Abstract
Background:
The relationship between Mediterranean diet (MedDiet) adherence and acute pancreatitis (AP) risk is largely unknown.
Objectives:
To investigate the associations between MedDiet adherence and AP risk and joint associations of genetic risk and MedDiet adherence with AP risk.
Design:
A prospective cohort study using data from UK Biobank, a large population-based prospective study that recruited over 500,000 participants aged 40–69 between 2006 and 2010 across the United Kingdom.
Methods:
We included 103,449 participants free of AP with typical dietary intake from 24-h dietary recalls. MedDiet adherence was measured via the Mediterranean Diet Adherence Screener (MEDAS) continuous score. Genetic predisposition to AP was estimated by polygenic risk score (PRS). Incident AP cases were identified via electronic medical records. Hazard ratios (HRs) with 95% confidence intervals (CIs) were estimated by Cox proportional hazards models. Mediation analyses were further applied to explore the mediating effects of the low-grade inflammation (INFLA) score and metabolic status.
Results:
Over a mean follow-up period of 10.4 years, 371 AP cases were documented. Higher MedDiet adherence defined by MEDAS continuous score was inversely associated with lower AP risk (highest vs lowest tertiles: HR 0.60, 95% CI 0.46–0.79, p < 0.001), with the INFLA score and metabolic status mediating 10% and 7.1% of the association, respectively. Although no interaction was observed between PRS and MedDiet adherence, participants with combined low genetic risk and the highest MedDiet adherence had the lowest risk of AP (HR 0.54, 95% CI 0.36–0.80, p = 0.002).
Conclusion:
The study suggests that higher adherence to the MedDiet is associated with a decreased risk of AP, which is partially mediated by inflammation and metabolic status, and may attenuate the deleterious impact of genetics on AP risk.
Introduction
Acute pancreatitis (AP) is an acute inflammatory disorder of the pancreas. 1 The global morbidity of AP is reported to be approximately 34 cases per 100,000 person-years, with evidence of a rising trend over time.2,3 Despite recent advances in therapy, the mortality rate of severe AP in the early phase has remained unchanged over the past few decades.4,5 This highlights the need to identify modifiable risk factors for the primary prevention of this disease.
Dietary factors play crucial roles in the prevention of AP. For example, some epidemiological evidence has demonstrated that high consumption of vegetables and fruits and moderate consumption of fish are associated with a reduced AP risk.6–8 In contrast, the high consumption of red and processed meat increases AP risk. 7 The causality involving fruits, red meat, and processed meat was further verified by a Mendelian randomization study. 9 However, focusing solely on individual food or nutrients often overlooks the complex interactions and synergies between them, and the overall diet pattern better reflects real-world practice. The Mediterranean diet (MedDiet) is characterized by high consumption of vegetables, cereals, fruits, legumes, and olive oil; moderate consumption of seafood, red wine, and nuts; and limited consumption of red meat, sweetened drinks, and sweets.10,11 Given the characteristics of the MedDiet, which overlaps with foods shown to have protective effects against AP, it is plausible that this dietary pattern could help prevent AP. However, evidence on the relationship between AP risk and the MedDiet is limited. Only a case‒control study with 50 AP patients and 50 controls indicated a decreased risk of AP among children with increased MedDiet adherence. 12 The MedDiet also reduced the incidence of common etiologies of AP, such as gallstones and hyperlipidemia,13–16 and the burden of inflammation.17–19 Additionally, precision nutrition on the basis of genetic background can offer personalized guidance for more effective AP prevention. 20 To date, several susceptibility loci associated with AP risk have been identified by recent genome-wide association studies (GWASs).21,22 Thus, exploring the joint effects and interactions of genetic risk and the MedDiet on AP risk is valuable.
In this study, we conducted a prospective cohort study using data from the UK Biobank, aiming to investigate the association between MedDiet adherence and the risk of AP. We also explored the potential mediating pathways and investigated the joint associations of genetic risk and MedDiet adherence with AP risk.
Methods
Study population
The UK Biobank, a large community-based prospective study, recruited over 500,000 participants aged 40–69 years between 2006 and 2010 across the United Kingdom. 23 Ethical approval for the UK Biobank was obtained from the North West-Haydock Research Ethics Committee (REC reference: 21/NW/0157). All participants provided informed consent at the time of recruitment. This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 24
In the present study, we excluded participants without at least two typical diet recalls (n = 398,449) to reduce irregular dietary reporting. We also excluded those with implausible energy intakes (<800 or >4200 kcal/day for men, <600 kcal/day or >3500 kcal/day for women; n = 135) 25 to reduce extreme dietary reporting errors. Participants with prevalent AP or chronic pancreatitis (n = 396) and those who developed AP within 1 year of follow-up (n = 32) were excluded to minimize the potential for reverse causality (Figure 1). The final analysis of MedDiet adherence and AP risk included 103,449 participants. Written informed consent was obtained from all participants prior to study initiation, with access to UK Biobank data approved by the UK Biobank Access Committee (Application number 79612). For analysis by genetic risk, we further excluded participants without genetic information (n = 16,614), leaving a sample of 86,835 individuals.

Flowchart of participant inclusion for this study.
Assessment of MedDiet adherence
Dietary intake was assessed via the Oxford Web-Q, an online 24-h dietary recall questionnaire that has been well validated in previous studies.26,27 The participants completed the 24-h dietary questionnaires between April 2009 and September 2010. They subsequently answered follow-up dietary questionnaires every 3–4 months from February 2011 to June 2012. 28 Repeated 24-h dietary questionnaires showed good test–retest reliability, and the mean dietary intake across assessments was used to reduce potential biases from daily dietary variations. 29 Specific dietary intake in grams was calculated by multiplying the quantity of consumed portions by the standard portion size in grams.25,30
MedDiet adherence was quantified via the Mediterranean Diet Adherence Screener (MEDAS) continuous score (Tables S1 and S2), which has been validated in the UK population. 28 The traditional MEDAS score is a 14-point score with a binary evaluation of food components including olive oil, vegetables, fruits, red meat, butter, margarine or cream, sweetened or carbonated drinks, wine (red and rose), legumes, seafood, sweets or pastries, nuts, white meat, and Sofrito (a fragrant Spanish cooking base). The participants were allocated one point if they met a dietary target and zero point if they did not. The MEDAS continuous score uses the same dietary targets but allocates points proportionally on the basis of the degree of adherence,10,28 thus increasing the sensitivity to detect differences in dietary quality. 31 Notably, the original MEDAS continuous score includes a food item requiring the consumption of olive oil ⩾ 4 tablespoons/day. Owing to the infeasibility of the amount of olive oil consumed, we assigned one point for consumption and zero point for nonconsumption, which has been well validated in previous research. 28 Consistent with a previous study, 32 the MEDAS continuous score was adjusted for total energy intake via the residual method.
Polygenetic risk score for AP
To capture the participants’ genetic load of AP risk, we used 50 independent genetic variants (r2 for linkage disequilibrium < 0.01) that were associated with AP (p < 5×10−5) from a genome-wide association meta-analysis of people of European ancestry
21
(Table S3). The polygenic risk score (PRS) for AP was established by summing the risk-increasing alleles for each single nucleotide genetic polymorphism (SNP) weighted by the effect size to AP (
Ascertainment of outcome
The diagnosis of AP was ascertained via the International Classification of Disease (ICD) Code, which was obtained through inpatient records (ICD-9: 577, ICD-10: K85) and death registries (ICD-10: K85) (Table S4). The primary care records obtained via Read codes were mapped to the ICD codes. The follow-up time of the participants was calculated from the date of the last completed dietary questionnaire to the occurrence of the first diagnosed case of AP, death, loss to follow-up, or end of follow-up (October 31, 2022 for England, August 31, 2022 for Scotland and 31 May 2022 for Wales), whichever occurred first.
Assessment of covariates and mediators
Covariates were selected based on prior knowledge,28,33 including age (continuous, in years), sex (male and female), ethnicity (white and others), education level (college and below college), smoking status (never, previous and current), drinking status (never, previous and current), total energy intake (continuous, in kcal/day), physical activity level (low, moderate, and high, by tertiles), body mass index (BMI, <25 kg/m2, 25–29.9 kg/m2, ⩾30 kg/m2), Townsend deprivation index (TDI, low, moderate, and high, by tertiles), Charlson comorbidity index (CCI, continuous), sleep duration (continuous, in hours/day), alcohol consumption (continuous, in g/week), hyperlipidemia (triglycerides ⩾1.7 mmol/L and < 1.7 mmol/L), baseline diabetes (with and without), and gallstones (with and without). Furthermore, metabolic status (healthy, unhealthy) and inflammation, represented by the low-grade inflammation score (INFLA score, continuous), were considered potential mediators. Table S5 provides more details on these covariates and mediators.
Statistical analyses
The baseline characteristics of the participants are presented by tertiles of the MEDAS continuous score (low, middle, and high). Descriptive statistics are presented as the means (standard deviations, SDs) for continuous variables and percentages for categorical variables. Group differences were assessed by ANOVA or the chi-square test. Missing values of covariates were imputed by using the mean for continuous variables and the mode for categorical variables. The “unknown” category was created for physical activity and metabolic status because of its high missing data rate (Table S6). Population attributable fractions (PAFs) were used to estimate the proportion of the AP cases attributable to gallstones (with and without), alcohol consumption (>100 g/week and ⩽100 g/week), and hyperlipidemia (triglycerides ⩾1.7 mmol/L and < 1.7 mmol/L) which are the most three common risk factors for AP (collectively accounting for over 70% AP cases). 34 The formula is PAF = (p(HR-1))/(p(HR-1) +1), where p is the prevalence of the risk factor and HR is the hazards ratio of the AP of the exposed over the non-exposed.
Associations between the MEDAS continuous score and AP risk were estimated via Cox proportional hazards models with estimation of HRs and 95% confidence intervals (CIs). The minimally adjusted model was adjusted for age, sex, and total energy intake. The fully adjusted model was further adjusted for ethnicity, education, TDI, smoking status, physical activity, and baseline gallstones. The proportional hazards assumption was tested via Schoenfeld residual methods (all p > 0.05). The lowest tertile of the MEDAS continuous score was treated as a reference. In addition, we assessed the impact of each food component of the MEDAS score on AP risk.
The associations between PRS and AP were estimated via the Cox proportional hazards regression model. The interaction between the MEDAS continuous score and PRS was subsequently examined by stratifying participants by PRS and using the multiplicative interaction model. We also estimated the joint associations of the MEDAS continuous score and genetic risk with AP.
Subgroup analyses were performed by stratifying participants by sex, age, education, smoking status, TDI, and physical activity to explore potential effect modifications. Mediation analyses were conducted to evaluate the mediating effects of the INFLA score and metabolic status on the association between the MEDAS continuous score and AP risk.
Several sensitivity analyses were conducted to verify the robustness of our findings. First, given that alcohol abuse is an established etiology for AP, 1 we repeated the analysis using the MEDAS continuous score by excluding wine consumption and further adjusting drinking status. Second, we used the MEDAS binary score and an alternative Mediterranean diet (aMed) score to assess MedDiet adherence.35,36 The details of the aMed scores are presented in Table S7. Third, we excluded participants who developed AP within 2, 3, or 4 years of follow-up to further minimize reverse causality. Fourth, we additionally adjusted the models for potential confounders, including BMI, CCI, sleep duration, and baseline diabetes. Fifth, to evaluate the impact of the imputation methods, we filled in missing values with multiple imputations. Sixth, propensity score matching (PSM) was further conducted to investigate the association of MEDAS continuous score with AP risk, to minimize potential confounding bias from group differences of baseline characteristic, especially from known risk factors of AP like gallstones, alcohol, and hyperlipidemia. The matching was constructed based on a 1:1 ratio of highest tertile of MEDAS continuous score with lowest tertile using the nearest neighbor method with a caliper width of 0.05 without replacement. The balance of variables between the groups before and after matching was assessed using standardized mean difference (SMD), with a value of less than 0.10 indicating balance.
Statistical analyses were performed via R 4.2.1. All the statistical tests were two-sided, and a p value < 0.05 was considered statistically significant.
Results
Baseline characteristics
Over a mean follow-up of 10.4 years, 371 cases of AP were documented (34 cases/100,000 person-years). The baseline characteristics of the study population stratified by the tertiles of the MEDAS continuous score are shown in Table 1. The mean age of the 103,449 participants was 59.72 years (7.83). Participants with higher adherence to the MedDiet were more likely to be older, female, highly educated, and demonstrated higher physical activity levels, lower BMI, lower prevalence of diabetes and gallstones, lower triglyceride levels, and higher alcohol consumption compared to those with lower adherence. The PAFs for AP cases demonstrated 4.0% (95% CI 1.3%–6.7%) for hyperlipidemia and 15.3% (95% CI 7%–23.6%) for gallstones. In contrast, alcohol consumption showed no contribution to the AP risk (PAF −6.8%, 95% CI −19.1% to 5.4%; Table S8).
Participant characteristics according to the tertiles of the Mediterranean Diet Adherence Screener continuous score (n = 103,449).

p Values were calculated via χ2 tests or analysis of variance.
INFLA, low-grade inflammation.
MedDiet adherence and incident AP
Higher adherence to the MedDiet defined by the MEDAS continuous score was associated with a lower risk of AP (per SD HR 0.81, 95% CI 0.73–0.91, p < 0.001; p-trend < 0.001) (Table 2 and Figure 2). Compared with participants in the lowest tertile of the MEDAS continuous score, those in the middle and highest tertiles had HRs of 0.80 (95% CI 0.63–1.01, p = 0.061) and 0.61 (95% CI 0.46–0.79, p < 0.001), respectively.
Association between the Mediterranean Diet Adherence Screener continuous score and the risk of acute pancreatitis (n = 103,449).

Model adjusted for age, sex, and total energy intake.
Model adjusted for age, sex, total energy intake, ethnicity, education, Townsend deprivation index, smoking status, physical activity, and gallstones.
CI, confidence interval; HR, hazard ratio; MEDAS, Mediterranean Diet Adherence Screener.
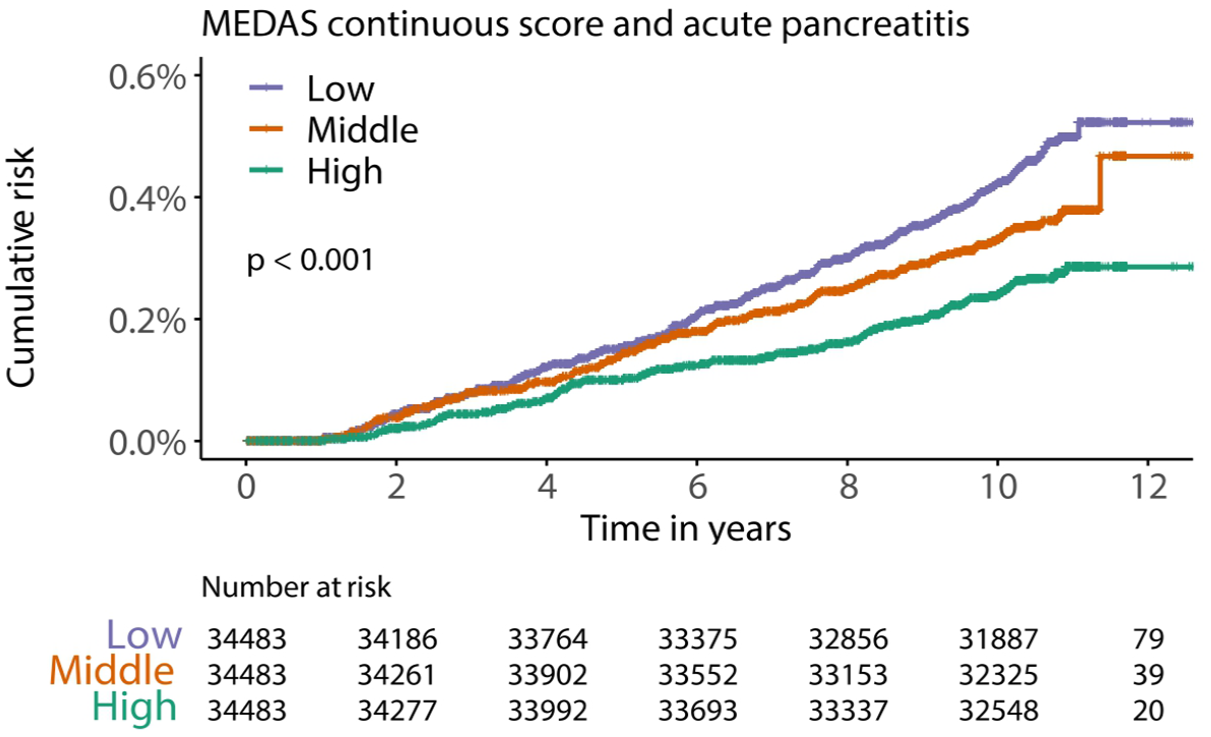
Association between the Mediterranean Diet Adherence Screener continuous score and the risk of AP using cumulative incidence curve.
Associations between each food component of the MEDAS score and the risk of AP are shown in Table S9. The participants who met the recommended intake of olive oil, wine, and sweetened or carbonated drinks had lower risks of AP, with HRs of 0.71 (95% CI 0.58–0.87, p < 0.001), 0.70 (95% CI 0.55–0.90, p = 0.005), and 0.68 (95% CI 0.55–0.84, p < 0.001), respectively.
MedDiet adherence, genetic risk, and incident AP
For genetic susceptibility, the PRS was significantly associated with AP risk (Figure S1 and Table S10). Compared with participants in the low genetic risk group, those in the high genetic risk group had a 31% increased risk of AP (HR 1.31, 95% CI 1.05–1.64, p = 0.018).
As shown in Table 3, the highest tertile of the MEDAS continuous score was associated with a reduced risk of AP compared with the lowest tertile in the high genetic risk stratum (HR 0.62, 95% CI 0.42–0.92; p = 0.016). However, no significant interaction was observed between PRS and MedDiet adherence (p-interaction = 0.709). When genetic risk and MedDiet adherence were combined, there was a monotonic association between decreasing genetic risk and increasing MedDiet adherence (Figure 3). Participants with both low genetic risk and the highest tertile of the MEDAS continuous score had the lowest risk of AP (HR 0.54, 95% CI 0.36–0.80; p = 0.002).
Subgroup analysis by genetic risk group.

Model adjusted for age, sex, total energy, education, Townsend deprivation index, smoking status, drinking status, physical activity, baseline gallstones, and first 20 principal components of ancestry.
CI, confidence interval; HR, hazard ratio.
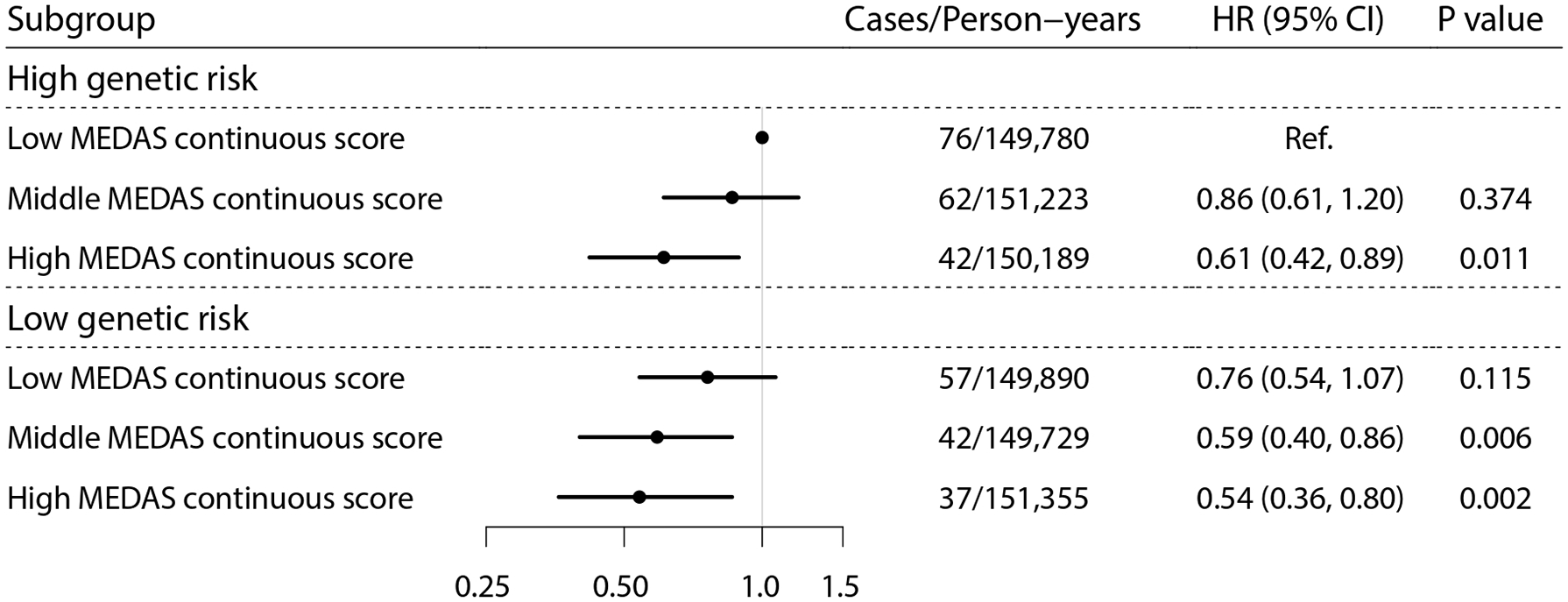
Joint associations between Mediterranean Diet adherence, genetic risk, and AP risk.
Subgroup and mediation analyses
In the subgroup analyses, the associations between the MEDAS continuous score and AP risk remained consistent, with no interactions across stratifications by sex, age, education, smoking status, TDI, or physical activity (all p-interactions > 0.05, Figure S2).
Mediation analyses revealed that 9.5% (95% CI 2.4%–21.9%) and 7.1% (95% CI 2.74%–18.0%) of the associations between the MEDAS continuous score and AP risk were mediated by the INFLA score and metabolic status, respectively (Figure 4 and Table S11).

The schematic diagram and mediation effects of the low-grade inflammation score (a) and metabolic status (b) on the relationship between the Mediterranean Diet Adherence Screener continuous score and risk of acute pancreatitis, and the y-axis represents the mediating effect, direct effect, and total effect. The mediation effects were individually examined in separate structural equation models.
Sensitivity analyses
The main results remained largely stable in the sensitivity analyses. Specifically, higher MedDiet adherence (highest vs lowest) was also associated with a lower risk of AP when excluding the wine component in the MEDAS continuous score (HR 0.65, 95% CI 0.50–0.84, p = 0.001), applying the MEDAS binary score (HR 0.66, 95% CI 0.51–0.86, p = 0.002), excluding participants with AP events that occurred within the first 2, 3, or 4 years of follow-up, further adjusted for BMI, CCI, sleep duration, and baseline diabetes, and replacing the missing values with multiple imputations (Tables S12–S14). Although the association was not statistically significant when using the aMed score (HR 0.80, 95% CI 0.62–1.03, p = 0.083), the results were consistent with the trends observed in other measures (Table S11). The PSM achieved satisfactory balance between highest tertile of MEDAS continuous score and the lowest tertile, with SMD < 0.10 for all baseline characteristic (Table S15). After PSM, the HR remained consistent with unmatched analysis (highest tertile vs lowest tertile: 0.71, 95% 0.54–0.93, p < 0.001), though slightly attenuated (Table S16).
Discussion
In this study, we found that higher adherence to the MedDiet was associated with a decreased risk of AP, which was partially mediated by inflammation and metabolic status. Individual components of the MedDiet, such as olive oil consumption, higher wine intake, and lower consumption of sweetened or carbonated drinks, appeared to drive the observed association. The inverse relationship between wine (red and rose) intake and AP risk contrasts with the known role of alcohol as an AP risk factor. Furthermore, the PAFs demonstrated that alcohol consumption made no contribution to AP risk. This paradox may be explained by the fact that the dominant sources of alcoholic beverages in the UK population—wine and beer—have previously been found unassociated with AP risk, 37 with their anti-inflammatory polyphenols potentially mitigate the harmful effects of alcohol.38,39 We observed no interaction effect between MedDiet adherence and genetics on AP risk, suggesting that MedDiet adherence affects AP risk independently of genetics. In the sensitivity analysis, most results were consistent, except when MedDiet adherence was measured by the aMed. Unlike MEDAS, aMed uses the median of study subjects as a cutoff. This approach may be influenced by “healthy-volunteer bias,” where individuals with healthier lifestyles, better education, and health are more likely to join the UK Biobank cohort.40,41 As a result, the effects of the MedDiet may be attenuated due to higher median dietary intake values.
Compared with previous studies
The clinical evidence regarding the association between MedDiet adherence and AP risk is rather limited. Only one study involving 50 children diagnosed with AP and their 50 healthy controls revealed that children with AP had lower adherence to the MedDiet than healthy controls did, 12 similar to our findings.
Some previous studies linking individual components of the MedDiet with AP risk also suggested the potential benefits of the MedDiet against incident AP. In a study involving 8019 participants, every two servings per day increase in vegetable consumption was associated with a 17% reduction in AP risk. 6 A multiethnic cohort study of 145,886 participants demonstrated that dietary fiber intake from fruits was inversely associated with AP risk, whereas red meat intake increased gallstone-related AP risk. 7 A two-sample Mendelian randomization study revealed similar effects of fruits and red meat on AP risk. 9 Furthermore, a cohort study from Sweden revealed that moderate fish consumption (1–3 servings/week) was associated with a decreased risk of non-gallstone-related AP. 8 Together, this evidence supports the potential benefits of the MedDiet on AP risk, and more studies are needed to clarify our findings.
Possible interpretations
Both inflammation and metabolic status were substantiated as possible mediators in the relationship between the MedDiet and incident AP in our analysis, which aligns with existing evidence. The MedDiet is rich in dietary fiber, which has been reported to enhance intestinal barrier function and reduce bacterial translocation.42,43 Furthermore, participants adhering to the MedDiet presented a significant increase in short-chain fatty acid levels, which are derived from dietary fiber fermentation.44–46 Short-chain fatty acids inhibit inflammatory signals, maintain intestinal barrier integrity, and promote the growth and diversity of the gut microbiota.47,48 In addition, plant foods and olive oil are rich in polyphenols that exhibit anti-inflammatory and antioxidant effects.49–51 Our analysis demonstrated that hypertriglyceridemia and gallstone disease accounted for 4.0% and 15.3% of AP risk, respectively. Furthermore, reduced triglyceride levels and gallstone incidence were observed in populations with higher MedDiet exposure at baseline. Hyperlipidemia is the third most common cause of AP. 52 Previous clinical evidence has also shown that adherence to MedDiet is associated with a reduction in triglyceride levels14–16; therefore, its lipid-lowering effect may be one of the potential mechanisms for preventing AP. Gallstones are another common etiology of AP. The dietary intake of foods rich in saturated fat and cholesterol, such as red meat and eggs, is linked to an increased risk of gallstones and consequently an elevated risk of biliary AP7,53. Higher adherence to the MedDiet is associated with a lower risk of gallstone diseases,13,54 suggesting that the protective effects of the MedDiet may be related to a decreased incidence of AP etiology.
Strengths and limitations
The major strengths of our study included its large sample size, long follow-up time, and highly reliable clinical data. However, the present study should be interpreted in light of several limitations. First, the majority of participants at baseline were predominantly middle-aged and older population, which may restrict the generalizability of findings to younger populations and other ethnicity. Notably, compared with younger populations, a disproportionate burden of AP is borne by middle-aged and older populations, manifested as increased prevalence, worsened clinical outcomes, and higher mortality risk,55–57 which underscores the imperative need for prevention measures for these vulnerable populations. Second, a subset of identified genetic variants of AP in our study has been replicated in other European-descent GWASs (e.g., rs75331444 in ABCG8, rs12688220 in NUP62CL),21,58 but their applicability to other geographical populations (e.g., African and Asian) remains undetermined (>78% of GWASs derive from European populations). 59 Third, the small number of incident AP cases may have limited the statistical power. However, the incidence rate observed in our analysis was comparable to that of the general UK population (56 cases/100,000 person-years), 60 which allows for the extrapolation of our results to the broader UK population. Fourth, measurement error and recall bias were inevitable in the dietary intake data extracted via 24-h dietary recall questionnaires. We have attempted to address this problem by including participants with at least two typical dietary assessments. Fifth, residual confounding and reverse causation, as with other observational studies, cannot be fully ruled out despite adjusting for potential confounders.
Conclusion
In this prospective cohort study, increased MedDiet adherence was inversely associated with AP risk, which was partially mediated by inflammation and metabolic status, and could counteract the harmful impact of genetic factors on the risk of AP. Further studies are warranted to validate our findings and elucidate the underlying mechanisms involved.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251346291 – Supplemental material for Mediterranean diet adherence and incident acute pancreatitis: a prospective cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848251346291 for Mediterranean diet adherence and incident acute pancreatitis: a prospective cohort study by Chunhua Zhou, Jiawei Geng, Zhipeng Wu, Lintao Dan, Hanyi Huang, Xixian Ruan, Jie Chen, Yao Zhang and Duowu Zou in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251346291 – Supplemental material for Mediterranean diet adherence and incident acute pancreatitis: a prospective cohort study
Supplemental material, sj-docx-2-tag-10.1177_17562848251346291 for Mediterranean diet adherence and incident acute pancreatitis: a prospective cohort study by Chunhua Zhou, Jiawei Geng, Zhipeng Wu, Lintao Dan, Hanyi Huang, Xixian Ruan, Jie Chen, Yao Zhang and Duowu Zou in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
This work was conducted via the UK Biobank Resource under application number 79612. We want to thank all the UK Biobank participants and the management team for their participation and assistance.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
