Abstract
Background:
Clostridioides difficile infection (CDI) treated with bezlotoxumab (BEZ) has been demonstrated to have a lower recurrence rate than placebo in clinical trials. However, real-world data on BEZ’s effectiveness remain limited and heterogeneous.
Objectives:
To evaluate the real-world effectiveness of BEZ in preventing CDI recurrence through a single-center retrospective cohort and a meta-analysis.
Design:
A retrospective cohort study and a meta-analysis.
Methods:
A retrospective study of patients treated with BEZ from 2017 to 2021 was performed at the Mayo Clinic. Demographics, CDI diagnostics, and several prespecified risk factors were analyzed. A literature search was conducted in August 2024 utilizing the Cochrane Central Register of Controlled Trials, Embase, Medline, Scopus, and Web of Science Core Collection. Studies reporting CDI resolution rates with BEZ were included. The random-effects model described by DerSimonian and Laird was used to calculate weighted pooled resolution rates (WPR) with 95% confidence intervals (CI). We assessed heterogeneity with the inconsistency index (I2) statistic.
Results:
Across 28 studies and our retrospective cohort, 2639 CDI patients were analyzed. Among 1786 patients treated with BEZ, 1450 achieved clinical resolution (WPR: 81.6%, 95% CI 77.2–85.6%), with significant heterogeneity (I² = 77.3%). In subgroup analysis, the pooled relative risk of recurrence was 0.56 (95% CI 0.36–0.88; p < 0.01) for BEZ with SoC compared to SoC alone. A WPR of 83.3% (95% CI 75.5%–91.1%) for patients receiving BEZ with SoC was observed when compared with a WPR of 70.8% (95% CI 62.7%–78.8%) in patients receiving SoC alone on subgroup analysis. In our cohort of 47 patients, the CDI resolution rate was 72.3% (34/47).
Conclusion:
Our retrospective study and meta-analysis demonstrate the real-world efficacy of BEZ in reducing CDI recurrence. The higher recurrence rates in our cohort likely reflect the high-risk nature of the population, including a greater proportion of immunocompromised patients.
Plain language summary
Clostridioides difficile infection (CDI) is a common and serious gut infection that often comes back after treatment. Bezlotoxumab (BEZ) has been shown to reduce the chances of recurrence. This study included a review of 47 patients treated with BEZ at the Mayo Clinic and combined their results with data from 28 other studies involving a total of 2639 CDI patients. Overall, 83.3% of patients treated with BEZ had their infections resolved, compared to 70.8% for patients treated with standard antibiotics alone. At the Mayo Clinic, 72.3% of the 47 patients had their CDI resolved with BEZ treatment. BEZ was found to reduce the risk of recurrence by nearly half (relative risk of 0.56). In conclusion, our study and analysis confirm that BEZ therapy is effective in reducing CDI recurrence in real-world settings. However, higher recurrence rates in high-risk patients, particularly those with weakened immune systems, highlight the importance of future studies to better understand the impact of additional health conditions.
Introduction
Clostridioides difficile is the most common cause of infectious diarrhea in hospitalized patients in the United States. 1 Globally, the incidence of healthcare-associated Clostridioides difficile infection (CDI) is estimated at 2.24 per 1000 admissions per year and 3.54 per 10,000 patient-days. While the overall incidence of CDI has declined in recent years, recurrence remains a significant hurdle in the treatment of CDI. Following an initial episode, up to 35% of patients experience a recurrence, and this risk increases to 65% in those with two or more prior infections. 2 Recurrent CDI (rCDI) is associated with significant clinical and economic burdens, including increased hospital readmissions, complications such as toxic megacolon and bowel perforation, and substantially higher healthcare costs. 3
Current strategies for managing CDI and preventing recurrence include optimizing antibiotic therapy and restoring a healthy gut microbiome. Standard-of-care antibiotics such as vancomycin or fidaxomicin are effective in treating acute CDI and have some impact on recurrence risk. For instance, fidaxomicin has been shown to modestly lower recurrence rates compared to vancomycin in initial CDI treatment.4,5 In patients with multiple recurrences, prolonged or tapered antibiotic regimens are often used in an attempt to suppress relapse. Additionally, fecal microbiota transplantation (FMT) has emerged as a highly effective treatment for patients with refractory or multiply recurrent CDI, with success rates often exceeding 80% in those scenarios. 6 However, FMT is typically reserved for patients after multiple relapses and comes with practical challenges in standardization and access. Even with these approaches, many high-risk patients still suffer repeat infections, underscoring the need for adjunctive therapies that specifically target recurrence mechanisms. Evolving data suggest microbiome-based therapies such as fecal microbiota transplantation, fecal microbiota, live-jslm, and fecal microbiota spores, live-brpk may be effective in patients with rCDI (recurrent Clostridioides difficile infection). 6
Bezlotoxumab (BEZ) is a novel human monoclonal IgG1 antibody that binds and neutralizes C. difficile toxin B, thereby preventing toxin-mediated damage to the colonic mucosa. It offers passive immunotherapy to reduce the risk of symptomatic recurrence. BEZ is approved for the prevention of rCDI in adults receiving standard-of-care (SoC) antibiotic therapy, such as oral vancomycin or fidaxomicin. In two large, double-blinded, randomized, placebo-controlled phase III trials, MODIFY I and MODIFY II, were conducted to investigate the clinical efficacy and safety of BEZ, to prevent rCDI in adult patients receiving SoC. 7 In these trials, BEZ significantly reduced the rate of recurrence compared to placebo (MODIFY I: 17% vs 28%; MODIFY II: 16% vs 26%, both p < 0.01). Post-hoc analyses of these trials identified older age (⩾65 years), severe CDI, immunocompromised status, and prior history of CDI in the last 6 months as key risk factors associated with the greatest benefit from BEZ. Patients with three or more of these risk factors experienced the greatest risk reduction in recurrence risk with BEZ. 8 Clinical guidelines subsequently incorporated BEZ as a recommended option for patients with a recent CDI recurrence or other high-risk features, to be administered during CDI treatment to avert further episodes. 9
Despite its proven efficacy in controlled trials, real-world data on BEZ are limited and based largely on small, heterogeneous cohort studies. The extent to which the clinical benefits observed in trials translate to routine practice remains unclear as individuals with severe CDI, multiple prior recurrences, or recent FMT were underrepresented or excluded in trials. Additionally, BEZ’s high cost and the need for insurance approval limited its uptake in many healthcare settings, meaning that data on its outcomes outside of clinical trials have been limited. Real-world evidence is needed to confirm BEZ’s effectiveness in broader patient populations and to inform clinical decision-making regarding which patients derive the most benefit.
Given this context, we conducted a retrospective cohort study to evaluate the real-world effectiveness of BEZ in preventing rCDI at our institution. To further contextualize our findings, we also performed a meta-analysis of available real-world studies, pooling data on recurrence outcomes with BEZ versus standard care. This combined approach was designed to clarify BEZ’s impact on rCDI prevention outside of trial settings. Therefore, in this study, we also aimed to report recurrence rates stratified by prespecified risk factors among patients with rCDI treated with BEZ at the Mayo Clinic.
Methods
Mayo Clinic Study
All patients who received BEZ per CDI indication from 2017 to 2021 were identified. The study was deemed exempt from requiring patient consent by the Mayo Clinic Institutional Review Board. Patients who had previously authorized consent to use their records for research purposes were included. Data regarding BEZ use were collected using the Mayo Data Explorer (MDE), a Mayo Clinic-specific data retrieval tool for querying a clinical data repository maintained by the Unified Data Platform. A CDI episode was defined as the presence of diarrhea (⩾3 unformed stools in 24 h) with laboratory confirmation of Clostridioides difficile. Recurrent CDI was defined as symptom recurrence or a positive stool test for C. difficile toxins or toxigenic C. difficile within 8 weeks after the completion of treatment for the initial episode. Additional information, including risk factors (age ⩾65 years, immunosuppression, severe CDI, prior CDI, and the number of prior CDI episodes), use of concomitant SoC, and recurrence after BEZ administration were acquired through chart review.
Analysis
Data, including patient demographics, CDI diagnostics, number of CDI episodes, antibiotics received, and the number of prespecified risk factors, were analyzed. Recurrence was defined as a subsequent CDI episode within 8 weeks (56 days) of the previous episode.
Averages are reported as median with interquartile range (IQR). Statistical analyses were performed with JMP version 16.2 (SAS Institute Inc.). As applicable, data analysis included descriptive statistics and t-tests for normally distributed continuous variables. Multivariable logistic regression analysis was performed to adjust for baseline characteristics. A Kaplan–Meier survival curve and Cox model were constructed to generate hazard ratios (HR) and confidence intervals (CI). Statistical significance was set at two-tailed p < 0.05.
Meta-analysis
All procedures used in this meta-analysis were consistent with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 10 (Supplemental Table 1). The protocol was not registered.
Selection criteria
Studies considered in this meta-analysis were any retrospective or prospective studies of patients with rCDI who were treated with SoC antibiotics, including vancomycin, metronidazole, or fidaxomicin, followed by BEZ. We excluded studies that did not evaluate CDI resolution as an outcome.
Data sources and search strategy
The literature was searched by a medical librarian for the concepts of bezlotoxumab for C. difficile treatment. Search strategies were created using a combination of keywords and standardized index terms. Searches were run on August 28, 2024, in Ovid Cochrane Central Register of Controlled Trials, Ovid Embase, Ovid Medline (including epub ahead of print, in-process & other non-indexed citations), Scopus, and Web of Science Core Collection (Science Citation Index Expanded & Emerging Sources Citation Index). After limiting results to the English language from the year 2015 onward, with some animal studies removed based on the exclusion criteria justified below, a total of 1206 citations were retrieved. Deduplication was performed in Covidence, leaving 565 citations. Full search strategies are provided in the Supplemental Table 2.
Two authors (K.S. and P.B.) independently reviewed the titles and abstracts. The selection was based upon the following parameters: the study was an original article or abstract, included human subjects, was not a case or case series (<5 patients), and included patients with CDI treated with BEZ plus metronidazole, vancomycin, fidaxomicin, or fecal microbiota transplantation. Lastly, any article known to the authors satisfying the inclusion and exclusion criteria was included in the analysis. Only observational studies (retrospective and prospective designs) were included, as we aimed to evaluate the real-world effectiveness of bezlotoxumab. Randomized controlled trials (RCTs) were excluded to focus specifically on routine clinical use.
Data abstraction
Data were abstracted to a pre-determined collection form by two investigators (K.S. and P.B.). Data collected for each study included study title, year of publication, study population characteristics including age and number of previous CDI episodes, any treatment control population, number of patients who received BEZ, number of patients with alternative therapy, and CDI clinical resolution rates as well as resolution rates with alternative treatment administered.
Quality assessment of included studies
The critical appraisal of studies was assessed by two reviewers independently (K.S. and P.B.). The Joanna Briggs Institute (JBI) Critical Appraisal Checklist for cohort studies was utilized to evaluate the methodological quality of each included study. The two reviewers independently assessed each of the studies included. If there were disagreements, the two reviewers discussed them and resolved them by reaching a consensus. The JBI Critical Appraisal Checklist included 11 items, each needing an answer of Yes, No, Unclear, or Not Applicable. Studies were classified as high quality if ⩾9 of 11 criteria were met, medium if 8/11, and low quality if ⩽7 of 11 criteria were met (Supplemental Table 3 and Table 1).
Methodological quality assessment.

NA, not applicable; N, no; U, unclear; Y, yes.
Outcomes assessed
Our primary aim was to assess the clinical resolution of CDI after BEZ in a real-world clinical setting, including our data. In this study, clinical resolution was defined as no recurrence during the follow-up period.
Statistical analysis
The primary outcome of our pooled analysis was CDI clinical resolution rates with BEZ, that is, resolution of CDI with no recurrence in the follow-up period. The random-effects model described by DerSimonian and Laird was used to calculate weighted pooled resolution rates (WPR) with corresponding 95% confidence intervals (CI). 11 Data were weighted based on the sample size in each trial to calculate WPR. The Freeman–Tukey double arcsine transformation was used to avoid giving weight to studies with prevalence estimates that were either too large or too small. We assessed heterogeneity within groups with the inconsistency index (I2) statistic, which estimates the proportion of total variation across studies due to heterogeneity in study patients, design, or interventions, rather than chance. I2 values >50% suggest substantial heterogeneity. All p-values are two-tailed and considered statistically significant if <0.05. Publication bias was assessed by visual inspection of funnel plots and numerically using Egger’s test and Begg’s test (Supplemental Figures 1 and 2). Calculations were performed and graphs were constructed with RStudio (Version 2024.04.2; 2009–2024 Posit Software, PBC).
Results
Mayo Clinic Study
We included 47 patients treated with BEZ (Table 2), where 59.6% (28/47) were female with a median age of 62 years (range: 23–93 years). Of these, 16 patients had two of the prespecified risk factors, 19 patients had three, 4 patients had four, and 8 had at least one risk factor. The median number of prior CDI episodes in this population was 3 (IQR 2–4). SoC antibiotics received along with BEZ therapy included vancomycin alone in 25 patients (53.2%) and fidaxomicin alone in 17 patients (36.2%). Three patients (6.3%) were treated with vancomycin and metronidazole, and two (2.1%) with vancomycin and fidaxomicin. No patient received metronidazole alone. Additionally, one patient (2.1%) received fecal microbiota transplantation. Four patients who died (8.5%) in our cohort from non-CDI-related causes showed resolution of CDI within 8 weeks after BEZ.
Baseline demographics and clinical characteristics of Mayo Clinic cohort.

Retrospective data
In our data, 34/47 patients (72.3%) had CDI resolution, and the other 13 patients experienced recurrence (27.6%). No recurrence was observed in patients with only one risk factor (0/8, 0%). Among patients with two risk factors, 3 of 16 (18.8%) experienced recurrence, compared to 9 of 19 (47.4%) with three risk factors and 1 of 4 (25%) with four risk factors (Figure 1). A Likelihood Ratio Chi-square test demonstrated a statistically significant difference in recurrence rates across these risk strata (p = 0.0267), suggesting that the likelihood of recurrence increases with higher baseline risk burden.
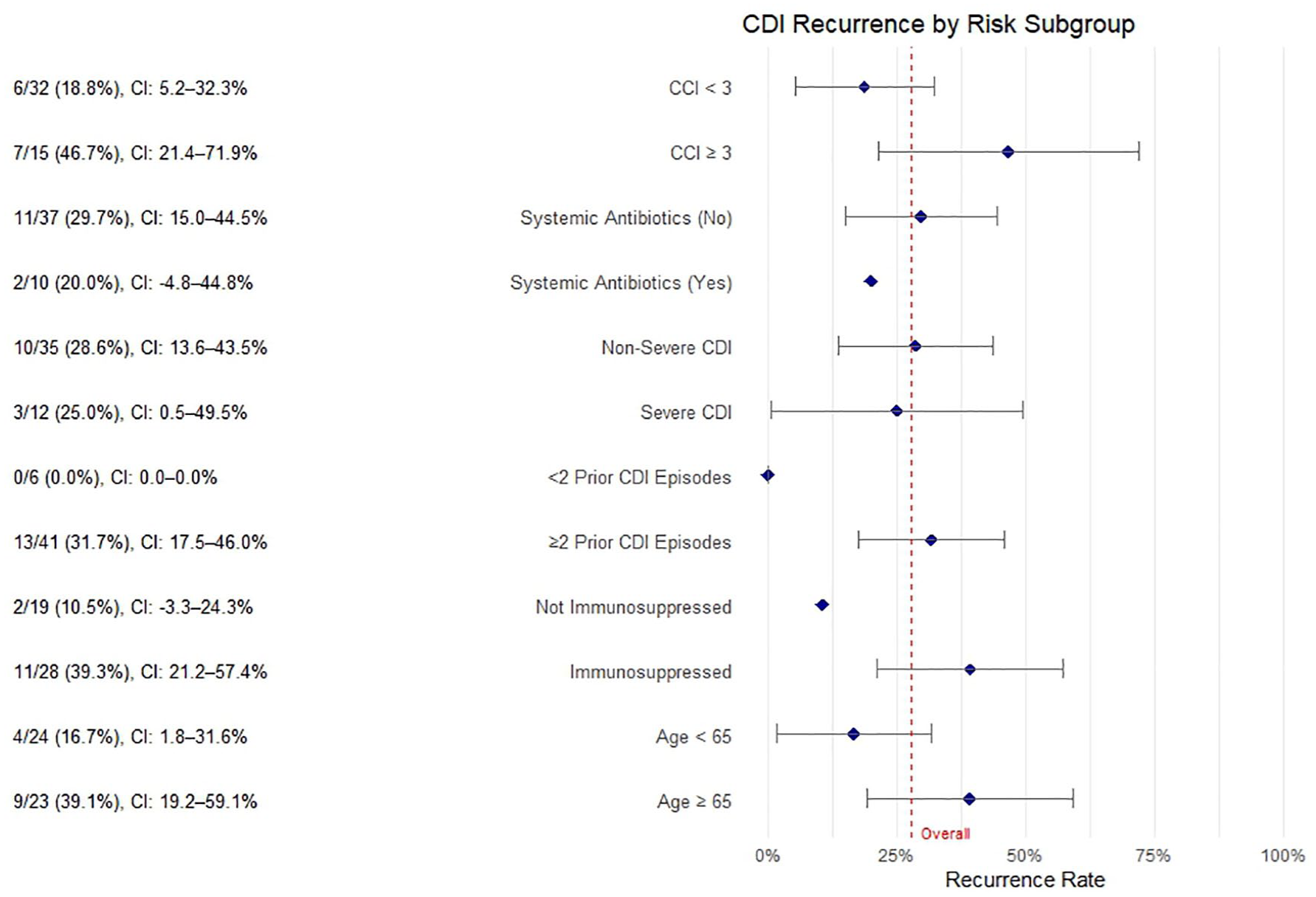
Clostridioides difficile recurrence rate in our cohort stratified by risk factors.
Among the prespecified risk factors, the history of prior CDI was the most common, observed in 40 of 47 patients (85.1%). This was followed by immunosuppressive therapy in 28 patients (59.6%), age ⩾65 years in 23 patients (49.0%), receipt of systemic antibiotics within 60 days of BEZ administration in 10 patients (21.3%), and severe CDI in 12 patients (25.5%). The median time to recurrence since BEZ administration was 95 days (IQR 52–216). Recurrence was observed in 28% (7/25) patients treated with BEZ following vancomycin, 29.4% (5/17) patients treated with BEZ following fidaxomicin, and one patient, 50% (1/2) treated with vancomycin and fidaxomicin both with BEZ.
In multivariable logistic regression with CDI recurrence as the outcome, age ⩾65 years (OR 9.27, 95% CI: 1.70–73.00, p = 0.018) and immunosuppressive therapy (OR 12.39, 95% CI: 2.11–123.07, p = 0.013) were independently associated with significantly increased odds of recurrence after BEZ administration. Although not statistically significant, prior CDI episodes (OR 7.48, p = 0.134) and severe CDI (OR 2.58, p = 0.318) showed trends toward higher recurrence risk. Systemic antibiotic use within 60 days after BEZ did not demonstrate a significant association with recurrence (OR 0.54, 95% CI: 0.06–3.47, p = 0.539). The model supports the role of known risk factors (Supplemental Table 4).
Meta-analyses
Search results
Through the search strategy, we obtained 1206 studies, of which 634 were duplicates and were removed. Titles and abstracts were screened from the remaining 565 studies. Of these, 383 were excluded, and 182 were included for full-text review. Eventually, 28 studies were included in the final analysis (Figure 2).

PRISMA flowchart: study selection process for meta-analysis of bezlotoxumab in reducing CDI recurrence.
Characteristics of all studies included in meta-analysis
The characteristics of the studies included are shown in Table 3. We included our data with the studies found through a literature search for analysis. Eighteen studies were conducted in the United States, one in Finland, five in Spain, one in Italy, one in Japan, and two did not specify the country. Recurrence with BEZ with SoC antibiotics was compared with SoC antibiotics (metronidazole, vancomycin, or fidaxomicin) alone in 10 studies and with fecal microbiota transplantation (FMT) with SoC antibiotics in 2 studies. The other 16 studies had no comparator arm. Recurrence was defined as a reappearance of diarrhea at a median of 84 days (interquartile range 70–90 days). All 28 included studies were observational in nature, with the majority being retrospective cohorts (n = 26) and a small number being prospective (n = 2).
Characteristics of included studies.

BEZ, bezlotoxumab; CDI, Clostridioides difficile infection; FMT, fecal microbiota transplantation; SOC, standard of care antibiotics.
Methodological quality of included studies
The quality of the studies included is shown in Table 1. Quality was high in seven studies, medium in 15, and low in 6.
Clostridioides difficile clinical resolution rates
The 28 included studies, along with our institutional data, comprised a total of 2639 patients with CDI. Of these, 1786 patients (including 47 patients from our cohort) received BEZ with SoC, 790 received SoC alone, and 63 received FMT with SoC antibiotics. Among patients treated with BEZ and SoC, 1450 achieved CDI resolution, yielding a WPR of 81.6% (95% CI 77.2%–85.6%; Figure 3). Significant heterogeneity was observed (I2 = 77.3%).

Forest plot: meta-analysis of CDI resolution rates with bezlotoxumab and standard-of-care therapy.
To explore sources of heterogeneity, several subgroup analyses were conducted. By study design, retrospective studies had a pooled resolution rate of 80.0% (95% CI: 77.0%–82.0%), while prospective studies showed a higher rate of 89.0% (95% CI: 83.0%–93.0%; Supplemental Figure 3). The difference was statistically significant in the fixed-effects model (χ² = 8.33, p = 0.004), but not in the random-effects model (χ² = 1.96, p = 0.16), suggesting study design may contribute to heterogeneity, but not conclusively.
In the sensitivity analysis by study quality, the WPR was 85.0% (95% CI: 82.0%–88.0%) for medium-quality studies, 80.0% (95% CI: 77.0%–83.0%) for high-quality studies, and 75.0% (95% CI: 70.0%–79.0%) for low-quality studies (Supplemental Figure 4). Again, subgroup differences were significant under the fixed-effects model (p < 0.001), but not under the random-effects model (p = 0.10).
In the subgroup analysis comparison of BEZ with SoC versus SoC alone, the WPR was 83.3% (95% CI 75.5%–91.1%) for BEZ with SoC, compared to 70.8% (95% CI 62.7%–78.8%) for SoC alone. The pooled relative risk ratio for recurrence was 0.56 (95% CI 0.35–0.88; p = 0.0119; Figure 4). When comparing BEZ with SoC to FMT with SoC, the relative risk was 0.69 (95% CI: 0.34–1.41; p = 0.31), indicating no significant difference.
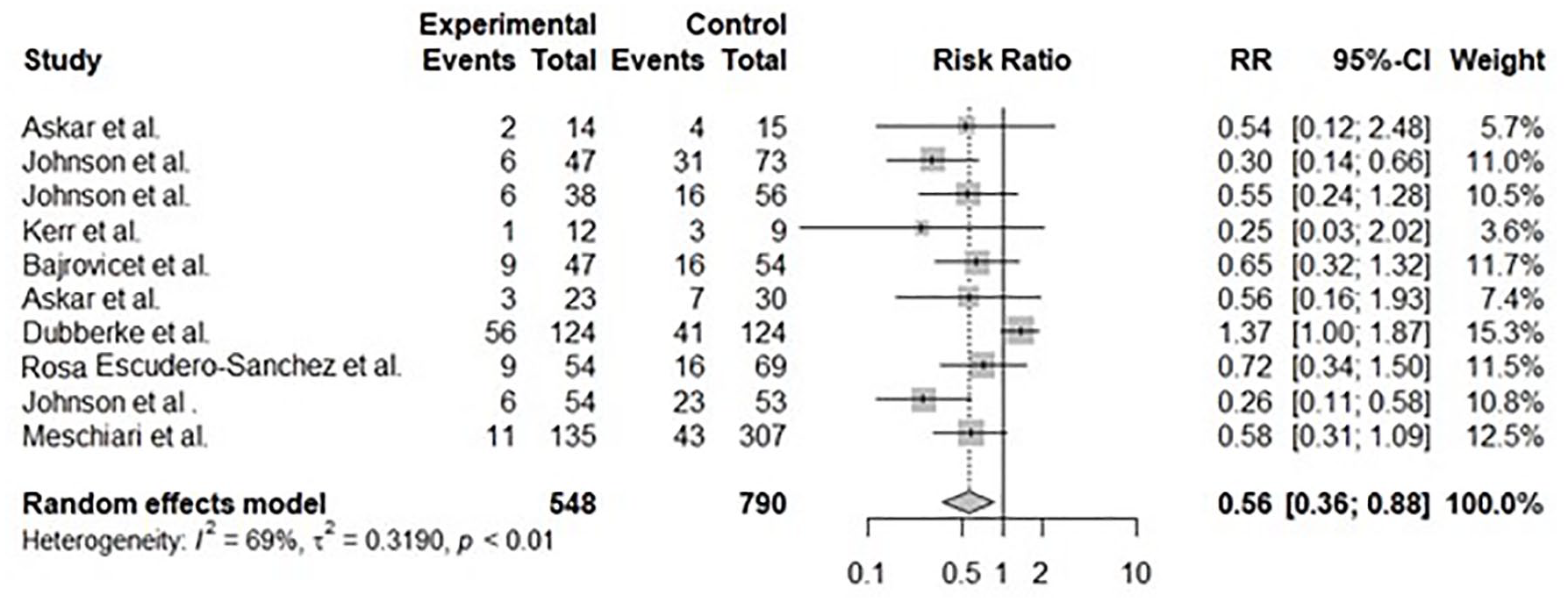
Forest plot: relative risk of CDI Recurrence with bezlotoxumab and standard-of-care therapy.
A subgroup analysis based on follow-up duration showed a WPR of 82.2% (95% CI: 79.0–85.3%) for studies with ⩽90 days of follow-up, and 78.0% (95% CI: 61.8%–89.1%) for studies with >90 days (Supplemental Figure 5). The difference was significant in the fixed-effects model (p = 0.04), but not under the random-effects model (p = 0.53), indicating a potential, but not definitive, impact of follow-up length on reported outcomes.
No significant funnel plot asymmetry was observed on Egger’s test for clinical resolution of CDI (z = −1.75, p = 0.0794) or on Begg’s test (Kendall’s tau = 0.1182, p = 0.3813), suggesting a low likelihood of substantial publication bias.
Discussion
Our study provides new evidence that BEZ meaningfully reduces recurrent C. difficile infection in real-world practice. This meta-analysis assesses BEZ’s real-world effectiveness, analyzing 28 studies and finding a high-resolution rate of 81.6%, higher than reported in clinical trials with a pooled 12-week sustained clinical cure rate of 64%. A systematic review by Mouhand et al. included 1472 patients receiving BEZ for rCDI prevention. Excluding data from MODIFY I and MODIFY II trials, they had a patient population of 691. 23 Our pooled relative risk of 0.56 agrees with the relative risk of 0.57 reported in their study. Our clinical resolution rate was also in agreement with the rCDI rate of 14.8% reported in a subgroup analysis of observational studies in their analysis and 15.8% in the overall analysis, including trial data and observational studies.
The patient population in our retrospective study was younger, more likely to be immunocompromised (59.6% vs 28%), and more likely to have severe CDI (25.5% vs 15.6%) compared to the trial population. The recurrence rate of 27.6% observed in our data is higher than the recurrence rates of 17% and 16% reported in MODIFY I and MODIFY II trials, respectively. The higher proportion of immunocompromised patients and individuals with severe CDI is likely to contribute to the higher recurrence rates in our data.
While our retrospective study lacks a comparator arm and cannot directly estimate clinical utility metrics, findings from the MODIFY trials offer meaningful guidance. Importantly, NNTs varied substantially based on patient risk strata: NNT = 48 in patients with no prespecified risk factors, NNT = 6 in ⩾1 risk factor, NNT = 7 in those with 1 or 2 risk factors, and NNT = 4 in those with ⩾3 risk factors. 8 This sharp gradient underscores the importance of targeting BEZ to high-risk populations, where its benefit is most clinically impactful.
Data comparing the efficacy of BEZ when administered with different established SoC antibiotics against each other are limited. Our analysis included two studies that examined BEZ alongside various SoC therapies.14,24 In the study by Escudero-Sanchez et al., the extended-pulse fidaxomicin regimen followed by BEZ had the highest resolution rate (100% from 4 patients), though this was not statistically significant (p = 0.40). Similarly, Hengel et al. 14 reported an 85% resolution rate when BEZ followed fidaxomicin (50/59), higher than rates after vancomycin or metronidazole, but also not statistically significant (p = 0.868). The lowest resolution rate was with metronidazole (67%, p = 0.405).
Limitations
Our study has several limitations. Firstly, all studies except the two that we included were retrospective. While our single-center retrospective cohort provides meaningful insights into the real-world use of BEZ in a high-risk population, we acknowledge the limitations related to sample size and lack of a control group. Secondly, heterogeneity in recurrence definitions and follow-up periods across studies may have influenced pooled estimates. Thirdly, many studies were published as abstracts, limiting available data for subgroup analysis. Additionally, the absence of detailed data on immunosuppressive types and concurrent medications limited our ability to explore their individual effects.
Finally, as a retrospective cohort, our study is inherently susceptible to bias, including confounding by indication, since BEZ was typically reserved for high-risk patients. We addressed this through multivariable logistic regression, which identified immunosuppressive therapy and prior CDI episodes as independent predictors of recurrence, underscoring their clinical relevance. However, limited data prevented us from assessing the impact of specific immunosuppressive agents or concurrent medications.
Our findings should be interpreted with caution, as unmeasured comorbidities—such as hepatic or renal impairment and inflammatory bowel disease—may have influenced outcomes. While patients receiving BEZ were likely at higher baseline risk, the observed reduction in recurrence suggests a meaningful benefit despite this potential bias. As BEZ is no longer commercially available, real-world analyses like ours remain valuable for informing future therapeutic strategies.
Conclusion
Our findings are in line with and expand upon the existing literature on BEZ. Previous randomized trials established proof of efficacy, and subsequent subgroup analyses from those trials indicated that patients with multiple risk factors derive substantial absolute benefit from BEZ. Our meta-analysis confirms that in a real-world setting, BEZ can achieve recurrence rates that mirror those seen in clinical trial participants. While recurrence rates were higher in our cohort, this reflects the complexity of patients typically considered for BEZ. Despite heterogeneity across studies in our meta-analysis, our findings support its role in targeted CDI management, especially in high-risk patients alongside standard-of-care antibiotics. As BEZ is no longer available, these data offer valuable insight into its past use and lessons for future therapies.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251346593 – Supplemental material for Real-world use of bezlotoxumab to prevent recurrent Clostridioides difficile infections: a single-center experience and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848251346593 for Real-world use of bezlotoxumab to prevent recurrent Clostridioides difficile infections: a single-center experience and meta-analysis by Kanika Sehgal, Parul Berry, Raseen Tariq, Darrell S. Pardi and Sahil Khanna in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
