Abstract
Background:
As an uncommon serological pattern, the effect of hepatitis B e antigen/anti-hepatitis B e (HBeAg/anti-HBe) coexistence on peginterferon-α (Peg-IFN-α) therapy in patients with chronic hepatitis B (CHB) remains unknown. Moreover, Peg-IFN-α is clinically limited due to several side effects. It is of significant value to early identify the favored population for Peg-IFN-α therapy in CHB patients.
Objectives:
This study aimed to analyze the impact of HBeAg/anti-HBe coexistence on the effectiveness of Peg-IFN-α and to construct a nomogram for predicting the occurrence of HBeAg seroconversion in treatment-naïve CHB patients with Peg-IFN-α therapy.
Design:
Retrospective, case–control study of treatment-naïve CHB patients with Peg-IFN-α at a tertiary care center.
Methods:
Data from HBeAg-positive treatment-naïve CHB patients were retrospectively analyzed. Clinical characteristics of the HBeAg/anti-HBe coexistence group were compared with those of the anti-HBe-negative group. In addition, univariate and multivariate logistic regression analyses were performed to identify independent risk factors for HBeAg seroconversion. The nomogram for the prediction of HBeAg seroconversion was constructed and evaluated.
Results:
A total of 140 HBeAg-positive CHB patients were enrolled. Patients with HBeAg/anti-HBe coexistence accounted for 11.4% of HBeAg-positive patients, and their hepatitis B surface antigen (HBsAg) and HBeAg levels were significantly lower than those of anti-HBe negative patients, but the HBeAg seroconversion rate was higher after Peg-IFN-α treatment. As revealed by multivariate logistic analysis, HBeAg/anti-HBe coexistence, baseline HBsAg, baseline HBeAg, and alanine aminotransferase ratio at baseline were independent correlates of HBeAg seroconversion. The nomogram model constructed based on these four independent correlates demonstrated good discrimination (area under the curve = 0.866), calibration, and clinical adaptability.
Conclusion:
HBeAg/anti-HBe coexistence is associated with a higher HBeAg seroconversion rate, and the nomogram model constructed based on HBeAg/anti-HBe coexistence performs well in predicting HBeAg seroconversion in treatment-naïve CHB patients treated with Peg-IFN-α therapy.
Introduction
Chronic hepatitis B (CHB) is an important public health problem, and approximately 257 million people worldwide are chronically infected with the hepatitis B virus (HBV). 1 It is estimated that there are 20–30 million cases of CHB in China, 2 and persistent infection can lead to a range of end-stage liver diseases, including cirrhosis, liver failure, and hepatocellular carcinoma (HCC). Currently, the first-line therapeutic agents for CHB mainly include nucleos(t)ide analogs (NAs) and peginterferon (Peg-IFN). Compared with NAs, Peg-IFN exhibits more potent and longer-lasting efficacy by directly inhibiting HBV viral replication and enhancing host immunity. In addition, Peg-IFN-α can mitigate the risk of cirrhosis and progression to HCC. 3 However, the efficacy of Peg-IFN remains constrained, with a hepatitis B e antigen (HBeAg) seroconversion rate of approximately 30%–40% and a hepatitis B surface antigen (HBsAg) clearance rate of less than 10% post-treatment. 4 Moreover, its clinical utility is limited by side effects such as peripheral hematopoiesis and flu-like symptoms, with early discontinuation reported in up to 20% of CHB cases. 5 Hence, early identification and screening of suitable candidates for Peg-IFN therapy are of crucial significance.
Simultaneous positivity for HBeAg and anti-hepatitis B e (anti-HBe) represents an atypical serological pattern in HBV infection, which is often viewed as a transitional stage from HBeAg(+) alone to anti-HBe(+) alone status. Moreover, this phenomenon may also be influenced by factors including detection technique sensitivity and gene locus mutations.6,7 While several studies have reported the prevalence of HBeAg/anti-HBe coexistence,7,8 few researches have investigated its impact on the antiviral therapy for CHB patients, particularly with regard to the Peg-IFN efficacy. HBeAg seroconversion, defined as the loss of HBeAg accompanied by the appearance of anti-HBe, is pivotal in HBeAg-positive patients and is a prerequisite for achieving clinical cure.8,9 Long-term studies have demonstrated that HBeAg seroconversion is correlated with reduced risks of cirrhosis, HCC, and liver-related mortality, as well as an increased likelihood of HBsAg clearance. 10 Despite various indicators reported to predict HBeAg seroconversion, such as low baseline HBsAg, quantitative hepatitis B core antibody, and hepatitis B core related antigen,11–13 these predictors often rely on technically demanding and costly methods, restricting their widespread clinical utility.
In this study, we retrospectively analyzed the clinical characteristics of treatment-naïve HBeAg-positive CHB patients to elucidate the impact of HBeAg/anti-HBe coexistence on HBeAg seroconversion during Peg-IFN therapy. Based on these findings, a nomogram model capable of predicting HBeAg seroconversion was constructed and evaluated, aiming to enhance the application of Peg-IFN for HBeAg-positive CHB patients.
Materials and methods
Study population and design
Nine hundred ninety CHB patients who received Peg-IFN-α (180 μg/week) from January 2018 to December 2023 at Fujian Medical University Affiliated First Quanzhou Hospital were retrospectively screened. The inclusion criteria were as follows: (1) the presence of serum HBsAg for 6 months or longer and HBeAg positivity; (2) aged 18–60 years; (3) treatment-naïve; and (4) a Peg-IFN-α treatment duration of 48 weeks with complete follow-up data. The exclusion criteria included the following: (1) co-infection with hepatitis A virus, hepatitis C virus, hepatitis D virus, hepatitis E virus, or human immunodeficiency virus; (2) discontinuation or change of the interferon treatment regimen; (3) history of comorbid liver cirrhosis, hyperthyroidism, goiter, autoimmune hepatitis, pregnancy, or any type of tumor; and (4) incomplete data or information from relevant examinations (Figure S1).
According to the Guidelines for the Prevention and Treatment of CHB (version 2022), 14 HBeAg seroconversion was defined as the transition from HBeAg positivity to HBeAg negativity, accompanied by the presence of anti-HBe. Based on the outcome after 48 weeks of Peg-IFN-α therapy, patients were categorized into two groups, namely, HBeAg seroconversion (SC) and without HBeAg seroconversion (without-SC) groups. Patients with baseline anti-HBe positivity were classified as having HBeAg/anti-HBe coexistence. This study was approved by the Ethics Committee of Biomedical Research Affiliated to Fujian Medical University (No. 2014-87). The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist for case–control studies was used to prepare the manuscript (Supplemental Table 1). 15
Data collection
General patient information included gender, age, use of NAs, and other relevant factors. HBV serological markers were assessed using the Architect i2000 SR platform and Abbott Architect reagents (Abbott Laboratories, Chicago, IL, USA). Serum HBV DNA levels were quantified via PCR using the Roche COBAS AmpliPrep/COBAS TaqMan system (Roche Diagnostics, Mannheim, Germany). Following initiation of Peg-IFN-α therapy, measurements of HBsAg, anti-HBs, HBeAg, anti-HBe, anti-HBc, HBV DNA, alanine aminotransferase (ALT), aspartate aminotransferase, albumin, globulin, total bilirubin, alkaline phosphatase, and gamma-glutamyl transferase were recorded at 12, 24, 36, and 48 weeks. The ALT ratio refers to the value of ALT divided by the upper limit of normal.
Statistical analysis
Using the SPSS 27.0 (IBM Corporation, USA) and R software 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria), statistical analysis was completed. Measurement data that followed a normal distribution were presented as mean ± standard deviation, and comparisons between groups were conducted using two independent samples t-tests. Measurement data that were not normally distributed were expressed as median (M) and interquartile range (M, P25, P75), with between-group comparisons made using the non-parametric Mann–Whitney U test. Count data were presented as frequencies or percentages (%), and comparisons were performed using the Chi-square test or Fisher’s exact probability method as appropriate. The cumulative HBeAg seroconversion rate was analyzed using Kaplan–Meier analysis and compared by the Log-rank test. Univariate and multivariate logistic regression analyses were conducted to identify independent risk factors for HBeAg seroconversion. The finally selected relevant factors were integrated into a nomogram. Receiver operating characteristic (ROC) curves were utilized to assess the discriminatory ability of the model. Calibration curves were plotted to evaluate the consistency between observed and predicted probabilities. Decision curve analysis (DCA) was employed to evaluate the net clinical benefit of the nomogram model. A significance level of p < 0.05 was considered statistically significant.
Results
Demographic characteristics
In this study, 140 treatment-naïve HBeAg-positive patients aged 18–60 years were enrolled, including 94 males. Among them, 59 patients received Peg-IFN-α therapy alone, and 16 patients exhibited HBeAg/anti-HBe coexistence. After 48 weeks of Peg-IFN-α therapy, 41 patients achieved HBeAg seroconversion, resulting in an HBeAg seroconversion rate of 29.3%. The SC and without-SC groups were compared, which revealed significantly lower baseline levels of HBsAg, HBeAg, and HBV DNA in the SC group. Meanwhile, there was a higher proportion of patients with HBeAg/anti-HBe coexistence in the SC group. However, age, gender, treatment strategy, baseline anti-HBs, baseline anti-HBc, or baseline hematological profile were not significantly different between the two groups (Table 1). During Peg-IFN-α therapy, the hematological profile generally underwent a more pronounced decline within 4 weeks and then showed a stable trend after 4 weeks. Besides, the counts of white blood cells, neutrophils, hemoglobin, and platelets were similar between the two groups at different time points (Figure S2).
Demographic, clinical, and laboratory characteristics of the HBeAg-positive CHB patients regarding HBeAg seroconversion.

HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HBV-DNA, hepatitis B virus-deoxyribonucleic acid; NAs, nucleos(t)ide analogs; SC, HBeAg seroconversion group; without-SC, without HBeAg seroconversion.
Comparison of clinical characteristics between anti-HBe(+) and anti-HBe(−) patients
Patients positive for anti-HBe, or those with HBeAg/anti-HBe coexistence, accounted for 11.4% of HBeAg-positive patients. Compared with patients negative for anti-HBe, anti-HBe(+) patients showed significantly lower levels of HBsAg and HBeAg, and a higher proportion of them received Peg-IFN-α combined with NAs treatment. Following Peg-IFN-α therapy, HBeAg/anti-HBe coexistence was associated with a significantly higher HBeAg seroconversion rate (Table 2). Kaplan–Meier survival analysis and the Log-rank test (χ² = 16.9, p < 0.001) indicated a significantly higher cumulative HBeAg seroconversion rate in patients with HBeAg/anti-HBe coexistence (Figure 1). Age, gender, baseline HBV DNA level, baseline ALT, and AST levels were similar between the two groups of patients.
Comparison of baseline clinical and laboratory characteristics between anti-HBe (+) and anti-HBe (−) patients.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HBV-DNA, hepatitis B virus-deoxyribonucleic acid; NAs, nucleos(t)ide analogs.
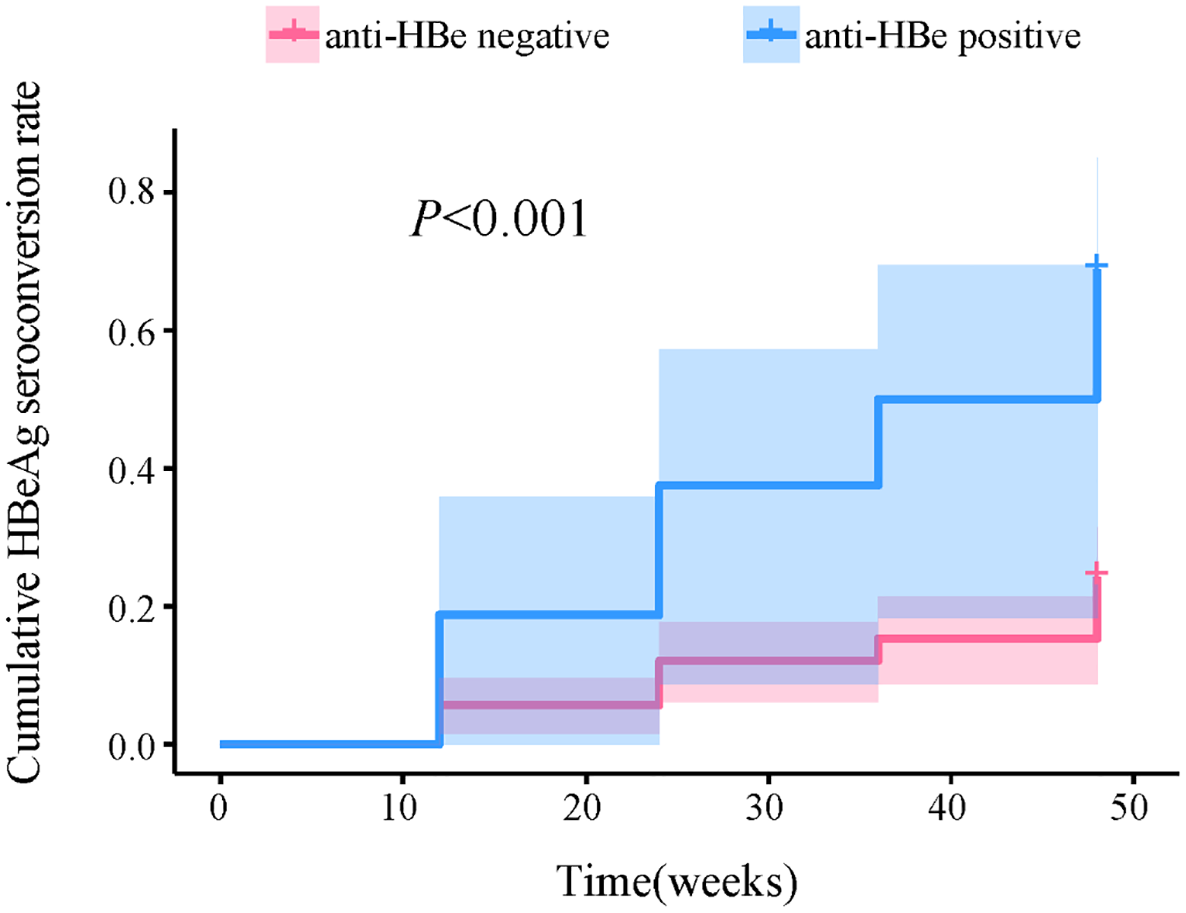
Comparison of cumulative HBeAg seroconversion rate between anti-HBe positive and anti-HBe negative group. Anti-HBe positive group refers to HBeAg/anti-HBe coexistence; anti-HBe negative group refers to without HBeAg/anti-HBe coexistence.
Construction of the nomogram model for predicting HBeAg seroconversion
To facilitate clinical utility, continuous variables including HBsAg, HBeAg, and HBV DNA were categorized into multiple categories. Univariate logistic regression analysis was initially conducted on baseline clinical indicators, followed by the inclusion of statistically significant factors (HBsAg, HBeAg, HBeAg/anti-HBe coexistence, HBV DNA, and ALT ratio) into multivariate logistic regression analyses. The results indicated that baseline HBsAg (odds ratio (OR) = 0.080, p = 0.038), baseline HBeAg (OR = 0.085, p = 0.036), HBeAg/anti-HBe coexistence (OR = 4.222, p = 0.027), and ALT ratio at baseline (OR = 2.451, p < 0.001) were independent correlates of HBeAg seroconversion (Table 3). Finally, the nomogram predicting HBeAg seroconversion for CHB patients was constructed based on these four baseline indicators, assigning a total score of 130 (Figure 2).
Univariate and multivariate analyses for the prediction of HBeAg seroconversion.

ALB, albumin, globulin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HBV-DNA, hepatitis B virus-deoxyribonucleic acid; NAs, nucleos(t)ide analogs; TBil, total bilirubin; ULN, upper limit of normal.

Nomogram for prediction of HBeAg seroconversion in treatment-naïve CHB patients with peginterferon-α.
Evaluation of the nomogram model
The predictive model was evaluated for its discrimination, calibration, and clinical validity. The ROC curve demonstrated an area under the curve (AUC) of 0.866, with a sensitivity of 85.4% and a specificity of 77.8%. Moreover, internal validation through 1000 bootstrap samples confirmed an AUC of 0.866, indicating robust discriminative capability (Figure 3). Furthermore, calibration curves illustrated good agreement between predicted and observed probabilities, with the Hosmer–Lemeshow goodness-of-fit test yielding χ² = 12.788, p = 0.119, suggesting adequate model calibration (Figure 4(a)). Also, DCA indicated substantial net clinical benefit across a range of threshold probabilities, enhancing the model’s clinical applicability (Figure 4(b)).

ROC curves for validating the discrimination power of the nomogram.

(a) An analysis of calibration curves for the nomogram prediction in the cohort. (b) Decision curve analysis of the nomogram in the cohort.
Discussion
HBeAg seroconversion is critical for the treatment and management of HBeAg-positive patients with CHB. HBeAg positivity signifies active HBV replication and is associated with a heightened risk of HCC. It has been reported that the relative risk of HCC in HBsAg-positive patients is 9.6%, which increases to 60.2% in those who are also HBeAg positive. 16 Hence, it is essential to monitor HBeAg status and recognize HBeAg seroconversion during CHB treatment. In our study, the incidence of HBeAg/anti-HBe coexistence was observed in 11.4% of HBeAg-positive CHB patients, consistent with previous reports by Mutimer et al. 8 After 48 weeks of Peg-IFN-α therapy, these patients exhibited a significantly higher HBeAg seroconversion rate than those negative for anti-HBe. Overall, 29.3% of CHB patients achieved HBeAg seroconversion by the end of treatment. Through univariate and multivariate logistic regression analyses of baseline clinical characteristics, HBeAg/anti-HBe coexistence, HBsAg, HBeAg, and ALT ratio were identified as the independent factors correlated with HBeAg seroconversion. Unlike previous studies, our research uniquely assessed the impact of HBeAg/anti-HBe coexistence on HBeAg seroconversion in treatment-naïve CHB patients with Peg-IFN-α therapy. A nomogram model was developed based on these factors, including HBeAg/anti-HBe coexistence, to predict HBeAg seroconversion. Upon evaluation, the model demonstrated favorable discrimination, calibration, and clinical applicability. This tool may assist clinicians in optimizing the management of CHB patients, thereby reducing unnecessary medical resource utilization and enhancing patient compliance.
The particular pattern of simultaneous HBeAg/anti-HBe positivity is not very common in clinical practice. During the course of HBV infection, with the dynamics of viral replication and body immunity, HBeAg levels tend to decrease, while anti-HBe begins to be produced and gradually increases. It is hypothesized that simultaneous positivity for both HBeAg and anti-HBe can be detected when the levels of HBeAg and anti-HBe are at a particular optimal ratio. 6 Moreover, different HBV gene subtypes may also have an impact on the formation of HBeAg/anti-HBe coexistence, with one study noting that subgenotype B2 is more common in patients with HBeAg/anti-HBe coexistence than those with genotype C. 17 Xue et al. 18 observed that patients with HBeAg/anti-HBe coexistence had significantly lower serum HBsAg levels, HBeAg levels, and HBV DNA levels, and a higher HBeAg seroconversion rate. Similarly, our cohort of patients with HBeAg/anti-HBe coexistence demonstrated a higher cumulative HBeAg seroconversion rate during Peg-IFN-α therapy. In addition, multivariate logistic regression analysis revealed that HBeAg/anti-HBe coexistence was an independent correlate of HBeAg seroconversion. However, the mechanism underlying the association of HBeAg/anti-HBe coexistence with HBeAg seroconversion remains unclear. It has been well known that the therapeutic efficacy of CHB, as a virus-host interaction disease, is not only related to the suppression of viral replication but also to the immune status of the patient’s body. Therefore, it may be ascribed to the formation of immune complexes between HBeAg and anti-HBe, and the deposition of these antigen–antibody immune complexes may stimulate the immune system response. 7 This process may also involve changes in immune cells, particularly cytokines secreted by CD8+ T lymphocytes, which are effective in suppressing HBV infection and thus promoting HBeAg seroconversion.19,20 However, this explanation only partially elucidates the observed results, and further studies are warranted to explore the exact mechanism of action regarding HBeAg/anti-HBe coexistence.
Several previous studies have reported a correlation of HBsAg or HBeAg with HBeAg seroconversion. Fried et al. 21 compared the effectiveness of HBeAg and HBV-DNA in predicting HBeAg seroconversion in patients treated with Peg-IFN-α. Among the 87 patients who achieved HBeAg seroconversion, the levels of HBeAg decreased during treatment and remained minimal during follow-up. The ROC curves were plotted, which indicated that quantitative HBeAg was a superior predictor of HBeAg seroconversion compared with HBV-DNA. Our study also found that baseline HBeAg levels (OR = 0.085, p = 0.036) were independently correlated with HBeAg seroconversion after 48 weeks of Peg-IFN-α treatment. In a prospective, multicenter study 13 of CHB patients treated with Peg-IFN-α for 48 weeks, the investigators concluded that HBsAg was an independent risk factor for HBeAg seroconversion (OR = 0.1704, p < 0.0001), consistent with our findings. In addition, we observed a correlation between baseline ALT levels and HBeAg seroconversion, with higher baseline ALT levels being associated with an increased likelihood of achieving HBeAg seroconversion, aligning with several prior reports.12,22 The mechanism of ALT in affecting HBeAg seroconversion may involve the elevated ALT levels that reflect host immune-mediated viral clearance, 23 accompanied by changes in cytokines and chemokines associated with antiviral resistance, such as the increased levels of C-X-C motif chemokine ligand 13 favoring HBV clearance in CHB patients. 24
There are several limitations in our study. First, it was a single-center retrospective study, which might have led to data omissions and other circumstances, such as missing data on lipid and liver stiffness tests, precluding their inclusion in statistical analysis. In addition, the constructed nomogram model requires further validation before its application in other hospitals can be considered reliable. Second, the sample size of our study cohort was relatively small, so the credibility of our results should be further verified. Moreover, the nomogram was internally validated only, lacking external validation. The assessment of results may therefore be biased, highlighting the need for more future large-scale, multicenter studies to assess the predictive efficacy of the model. Furthermore, the mechanism underlying the effect of HBeAg/anti-HBe coexistence on HBeAg seroconversion remains unclear and warrants exploration through further basic experiments. Finally, given that all participants received antiviral therapy, the rate of spontaneous HBeAg seroconversion in patients and the effect of Peg-IFN-α on it could not be evaluated. We did not analyze HBV genotypes, and the applicability of our nomogram model to CHB patients with different genotypes requires validation.
Conclusion
In summary, among treatment-naïve HBeAg-positive CHB patients, those with HBeAg/anti-HBe coexistence exhibit a higher rate of HBeAg seroconversion following Peg-IFN-α therapy. The nomogram model, incorporating HBeAg/anti-HBe coexistence, baseline HBsAg, baseline HBeAg, and ALT ratio at baseline, demonstrates good predictive performance, enabling early individualized assessment of the likelihood of achieving HBeAg seroconversion in CHB patients treated with Peg-IFN-α.
Supplemental Material
sj-doc-1-tag-10.1177_17562848251318037 – Supplemental material for Effect of HBeAg/anti-HBe coexistence on HBeAg seroconversion in treatment-naïve chronic hepatitis B patients with peginterferon-α
Supplemental material, sj-doc-1-tag-10.1177_17562848251318037 for Effect of HBeAg/anti-HBe coexistence on HBeAg seroconversion in treatment-naïve chronic hepatitis B patients with peginterferon-α by Kaimin Song, Huitong Wang, Dawu Zeng, Yunyun Qian, Zhixiang Guo, Huatang Zhang, Yijuan Zheng, Yongjun Zhou, Xueping Yu and Zhijun Su in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251318037 – Supplemental material for Effect of HBeAg/anti-HBe coexistence on HBeAg seroconversion in treatment-naïve chronic hepatitis B patients with peginterferon-α
Supplemental material, sj-docx-2-tag-10.1177_17562848251318037 for Effect of HBeAg/anti-HBe coexistence on HBeAg seroconversion in treatment-naïve chronic hepatitis B patients with peginterferon-α by Kaimin Song, Huitong Wang, Dawu Zeng, Yunyun Qian, Zhixiang Guo, Huatang Zhang, Yijuan Zheng, Yongjun Zhou, Xueping Yu and Zhijun Su in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
