Abstract
Background:
Managing extraintestinal manifestations (EIMs) in inflammatory bowel disease (IBD) patients remains challenging due to considerable heterogeneity in diagnostic criteria and the lack of a standardised definition and validated diagnostic pathways. Delays in recognising and treating EIMs can lead to significant disease progression. Therefore, early detection and treatment are crucial.
Objectives:
We aimed to assess the effectiveness of a dedicated immune-mediated inflammatory diseases (IMIDs) clinic in reducing EIM diagnostic delays and improving patients’ outcomes.
Design:
A single-centre observational study was conducted, including IBD patients presenting with EIMs red flags.
Methods:
We compared the EIMs diagnostic delay between patients who attended a multidisciplinary IMID outpatient clinic (IMID-G) and those who attended individual referral specialists representing the standard outpatient clinic group (SOC-G). We further evaluated the impact of diagnostic timing on 18-month clinical outcomes, including therapeutic changes, steroid and immunosuppressant use and biological therapy switch/swap.
Results:
We enrolled 238 IBD patients, 127 in the IMID-G and 111 in the SOC-G. The average time to EIM diagnosis was 2.48 ± 1.8 and 5.36 ± 2.3 months for the IMID and SOC-Gs (Δ = 2.88 months, p = 0.005). The majority of patients received a diagnosis of peripheral arthritis (IMID-G = 37.5%; SOC-G = 33.7%) and spondyloarthropathy (IMID-G = 32.1%; SOC-G = 33.7%). No significant difference was observed in the rates of EIMs between the two groups (88.2% in IMID-G vs 92.8% in SOC-G, p = 0.27). Regarding therapeutic changes, the IMID-G reported a mean time to the first therapeutic change driven by the specialist referral of 2.96 ± 1.8 months, compared to 6.09 ± 2.5 months in the SOC-G, showing a significant difference (p = 0.007). The IMID-G had a higher frequency of biological therapy switching/swapping and adding immunosuppressive treatment than the SOC-G (p = 0.008 and p = 0.04, respectively). Survival curves revealed a significant reduction in diagnostic delay and time to treatment in the IMID-G compared to the SOC-G (log-rank test, p < 0.001).
Conclusion:
Attending a dedicated IMID clinic can enhance the diagnostic process for EIMs in IBD patients, thereby reducing diagnostic delays and allowing early interventions to avoid disease progression.
Plain language summary
An outpatient model involving specialists in immune-mediated inflammatory diseases (IMIDs) – gastroenterologists, dermatologists, and rheumatologists – enhances the diagnostic process through early referral strategies and a multidisciplinary approach. This model promotes teamwork and improves communication, reducing diagnostic delays and enabling timely access to effective treatments, potentially minimising the risk of disease progression and thereby improving patient outcomes
Keywords
Introduction
Inflammatory bowel diseases (IBD), encompassing Crohn’s disease (CD) and ulcerative colitis (UC), are chronic immune-mediated inflammatory diseases (IMIDs) whose symptoms mainly affect the gastrointestinal tract.1,2 However, up to 47% of patients may exhibit extraintestinal manifestations (EIMs).3 –5 The factors contributing to EIMs development are not entirely understood. Still, an altered immune response driven by gut inflammation may facilitate leukocyte trafficking to other organs in genetically predisposed patients after exposure to environmental or microbial triggers.3,6
Managing EIMs remains challenging for clinicians due to considerable heterogeneity in diagnostic criteria as well as the lack of standardised and validated diagnostic pathways.7 –10 Despite numerous clinical screening tools and red flags 11 have been proposed for the diagnosis of EIMs in patients with IBD, 10 variability still persists depending on differences in guideline interpretation, clinical expertise across specialties and resource availability, and ultimately contributing to diagnostic delays. Delays can occur at various levels prior to diagnosis. Still, the most common causes include patients taking time to approach their physicians, misinterpreting signs and symptoms, and lengthy waiting times for specialists (system delays). 12 Consequently, diagnostic delays are associated with worse functional outcomes, greater structural progression, diminished quality of life and lower treatment response rates. 10
A multidisciplinary approach has the potential to significantly improve the management of EIMs by ensuring comprehensive care through collaboration between gastroenterologists, rheumatologists and dermatologists.13,14 However, little is known about whether early referral through a multidisciplinary approach has translated into more rapid treatment and disease control in routine care.
Therefore, we conducted a prospective study to assess the impact of dedicated IMIDs outpatient clinics – including specialist gastroenterologists, dermatologists and rheumatologists – on reducing diagnostic delays in EIMs and optimising therapeutic management, ultimately improving patient outcomes.
Patients and methods
We conducted a single-centre observational prospective study with a retrospective case-matched analysis involving patients aged ⩾18 years and diagnosed with IBD, confirmed in accordance with current diagnostic guidelines15,16 regularly followed up at the IBD Unit of ‘Federico II’ University Hospital. Consecutive eligible patients presenting with symptoms suggestive of red flags for EIMs,11,17,18 who were referred for outpatient evaluations at the dedicated IMIDs clinics between January 2018 and June 2022, were included. The IMID clinic is a dedicated outpatient service offering four clinic visits per week available and fixed, scheduled time slots for IBD patients presenting with rheumatological or dermatological EIMs. Gastroenterologists, rheumatologists and dermatologists collaborate, by sharing diagnostic and therapeutic approaches and discussing complex cases in multidisciplinary meetings (Figure 1).

Multidisciplinary outpatient clinic process.
A retrospective analysis was performed using clinic medical records from a historical database to identify a case-matched cohort with similar clinical characteristics regarding red flags for EIMs and features of IBD. All patients in the retrospective cohort had undergone conventional visits with individual referral specialists between January 2017 and June 2022.
The decision to refer patients to the IMID clinic was made by their treating gastroenterologists and based on patients’ preferences. Patients who required a more detailed assessment of potential EIMs were generally referred to the IMID clinic for a comprehensive evaluation. Cases and controls were matched based on the IBD diagnosis and the presence of red flags for EIMs, such as joint pain, skin lesions, uveitis or other symptoms suggestive of systemic involvement outside the gastrointestinal tract, ensuring that both groups had comparable clinical profiles without significant differences.
All patients were then followed up for 18 months.
Demographics and clinical data
Demographic, clinical data and medical history were obtained for each patient. CD clinical activity was evaluated according to the Harvey–Bradshaw Index (HBI) score, while UC clinical activity was assessed using the partial MAYO (pMAYO) score. Patients were considered clinically active if they had an HBI ⩾5 or a pMAYO ⩾2. Endoscopic activity was assessed through the Simple Endoscopic Score for Crohn’s Disease or Rutgeerts Score for CD and the Mayo Endoscopic Score for UC. Red flags for rheumatological and dermatological EIMs were identified based on the most updated expert consensus available at enrolment start.11,17,19,20 These red flags are signs or symptoms that may indicate a potential diagnosis of articular or dermatological EIMs in IBD patients, prompting timely referral to the appropriate clinical specialist (Supplemental Table 1).
EIM diagnosis was made by the specialist (either a rheumatologist or dermatologist) based on the latest version of specific guidelines21 –23; when necessary, specialists also recommended imaging or pathological assessments for further evaluation.
Clinical outcomes at 18 months included a switch or change in biologic therapy, systemic corticosteroids (CCS) administration at least once for EIMs or the addition of conventional immunosuppressants (ISS). The study flow is summarised in Figure 2.

Study flow.
Endpoints
The primary objective of the present study was to compare the diagnostic delay of EIMs between the IMIDs clinics group (IMID-G), attending dedicated IMIDs clinics and the standard outpatient clinic group (SOC-G), receiving conventional visits from individual referral specialists. Diagnostic delay, expressed in months, was defined as the interval between symptom onset and the confirmed diagnosis of EIMs. 24 Secondary endpoints included evaluating the impact of a multidisciplinary IMIDs clinic on clinical outcomes at 18 months, specifically regarding therapeutic changes, steroid and immunosuppressant use and switch/swap to biological therapy.
Statistical analysis
Continuous variables were expressed as mean with standard deviations or median with interquartile ranges (IQR), while categorical variables were as percentages and proportions. Chi-square, T-student or U Mann–Whitney test was calculated with a statistical significance threshold of p < 0.05. Survival curves with log-rank tests were plotted to evaluate the impact of each therapeutic path on diagnostic delay. Data were included in an Excel spreadsheet, and statistical analysis was performed using SPSS Statistics v.26 software (IBM – USA). All analyses were pre-specified in the study design to ensure consistency and reduce the risk of data-driven findings.
Ethics
The study was approved by the local Ethics Committee (cod.169/24) and conducted in accordance with the principles of the Declaration of Helsinki and good clinical practice. Drafting the current manuscript followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) 25 statements checklist (Supplemental Table 2).
Results
Baseline characteristics and time to diagnosis of EIMs
We enrolled 238 IBD patients, 145 (60.9%) of whom were female. The median age was 42 (IQR 32–54), and the median disease duration was 12 years (IQR 5–19). In all, 127 patients (53.4%) belonged to the IMID-G, and 111 (46.6%) to the SOC-G. Demographic and clinical characteristics are summarised in Table 1. The two groups did not differ regarding IBD features, UC extension, CD location and behaviour. However, IMID-G patients had a statistically significant longer disease duration (Δ = 60 months; p < 0.001). In addition, IMID-G had significantly higher HBI (Δ = 1; p = 0.003) values, and 45 (97.8%) patients were endoscopically active, compared to 20 (71.4%) in SOC-G (p < 0.001). UC clinical activity did not differ between the two groups (p = 0.32), while IMID-G had significantly lower rates of endoscopic activity (p = 0.02). With respect to therapy, at baseline, 66 (51.9%) IMID-G patients were under treatment with biological therapy, while 60 (54.1%) were in SOC-G (p = 0.74).
Baseline demographic characteristics of the two cohorts.
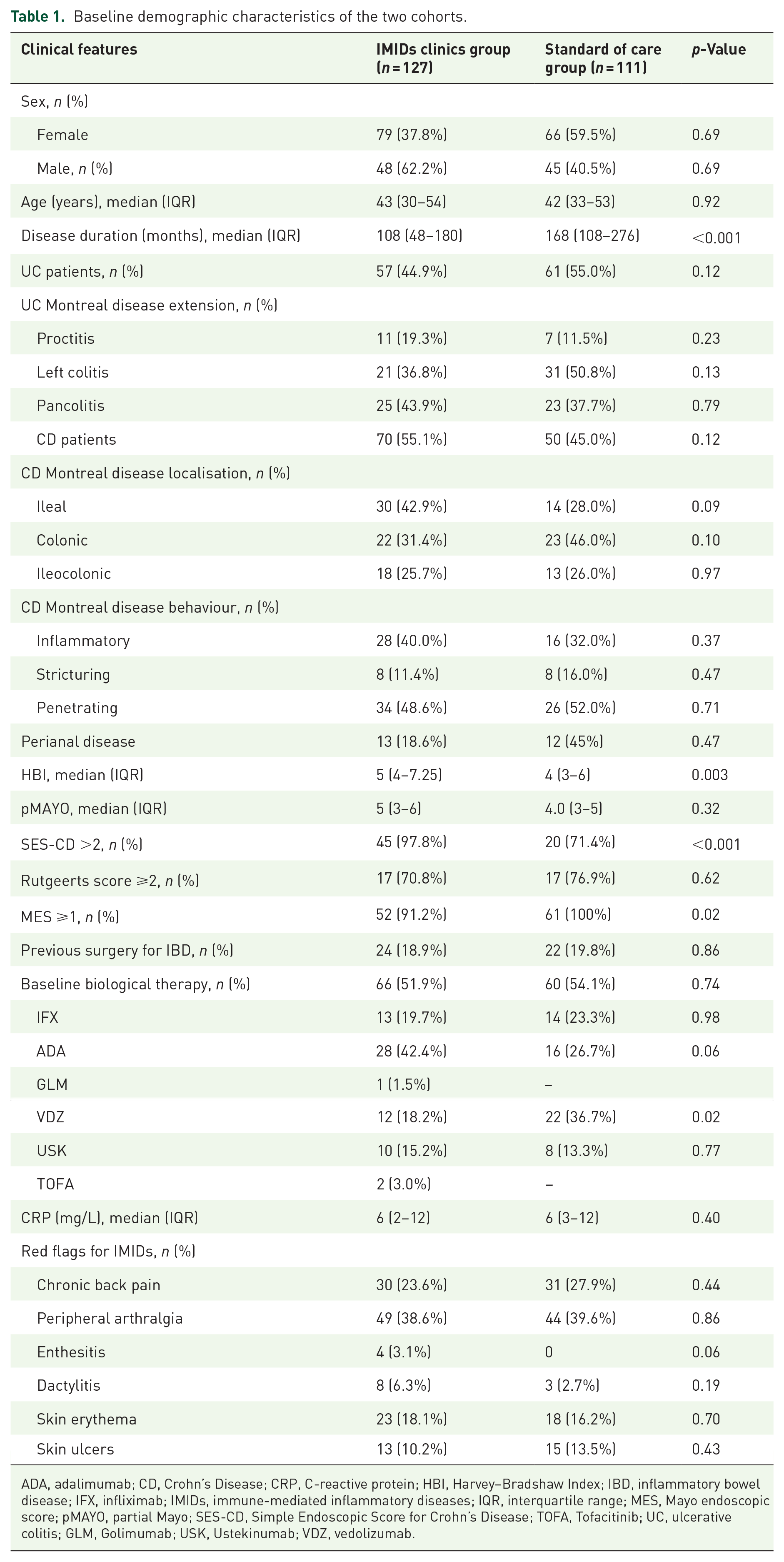
ADA, adalimumab; CD, Crohn’s Disease; CRP, C-reactive protein; HBI, Harvey–Bradshaw Index; IBD, inflammatory bowel disease; IFX, infliximab; IMIDs, immune-mediated inflammatory diseases; IQR, interquartile range; MES, Mayo endoscopic score; pMAYO, partial Mayo; SES-CD, Simple Endoscopic Score for Crohn’s Disease; TOFA, Tofacitinib; UC, ulcerative colitis; GLM, Golimumab; USK, Ustekinumab; VDZ, vedolizumab.
In both IMID-G and SOC-G, the most common red flags reported were peripheral arthralgia and chronic back pain. However, the time to diagnosis differed significantly between the two groups (p = 0.005), with a mean difference of Δ = 2.88 months. In both groups, the most common diagnoses were peripheral arthritis (PA), affecting 42 patients (37.5%) in the IMID group and 31 patients (33.7%) in the SOC-G group, and spondyloarthropathy (SpA), affecting 36 patients (32.1%) in the IMID group and 31 patients (33.7%) in the SOC-G group.
Clinical outcomes
After 18 months, 112 (88.2%) patients in IMID-G and 92 (82.8%) in SOC-G received a diagnosis of EIMs (p = 0.27). In relation to therapeutic changes, the IMID-G reported a mean time to the first therapeutic change driven by the specialist referral of 2.96 ± 1.8 months, compared to 6.09 ± 2.5 months in SOC-G, showing a significant difference (p = 0.007). Higher frequencies of biological therapy switch/swap and addition of ISS were observed in the IMID-G compared to the SOC-G (11.8% vs 4.5%, p = 0.008; 26.8% vs 17.3%, p = 0.04, respectively). Among the 15 (11.8%) patients in the IMID-G who switched or swapped therapy, 5 (33.3%) switched from adalimumab to infliximab, 7 (46.7%) swapped from vedolizumab to infliximab and 3 (20%) from ustekinumab to tofacitinib. Meanwhile, among the five (4.5%) patients in the SOC-G who changed therapy, two (40%) switched from adalimumab to infliximab, while three (60%) swapped from vedolizumab to adalimumab.
Conversely, the addition of CCS did not reach statistical significance despite being slightly more frequent in IMID-G (38.6% vs 44.1%, p = 0.69; Table 2). Survival curves showed a significantly higher cumulative rate of diagnosis and therapeutic change in IMID-G compared to SOC-G (log-rank test, respectively, p < 0.001 (Figure 3) and p < 0.001 (Figure 4)).
Outcomes at the end of follow-up.

CCS, corticosteroids; IMIDs, immune-mediated inflammatory diseases; ISS, immunosuppressive drugs; SD, standard deviation.
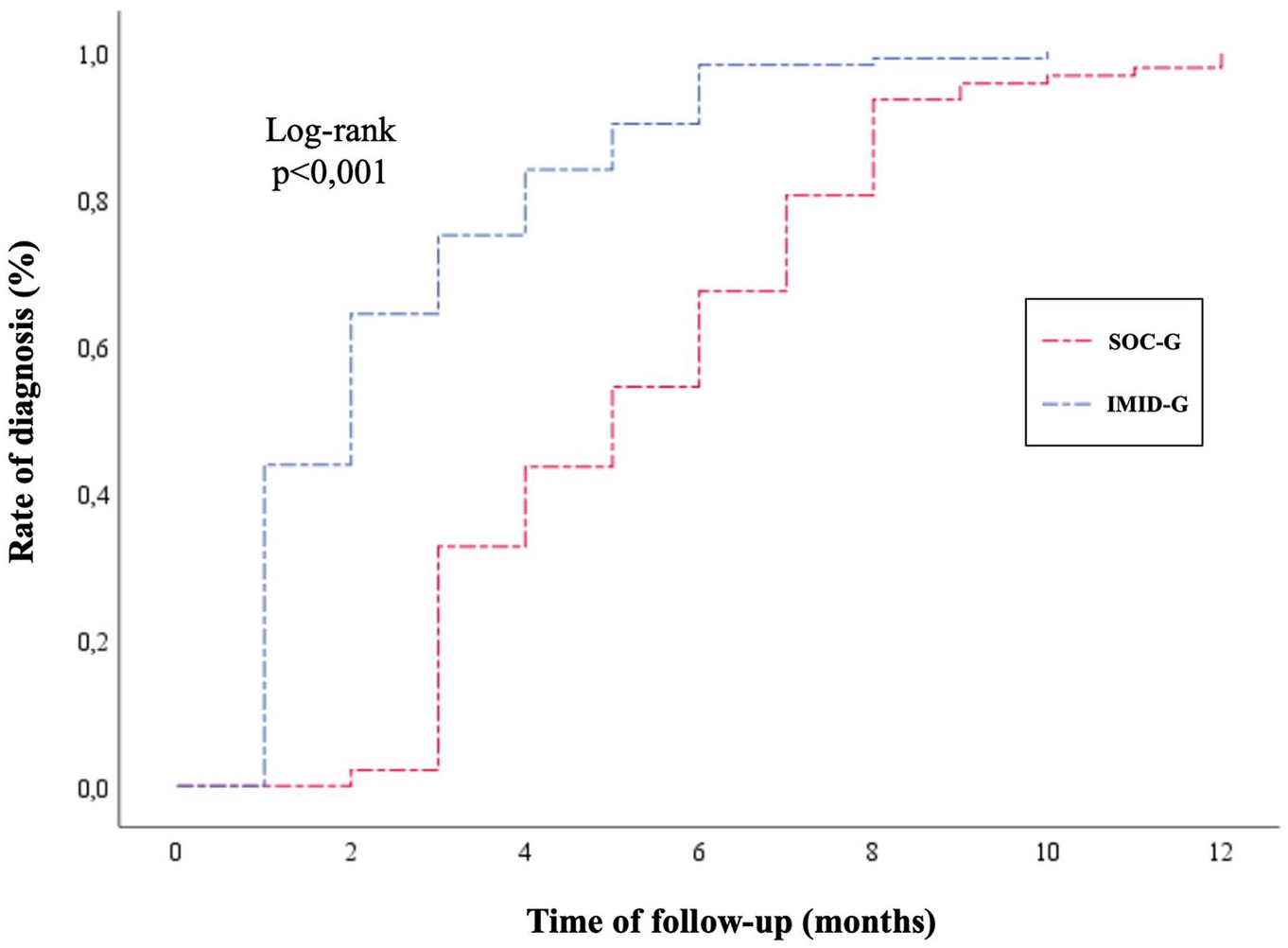
Kaplan–Meier survival curve for EIMs diagnosis in IMID-G and SOC-G.
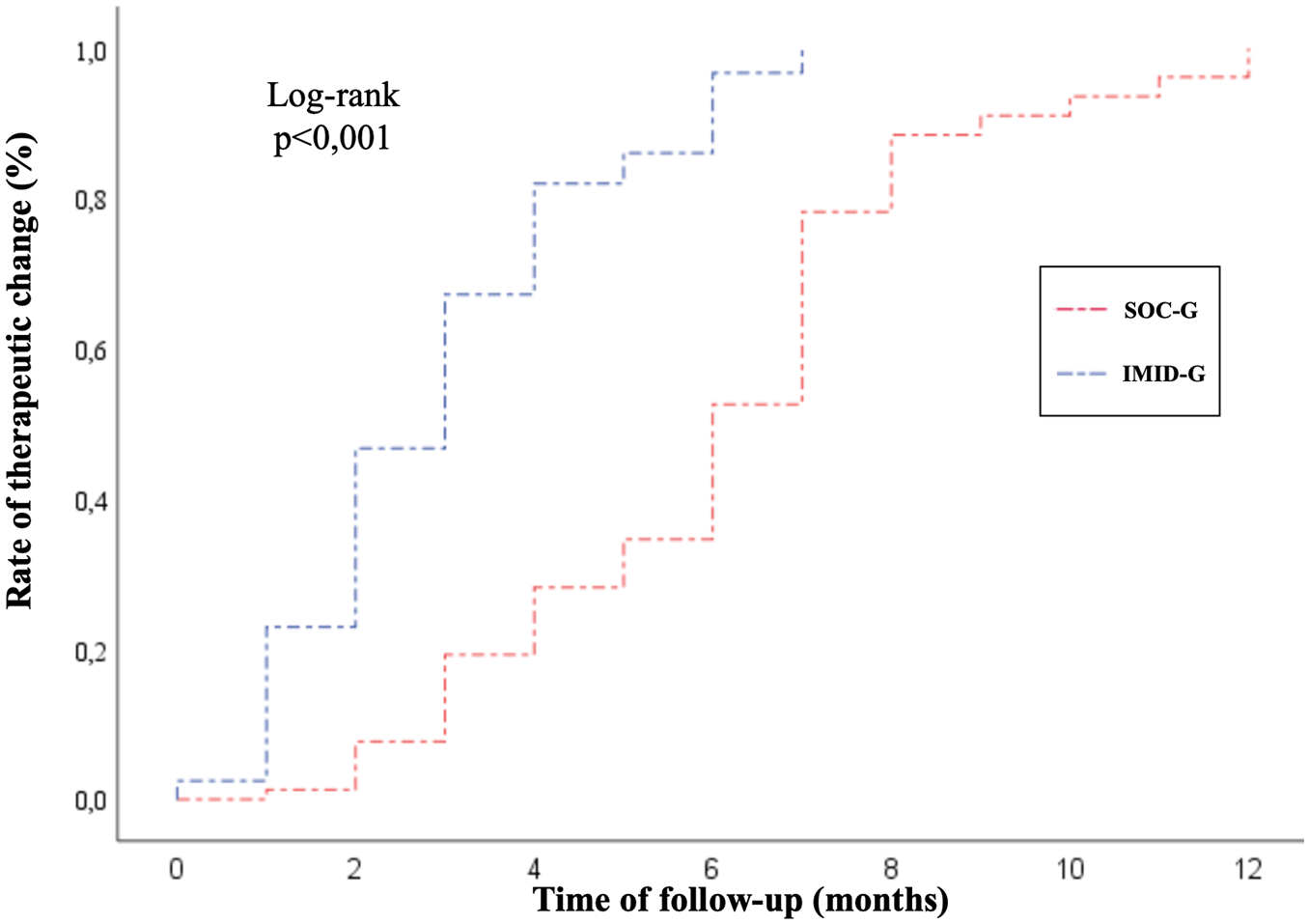
Kaplan–Meier survival curve for first therapeutic change in IMID-G and SOC-G.
Discussion
A standardised pathway for assessing and diagnosing IBD-EIMs is currently lacking. 10 However, the recent SPIRIT consensus from the International Organization for the Study of Inflammatory Bowel Diseases has included EIMs as an outcome measure in IBD disease-modification trials, recognising their importance in changing the disease course in the long term and blocking disease progression. 26 This emphasises the need for a standardised approach to diagnosis and assessment, achieved through multidisciplinary collaboration that integrates existing classification and consensus criteria, physician assessments and patient-reported symptoms.
In this study, we proposed a multidisciplinary outpatient clinic involving IMIDs specialists and evaluated its effectiveness in reducing diagnostic delays of EIMs compared to standard specialist referrals. Our findings show a significantly shorter diagnostic delay in the IMID-G compared to the SOC-G (2.48 ± 1.8 vs 5.36 ± 2.3, p = 0.005).
Notably, diagnostic delays may result in a missed window of opportunities for early treatment, which is particularly concerning given the impact of IMIDs on IBD suboptimal disease control and an increased risk of complications. 27
Thus, EIMs still represent a challenge in clinical practice. The occurrence of EIMs varies widely in both presentation and timing, with up to 25% of cases appearing before IBD diagnosis. 3 Factors such as ongoing therapy can further complicate the clinical picture. Some therapies, like anti-TNF treatment, may trigger extraintestinal diseases, such as psoriatic skin disease, and at the same time can also inhibit the development of such conditions.28 –30 These factors underscore the importance of thorough comprehensive evaluation and early referrals to specialists with the expertise necessary for the diagnostic process.
Accordingly, the IMID clinic has helped reduce patient waiting times, offer prompt access to diagnostic tools and enhance communication among specialists, enabling collaborative clinical decision-making. Established in 2018, it offers 4 visits/week. Given its fixed and scheduled availability, patients could only be seen during these time slots, reflecting the limited scheduling options and availability that patients referred to individual specialists face in the regular outpatient setting.
The most frequently diagnosed EIMs were articular, with PA and SpA being the most common in both groups, accounting for 32.1% of the IMID-G and 33.7% of the SOC-G.
Even though we did not find significant differences in the overall rate of EIMs diagnosis between the two groups by the end of follow-up, the time from symptom onset to the first therapeutic intervention by the specialist was significantly shorter in IMID-G compared to SOC-G (2.96 ± 1.8 vs 6.09 ± 2.5, p = 0.007). Indeed, the biological therapy switch rate and immunosuppressive agents’ introduction were higher in the IMID-G (11.8% vs 4.5%, p = 0.008; 26.8% vs 17.3%, p = 0.04, respectively). This implies that therapeutic adjustments were made more rapidly and effectively, which helped limit the disease course.
Of note, this may also be attributed to the expertise of specialists at tertiary care centres in managing advanced therapies, underscoring the necessity for dedicated structures and care pathways in IBD to ensure high-quality care.31,32
Recently, significant efforts have focused on standardising the use of quality indicators for IBD. 32 Coordinated care models, which integrate a multidisciplinary approach to structured clinical pathways for diagnosing, monitoring and treating IBD, are now considered the main standard of care. In this context, IMID multidisciplinary teams benefit from sharing management algorithms, ensuring effective management of this complex disease. Furthermore, it minimised care fragmentation and increased the speed of treatment initiation compared to individual referral specialists, who often face limitations in prescribing advanced therapies.
The concept of a dedicated clinic for IMIDs has been tested in other settings. For instance, in a prospective study by Luchetti et al. 33 involving 482 patients with psoriatic arthritis, a dermo-rheumatologic clinic achieved optimal disease management. In a gastroenterological context, a pragmatic trial conducted in 2021 34 explored the effects of an interdisciplinary combined clinic versus usual care for individuals with co-occurring IMIDs over a 6-month intervention and 6-month follow-up period, using the SF-36 Physical Component Summary as the primary outcome. At 6-month follow-up, patients in the dedicated clinic reported significantly better quality of life than those in the usual care group. However, the 24-week duration of the intervention may be insufficient to provide the full benefit and long-term therapeutic implications.
Noteworthy, our study has several strengths. It is the first to examine the impact of a multidisciplinary clinic on EIM diagnostic delays in an IBD context, including a large cohort of IBD patients and an extended follow-up period. Furthermore, the protocol’s high reproducibility suggests the potential to expand this approach to involve other specialists, such as ophthalmologists.
Nevertheless, it also has limitations. The study was conducted in a single tertiary centre specialising in IBD, which may not fully reflect the broader clinical practice encountered in more diverse or less specialised healthcare settings. Additionally, the lack of randomisation in assigning patients to the IMID-G and SOC-G groups could introduce bias. However, the groups were carefully selected to reflect real-world clinical practice closely.
The hybrid nature of the analysis using a historical case-matched cohort restricted our ability to assess the effects of treatments as dynamic immune modifiers and investigate specific outcomes, such as quality of life. The therapeutic landscape during the historical cohort’s treatment period may not fully reflect the treatment options available in the IMID-G, introducing a potential bias in comparing clinical outcomes between the two groups. Finally, the lack of standardised assessment criteria for evaluating the clinical activity of EIMs hindered a comprehensive evaluation of clinical outcomes in both groups. Indeed, we did not use formal scoring systems for EIMs, but we relied on clinical judgement and physician assessments of response to therapy and remission status for each EIM. While standardised assessment tools (such as those used for RA, psoriasis or SpA) are available and frequently utilised in tertiary care centres or specialised settings, their use is not as widespread or consistent in general practice or individual referral settings. In our study, patients in the SOC-G, who were seen through individual referrals, did not consistently undergo these standardised assessments because they were evaluated in the context of separate, independent specialities without a coordinated approach. We recognise that standardised outcome measures could provide more robust data. Yet, in the current study, the lack of consistent use of such assessments in the control group limited their applicability.
Conclusion
In conclusion, attendance at dedicated IMIDs clinics has been shown to improve the diagnostic process through an early referral strategy and a multidisciplinary approach that fosters teamwork and enhances communication among specialists. This, in turn, reduces diagnostic delays and facilitates an early use of highly effective treatment aimed at decreasing the impact of IBD on patients’ lives, reducing the risk of disease complications and potentially avoiding disease progression. Large-scale longitudinal data and randomised controlled trials, including patient-reported outcome measures, are awaited to validate the efficacy of an interdisciplinary combined clinic in daily practice. In addition, future studies would benefit from incorporating more structured assessments of EIMs or specific validated scoring systems for individual EIMs. These could provide a clearer, more standardised way to evaluate whether the IMID clinic model truly improves outcomes, both from the perspective of the treating physician and the patient and ultimately its long-term impact on structural damage.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251323529 – Supplemental material for Reducing diagnostic delays of extraintestinal manifestations in inflammatory bowel disease: a comparative study of a multidisciplinary outpatient clinic versus conventional referral specialists
Supplemental material, sj-docx-1-tag-10.1177_17562848251323529 for Reducing diagnostic delays of extraintestinal manifestations in inflammatory bowel disease: a comparative study of a multidisciplinary outpatient clinic versus conventional referral specialists by Olga Maria Nardone, Giulio Calabrese, Alessia La Mantia, Guido Daniele Villani, Matteo Megna, Sara Cacciapuoti, Francesca Foglia, Rosario Peluso, Ermelinda D’Alessandro, Mario Ferrante, Anna Testa, Alessia Dalila Guarino, Antonio Rispo and Fabiana Castiglione in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251323529 – Supplemental material for Reducing diagnostic delays of extraintestinal manifestations in inflammatory bowel disease: a comparative study of a multidisciplinary outpatient clinic versus conventional referral specialists
Supplemental material, sj-docx-2-tag-10.1177_17562848251323529 for Reducing diagnostic delays of extraintestinal manifestations in inflammatory bowel disease: a comparative study of a multidisciplinary outpatient clinic versus conventional referral specialists by Olga Maria Nardone, Giulio Calabrese, Alessia La Mantia, Guido Daniele Villani, Matteo Megna, Sara Cacciapuoti, Francesca Foglia, Rosario Peluso, Ermelinda D’Alessandro, Mario Ferrante, Anna Testa, Alessia Dalila Guarino, Antonio Rispo and Fabiana Castiglione in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
