Abstract
Background:
Recurrent Clostridioides difficile infection (rCDI) occurs in up to 35% of patients with CDI, and further recurrence is common. Fecal microbiota, live-jslm (RBL) is safe and effective for preventing rCDI in adults following antibiotic treatment for rCDI when administered rectally. There is clinical interest in alternative routes of administration.
Objectives:
CDI-SCOPE assessed the safety and clinical effectiveness of RBL when administered via colonoscopy to adults with rCDI.
Design:
Single-arm, exploratory phase IIIb trial conducted at 12 sites in the United States.
Methods:
Eligible adults with rCDI received one administration of RBL consisting of one 150-mL dose of RBL, delivered via colonoscopy to the right side of the colon. The primary endpoint assessed RBL-related treatment-emergent adverse events (TEAEs) within 8 weeks of RBL administration or until confirmed treatment failure. Secondary endpoints included treatment success (absence of CDI recurrence for 8 weeks following RBL administration), physician experience administering RBL via colonoscopy, and physician perception of participant benefit.
Results:
Of 54 participants screened, 41 were enrolled and received RBL via colonoscopy; 39 participants completed the 8-week visit. Five TEAEs in four participants (9.8%) were assessed as related to RBL, all of which were gastrointestinal and mild in severity. Overall, 18 participants (43.9%) experienced 33 TEAEs within 8 weeks, most of which were of mild (25/33; 75.8%) or moderate (5/33; 15.2%) severity. No TEAEs led to intensive care unit admission or death. Overall, 39 participants (95.1%) experienced treatment success; 2 participants (4.9%) withdrew consent and had an indeterminate outcome. Among physicians, 90.2% of investigators indicated a “positive” or “very positive” experience administering RBL by colonoscopy. All physicians assessed participant benefit as “much” or “very much” improved.
Conclusion:
This single-arm study suggests RBL administered via colonoscopy is practical, safe, and effective for preventing CDI recurrence following antibiotic treatment in adults.
Trial registration:
ClinicalTrials.gov: NCT05831189.
Plain language summary
Some patients who have had Clostridioides difficile infection (CDI) keep being reinfected. Fecal microbiota, live-jslm (REBYOTA®, abbreviated here as RBL) is a microbiome-based product that prevents another CDI reinfection after antibiotic treatment. RBL is given via the rectum but can also be given during a colonoscopy. The CDI-SCOPE trial looked at how safe RBL was and how well it prevented more CDI reinfections. Forty-one adults who had CDI more than once were given one dose of RBL during a colonoscopy. Investigators then monitored participants for side effects or CDI reinfections. After 8 weeks, four people (9.8%) had mild gastrointestinal side effects considered related to RBL, and none had confirmed CDI reinfections. The trial showed that RBL is safe and effective at preventing CDI reinfection in adults when given during colonoscopy.
Introduction
The leading cause of antibiotic- and healthcare-associated diarrhea in the United States is Clostridioides difficile infection (CDI).1,2 With a crude overall incidence rate of 116.1 cases per 100,000 persons in the United States, CDI has been identified as an urgent public health threat by the US Centers for Disease Control and Prevention.3 –5 Of patients who develop CDI, up to 35% will have a recurrent C. difficile infection (rCDI) and, of these, up to 60% will have more than one subsequent recurrence. 3 The largest risk factors for rCDI include advanced age (⩾65 years), antibiotic use, underlying comorbidities, and recent hospital admission.6 –9
CDI pathogenesis involves dysbiosis of the gut microbiota, leading to colonization and proliferation of toxin-producing C. difficile, resulting in the symptoms of CDI. 10 The standard-of-care (SOC) treatments for rCDI are antimicrobials, including vancomycin and fidaxomicin, which can exacerbate dysbiosis associated with rCDI.11 –13 Administration of fecal microbiota-based therapies derived from healthy donors after completion of SOC antimicrobials has been shown to decrease rCDI frequency through diversifying the gut microbiome and correcting the underlying problematic dysbiosis. 14 The use of fecal microbiota-based therapies to manage rCDI is supported by 2024 American Gastroenterological Association clinical guidelines. 15
Fecal microbiota, live-jslm (REBYOTA®, abbreviated here as RBL; previously known as RBX2660) was approved in November 2022 by the US Food and Drug Administration (FDA) as the first single-dose, broad consortium, microbiota-based product indicated for rCDI prevention in adults following SOC antibiotic treatment for rCDI.16,17 The safety and efficacy of RBL in preventing rCDI have been evaluated in five prospective clinical trials, including two completed placebo-controlled trials (phase IIb PUNCH CD2 and phase III PUNCH CD3) and three completed open-label trials (phase II PUNCH CD, phase II PUNCH open-label, and phase III PUNCH CD3-OLS), as well as one retrospective study.6,11,18 –22 The efficacy of RBL to prevent rCDI following SOC antibiotics was demonstrated using a Bayesian analysis in the PUNCH CD3 phase III trial, in which 70.6% of participants administered RBL experienced treatment success (no CDI recurrence at 8 weeks) compared with 57.5% in the placebo group, with a posterior probability of superiority of 0.991. 22 Results were consistent in a subsequent open-label phase III study, PUNCH CD3-OLS, with a treatment success rate of 73.8% in nearly 700 participants. 6 RBL has a favorable safety profile, with most observed treatment-emergent adverse events (TEAEs) assessed as mild or moderate in severity and no potentially life-threatening TEAEs assessed as related to RBL.22,23 Collectively, these studies provide comprehensive evidence supporting the safety and efficacy of RBL.
While clinical data demonstrate that rectal administration of RBL is safe and efficacious, there is clinical interest in alternative methods of delivery. Administration via colonoscopy allows for targeted delivery of microbiota therapy while allowing assessment of the colonic mucosa that may be particularly important in patients with comorbidities (e.g., inflammatory bowel disease). Some practitioners may prefer administration via colonoscopy over rectal administration due to integration opportunities with routine gastroenterology clinical practice workflows. Additionally, both providers and patients may benefit from the efficiency of having only one procedure for administration rather than requiring multiple visits for management. The therapeutic potential of RBL administration via colonoscopy was observed in a retrospective analysis in which a small number of participants (n = 10) received RBL by this route under FDA enforcement discretion. 24 Among these participants, no new safety signals for RBL were identified, and 80% had treatment success. The CDI-SCOPE trial (NCT05831189) was conducted to further assess the safety and clinical effectiveness of RBL when administered to adults with rCDI via colonoscopy. Here, 8-week outcomes from participants in CDI-SCOPE are reported.
Methods
Trial design and conduct
CDI-SCOPE was a single-arm, exploratory phase IIIb trial conducted at 12 sites in the United States that assessed the safety and clinical effectiveness, and explored the real-world feasibility of administering RBL by colonoscopy to adults with rCDI (Figure S1). Follow-up visits occurred at 1, 2, 4, and 8 weeks, and 3 and 6 months after administration. The protocol was approved by the relevant institutional review boards, and the trial was conducted in accordance with the ethical principles of the Declaration of Helsinki, Good Clinical Practice guidelines, and requirements of publicly registered clinical trials. Written informed consent was obtained for all participants prior to study commencement. CONSORT guidelines were followed while developing this report. 25
Participant eligibility and screening
Adults (aged ⩾ 18 years) with a diagnosis of rCDI (⩾1 recurrence or ⩾2 episodes and a positive stool test for C. difficile toxin or toxigenic C. difficile), a current prescription for antibiotics to control CDI-related diarrhea, and who were a candidate for colonoscopy were eligible for screening. The antibiotic course for the qualifying rCDI episode must have included a minimum of 10 consecutive days but not more than 60 total days before initiating a 24- to 72-h washout period (i.e., the time between administration of SOC antimicrobials and administration of RBL). The washout period permitted the participant to pass the majority of the antimicrobial from their system to ensure minimal to no residual impact on the microbiota-based therapeutic used to prevent recurrence. CDI symptoms must have been under control prior to and during the antibiotic washout period, as recorded by participants in an electronic stool diary. Participants were ineligible for the trial if they were using systemic antibiotics for an indication other than rCDI, had symptoms caused by a confirmed intestinal pathogen other than C. difficile, had current uncontrolled diarrhea unrelated to CDI, had CDI unresponsive to antibiotic therapy, had active/fulminant colitis, or had received any microbiota-based therapies (e.g., fecal microbiota transplantation, RBL) within 6 months before screening or between screening and baseline. The full inclusion and exclusion criteria can be found in the supplement.
RBL administration
Following the antibiotic washout period, bowel preparation for colonoscopy was conducted using a method determined at the investigator’s discretion. All eligible participants received one administration of RBL consisting of one 150-mL dose delivered via colonoscopy. Each RBL dose consisted of a microbiota suspension of 1 × 108 to 5 × 1010 colony-forming units/mL from human stool supplied by prescreened healthy donors. The suspension was manufactured under good manufacturing practices to ensure consistency and quality and was pathogen tested to promote participant safety. RBL was administered to the right side of the colon between the ileocecal valve and the hepatic flexure of the colon.
Trial outcomes
The primary endpoint assessed RBL-related TEAEs within 8 weeks of RBL administration or until confirmed treatment failure. Recurrence of CDI within 8 weeks after RBL administration was a secondary endpoint. Treatment failure (i.e., recurrence) was defined as the presence of CDI diarrhea and a positive test for C. difficile toxin (per the site’s standard practice) within 8 weeks after RBL administration, while treatment success was defined as the absence of CDI diarrhea for 8 weeks after RBL administration. Additional secondary endpoints included physician experience and physician perception of participant benefit (both described in detail in the next section), safety up to 6 months after RBL administration, time to CDI recurrence, and participant quality-of-life changes following administration of RBL collected through a qualitative telephone interview. Adverse events (AEs) were coded using Medical Dictionary for Regulatory Activities version 25.1. TEAEs were defined as any new AE or worsening of a pretreatment AE or preexisting medical condition that occurred after the start of RBL administration through the end of trial participation.
RBL practical usability and physician experience
Due to variability in how gastroenterologists perform colonoscopies based on institutional procedures, clinical practices, and available equipment, the method of RBL administration was largely conducted according to investigator discretion. To capture the subjective experience on the usability and practicality of RBL administration via colonoscopy, all investigators completed a physician experience questionnaire (Table S1). In addition, investigators rated the total improvement of participants compared with baseline according to the Clinician Global Impression of Improvement (CGI-I) scale at the 8-week follow-up visit or at a CDI recurrence visit for participants who had experienced recurrence. The CGI-I scale contains seven possible scores: very much improved, much improved, minimally improved, no change, minimally worse, much worse, and very much worse.
Statistical analysis
As this trial was exploratory without prespecified statistical hypotheses, a formal sample size calculation was not performed. The planned enrollment of approximately 40 participants was deemed sufficient for descriptive characterization of safety and effectiveness. Data were reported using descriptive statistics. Safety, effectiveness, and baseline analyses were conducted for all participants who received RBL. Categorical data collected during the trial were summarized using counts and percentages, while continuous data were presented using the number of participants, mean, standard deviation (SD), median, and range. If participants were deemed to have treatment failure or an indeterminate treatment outcome, TEAEs were included if the time of onset occurred after RBL administration and before the time of onset of CDI symptoms. If the time of CDI symptom onset was missing, 8-week follow-up visit data were used. If data from the 8-week follow-up were also missing, TEAEs with onset between the date of RBL administration and up to 56 days thereafter were included.
Results
Participant baseline demographics and CDI history
Of the 54 participants screened between May 2023 and June 2024, 41 were enrolled and received RBL via colonoscopy; 39 participants finished the 8-week visit and 2 withdrew prior to week 8 (Figure S2). Most participants were White (95.1%) and female (87.8%), having a mean (SD) age of 61.2 (14.9) years and mean (SD) body mass index of 26.3 (4.8; Table 1). Among enrolled participants, the mean (SD) number of previous CDI episodes was 3.2 (1.8), with mean (SD) duration of the last CDI episode of 24.2 (11.1) days. The most commonly prescribed antibiotics during participants’ most recent CDI episode were vancomycin (n = 29; 63.0%) and fidaxomicin (n = 16; 34.8%). Most participants were not hospitalized due to their last CDI episode (n = 38; 92.7%).
Participant baseline demographics and CDI history.

The last episode was also the qualifying episode; duration was defined as CDI symptom start date to CDI symptom end date.
A participant could have had more than one antibiotic prescribed per CDI episode.
CDI, Clostridioides difficile infection; RBL, fecal microbiota, live-jslm; SD, standard deviation.
Safety
For the primary endpoint, a total of five TEAEs considered related to RBL were reported in 4 (9.8%) participants within 8 weeks of RBL administration, all of which were gastrointestinal disorders (Table 2; abdominal distension occurred in 2 (4.9%) participants, while constipation, diarrhea, and flatulence each occurred in 1 (2.4%) participant) and assessed as mild in severity. Overall, 33 TEAEs occurred in 18 (43.9%) participants within 8 weeks, most of which were mild (25/33; 75.8%) or moderate (5/33; 15.2%) in severity (Table 3), and gastrointestinal in nature (Table 4). TEAEs experienced by ⩾5% of participants included abdominal distension and constipation (7.3% each). Serious TEAEs occurred in two participants; one participant experienced a flare of preexisting ulcerative colitis that was severe in intensity, and one participant experienced a brain neoplasm that was considered life-threatening. Additionally, one participant experienced a severe TEAE of abdominal pain. None of the serious or severe TEAEs were considered related to RBL. No TEAEs leading to death or intensive care unit admission were reported.
TEAEs assessed as related to RBL occurring within 8 weeks of RBL administration.
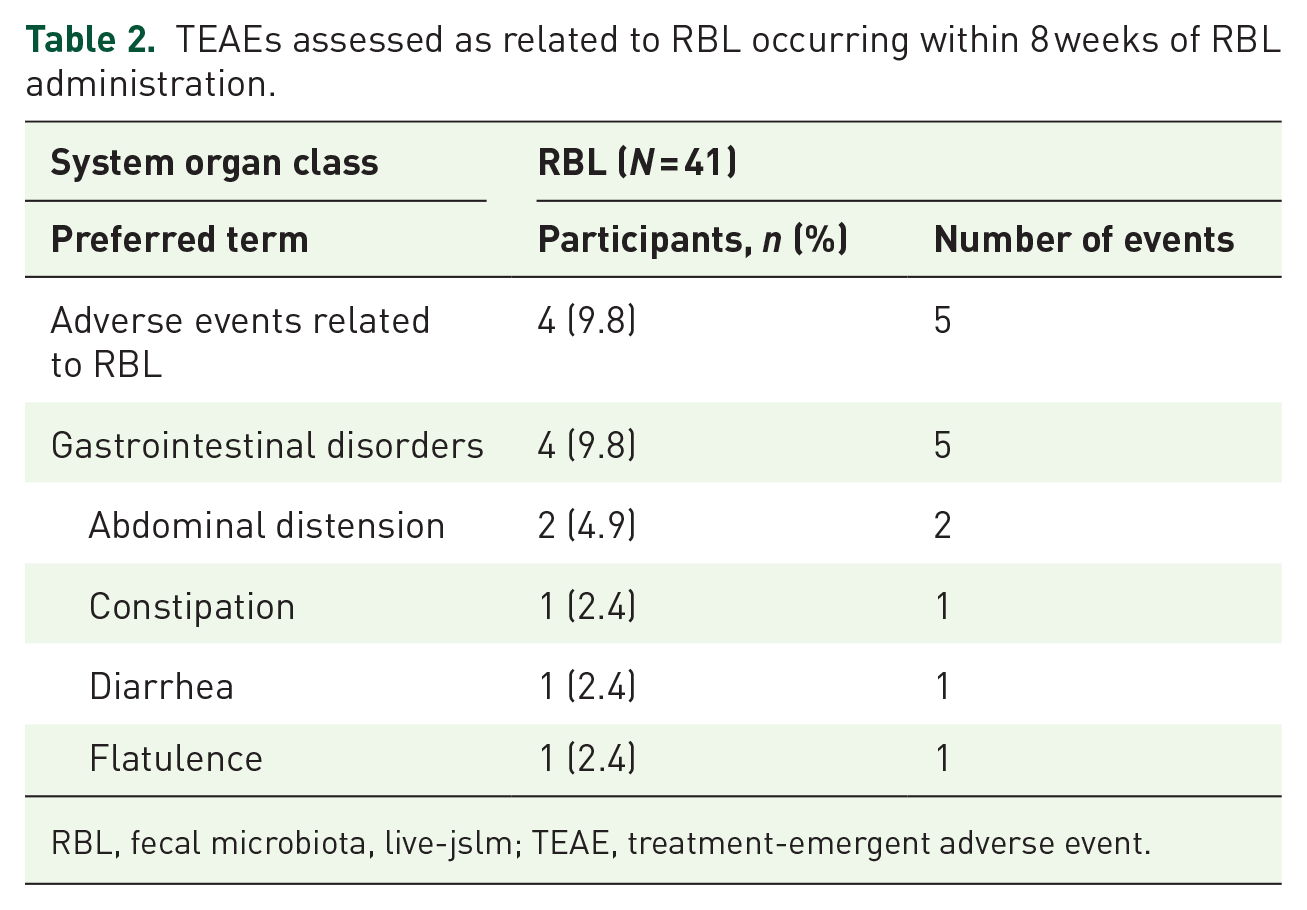
RBL, fecal microbiota, live-jslm; TEAE, treatment-emergent adverse event.
Safety summary of TEAEs occurring through 8 weeks after RBL administration.

Participant had a brain neoplasm, which was unrelated to RBL.
Participant had a flare of preexisting ulcerative colitis during the study; assessed as unrelated to RBL.
RBL, fecal microbiota, live-jslm; TEAE, treatment-emergent adverse event.
TEAEs assessed as not related to RBL occurring within 8 weeks of RBL administration.
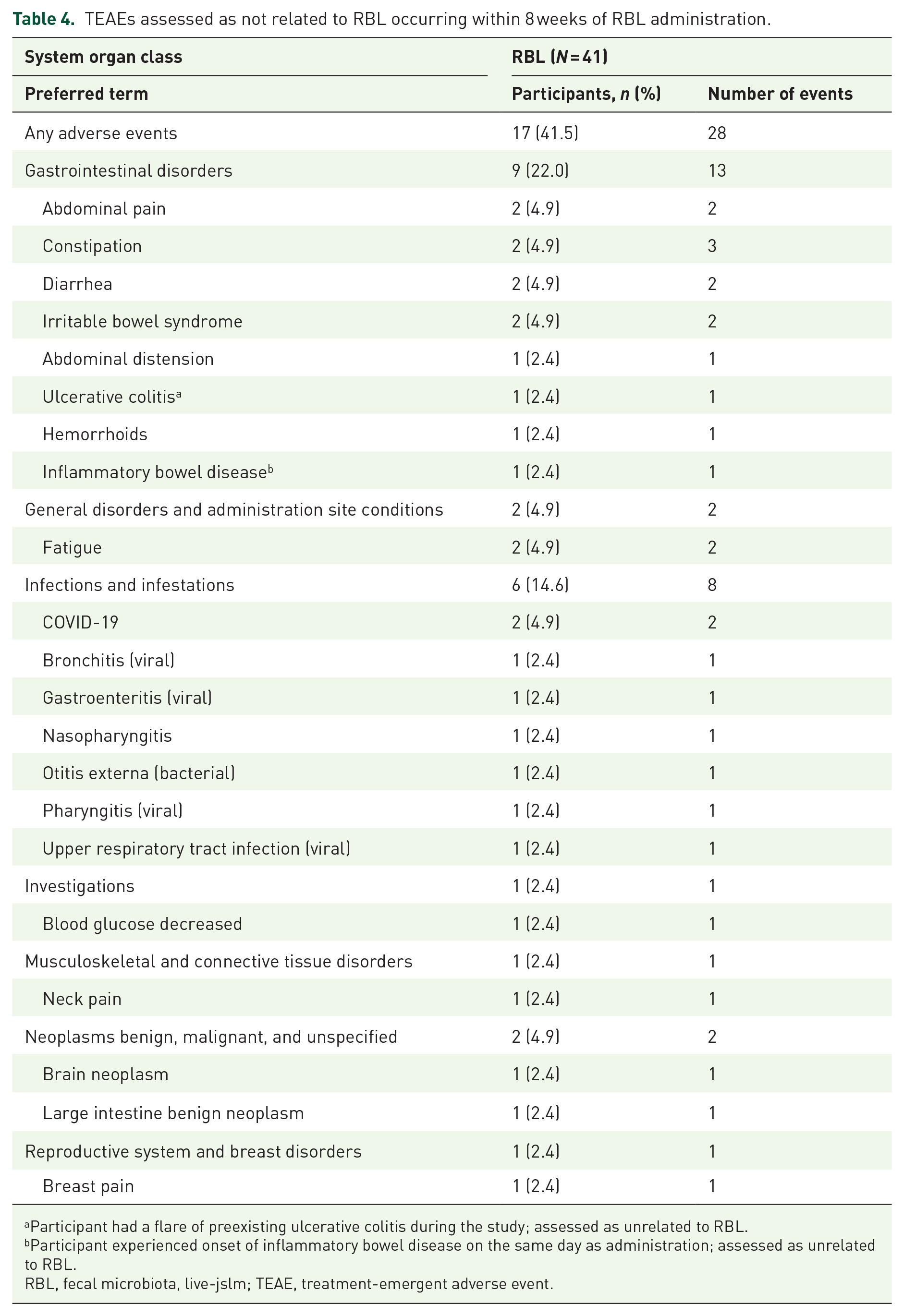
Participant had a flare of preexisting ulcerative colitis during the study; assessed as unrelated to RBL.
Participant experienced onset of inflammatory bowel disease on the same day as administration; assessed as unrelated to RBL.
RBL, fecal microbiota, live-jslm; TEAE, treatment-emergent adverse event.
Effectiveness
Overall, 39 participants (95.1%) experienced treatment success (no CDI recurrence) through 8 weeks after RBL administration by colonoscopy (Figure 1). The remaining two participants (4.9%) had an indeterminate outcome (i.e., neither the protocol-specified definition for treatment success nor treatment failure was met). These two participants withdrew consent prior to the 8-week follow-up assessment, one due to an unrelated serious AE (brain neoplasm) and one due to scheduling conflicts; neither reported any CDI symptoms up to the time of withdrawal. Since no participants experienced CDI recurrence through 8 weeks after RBL administration delivered by colonoscopy, no data were available for time to CDI recurrence.

Clinical effectiveness of RBL administered by colonoscopy after 8 weeks.
Physician experience
Physicians’ experiences were predominantly favorable, with 90.2% of the investigators indicating they had a “positive” or “very positive” overall experience when administering RBL via colonoscopy (n = 23 and n = 14, respectively; Figure 2(a)). Factors such as the ease of passage through the colonoscope and time required for material preparation were cited as contributing to these “positive” or “very positive” experiences by 36.6% (n = 15) and 29.3% (n = 12) of physicians, respectively. One investigator (2.4%) reported a “somewhat negative” experience, which was attributed to challenges with the connectivity of RBL to the colonoscope. Among the 39 participants who completed their 8-week follow-up visits, all (100%) were evaluated by the physicians as either “very much improved” or “much improved” according to CGI-I scores (n = 26; 66.7% and n = 13; 33.3%, respectively; Figure 2(b)). Overall, physicians reported that administering RBL via colonoscopy was straightforward and described the process as quick, easy, and well-tolerated by participants.

(a) Physician experience on the usability of RBL in clinical practice. (b) Physician perception of participant benefit at 8 weeks after RBL administration.
Physician-reported approach to RBL administration
Most participants (n = 35; 85.4%) were given osmotically acting laxatives (e.g., polyethylene glycol (PEG) 3350, PEG 3350/potassium chloride/sodium bicarbonate/sodium chloride/sodium sulfate) and 23 (56.1%) were given contact laxatives (e.g., bisacodyl, citric acid/magnesium oxide/sodium picosulfate) for bowel preparation prior to RBL. Two participants (4.9%) received loperamide. A more detailed description of bowel preparation medications can be found in the supplement (Table S2). With respect to RBL administration, a commonly reported approach involved cutting the product bag and drawing up the thawed suspension into syringes to inject via the colonoscope. Forty participants (97.6%) received RBL in the right side of the colon (cecum or ascending colon), while one participant (2.4%) received RBL in the sigmoid colon.
Thirty-one participants (75.6%) received the full 150-mL RBL dose with no product loss, 9 (22.0%) experienced a small amount of product loss (1–25 mL), and 1 participant experienced a moderate amount of product loss (26–50 mL). The amount of RBL received did not have an impact on treatment success rates (Figure S3).
Discussion
In CDI-SCOPE, RBL administered via colonoscopy appeared both safe and effective in preventing CDI recurrence within 8 weeks of administration. The primary endpoint, which focused on the safety profile as measured by RBL-related TEAEs, indicated RBL was well-tolerated, with no safety concerns arising during the trial. All 39 participants in CDI-SCOPE who completed the 8-week follow-up visit experienced treatment success, demonstrating the clinical effectiveness of single-dose RBL when administered by colonoscopy.
Participant demographics and CDI history in CDI-SCOPE were similar to participant populations in previous RBL trials, with a majority of participants being White females who experienced at least two CDI recurrences and who received vancomycin to treat their most recent CDI episode. 23 The safety profile of RBL in CDI-SCOPE was consistent with trials in the clinical development program involving rectal administration of RBL, which also demonstrated RBL to be well-tolerated, with a majority of TEAEs graded as mild or moderate in severity and gastrointestinal in nature.6,11,18 –22 Notably, the rate of treatment success observed in CDI-SCOPE (95.1%) was higher than that observed in clinical trials of single-course rectal administration of RBL (70.6%–73.8%).6,22 The use of bowel preparation, which is proposed to expedite antibiotic washout, 26 may improve the effectiveness of RBL when administered via colonoscopy. A previous analysis of phase II and phase III RBL studies, in which bowel preparation was not utilized, indicated that the use of a 72-h antibiotic washout resulted in higher treatment success versus placebo relative to 48 h or less, which may indicate an impact of residual SOC antibiotic in the gastrointestinal tract. 8 Furthermore, colonoscopy allows for administration of RBL to the right colon, which may result in broad colonic exposure due to peristalsis, potentially contributing to the 95.1% treatment success rate observed in CDI-SCOPE.
The 8-week results from CDI-SCOPE support the hypothesis that administering RBL via colonoscopy is safe and effective at preventing rCDI and build upon previous preliminary evidence of RBL outcomes when delivered by colonoscopy. In a retrospective subgroup analysis, 8 of 10 participants achieved treatment success, while the remaining 2 participants had incomplete medical records and were conservatively considered treatment failures. 24 In that analysis, RBL was administered under the FDA’s enforcement discretion policy via colonoscopy using a routine practice framework for colonoscopy administration. Details on specific administration methods and participant selection data were not collected in that study, which CDI-SCOPE began to address with a prospective trial design.
Colonoscopic administration of RBL was regarded positively by physicians, both in relation to the benefits for participants and the practicality of the administration process within the CDI-SCOPE trial. Most physicians noted minimal challenges with colonoscopic administration of RBL, especially when utilizing syringes to transfer and inject the RBL suspension, which passed easily through the colonoscope channel. The ease of colonoscopic administration carries further benefits for patients, especially elderly patients, who may retain RBL more easily when administered via colonoscopy. This favorable physician perspective complements the clinical effectiveness and safety of RBL administered via colonoscopy. Participant experience with RBL administration via colonoscopy in CDI-SCOPE will be reported separately.
Several limitations of this study must be acknowledged. First, the trial was a single-arm open-label design, which introduces the potential for bias in both treatment administration and outcome assessment. The absence of a control group limits the ability to draw definitive conclusions regarding the comparative efficacy of RBL relative to other available treatments. Second, the sample size of this study was small, which restricts the generalizability of the results. Participant characteristics varied, including with respect to their CDI histories (e.g., number of previous recurrences and antibiotic treatment received). A larger cohort could be necessary to confirm the observed outcomes and assess the broader applicability of RBL in diverse patient populations. Furthermore, despite several efforts to be more inclusive, the trial population was somewhat homogeneous in terms of sex and race (similar to other previous RBL trials), which could limit the generalizability of the findings to a wider demographic. Third, relatedness of TEAEs was limited to their potential association with RBL, and potential associations with other variables such as preexisting conditions were not captured. While this report focuses on the safety and prevention of recurrence within 8 weeks of follow-up, key results from the 6-month follow-up period, which will be presented in future publications, will be important in determining whether any long-term safety concerns arise. Fourth, the absence of formal statistical hypothesis testing and procedural variability with regard to colonoscopy administration limit the ability to draw definitive conclusions regarding the clinical effectiveness of RBL. Last, it is important to note that RBL was not used for the treatment of active CDI, including fulminant disease as well as antibiotic-refractory CDI.
Conclusion
These exploratory findings suggest that RBL administered via colonoscopy is a practical, safe, and effective option for preventing CDI recurrence in adults. The safety findings were consistent with previous RBL trials despite the difference in administration method. Results support the potential clinical utility of RBL delivered by colonoscopy to prevent CDI recurrence.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251339697 – Supplemental material for Safety and effectiveness of fecal microbiota, live-jslm (REBYOTA®) administered by colonoscopy for prevention of recurrent Clostridioides difficile infection: 8-week results from CDI-SCOPE, a single-arm, phase IIIb trial
Supplemental material, sj-docx-1-tag-10.1177_17562848251339697 for Safety and effectiveness of fecal microbiota, live-jslm (REBYOTA®) administered by colonoscopy for prevention of recurrent Clostridioides difficile infection: 8-week results from CDI-SCOPE, a single-arm, phase IIIb trial by Sahil Khanna, David Yoho, Daniel Van Handel, Brian J. Clark, Tahany Awad, Beth Guthmueller, Daniel Armandi, Whitfield Knapple, Nasia Safdar, Brian Baggott, Karen Simon and Paul Feuerstadt in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-2-tag-10.1177_17562848251339697 – Supplemental material for Safety and effectiveness of fecal microbiota, live-jslm (REBYOTA®) administered by colonoscopy for prevention of recurrent Clostridioides difficile infection: 8-week results from CDI-SCOPE, a single-arm, phase IIIb trial
Supplemental material, sj-pdf-2-tag-10.1177_17562848251339697 for Safety and effectiveness of fecal microbiota, live-jslm (REBYOTA®) administered by colonoscopy for prevention of recurrent Clostridioides difficile infection: 8-week results from CDI-SCOPE, a single-arm, phase IIIb trial by Sahil Khanna, David Yoho, Daniel Van Handel, Brian J. Clark, Tahany Awad, Beth Guthmueller, Daniel Armandi, Whitfield Knapple, Nasia Safdar, Brian Baggott, Karen Simon and Paul Feuerstadt in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors thank all the participants and their families and caregivers as well as the investigators and site staff. We acknowledge the contributions of investigators Humberto Aguilar, Tauseef Ali, Clint Behrend, and Jonathan Rosenberg to this trial. Medical writing support, under the guidance of the authors, was provided by Benjamin G. Richardson, PhD, and Rachel Hood, PhD (ApotheCom, Yardley, PA, USA), and was funded by Ferring Pharmaceuticals, Inc., Parsippany, NJ, USA.
Declarations
Supplemental material
Supplemental material for this article is available.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
