Abstract
The immunotherapy landscape for gastrointestinal (GI) tumors is rapidly evolving. There is an urgent need for reliable biomarkers capable of predicting treatment outcomes to optimize therapeutic strategies and enhance patient prognosis. This review presents a comprehensive overview of biomarkers associated with the immunotherapy response of GI tumors, covering advances in molecular genetics, histopathological markers, and imaging. Key molecular biomarkers, such as microsatellite instability, tumor mutational burden, and programmed death-ligand 1 expression, remain critical for identifying patients likely to benefit from immune checkpoint inhibitors. The significance of tumor-infiltrating lymphocytes, notably the CD8+ T cell to regulatory T cell ratio, as a predictor of immunotherapy response is explored. In addition, advanced imaging techniques, including computed tomography (CT), magnetic resonance imaging, and positron emission tomography-CT, facilitate the noninvasive evaluation of tumor biology and therapeutic response. By bridging molecular and imaging data, this integrated strategy enhances precision in patient selection, treatment monitoring, and adaptive therapy design. Future studies should aim to validate these biomarkers in larger, multicenter cohorts and focus on clinical translation to advance precision medicine in GI oncology.
Introduction
Gastrointestinal (GI) cancers, including esophageal, gastric, and colorectal cancers, are major causes of cancer-related deaths globally. 1 Despite progress in surgery, chemotherapy, and radiotherapy, advanced GI cancers still have poor outcomes due to genetic complexity and tumor microenvironment (TME) heterogeneity,2,3 sparking increasing interest in immunotherapy as a novel treatment approach. Immunotherapy, particularly through immune checkpoint inhibitors (ICIs),4,5 has revolutionized the treatment landscape for these malignancies.6,7 Nevertheless, variability in patient responses to immunotherapy underscores the urgent need for predictive biomarkers capable of identifying patients most likely to benefit from such treatments.8–10
Predictive biomarkers are now a key focus in GI oncology research.11,12 Key biomarkers have under investigated, including molecular genetic factors such as microsatellite instability (MSI) and mismatch repair deficiency (dMMR), 13 protein expression levels such as programmed death 1 (PD-1) and programmed death-ligand 1 (PD-L1). 14 In recent years, imaging-based biomarkers have gained significant attention.15–17 This review aims to integrate current research findings on predictive biomarkers for immunotherapy in GI tumors, assess their role in elucidating tumor–immune interactions, emphasize their predictive potential and clinical application, and explore their potential in guiding individualized treatment strategies (Figure 1).

The main content of this review (By Figdraw).
TME and mechanisms of immunotherapy
The TME is a complex and dynamic ecosystem composed of cancer cells, immune cells, fibroblasts, stromal elements, and vascular components,18,19 which play crucial roles in tumor progression and therapeutic response.20,21 Immune cells, such as CD8+ cytotoxic T lymphocytes and natural killer (NK) cells, help eliminate malignant cells. However, tumors can evade immune surveillance by recruiting regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs), fostering an immunosuppressive environment,22–24 and upregulating immune checkpoint molecules such as PD-1/PD-L1.25–27
Interaction between immune cells and the TME
The interaction between immune cells and the TME plays a pivotal role in determining the success of immunotherapy.28,29 For instance, high densities of effector T cells, particularly CD8+ T cells, which can recognize and kill tumor cells, are often associated with a favorable prognosis and robust responses to ICIs.30,31 By contrast, the recruitment of Tregs and MDSCs, which suppress T-cell proliferation and activation, contributes to an immunosuppressive TME that promotes immune escape by tumor cells, often correlating with resistance to immunotherapy.32–34 Understanding these interactions is critical for developing therapeutic strategies that can modulate the TME to enhance immunotherapy efficacy.
Mechanisms of immune escape
Tumors employ various mechanisms to evade immune detection and destruction, including (1) the upregulation of immune checkpoint molecules (e.g., PD-1, PD-L1), which inhibit T-cell activation and proliferation 27 ; (2) the secretion of immunosuppressive cytokines, which alter the phenotype of immune cells within the TME35,36; and (3) the induction of immune tolerance via the recruitment of immunosuppressive cells such as Tregs and MDSCs.37,38 Targeting these mechanisms has become a primary focus of immunotherapeutic strategies, including the development of ICIs that block the inhibitory signals of the PD-1/PD-L1 and CTLA-4 pathways, thereby restoring T-cell activity and promoting antitumor responses.
As our understanding of the TME and its role in immune evasion grows, there is a rising interest in exploring molecular and histopathological biomarkers for their potential to predict responses to immunotherapy in GI tumors.
Novel immunotherapies
Beyond immune checkpoint blockade, novel immunotherapies such as CAR T-cell therapy, PI3K inhibitors (TQ-B3525), EZH2 inhibitors, anti-CD20/79B antibodies, and dual-targeting agents (e.g., CD20/CD3 bispecific antibodies) have shown efficacy in follicular lymphoma by remodeling the tumor microenvironment. Importantly, these therapies hold emerging potential for gastrointestinal cancers.39,40 Immunomodulators like lenalidomide further expand therapeutic options by enhancing antitumor immunity through cytokine regulation.
Future research should prioritize integrating novel agents (e.g., dual-targeting antibodies, CAR T cells) with conventional immunotherapies to overcome resistance mechanisms. In EBV-associated tumors (e.g., cholangiocarcinoma), EBV-driven PD-L1 overexpression alters the immune microenvironment, impacting checkpoint blockade. Strategies combining pembrolizumab/rituximab (effective in relapsed lymphoma) and CD79a/CD79b co-targeting in B-cell malignancies may guide gastrointestinal cancer therapy.41–43
Molecular and histopathological biomarkers for predicting immunotherapy response in GI tumors
Multiple histopathological and molecular biomarkers have been explored to predict the efficacy of immunotherapy in GI tumors.44–47 This section categorizes these biomarkers and emphasizes their clinical relevance and potential applications.
Microsatellite instability and mismatch repair deficiency
MSI and dMMR are critical biomarkers for immunotherapy sensitivity, particularly in colorectal cancer.48–50 MSI-High (MSI-H) and dMMR tumors generally exhibit a high mutational burden, leading to increased neoantigen production, which enhances tumor visibility to the immune system. This makes MSI-H and dMMR tumors particularly suitable for immune checkpoint blockade (ICB) therapy.51,52
In clinical settings, MSI-H and dMMR statuses are typically determined via polymerase chain reaction or immunohistochemistry (IHC) methods. 53 Numerous studies have demonstrated that MSI-H patients exhibit significantly higher response rates to PD-1 inhibitors compared to MSS patients.54–56 Consequently, the MSI and dMMR statuses are crucial in selecting GI cancer patients for ICI therapy.
Although MSI and dMMR are strong predictive factors in certain tumor types, such as colorectal cancer, their predictive value in other gastrointestinal cancers may not be well-defined. In addition, testing heterogeneity and the need for standardized assessment also limit their broader clinical application.
TME and immune cell infiltration
The TME, which consists of cancer cells, immune cells, fibroblasts, and vascular elements, influences tumor response to therapy through its inherent heterogeneity and dynamic nature.18,19 The infiltration pattern of various immune cells, particularly CD8+ T cells, Tregs, and MDSCs, is a key determinant of patient outcomes. High densities of CD8+ T cells are associated with favorable immunotherapy responses, 57 while increased levels of Tregs and MDSCs often indicate an immunosuppressive TME, leading to resistance to therapy.37,38
Research has shown that TME subtypes are crucial in determining immunotherapy outcomes. For example, “hot” tumors, characterized by high infiltration of CD8+ T cells and other effector immune cells, are more likely to respond to ICIs, while “cold” tumors, lacking these effector cells, are typically resistant.58,59 Thus, evaluating TME status, including the density of CD8+ T cells and the distribution of suppressive cells, can serve as a powerful indicator of treatment efficacy.
Although TME and immune cell infiltration are promising areas of research, their clinical application requires further standardization and validation. A more profound understanding of intratumoral and intertumoral heterogeneity is also needed, as this variability can lead to inconsistent predictions of treatment responses.
Tumor mutational burden
TMB, representing the total number of somatic mutations within a tumor genome, is a significant predictor of immunotherapy response.60,61 Tumors with high TMB tend to produce more neoantigens, which can be recognized by T cells, leading to a stronger antitumor immune response. High TMB has been associated with improved responses to ICIs and favorable prognoses in multiple solid tumors, including GI cancers.62,63
However, the clinical implementation of TMB as a biomarker remains challenging due to variations in TMB thresholds across different cancer types and inconsistencies in its predictive value. 64 Standardization of TMB assessment and the development of consensus guidelines are necessary to fully realize its potential in clinical practice.
Although TMB has the potential to predict immunotherapy response, its clinical application remains limited by a lack of standardization and technical challenges in assessment.
PD-1 and PD-L1 expression
PD-1 and PD-L1 expression, assessed via IHC, are studied as immunotherapy biomarkers.65–67 High PD-L1 expression is frequently associated with favorable responses to ICIs.68–71 However, the predictive role of PD-L1 in GI tumors remains controversial, not only due to the variability in its expression but also because of multiple factors, including TME, genetic heterogeneity, types of immunotherapy, and patient immune status. The comprehensive impact of these complex factors means that the predictive value of PD-L1 needs a more holistic assessment. Furthermore, PD-L1 expression levels are often quantified using different scoring systems (e.g., combined positive score and tumor proportion score), which increases the complexity of interpretation.72–74
PD-1 and PD-L1 expression have become key biomarkers in the field of immunotherapy, but they still face challenges in clinical practice. For example, accurate PD-L1 assessment often requires specialized techniques and expertise. In addition, the use of different scoring systems complicates the interpretation of PD-L1 staining results.
Tumor spatial heterogeneity
Tumor spatial heterogeneity (ITH) describes the diversity within a tumor across various regions in terms of genetics, immune cell composition, microenvironmental characteristics, and drug delivery. 75 This heterogeneity influences tumor response to immunotherapy, as different areas may have varying mutational burdens, immune cell densities, and microenvironmental conditions, such as hypoxia and differences in nutrient supply (Figure 2). For example, areas with greater infiltration of CD8+ T cells may be more sensitive to ICIs, whereas hypoxic regions may be more immunosuppressive. In addition, vascular and stromal differences within the tumor can lead to uneven drug distribution, affecting treatment outcomes.76–78 Therefore, understanding ITH is crucial for developing personalized treatment strategies that target the specific characteristics of different tumor regions, which can improve the efficacy and selectivity of immunotherapy. Radiomics technology, through the extraction of imaging features (e.g., texture and density), can quantify this heterogeneity and provide insights into underlying genetic and immune patterns.79,80

Schematic of tumor spatial heterogeneity (By Figdraw). Region A is enriched with CD8+ T cells and oxygenated, rendering it sensitive to ICIs. Region B is hypoxic and dominated by immunosuppressive cells (Tregs and MDSCs), leading to therapy resistance. Uneven vascular networks further contribute to inefficient drug delivery.
Assessing ITH requires the use of advanced technologies, such as next-generation sequencing and multiparametric imaging, and standardized methods for evaluation. Moreover, the complexity of ITH data can be challenging to interpret. Continued research and technological advancements are needed to overcome these challenges and fully realize the potential of ITH in clinical oncology.
Imaging technologies for predicting the immunotherapy response
Medical imaging is one of the key factors informing medical science. Its superiority lies in its ability to noninvasively assess the characteristics of human tissue and organs.81–83 Thus, it is routinely used in clinical practice for oncologic diagnosis as well as predicting immunotherapy responses in GI tumors.84–86 Computed tomography (CT) has the advantages of fast imaging speed and high spatial resolution of images (Figure 3). Magnetic resonance imaging (MRI) has a high resolution of soft tissue, to reflect tumor functional status and microenvironmental changes (Figure 4). Positron emission tomography-CT (PET-CT) employs metabolic tracers (e.g., 18F18F-FDG, 68Ga68Ga-FAPI-04) to map tumor activity and immune cell infiltration by targeting glucose metabolism or fibroblast activation protein (FAP).
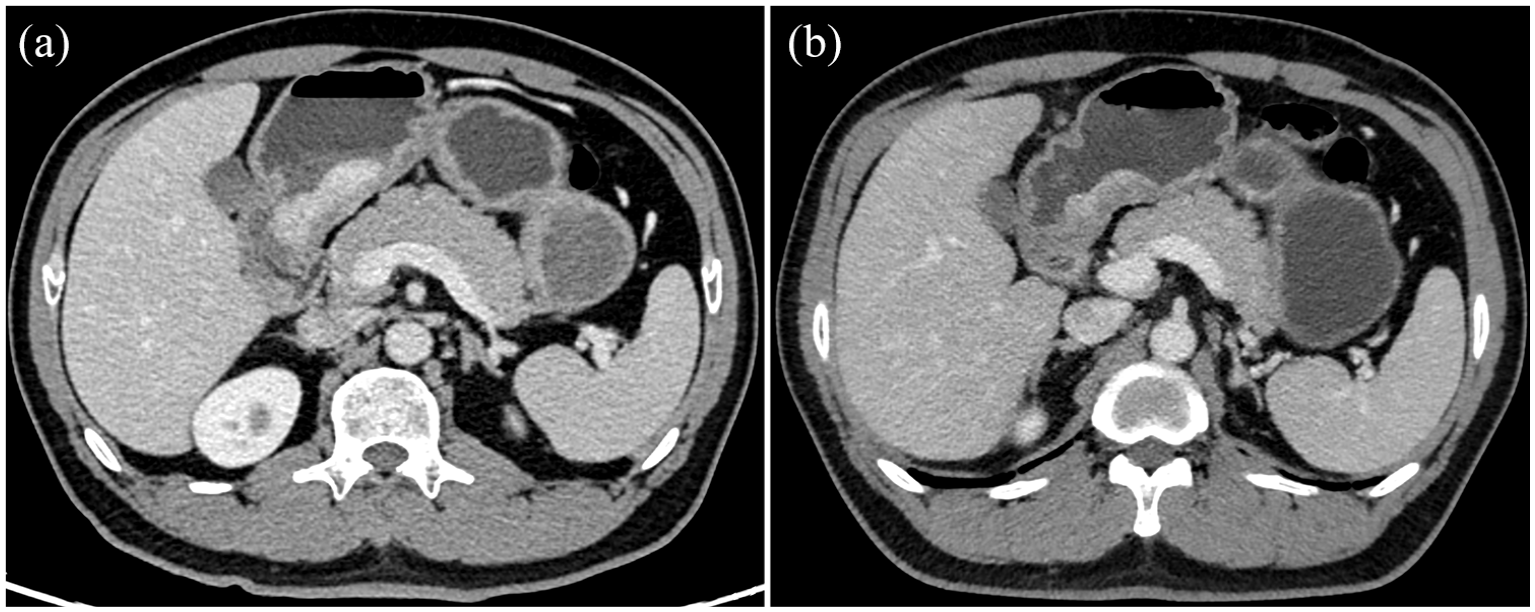
Male, 48-year-old, diagnosed with Gastric Mucosa-associated low-differentiated adenocarcinoma (immunohistochemistry results: pMMR, HER2 (0), PD-L1 22C3 (combined positive score < 1)). Compared to pre-treatment portal venous phase CT image (a), the range of the lesion has significantly decreased after three cycles of immunochemotherapy (b).
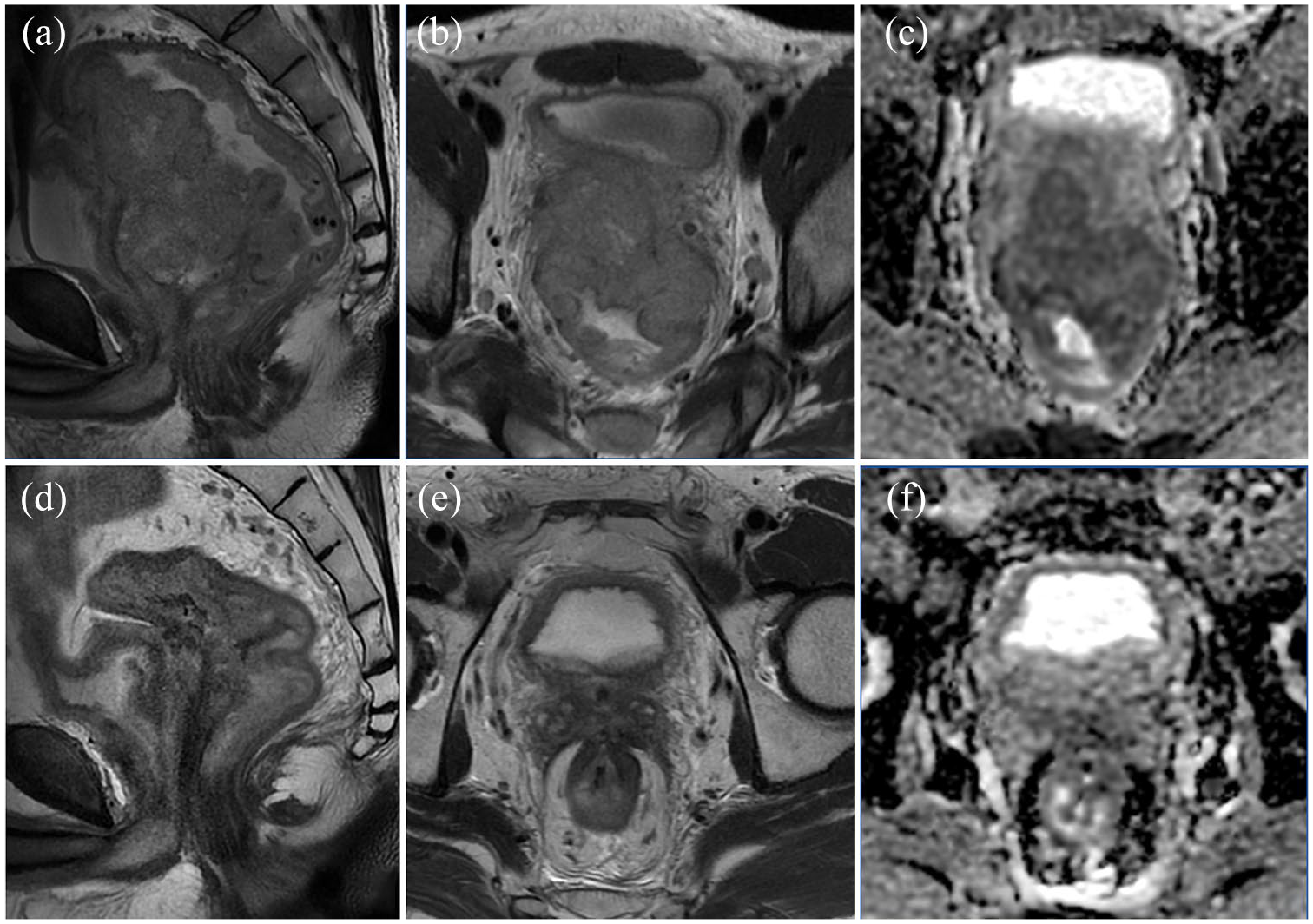
Advanced rectal cancer with positive circumferential resection margin (CRM+), invasion of the prostate, seminal vesicles, and posterior bladder wall, bilateral lateral lymph node metastasis (a, b), and significantly restricted diffusion on DWI (c). NGS: Microsatellite instability MSI-High; Germline MSH6 mutation p.Y969C (Deficient Mismatch Repair, dMMR). Compared to the baseline high-resolution MRI of the rectum (a, b, c), there is significant regression of the rectal tumor with a small amount of fibrosis, and the lateral lymph nodes have decreased in size (d, e), with no significant restriction on DWI diffusion (f) after immunotherapy combined with radiotherapy.
Recent advances in medical image acquisition and analysis have allowed for high-quality images with isotropy to be obtained. These images contain not only general morphological information but also rich data. Radiomics has been recognized as an important imaging technology in oncology. It can convert CT images into high-throughput quantitative data, which can reflect intratumor heterogeneity and be associated with tumor molecular and immune profiles. 87 Radiogenomics, the linkage of imaging features to gene expression, further enhances precision. Such integration bridges macroscopic imaging findings with microscopic genetic alterations, enabling dynamic monitoring of tumor evolution during therapy.88–90
Imaging characterization of the molecular and histological features
Advanced imaging techniques characterizing molecular genetics have opened new avenues for predicting the efficacy of immunotherapy in GI tumors. 91 Table 1 encapsulates various studies utilizing imaging biomarkers to characterize the molecular genetics of GI tumors, underscoring their correlation with treatment response.85,92–107 These studies highlight the potential of imaging to reflect underlying molecular-genetic traits such as the TME, MSI, TMB, and PD-L1 expression.
Correlation studies of imaging characterization for molecular genetics.

18F-FDG PET/CT, 2-deoxy-2-[18F] fluoro-D-glucose positron emission tomography/CT; ADC, apparent diffusion coefficient; AGC, advanced gastric cancer; AUC, area under the receiver operating characteristic curve; CC, colon cancer; CECT, contrast-enhanced CT; CPS, combined positive score; CRC, colorectal cancer; CRLM, colorectal cancer liver metastases; CT, computed tomography; DFS, disease-free survival; EMVI, extramural venous invasion; ESCC, esophageal squamous cell carcinoma; FAP, fibroblast activation protein; FAPI, fibroblast activation protein inhibitor; FDG, fluorodeoxyglucose; GC, gastric cancer; GEC, gastroesophageal cancer; ICI, immune checkpoint inhibitor; IHC, immunohistochemistry; KRAS, Kirsten ras sarcoma viral oncogene; LRS, lymphoid radiomic score; MRI, magnetic resonance imaging; MRS, myeloid radiomic score; MSI, microsatellite instability; MTV, metabolic tumor volume; NLR, neutrophil-to-lymphocyte ratio; OS, overall survival; PD-L1, programmed death-ligand 1; PET, positron emission tomography; RC, rectal cancer; rs, Spearman rank correlation coefficient; SUV, standardized uptake value; SUVmax, maximum standardized uptake value; SUVmean, mean standardized uptake value; SUVpeak, peak standardized uptake value; SUVsd, standardized uptake value standard deviation; TITreg, tumor-infiltrating regulatory T cells; TLF, total lesion FAP; TMB, tumor mutational burden; TME, tumor microenvironment.
Jiang et al. 92 developed a radiomics model using CT images to assess the TME in gastric cancer patients. The model achieved an AUC of 0.937 in the training cohort and 0.909 in the validation cohort, demonstrating its potential as a noninvasive predictor of TME composition. Similarly, Sun et al. 93 established radiomic scores for lymphoid and myeloid contexts in gastric cancer, which correlated with IHC-derived immune contexts and were predictive of disease-free survival (DFS) and overall survival (OS). Huang et al. 94 demonstrated a correlation between a CT-based radiomic score and DFS and OS, achieving AUCs of 0.795 and 0.861, respectively. Collectively, these studies suggest that radiomics may effectively capture a tumor’s immune profile, thereby enhancing treatment prediction for GI cancers.
Recently, researchers developed a “Radscore” model combining radiomics and clinical features to predict the MSI status in gastric cancer patients, achieving an AUC of 0.836 in the training cohort and 0.834 in the validation cohort. The model may serve as a noninvasive biomarker. 97 Li Z et al. revealed that magnetic resonance imaging (MRI)-based provides greater accuracy in assessing MSI in rectal cancer than T2-weighted imaging or apparent diffusion coefficient (ADC) imaging alone, with AUCs ranging from 0.908 to 0.926. 98 In addition, Lee et al. 101 demonstrated that 18F-FDG positron emission tomography CT (PET/CT) could assess kirsten ratsarcoma viral oncogene homolog mutations and MSI status in colorectal cancer with moderate accuracy. Collectively, these findings underscore the expanding role of advanced imaging in predicting treatment response in patients with GI tumors.
Hoshino et al. demonstrated the potential of radiomic analysis to predict TMB variations between primary and metastatic lesions in colorectal cancer, providing valuable insights into the role of radiogenomics in assessing TMB status. 102 Yang et al. identified a CT-detected extramural venous invasion-related gene signature, associating a high extramural venous invasion score with reduced MSI, lower TMB, and an increased tendency for immune escape, which correlates with a poorer response to ICI treatment in gastric cancer patients. 85 These studies highlight the potential of imaging as a predictive tool for immunotherapy response, complementing molecular genetic data.
Traditionally, PD-L1 expression has been evaluated via IHC, which involves the assessment of staining intensity and distribution in tumor biopsies. Complementary molecular techniques, such as Western blotting for direct protein quantification and RT-PCR for mRNA analysis, have also been routinely employed to validate PD-L1 levels. While IHC remains the clinical gold standard for PD-L1 assessment, its interpretation is complicated by variability in scoring systems and tissue heterogeneity. Noninvasive imaging approaches, such as radiomics and molecular PET/CT, are now being explored to overcome these limitations and provide spatially resolved insights into PD-L1 expression across entire tumors. Xie et al. 104 utilized contrast-enhanced CT in gastric cancer patients to build a radiomics model, achieving an AUC of 0.806 for predicting high PD-L1 levels and thus aiding immunotherapy decision-making. Qiao et al. 105 reported a positive correlation between 18F-FDG PET/CT maximum standardized uptake value (SUVmax) and PD-L1 expression in colon cancer liver metastases. Zhao et al. 106 identified standardized uptake value standard deviation (SUVsd) as the best predictor for PD-L1 positivity via 18F-FAPI-04 PET/CT, with an AUC of 0.882, underscoring the potential of molecular imaging in assessing PD-L1 expression for immunotherapy selection. Although current imaging techniques cannot directly quantify PD-L1, advanced modalities such as PET/CT and MRI provide valuable insights into the mutational landscape and immune cell infiltration of tumors.
By integrating various imaging modalities (e.g., CT, MRI, and PET/CT), researchers have preliminarily achieved noninvasive prediction of key molecular features in gastrointestinal tumors, such as microsatellite instability (MSI), tumor mutational burden (TMB), and PD-L1 expression. These imaging biomarkers not only overcome the sampling bias and invasiveness associated with traditional biopsies but also enable dynamic monitoring of tumor microenvironment evolution through multi-timepoint imaging analyses.
The integration of imaging biomarkers with molecular and histopathological data can refine predictive models. Future research should focus on validating these imaging biomarkers in larger-scale, multicenter cohorts to ensure their reproducibility and external validity.
Imaging prediction for immunotherapy outcomes
The efficacy of immunotherapy in GI tumors has been a significant focus of oncological research, with ICIs at the forefront. However, variability in patient response to these therapies necessitates the development of predictive biomarkers to enhance treatment personalization. Imaging techniques have emerged as valuable tools in this domain, providing insights into treatment outcomes and guiding patient selection for immunotherapy (Table 2).108–125
Correlative studies of imaging prediction for immunotherapy outcomes.

[18F]AlF-mNOTA-GZP, [18F]Aluminum Fluoride-Conjugated Modified NOTA Chelator with Targeting Ligand GZP; 2D, two-dimensional; AUC, area under the receiver operating characteristic curve; BOLD-MRI, blood oxygenation level-dependent magnetic resonance imaging; CC, colon cancer; CECT, contrast-enhanced CT; CpG-ODN, CpG-oligodeoxynucleotides; CRC, colorectal cancer; CT, computed tomography; dMMR, mismatch repair deficiency; DoR, depth of response; ECPI-Score, ESCC preoperative imaging score; EMVI, extramural venous invasion; ETS, early tumor shrinkage; FAPI, fibroblast activation protein inhibitor; FDG, fluorodeoxyglucose; GC, gastric cancer; HR, hazard ratio; ICB, immune checkpoint blockade; ICIs, immune checkpoint inhibitors; IORT, immunotherapy combined with radiotherapy; irAEs, immune-related adverse events; irPFS, immunotherapy-related progression-free survival; IVIM-DWI, intravoxel-incoherent-motion diffusion-weighted imaging; MRI, magnetic resonance imaging; MSI, microsatellite instability; MSS, microsatellite stability; N/A, not available; NCI, neoadjuvant chemotherapy combined with immunotherapy; nCRT, neoadjuvant chemoradiotherapy; NICT, neoadjuvant immunochemotherapy; NIT, neoadjuvant immunotherapy; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio; OS, overall survival; pCR, pathological complete response; PD-1, programmed death 1; PET, positron emission tomography; PFS, progression-free survival; RECIST, response evaluation criteria in solid tumors; RFS, recurrence-free survival; RS, radiomic score; SAI, subcutaneous adipose index; SAR, sarcopenia; SMI, skeletal muscle index; TME, tumor microenvironment; VAI, visceral adipose index. VEGFR-2, vascular endothelial growth factor receptor 2.
Fucà et al. 108 found that early tumor shrinkage and the depth of response in 169 CRC patients treated with ICIs were associated with improved survival, thereby guiding treatment strategies. Huang et al. 109 showed that radiomic features could predict 12-month and 24-month irPFS in patients with gastric cancer with high accuracy. Rong et al. 123 demonstrated that 68Ga-FAPI-04 PET/CT could noninvasively predict the efficacy of ICIs in patients with gastric cancer, facilitating pretreatment patient stratification. Conversely, Fox et al. 119 reported that CT, MRI, and endoscopy were insufficient for predicting outcomes in dMMR CRC patients, underscoring the need for better biomarkers. These findings suggest that imaging biomarkers may optimize treatment strategies and outcomes for GI tumors treated with ICI therapy, warranting further validation in future studies.
Combining immunotherapy with chemotherapy and radiotherapy shows promise in improving cancer treatment efficacy. 126 Imaging plays a crucial role in predicting responses to these combined therapies, providing valuable information for treatment planning and patient outcomes. Ruan et al. included 192 patients with esophageal squamous cell carcinoma undergoing neoadjuvant immunochemotherapy which were analyzed to assess dynamic radiologic features for predicting pathologic complete response (pCR). Notably, higher pCR rates were observed in patients without vascular signs. 111 Kim et al. identified an association between sarcopenia, as measured by the skeletal muscle index, and shorter PFS in gastric cancer patients on PD-1 inhibitors, underscoring the skeletal muscle index`s value as a prognostic marker. 116 Xu et al. 120 utilized intravoxel incoherent motion diffusion-weighted imaging (IVIM-DWI) and blood oxygenation level-dependent MRI (BOLD-MRI) to demonstrate increased tumor inhibition with combined vascular endothelial growth factor receptor -2 and PD-1 blockade, emphasizing MRI`s role in assessing treatment efficacy. Qi et al. built a machine-learning model to predict pCR in patients with esophageal squamous cell carcinoma by integrating radiomics from 18F-FDG PET/CT and clinical data, achieving an AUC of 0.852. 121
Dynamic imaging biomarkers, such as alterations in metabolic activity or tumor morphology reflected by standardized uptake values (SUVs) from PET scans, hold the potential for predicting responses to ICIs.122,127 Particularly, 68Ga-FAPI-04 PET imaging, which targets fibroblast-activation protein, represents a novel approach that offers insights into the tumor immune microenvironment (TIME). Elevated 68Ga-FAPI-04 uptake is associated with poorer outcomes and diminished ICI therapy efficacy, suggesting its utility as an independent predictor of treatment outcomes. 123 However, this technology faces challenges regarding accessibility and cost and requires further validation before broader patient implementation. In clinical settings, 68Ga-FAPI-04 PET has the potential to complement existing biomarkers, enabling personalized treatment strategies for patients most likely to benefit from ICIs.
Recent studies indicate that body composition, particularly adipose tissue distribution, significantly influences the success of immunotherapy. Imaging techniques have revealed a correlation between an individual’s adipose composition and the effectiveness of ICIs.117,128 These findings may help predict which patients are likely to respond well to specific immunotherapies, potentially personalizing treatment plans and enhancing the effectiveness of immunotherapy. In addition, the influence of adipose tissue on the TME might uncover new targets for treatment and the role of lifestyle changes in improving immunotherapy outcomes.
Innovative imaging technologies such as immuno-PET and hyperpolarized (HP) 13C-MRI show considerable potential for noninvasively monitoring the effects of immunotherapy on tumors. Immuno-PET uses monoclonal antibodies to track specific immune receptors or cells, predicting immunotherapy responses by quantifying tracer uptake.129,130 By contrast, HP 13C-MRI enhances the signal of 13C-labeled molecules, enabling real-time tracking of metabolic processes. This technique facilitates the study of cancer metabolism and early detection of responses to immunotherapy.131,132
Through fusion of these multidimensional data, imaging not only provides real-time guidance for immunotherapy initiation (e.g., metabolic decline preceding morphological changes) but also dynamically optimizes combination strategies (e.g., timing anti-angiogenic agents or radiotherapy), driving a paradigm shift in imaging from “post-treatment evaluation” to an “integrated tool for prediction, monitoring, and intervention.”
Future studies should prioritize validating these imaging biomarkers in prospective trials and integrating them into clinical workflows. Furthermore, longitudinal imaging studies are needed to assess dynamic changes in tumor characteristics (e.g., metabolic activity, immune cell infiltration) and correlate these changes with adaptive immune responses (e.g., T-cell receptor diversity, cytokine profiles), thereby uncovering resistance mechanisms and informing personalized therapeutic adjustments.
Challenges and future directions
Despite the significant potential of integrating molecular genetics and advanced imaging biomarkers into personalized immunotherapy for GI tumors, challenges remain in their clinical application: (1) many studies rely on single-center data, limiting generalizability due to homogeneous patient populations and institution-specific protocols. Multi-institutional collaborations are needed to enhance reproducibility and external validity. (2) Radiomic models are sensitive to differences in imaging equipment, reconstruction algorithms, and segmentation methods. Standardized guidelines are essential to harmonize radiomic analyses. (3) Limited cohorts reduce statistical power and increase overfitting risks, particularly in machine learning models. Larger, prospective datasets are required to validate findings. (4) Intratumoral and intertumoral heterogeneity challenge both molecular and imaging biomarkers. For instance, radiomic features averaged across entire tumors may obscure regional differences. Longitudinal studies with dynamic observation are needed to capture changes in the tumor microenvironment during therapy. (5) Discrepancies in endpoint definitions and follow-up durations hinder unified interpretation of biomarker efficacy. Long-term survival data are often lacking, limiting insights into durable immunotherapy benefits.
To overcome these challenges, future research should prioritize the following: (1) multi-center collaborations to ensure diverse and representative patient cohorts. (2) Standardization of imaging protocols and radiomic feature extraction. (3) Large-scale, prospective trials to validate integrated biomarker models. (4) Dynamic monitoring of tumor biology using advanced imaging techniques such as hyperpolarized 13C-MRI, immuno-PET.
Conclusion
This review highlights the transformative potential of integrating molecular genetics and advanced imaging biomarkers into personalized immunotherapy for GI tumors. Molecular biomarkers, such as MSI, TMB, and PD-L1 expression, have become indispensable tools for patient selection and treatment optimization. Meanwhile, advanced imaging technologies, including radiomics and PET/CT, provide noninvasive tools to assess tumor biology and predict therapeutic responses. Despite the challenges, through multi-center collaborations, standardized protocols, and large-scale prospective trials, these biomarkers are expected to play a significant role in future clinical practice, ultimately improving patient outcomes.
