Abstract
Background:
Acute pancreatitis (AP) has a high incidence, and patients can develop recurrent acute pancreatitis (RAP) and chronic pancreatitis (CP) after AP.
Objectives:
We aimed to estimate the pooled incidence rates (IRs), cumulative incidences, and proportions of RAP and CP after AP.
Design:
A systematic review and meta-analysis of studies reporting the proportion of RAP and CP after AP.
Data sources and methods:
The systematic search was conducted in three (PubMed, EMBASE, and CENTRAL) databases on 19 December 2023. Articles reporting the proportion of RAP or CP in patients after the first and multiple episodes of AP were eligible. The random effects model was used to calculate the pooled IR with 95% confidence intervals (CIs). The I2 value assessed heterogeneity. The risk of bias assessment was conducted with the Joanna Briggs Institute Critical Appraisal Tool.
Results:
We included 119 articles in the quantitative synthesis and 29 in the IRs calculations. Our results showed that the IR of RAP in adult patients after AP was 5.26 per 100 person-years (CI: 3.99–6.94; I2 = 93%), while in children, it was 4.64 per 100 person-years (CI: 2.73–7.87; I2 = 88%). We also found that the IR of CP after AP was 1.4 per 100 person-years (CI: 0.9–2; I2 = 75%), while after RAP, it increased to 4.3 per 100 person-years (CI: 3.1–6.0; I2 = 76%). The risk of bias was moderate in the majority of the included studies.
Conclusion:
Our results showed that RAP affects many patients with AP. Compared to patients with the first AP episode, RAP leads to a threefold higher IR for developing CP.
Trial registration:
Our protocol was registered on PROSPERO (CRD42021283252).
Introduction
The incidence of acute pancreatitis (AP) and chronic pancreatitis (CP) is well known; AP ranges from 13 to 45 cases per 100,000 persons per year, while CP ranges from 5 to 12 cases per 100,000 persons per year. 1 AP is one of the most common gastrointestinal tract diseases. It requires hospital admissions and is associated with significant morbidity, mortality, and prolonged hospital stay. 2 CP is a severe condition that significantly impairs quality of life and reduces life expectancy, and is currently an incurable disease. 3 Moreover, patients with CP often experience pain, stigma, unemployment, and depression. 4
It is now widely accepted that AP, recurrent acute pancreatitis (RAP), and CP can be a disease continuum, with recurrences of AP leading to CP. 5 RAP is a clinical condition characterized by repeated episodes of AP. Its diagnosis can, therefore, only be made retrospectively after at least the second episode of AP and can represent an intermediate step between AP and CP according to the sentinel acute pancreatitis event model.6,7 Besides this, RAP increases morbidity and healthcare costs with each recurrent episode, and it is the most important risk factor for progression to CP.7,8
As the progression of AP is time-dependent, the recurrence rate and the progression rate to RAP and CP vary as a function of the length of follow-up time. In addition, the etiology and severity of the first AP episode also impact progression. 7
Therefore, this study aims to better understand AP progression into RAP and CP by investigating the incidence rate (IR), the cumulative incidence, the recurrence rate, and the progression rate based on the etiology and severity of the first AP episode.
Methods
In our systematic review and meta-analysis, we followed the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 Statement. 9 We registered our meta-analysis protocol on the International Prospective Register of Systematic Reviews (PROSPERO registration number CRD42021283252), and we fully adhered to it.
Search strategy
We systematically searched three major medical databases: MEDLINE (via PubMed), Cochrane Library (CENTRAL), and EMBASE on 19 December 2023, restricting our search to articles published after 1992, with the following search key: acute AND (chronic OR recurrent) AND pancreatitis.
Eligibility criteria
To identify all eligible studies, we used the condition–context–population (CoCoPop) framework. 10 We included all the studies reporting on (1) patients diagnosed with AP according to the Atlanta Classification 11 (in the presence of two of the following three criteria: abdominal pain consistent with the disease, serum amylase, or lipase more than three times the upper limit of normal, and characteristic findings on abdominal imaging); (2) the proportion of RAP or CP in patients after first or multiple episodes of AP. Our primary outcomes were the IRs of RAP and CP after the first AP and the IR of CP after RAP. Our secondary outcomes were the cumulative incidences, and the proportion of RAP and CP after the first AP and the proportion of CP after RAP. We used articles where consecutive patients with the first AP episode were included to calculate RAP. We used two types of articles to calculate CP, but we analyzed them separately: (1) articles with consecutive patients with first AP; (2) articles with consecutive patients with RAP. The use of ‘AP’ in the manuscript refers to patients with a first episode of AP in every case. There were no restrictions regarding minimum follow-up duration; the minimum study population were 10. We excluded conference abstracts, case reports, review articles, in vitro, and animal studies.
Study selection and data extraction
We followed the Cochrane Handbook recommendations for study selection and data extraction. 12 A reference management program (EndNote X9; Clarivate Analytics, Philadelphia, PA, USA) was used for article selection. As the Atlanta Classification was developed in 1992, 11 we limited our search to articles published after 1 January 1992. After removing duplicates, two independent authors (E-BG and DP) selected the articles by title and abstract and then by full text. Cohen’s Kappa coefficient 13 was calculated to assess the agreement rate after each selection step. Disagreements were resolved by a third reviewer (BT).
Two independent authors (E-BG and DP) extracted the data into a standardized Excel spreadsheet (Office 365; Microsoft, Redmond, WA, USA). A third author (BT) resolved any disagreements. The following data were collected: first author, year of publication, study design, study period, study location, number of centers included, sample characteristics (sample sizes, age, and percentage of participating males), mean follow-up time, and the proportion of RAP and CP (overall cases, based on etiology and severity). After data extraction, we included articles with higher sample sizes in the analysis in case of overlapping populations. We contacted the authors in case of missing data.
Risk of bias
Two independent authors (E-BG and DP) assessed the methodological quality of included studies using the Joanna Briggs Institute Prevalence Critical Appraisal Tool. 14 A third author (BT) resolved any disagreements. The studies were evaluated based on nine criteria (I, sample frame; II, sampling; III, sample size; see details of criteria in the Supplemental Material), and each criterion was rated as ‘yes’, ‘no’, ‘unclear’, or ‘not applicable’.
Data synthesis
All statistical analyses were made with R (R Core Team 2021, v4.1.1) 15 using the meta 16 and dmetar 17 packages. The results were graphically summarized using forest plots for outcomes with at least three articles.
The IR with a 95% confidence interval (CI) was used for the effect size measure. Usually the IR is calculated from the number of new cases during an observed time period, using individual follow-up data, however these were not reported in the articles, therefore the IRs were calculated using the total number of patients, the number of patients with the event of interest, and the mean follow-up time instead of individual follow-up times. As considerable between-study heterogeneity was anticipated, a random effects model was used for data synthesis. The Higgins & Thompson’s I2 statistics were used to describe the between-study heterogeneity. 18 Funnel plots and Egger’s tests were used to assess publication bias 19 in cases with at least 10 articles per outcome. Leave-one-out analysis was used to evaluate whether a single study could have a marked impact on overall heterogeneity and IR in cases with at least eight articles per outcome. We followed the recommendations of Inthout et al. 20 by reporting the prediction intervals where it was applicable. We used the meta-regression of the random effects model to identify whether the age, sex, and severity had a confounding effect on the pooled IR in cases with at least 10 articles per outcome. To provide a more straightforward interpretation of our results, we calculated the estimated 5-year cumulative incidences 21 in Figure 1. The calculations were made using the formula by Rothman et al. 21 (CI = 1 − e(−IR×T), where ‘e’ = 2.71828; e, Euler number; IR, incidence rate; T, 5 years). Moreover, to provide a fully comprehensive picture of the progression of AP into RAP and CP, we also calculated the recurrence and progression rates of AP to CP (Figure 1) using proportional meta-analysis calculations (Supplemental Figures S1–S4). Proportion with a 95% CI was used for the effect size measure. The total number of patients and those with the event of interest were extracted from each study to calculate the proportion.

Summary forest plot showing all the IR and proportion results regarding AP recurrence and progression into CP. Each row represents a separate forest plot. The numbers in the cumulative incidence column were calculated from the incidence rate results (see Methods).
Results
Search and selection
Our systematic search resulted in a total of 18,483 records. After the selection by title, abstract, and full-text, 119 were eligible for qualitative synthesis and quantitative synthesis. Of these, 119 studies were used for proportion calculations, and 292,22–49 studies detailed the mean follow-up time that could be used for IR calculations. Details of the selection process are shown in Figure 2. Study characteristics and patient baseline characteristics for the studies included in the IR calculations are summarized in Tables 1 and 2.2,22–49
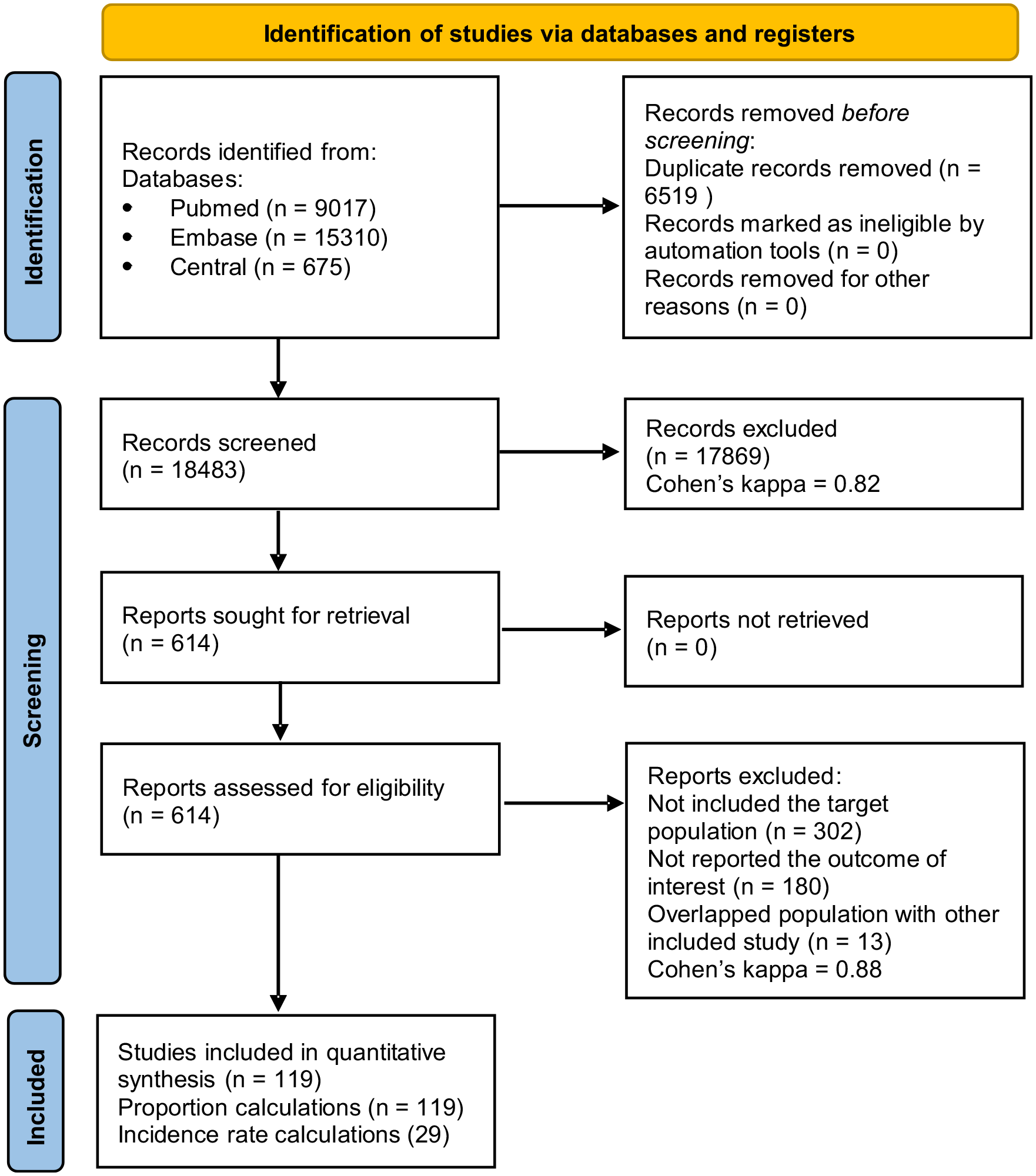
PRISMA flowchart of the included studies in the meta-analysis.
Characteristics of included studies.

AP, acute pancreatitis; CP, chronic pancreatitis; N, number; NR, not reported; RAP, recurrent acute pancreatitis; USA, United States.
Baseline characteristics of included patients.

AP, acute pancreatitis; HTG, hypertriglyceridemia; i, interquartile range; m, median; N, number; NR, not reported; r, range; ±SD, standard deviation.
Overall IRs of RAP and CP
First, we looked at the overall IR of RAP and CP in AP patients. We found that IRs of RAP after first AP were 5.26 per 100 person-years (95% CI 3.99–6.94; I2: 93%) in adults compared to children with 4.64 per 100 person-years (95% CI 2.73–7.87; I2: 88%), however this difference was not statistically significant (p = 0.671) [Figure 3 (1)]. As expected, the overall progression rate to CP in adults was threefold higher after RAP with 4.31 per 100 person-years (95% CI 3.10–5.99; I2: 76%) compared to CP after the first AP with 1.38 per 100 person-years (95% CI 0.97–1.96; I2: 75%) [Figure 3 (2)]. Further details of the results can be seen in Figure 3.

Forest plots showing: (1) the IRs of RAP in adults and children after an episode of AP; (2) the IRs of CP after AP and RAP in adults; (3) the IRs of RAP in adults by etiology after an episode of acute pancreatitis; (4) the IRs of CP in adults by etiology; (5) the IRs of RAP in adults by severity.
IRs of RAP and CP in different etiologies
To have a deeper insight into AP progression, we calculated RAP and CP IRs by etiology and severity. The IRs of RAP, based on the etiology [Figure 3 (3)] of the first AP episode, were as follows in descending order: 8.58 per 100 person-years in HTG-induced AP (95% CI 6.86–10.72; I2: 16%); 6.34 per 100 person-years in alcohol-induced AP (95% CI 4.80–8.37; I2: 79%); 4.86 per 100 person-years in idiopathic AP (95% CI 4.19–5.64; I2: 33%); and 3.03 per 100 person-years in biliary AP (95% CI 2.40–3.81; I2: 77%).
The IRs of CP by etiology [Figure 3 (4)] were as follows in descending order: 2.66 per 100 person-years in alcohol-induced AP (95% CI 1.58–4.48; I2: 66%); 1.10 per 100 person-years in idiopathic AP (95% CI 0.80–1.51; I2: 0%); and 0.33 per 100 person-years in biliary AP (95% CI 0.13–0.80; I2: 81%).
IRs of RAP in different severities of AP
Based on the severity [Figure 3 (5)] of the first AP episode, the IRs of RAP changed as follows: 7.56 per 100 person-years in moderate AP (95% CI 4.63–12.34; I2: 88%); 4.48 per 100 person-years in mild AP (95% CI 3.13–6.43; I2: 90%); and 4.90 per 100 person-years in severe AP (95% CI 3.66–6.55; I2: 0%). The IR of CP could not be analyzed by severity because there was insufficient data.
Proportions calculations
The proportion calculations were made on 119 articles; details can be seen in the Supplemental Material (Supplemental Figures S1–S4) and their summary in Figure 1. Here we show the results of the same outcomes as the IR calculations. The overall (only includes articles with consecutive AP patients with all etiologies) recurrence rate of AP was 20% in the adult and 23% in the pediatric populations (p = 0.227). The same adult–pediatric comparisons based on etiology were the following: idiopathic 21% and 28% (p = 0.125); biliary 8% and 15% (p = 0.055). The overall progression rate into CP was 8% after AP and 24% after RAP. The recurrence rates of AP in adults based on etiology were as follows: 28% in hypertriglyceridemia-induced AP; 24% in alcohol-induced AP; 21% in idiopathic AP; and 8% in biliary AP. The progression rates into CP in adults based on etiology were as follows: 18% in alcohol-induced AP; 7% in idiopathic AP; and 2% in biliary AP. The recurrence rates of AP in adults based on the severity of the first AP episode were as follows: 21% in moderate AP; 20% in mild AP; and 13% in severe AP.
Risk of bias assessment
The overall risk of bias for each outcome was moderate among the included studies. Most of the studies were downgraded because of an inadequate follow-up evaluation of all patients and sample sizes. The summary of the risk of bias assessment is shown in Supplemental Tables S2–S15.
Investigating heterogeneity
Several methods were used to investigate heterogeneity. We performed three meta-regression analyses for the overall IR of RAP in adults, considering as moderators the mean age, proportions of severe AP patients, and proportions of male patients, but these did not significantly influence the observed differences in the IRs of RAP (Supplemental Figure S5). We also performed leave-one-out influential analyses for the IR of RAP in adults and for alcoholic and biliary etiologies (Supplemental Figure S5), but they reduced heterogeneity only marginally. We subgrouped the articles into different blocks according to etiology and severity and whether they included an adult or pediatric population. We did not pool articles containing AP patients with one etiology with articles containing patients with all types of etiologies. Additionally, we investigated several potential factors (see Figure 4) that may have a role in increasing the heterogeneity, and we found that alcohol consumption [6.87 in ‘alcohol consumption – yes’ group versus (versus) 4.22 in ‘alcohol consumption – no’ group, p < 0.01], smoking (6.76 in ‘smoking – yes’ group versus 4.23 in ‘smoking – no’ group, p = 0.02), the absence of cholecystectomy (1.67 in ‘cholecystectomy – yes’ group versus 3.92 in ‘cholecystectomy – no’ group, p = 0.038), sample size <500 (7.48 in ‘sample size <500’ group versus 4.17 in ‘sample size >500’ group, p = 0.01) were associated with significantly higher IRs of RAP (see Figure 4). On the other hand, the study type (5.07 in ‘Retrospective cohort’ group versus 5.55 in ‘Prospective cohort’ group, p = 0.73), geographical area (4.98 in ‘Europe’ group versus 6.20 in ‘Asia’ group, p = 0.46), number of centers (5.93 in ‘Unicentric’ group versus 3.71 in ‘Multicentric’ group, p = 0.09) did not significantly increase the heterogeneity. The differences in CP definitions also increased the heterogeneity regarding IRs of CP after AP (1.75 in ‘M-ANNHEIM diagnostic criteria’ group versus 1.06 in ‘Other diagnostic criteria’ group, p = 0.03). These analyses were made in the adult population with articles containing all etiologies of AP except the cholecystectomy analysis, where we used only biliary etiology AP patients. The results were presented as events/100 person-years (all details of the results can be seen in Figure 4).

Forest plots showing the association between several factors and the IRs of (RAP or CP) after AP. Investigated factors: (1) alcohol consumption; (2) study type; (3) number of centers; (4) cholecystectomy; (5) geographical area; (6) CP definition; (7) smoking; (8) sample size.
Publication bias
Publication bias could be assessed for three outcomes (Supplemental Figure S5). The Egger’s test p value was greater than 0.01 in all three cases (Supplemental Figure S5) suggesting that there was no statistically significant evidence of publication bias. Though the Egger’s test found no evidence of publication bias in small studies, it’s important to note an observed asymmetry in effect sizes in the middle range of the funnel plot for the IR of RAP in adults with alcoholic AP, as shown in Supplemental Figure S5, subgraph 8.
Discussion
This study is the first systematic review and meta-analysis assessing the IR of RAP and CP after AP. As we know, the transition rate of AP to RAP and CP is time-dependent. By using IRs in this meta-analysis, we overcame the differences due to the variation of follow-up times to obtain the most accurate estimate of the transition of AP into RAP and CP as a function of time. For the purpose of comprehensiveness, we also assessed the proportions of AP progression into RAP and CP without taking into account the time factor.
Our findings showed that the overall RAP IR after AP in the adult population does not significantly differ compared to the pediatric population. In the adult population, we found a threefold higher IR of CP after RAP compared to CP after AP (4.31 per 100 person-years versus 1.38 per 100 person-years; p = 0.000). The higher IR of CP following RAP may be attributed to a combination of pathophysiological, genetic, and environmental factors. It is hypothesized that repeated episodes of RAP lead to ongoing inflammation and subsequent fibrosis, progressively damaging pancreatic tissue and increasing the risk of irreversible transition to CP. 50 It was previously reported that the presence of PRSS1 genetic mutation was associated with a faster progression to CP. 51 As we also showed in our study, environmental factors such as alcohol consumption and smoking increase the likelihood of RAP, therefore accelerating the transition to CP. 52
We found that HTG-induced AP had the highest recurrence rate, similar to data in the literature. 53 This high recurrence rate may be related to the fact that HTG-induced AP is often associated with multiple risk factors like metabolic diseases, obesity, and alcohol intake. Also, poor lipid control and follow-up after discharge can explain the bad prognosis. 53
In our study, alcohol-induced AP had the second-highest recurrence rate and the highest progression rate into CP, and these rates are consistent with the literature where it was described that these patients tend to continue drinking against medical advice.35,54 This highlights the importance of using alcohol cessation programs and psychological interventions in these patients.55,56
The high recurrence and progression rate in idiopathic acute pancreatitis (IAP) can be explained partly by the underdiagnosis of its underlying cause. Several studies examined the underlying causes of IAP.38,57 In one systematic review, 13 studies with idiopathic AP cases were analyzed. Results showed that endoscopic ultrasonography found more diagnostic information at 61% of patients with IAP, of whom 41% had biliary tract disease. 57 Use of appropriate treatment such as cholecystectomy for biliary cause may reduce recurrence and progression in these cases.
The lowest IR of RAP and CP was observed in the case of biliary etiology. In this population, most patients received cholecystectomy or endoscopic sphincterotomy after the first episode. The presence of cholecystectomy was associated with lower IR in our study. This lower IR indicates that treatment modalities depending on the healthcare system achieved better long-term results than treatments where patients should change their lifestyle to prevent RAP.
As for severity, in most of the included articles, the definitions for AP severity were consistent with the definitions in the Revised Atlanta Classification: severe (persistent organ failure), moderately severe (transient organ failure or local or systemic complications), mild (absence of organ failure, absence of local and systemic complications). 58 In our study we found that moderately severe AP was associated with the highest recurrence rate, followed by mild and severe AP. Our result confirms the findings of a previous meta-analysis by Sankaran et al. 7 They found that the severity of the first episode of AP was not necessarily a determinant of progression. 7 However, another study by Bertilsson et al. 24 found that severe first AP was associated with a higher recurrence rate and progression rate to CP. Our findings can be partly explained by the high mortality rate in severe cases and the high degree of irreversible destruction of pancreatic tissue caused by severe AP, leading to CP directly.
Strengths
Considering the strengths of our meta-analysis, we followed a rigorous methodology. Our results represent the best currently available estimate of the IRs using the literature. We studied the progression of AP into RAP and CP in detail based on etiology and severity, which can help clinicians in risk stratification for the progression of pancreatitis. Using our results, we have enabled the comparison of the incidence of CP after AP with the incidence of CP in the general population for the first time. Additionally, we provided a comprehensive analysis assessing 119 articles reporting the recurrence rate and progression rate of AP, and these results supported the findings of our IR calculations. Here we could analyze more outcomes based on etiology or geographical area.
Limitations
Our study has several limitations. Firstly, the majority of the included studies were retrospective. Secondly, we detected high heterogeneity between the included studies. This can be partially explained by the identified influential factors such as the proportion of drinkers, smokers, cholecystectomy as these were different across the studies. The different sample sizes of the studies and the different definitions of CP also increased the heterogeneity. Thirdly, we could only analyze the presence of RAP but could not explore in detail the number of RAP episodes after AP and their effect on progression, as these were not reported in the studies.
Implications for research and practice
Our study highlights the need for studies with better follow-up strategies, where patients are followed up more closely, for more extended periods, and where individual follow-up times are reported, not just average follow-up times. We also emphasize the need to develop new and better interventions, and, importantly, we highlight the better use of existing ones like brief and repeated psychological interventions, alcohol and smoking cessation programs, and deeper investigation of the underlying causes of the etiology of AP to reduce recurrence and progression rates. Additionally, better patient education and evidence-based patient care are crucial in preventing RAP and CP.
Conclusion
The IR of RAP is not significantly different between adult and pediatric patients after their first episode of AP. In adults, the IR of RAP is highest in cases induced by hypertriglyceridemia and alcohol, followed by idiopathic and biliary AP, while the IR of CP after the first AP episode is highest in alcoholic AP, followed by idiopathic and biliary AP. Compared to patients with the first AP episode, RAP leads to a threefold higher IR of developing CP.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241255303 – Supplemental material for Incidence of recurrent and chronic pancreatitis after acute pancreatitis: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848241255303 for Incidence of recurrent and chronic pancreatitis after acute pancreatitis: a systematic review and meta-analysis by Endre-Botond Gagyi, Brigitta Teutsch, Dániel Sándor Veres, Dániel Pálinkás, Nóra Vörhendi, Klementina Ocskay, Katalin Márta, Péter Jenő Hegyi, Péter Hegyi and Bálint Erőss in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
