “Practice.” These keywords were used to locate relevant articles in the PubMed, Embase, and Web of Science databases. The search extended up to December 2023. The inclusion criteria included: (i) relevant clinical or basic research; and (ii) papers published in peer-reviewed journals without language restrictions. Exclusion criteria were: (i) papers lacking full-text availability; (ii) studies with low relevance to the topic; (iii) studies with unclear descriptions or insufficient evidence-based ratings. We identified 940 papers through this search process, from which 90 met our selection criteria and were included in this review. Figure 1 shows the distribution of the 20 countries engaged in peri-endoscopic and antithrombotic research. The United States produced the highest number of documents (n = 29, 33.0%), followed by Japan (n = 13, 14.8%), and Italy (n = 7, 7.9%).

Distribution of countries engaged in peri-endoscopic and antithrombotic research.
Balancing risk and benefit: Managing antithrombotic therapy in endoscopic procedures
Over recent decades, advancements in antiplatelet and anticoagulant therapies have significantly evolved, introducing new agents and shifting treatment paradigms.9,10 Aspirin (ASA) remains the most commonly prescribed antiplatelet agent, reducing vascular mortality by 15% and nonfatal vascular events by 30%, as evidenced by a review of 25 randomized controlled trials. 11 The advent of ADP receptor antagonists, including ticlopidine, clopidogrel, prasugrel, and ticagrelor, marked a significant development in antiplatelet therapy.12,13 The dual antiplatelet therapy (DAPT) of clopidogrel and aspirin, particularly in non-ST elevation myocardial infarction patients, demonstrated reduced mortality rates and ischemic events. 14
Heparin, the first medically used anticoagulant, significantly reduced the risk of fatal pulmonary embolism in a 1975 multicenter study. 15 However, its limitations, including a narrow therapeutic window and variable response, led to the development of low-molecular-weight heparin (LMWH), offering advantages over unfractionated heparin. 16 In oral anticoagulation, dicoumarol and subsequently warfarin were milestones, despite warfarin’s drawbacks such as a narrow therapeutic index and frequent monitoring requirements. 17 The introduction of direct oral anticoagulants (DOACs) represented a paradigm shift, offering reduced stroke, mortality, and major bleeding risks, albeit with limitations such as cost and absence of an antidote. 18
In the context of endoscopic procedures, including polypectomy, endoscopic mucosal resection, endoscopic submucosal dissection, and sphincterotomy, the risk assessment of GIB and thromboembolism for patients on antithrombotics is crucial in these high-bleeding risk procedures. 19 Aspirin has been associated with a marginally increased risk of post-polypectomy bleeding (PPB), whereas clopidogrel users exhibited a higher rate of PPB. 20 The use of DAPT complicates clinical management due to increased risks of PPB and thrombotic events. 21 Warfarin and DOACs are identified as independent risk factors for PPB, necessitating careful cessation strategies, including heparin bridge therapy (HBT). However, evidence suggests that HBT may not be superior to non-HBT strategies in preventing thrombotic events and may even increase the risk of PPB. 22
The comparison between warfarin and DOACs regarding PPB risk remains under investigation, with mixed outcomes reported across studies. 23 Nevertheless, temporary cessation of DOACs for two half-lives before procedures is recommended, aiming to balance the risk of PPB against thromboembolic complications. 24 Recent evidence supports a 1-day interruption of DOACs as a safe practice before polypectomy, minimizing PPB risk while maintaining therapeutic efficacy. 25 These evolving practices underscore the need for ongoing research and guideline updates to navigate the complex interplay between antithrombotic therapy and endoscopic procedures.
Guidelines on the management of antithrombotic agents in the peri-endoscopic period
The management of antithrombotic therapy during endoscopic procedures is a critical area addressed by various international guidelines. The European Society of Gastrointestinal Endoscopy (ESGE), the British Society of Gastrointestinal Endoscopy (BSGE), 26 ASGE, 27 the Asian Pacific Association of Gastroenterology (APAGE), the Asian Pacific Society for Digestive Endoscopy (APSDE), 28 and the Japan Gastroenterological Endoscopy Society (JGES) 29 have all published guidelines to assist physicians in making informed decisions.
The bleeding risk of endoscopic procedures is widely discussed in different guidelines to estimate the necessity and safety of ceasing antithrombotic agents. ASGE, BSGE, ESGE, and APAGE classified procedures according to bleeding risk, while JGES stratified in terms of the purpose of endoscopy and bleeding risk. Detailed classification is outlined in Supplemental Table 1.
Similar to the bleeding risk classification mentioned above, there are different contents in the risk classification of thromboembolic events after discontinuation of antithrombotic drugs among guidelines. Detailed stratification is shown in Supplemental Tables 2 and 3. We provide a brief summary of the guideline-based risk stratification for endoscopic bleeding and post-cessation thromboembolic events below:
Bleeding risk stratification ● ESGE/BSGE/ASGE/APAGE classify procedures into low-, intermediate-, and high-risk categories based on bleeding severity (e.g., polypectomy = high risk). ● JGES uniquely adopts a dual stratification system: ○ Purpose-based classification (diagnostic vs therapeutic procedures). ○ Bleeding risk categorization (low/moderate/high). This hybrid approach reflects Japan’s emphasis on procedural context in risk assessment.
Thromboembolic risk assessment ● ASA/Warfarin Discontinuation:
○ Western Guidelines (ESGE, ASGE): Recommend continuing ASA for all procedures except high-risk interventions. ○ JGES permits ASA discontinuation for routine procedures (e.g., biopsy) due to lower observed bleeding risk in Japanese cohorts.
DAPT Management:
○ ESGE/BSGE: Advocate DAPT continuation in high-ischemic-risk patients (e.g., recent PCI). ○ JGES: Favors cilostazol substitution for short-term procedures. ○ APAGE/APSDE: Recommend individualized risk-benefit assessment, influenced by regional variations in stent implantation practices.
Different interruption and resumption timings of antithrombotic drugs under risk classifications
Peri-procedural management of antithrombotics ● Low-risk procedures: ○ Consensus across guidelines: Continue ASA (monotherapy or as part of DAPT) and warfarin. ○ DOACs (ASGE/JGES/APAGE) ● Generally recommend continuation. ● BSGE Exception: Suggests discontinuation, with timing adjusted by creatinine clearance. ● High-risk procedures: ○ ASA (all guidelines): Continued. ○ Non-ASA Antiplatelets (e.g., thienopyridines): Discontinued (detailed in Supplemental Table 4). ○ Warfarin (all guidelines): Held preprocedure with LMWH/heparin bridging. Restarted post-procedure, resumption timing varies among the guidelines. ○ JGES-Specific Approach: Supports cilostazol as an alternative to ASA in select cases. ○ DOACs: ● ASGE/ESGE/APAGE: Discontinuation based on CrCl, no bridging recommended (Supplemental Table 6). ● JGES: Bridging advised in certain cases. ● Post-procedural resumption (all guidelines): Once hemostasis confirmed.
Bridging and international normalized ratio (INR) monitoring ● Warfarin bridging (all guidelines): ○ LMWH/heparin substitution before high-risk procedures. ○ Variability in bridging duration (Supplemental Table 5). ● INR assessment (all guidelines): ○ Preprocedure INR check mandatory to guide discontinuation. ○ Heparin stopped once INR therapeutic post-procedure.
In addition to the comparative analyses presented above, Supplemental Table 4 details the management of thienopyridine before and after high-risk bleeding endoscopic procedures.
For high-risk procedures, all guidelines recommend withholding warfarin before the procedure and substituting it with LMWH or heparin. All guidelines recommend restarting the warfarin after the procedure since studies show there is an association between the resumption of warfarin and the reduction of thromboembolic events. 30 Guidelines differ in minor details concerning the length of drug discontinuation, the estimation of thromboembolic risk, and the need for bridging from warfarin to LMWH 31 (Supplemental Table 5). All guidelines recommend that doctors check the INR prior to the procedure to determine whether to discontinue therapy. Following the procedures, heparin should be discontinued once the INR returns to the therapeutic range. The ASGE emphasizes that in moderate-risk patients, the decision to use bridging therapy and its intensity should be tailored to individual patient needs and consider the patient’s preferences. Heparin may be resumed once hemostasis is confirmed. In Japan, doctors have observed a high number of thromboembolic complications following the withdrawal of antithrombotic agents. As a result, there is a tendency to shorten the discontinuation period of antithrombotic agents, even for routine examinations, biopsies, or endoscopic treatments. 32
The recommendations for DOACs from the ASGE, ESGE, and APAGE are consistent (Supplemental Table 6). Generally, different withdrawal times are recommended based on varying creatinine clearance rates, and bridging therapy is generally not advised. However, the JGES does recommend bridging, and discontinuation of antithrombotics is not suggested in low-risk conditions. All four guidelines agree that medications should be resumed post-procedure once hemostasis is confirmed. Discussions concerning DOACs have primarily focused on dabigatran, with limited supporting data available for other drugs. 33
To sum up, guidelines between east and west had a unifying trend, but discrepancies among them still remained, which is largely because of areal variation and insufficient evidence. Future guidelines should be adapted for daily clinical practice of incorporating novel endoscopic procedures and agents. 34
Impact of guideline discrepancies on clinical practice
Current evidence highlights that disparities between guidelines significantly impact peri-endoscopic antithrombotic management, though robust comparative data remain limited. Key discrepancies—such as divergent recommendations on DOAC cessation windows and bridging strategies—create systemic challenges. These variations lead to inconsistent risk stratification, particularly in high-risk populations, and may result in suboptimal thromboembolic or bleeding outcomes. Regional path dependencies, such as entrenched practices in East Asia favoring abbreviated cessation periods, further complicate care transitions and interventional planning. In addition, ambiguous responsibility allocation between specialties (e.g., cardiologists vs gastroenterologists) exacerbates interprofessional discord, while disparities in healthcare infrastructure and cultural attitudes toward guideline adherence limit equitable implementation. Addressing these gaps requires standardized, evidence-based frameworks and cross-disciplinary collaboration to harmonize practices across diverse clinical ecosystems.
Evaluating adherence to guidelines in endoscopic practices
Compliance with current guidelines for the use of antithrombotics before endoscopy remains unsatisfactory, especially as unnecessary discontinuation of antithrombotics remains high. In 1996, Kadakia et al. first explored the practice of using antithrombotic medications during endoscopic procedures. Their study revealed that physicians discontinued aspirin in 51% of cases before gastroscopy and 81% before colonoscopy, while discontinuation rates for warfarin ranged between 51%–60% and 71%–82%, respectively. 35 Due to the considerable variability in the management of antithrombotic and anticoagulant agents around the time of endoscopy, the ASGE responded by publishing the inaugural guideline in 1998. 36 The guideline aimed to standardize the management of anticoagulant and antiplatelet therapy during endoscopic procedures. Subsequent research has focused on adherence to the guidelines, revealing persistently suboptimal compliance by both physicians and patients since 2000. In elective gastrointestinal endoscopy scenarios, there is a notable trend of unnecessary discontinuation of anticoagulants and antiplatelet medications, with warfarin being ceased more frequently than aspirin without clinical indication. 31 In contrast, for procedures with high-bleeding risks, such as polypectomy, adherence to guidelines is better, as practitioners prioritize the avoidance of thromboembolic risks over bleeding risks. 27 Current literature is deficient in evaluating patient adherence to recommended practices, particularly concerning the use of LMWH bridging and the duration of warfarin discontinuation. The studies and surveys reporting the management of antithrombotics in patients undergoing elective endoscopy are summarized in Figure 2.20,31,37–47 Current evidence reveals significant discrepancies between guideline-recommended antithrombotic management and clinical practice. Population-based studies demonstrate alarming rates of non-indicated discontinuation, with 41.33% (88/213) of patients experiencing unnecessary antithrombotic cessation. Even among high-risk cohorts requiring continued antiplatelet therapy, 32.5% of aspirin users experienced protocol deviations contrary to evidence-based recommendations. Notably, short-term follow-up durations (median 30 days) and limited sample sizes precluded thromboembolic complication detection in these studies.
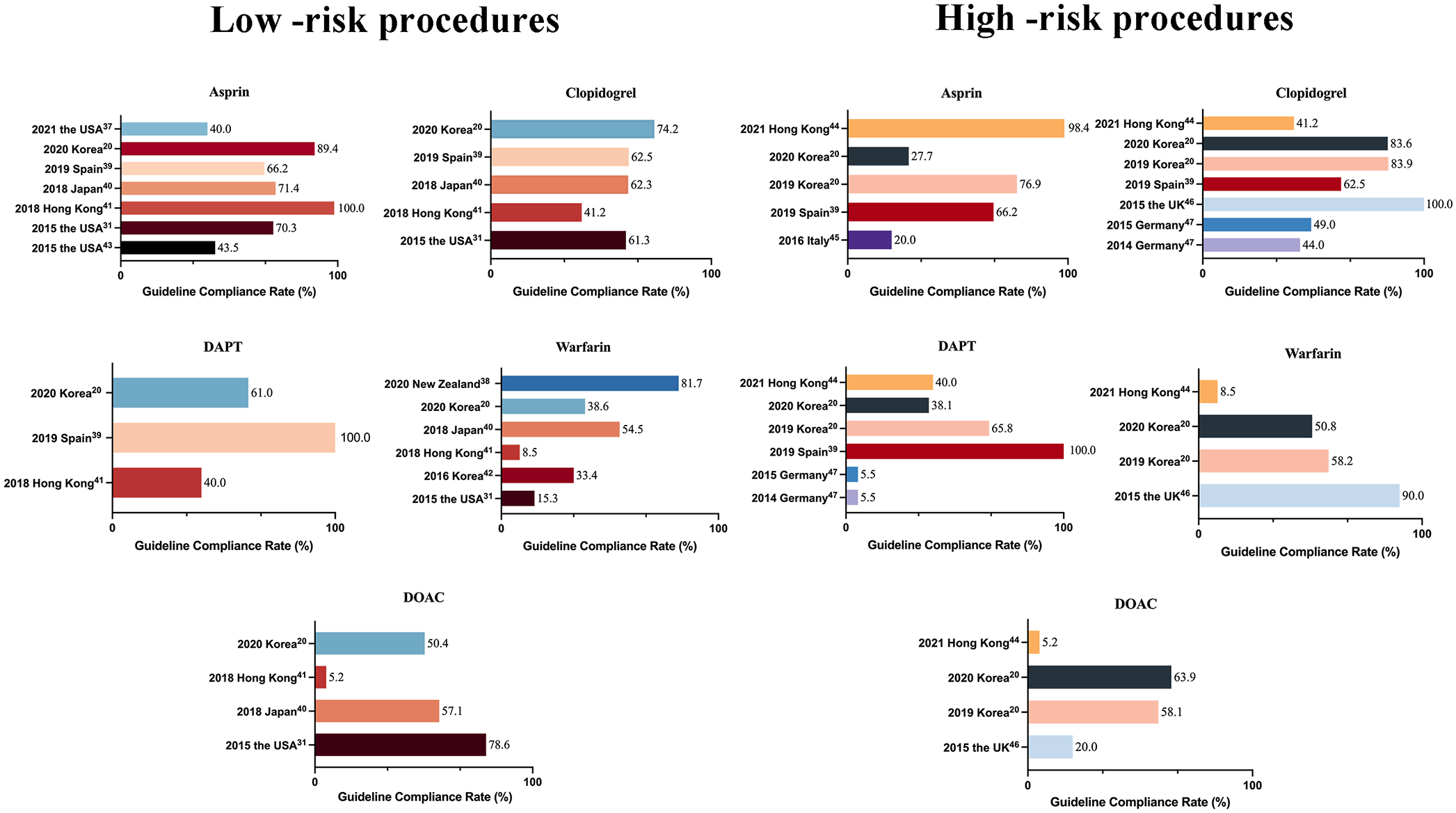
Adherence to antithrombotic guidelines among patients undergoing elective endoscopy. The chart above presents the guideline compliance rates for various antithrombotic medications across different countries and years.
Paradoxically, emerging data document preventable thrombotic events from overly aggressive drug cessation. A retrospective cohort analysis identified two serious adverse outcomes: (1) ischemic stroke occurring 30 days post-elective surgery in a patient with long-term aspirin therapy discontinued during preoperative preparation; (2) recurrent venous thromboembolism in a DOAC user maintained off therapy for 12 weeks despite guideline-recommended 1-week interruption.
These findings highlight critical knowledge gaps in antithrombotic stewardship. Current literature remains inconclusive regarding subgroup-specific risks due to: (1) lack of dedicated risk stratification frameworks; (2) insufficient power in existing trials to detect rare thrombotic events; and (3) heterogeneity in defining “inappropriate” discontinuation across studies. Prospective multicenter registries with standardized outcome definitions and extended follow-up (⩾90 days post-procedure) are urgently required to: (a) quantify bleeding-thrombosis trade-offs in real-world practice; (b) establish evidence-based discontinuation thresholds; (c) develop predictive models for high-risk subpopulations.
Potential barriers
Physician recommendations exert considerable influence on patient medication management, especially in the context of comorbidities such as cardiovascular conditions and the administration of antithrombotic therapy during endoscopic procedures, which represents a significant clinical challenge. A subset of endoscopists remains inadequately informed about current protocols, with more seasoned practitioners often relying on prior experience over contemporary guidelines, resulting in persistently low adherence rates.48–50 A 2015 survey across 317 US endoscopy centers revealed that merely 45% adhered to guidelines recommending the continuation of aspirin therapy preoperatively. 43 This trend has not seen substantial improvement; a 2021 study conducted by the University of Virginia found that 60% of patients (26 out of 43) discontinued aspirin use prior to endoscopic procedures, with 19% (5 out of 43) ceasing usage for over 5 days, primarily following medical advice from specialties including gastroenterology and cardiology. 37 This indicates a pervasive caution within the medical community toward procedural bleeding risks, overshadowing the potential cardiovascular risks associated with discontinuation of therapy. This issue is not isolated to one region, with similar patterns observed in diverse geographical contexts, including the United States, Japan, and Italy,32,37,45 underscoring a global trend of suboptimal guideline adherence.
The diversity in guidelines and their successive updates can sometimes generate confusion among clinicians. Notably, until 2015, Korean guidelines had not addressed warfarin management before gastroscopy. Son et al. highlighted variations in practice among national cardiac surgeons regarding warfarin cessation prior to gastroscopy. 42 Despite evidence suggesting that bridging therapy does not reduce thrombotic risk while increasing bleeding likelihood in patients with non-valvular AF, Slivnick et al. observed that endoscopists were more likely to employ LMWH bridging than cardiologists (30% vs 18%), underscoring the complexities faced by endoscopists in navigating anticoagulation management.51,52
A 2018 survey among cardiologists, endoscopists, and interns revealed varied opinions on the responsibility for anticoagulant guidance, with a general consensus favoring cardiologists, though no formal responsibilities had been designated, leading to ambiguity and occasional inaccuracies in antithrombotic therapy records. 53 This lack of clarity contributes to non-standardized approaches to anticoagulant management before endoscopic procedures.
Comparative analysis of guideline compliance between Eastern and Western contexts indicates better adherence in Western settings, partially attributed to the distribution of medical resources. 54 In Western healthcare systems, concentrated urban medical resources facilitate access to specialized care and advanced medications, improving guideline adherence. 55 However, the high costs of DOACs remain a significant barrier, affecting patient accessibility despite extensive healthcare coverage. 56 Conversely, in Eastern systems, particularly in countries such as China and India, the disparity between urban and rural healthcare access affects antithrombotic management. 57 This geographical inequality limits rural patients’ access to modern medications and endoscopic techniques, compounded by significant out-of-pocket expenses for newer drugs. 58
Cultural perspectives also play a role, with Western individualistic healthcare approaches promoting adherence to evidence-based medical practices, including antithrombotic therapy. In contrast, Eastern cultures, valuing traditional and holistic health practices, may exhibit skepticism toward Western medications, affecting adherence rates to antithrombotic guidelines. 59 The influence of family and community in healthcare decisions further differentiates patient attitudes toward treatment in these regions. 60
Strategies and perspectives for implementation of the guidelines
In light of the above barriers, we recommend several strategies (Figure 3) to improve adherence to the guidelines (or their region-specific modification) and access to guideline-recommended therapies.

Strategies for implementation of guidelines. The diagram illustrates multifaceted strategies for the implementation of anticoagulant medication guidelines aimed at enhancing the use and understanding among healthcare providers and patients and improving the healthcare environment.
The endoscopist plays a leading role in the patient’s treatment, enhancing guideline adherence in outpatient endoscopic treatments hinges on improving communication between patients and healthcare providers. 61 Often, patients on regular anticoagulant therapy receive their primary guidance from cardiologists. However, discrepancies in clinical recommendations between gastroenterologists and anticoagulation specialists often result in therapeutic confusion and medication non-adherence. 62 As Plumé et al. observed, 29.7% of patients receive contradictory instructions regarding anticoagulant management across different endoscopic procedures, leading to inconsistent pharmacological protocols. 39 This underscores the necessity for multidisciplinary collaboration in long-term anticoagulation management, particularly through enhanced specialist-endoscopist communication, structured pre-procedural consultations, and the establishment of integrated perioperative anticoagulation teams to standardize therapeutic decision-making. Concurrently, each endoscopic facility should implement standardized communication frameworks to mitigate prescription discrepancies. 63 Optimizing patient education requires moving beyond traditional paper-based modalities, as the evidence demonstrates the superior efficacy of direct communication strategies. Telephone-based pre-procedural counseling achieves better patient engagement than static pamphlets, particularly given the frequent delays in outpatient endoscopy scheduling that contribute to educational material neglect. 64 In addition, targeted competency-based training programs for healthcare providers—especially primary care physicians—should be prioritized to improve anticoagulation management proficiency, with particular emphasis on perioperative management principles. 49
The implementation of auxiliary technologies such as Best Practice Alert systems in electronic medical records has been shown to improve adherence to medication guidelines among endoscopy patients significantly. These systems, informed by expert protocols, assist endoscopists in making informed prescribing decisions, increasing both patient and physician satisfaction in anticoagulant management. 65 The development of apps and online tools aimed at reducing the anticoagulation knowledge burden for endoscopists represents a promising advancement.66,67 The integration of artificial intelligence and telemedicine into patient management systems holds great promise for the future, offering personalized and real-time adjustments to anticoagulation therapy. These technological advancements could lead to more dynamic and effective healthcare, improving patient outcomes and enhancing the efficiency of medical care.68,69
Reducing the guideline compliance gap between Eastern and Western countries involves several strategies. It is essential to foster collaborative research initiatives, for international conferences and workshops facilitate the exchange of knowledge and experiences, promoting a global understanding of anticoagulation management. 70 Another pivotal aspect of future research is the standardization of practices and protocols in the peri-endoscopical management of antithrombotic agents. This requires a concerted effort to harmonize guidelines based on evidence gathered from both Eastern and Western clinical practices. Collaborative research initiatives, leveraging diverse healthcare systems, enhance the quality and applicability of research across settings. Finally, the standardization of antithrombotic management protocols, guided by global evidence and adapted to local practices, is essential. International gastroenterology societies could spearhead these standardization efforts, fostering unified guidelines and practices worldwide. 71
Summary
This comprehensive review synthesizes contemporary evidence on pre-endoscopic antithrombotic management, offering an evidence-based framework to guide the development of harmonized perioperative antithrombotic protocols and improve patient outcomes.
Optimizing adherence to antithrombotic guidelines requires targeted strategies in four priority domains: evidence-based guideline refinement, digital health system integration, institutional policy reinforcement, and structured medical education. Current gaps include limited comparative analyses of guideline compliance between nations with and without national antithrombotic protocols. While international guidelines establish foundational standards, their variable applicability to regional populations necessitates developing evidence-based consensus frameworks through multinational expert collaborations prior to endoscopic procedures.
The complexity of antithrombotic recommendations poses implementation challenges, particularly for endoscopists managing high-risk patients. This underscores the urgency for integrating AI-driven decision-support algorithms into clinical workflows, particularly through clinical decision-support systems that automate risk stratification and therapeutic recommendations.
Effective perioperative antithrombotic management demands institutional commitment through multidisciplinary anticoagulation task forces. Establishing specialized pre-endoscopy care centers, embedded within hospital quality improvement frameworks, could standardize protocol implementation while enhancing coordination between gastroenterologists, hematologists, and primary care providers.
Finally, competency-based medical education reform should prioritize antithrombotic stewardship training. Simulation-based mastery learning for trainees, combined with just-in-time educational tools for practicing clinicians, may reduce practice variability and improve adherence to best practices.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251346869 – Supplemental material for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications
Supplemental material, sj-docx-1-tag-10.1177_17562848251346869 for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications by Jiaxuan Zuo, Wei Jiang, Lumei Wang, Kaiqi Yang, Peng Li, Shutian Zhang and Rui Cheng in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251346869 – Supplemental material for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications
Supplemental material, sj-docx-2-tag-10.1177_17562848251346869 for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications by Jiaxuan Zuo, Wei Jiang, Lumei Wang, Kaiqi Yang, Peng Li, Shutian Zhang and Rui Cheng in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848251346869 – Supplemental material for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications
Supplemental material, sj-docx-3-tag-10.1177_17562848251346869 for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications by Jiaxuan Zuo, Wei Jiang, Lumei Wang, Kaiqi Yang, Peng Li, Shutian Zhang and Rui Cheng in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848251346869 – Supplemental material for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications
Supplemental material, sj-docx-4-tag-10.1177_17562848251346869 for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications by Jiaxuan Zuo, Wei Jiang, Lumei Wang, Kaiqi Yang, Peng Li, Shutian Zhang and Rui Cheng in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-5-tag-10.1177_17562848251346869 – Supplemental material for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications
Supplemental material, sj-docx-5-tag-10.1177_17562848251346869 for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications by Jiaxuan Zuo, Wei Jiang, Lumei Wang, Kaiqi Yang, Peng Li, Shutian Zhang and Rui Cheng in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-6-tag-10.1177_17562848251346869 – Supplemental material for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications
Supplemental material, sj-docx-6-tag-10.1177_17562848251346869 for Contemporary approaches in the peri-endoscopic management of antithrombotic agents: current paradigms, challenges, and prognostications by Jiaxuan Zuo, Wei Jiang, Lumei Wang, Kaiqi Yang, Peng Li, Shutian Zhang and Rui Cheng in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
