Abstract
Background:
The comparable evaluation of computed tomography enterography (CTE), enteroscopy, and intestinal ultrasound in small bowel Crohn’s disease (CD) is imprecise.
Objectives:
The purpose of this study was to analyze the findings of enteroscopy, CTE, and intestinal ultrasound to determine the advantages and disadvantages of each method for the evaluation of small bowel CD.
Design:
It was a single-center, observational, retrospective study.
Methods:
The differences in localization of disease lesions, mucosal inflammation, and transmural inflammation between enteroscopy, CTE, and intestinal ultrasound for evaluation of small bowel CD were compared.
Results:
A total of 198 patients with small bowel CD were included in the analysis. CTE and intestinal ultrasound had a lower detection rate of upper intestinal lesions compared with enteroscopy (p < 0.05). Enteroscopy was better than CTE and intestinal ultrasound in the detection of stenosis (p < 0.001), and the assessment of fistula by CTE was better than that by enteroscopy and intestinal ultrasound (p < 0.05). Enteroscopy, CTE, and intestinal ultrasound differed in the assessment of inflammatory activity, and the agreement of the three methods was poor (all intra-class correlation coefficient <0.75).
Conclusion:
Enteroscopy is superior to CTE and intestinal ultrasound for the assessment of upper intestinal CD lesions. Enteroscopy, CTE, and intestinal ultrasound were not consistent in evaluating inflammatory activity, and the three methods may need to be combined for an accurate assessment.
Plain language summary
Enteroscopy is superior to CTE and intestinal ultrasound for the assessment of upper intestinal lesions. About inflammatory activity, the three need to be combined and complement each other to achieve a comprehensive assessment of CD patients and play a better role in guiding the clinic.
Introduction
Crohn’s disease (CD) is a chronic inflammatory bowel disease that can affect any part of the gastrointestinal tract. 1 Approximately 30% of patients exhibit isolated small bowel involvement. 2 The anatomical complexities and structural limitations of the small intestine, including its length and intricate curves, pose significant challenges in accurately diagnosing small bowel CD. 3 As a result, there is a high rate of underdiagnosis, which is independently correlated with poorer outcomes. 4 Delays in diagnosis can lead to complications that require surgery and increased healthcare costs, emphasizing the critical need for timely detection and effective management of this condition.
Advances in endoscopic techniques and imaging methods, such as dedicated capsule endoscopy, have improved the detection of CD; however, each diagnostic method has substantial limitations. 5 Enteroscopy is highly effective for visualizing lesions in the proximal small intestine; however, it is invasive and its reach is limited to the proximal small intestine. 6 Computed tomography enterography (CTE) offers broader visualization but exposes patients to ionizing radiation and subtle inflammatory changes are difficult to identify. 7 Intestinal ultrasound provides a radiation-free alternative, but has lower sensitivity and specificity compared to the other methods, particularly for visualizing deeper lesions and complications. 8
Therapeutic goals for CD have evolved to prioritize not only clinical remission and endoscopic mucosal healing but also transmural healing which is essential for improving patient outcomes. 9 Despite the increasing incidence of small bowel CD and the recognition of its unique challenges, comparative studies that evaluate the strengths and weaknesses of the three aforementioned imaging methods in a systematic manner are scarce. This study aims to bridge this gap by conducting a comprehensive comparison of enteroscopy, CTE, and intestinal ultrasound in detecting lesions, assessing inflammatory activity, and identifying complications across different segments of the small intestine in patients with small bowel CD.
Materials and methods
Patients
This was a retrospective, observational study. Case data of patients hospitalized at the Sixth Hospital of Sun Yat-sen University from April 2020 to October 2022 were retrospectively analyzed. This study was approved by the Medical Ethics Committee of the Sixth Affiliated Hospital of Sun Yat-sen University (No. 2022278). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 10
Inclusion criteria
Patients who met the diagnostic criteria for CD in the American College of Gastroenterology guidelines and were diagnosed with small bowel CD were eligible for inclusion. Included patients were required to have undergone enteroscopy, CTE, and intestinal ultrasound with an interval of <2 weeks between CTE and intestinal ultrasound, and <4 weeks between CTE and intestinal ultrasound, and enteroscopy. An interval of <2 weeks between CTE and intestinal ultrasound, and <4 weeks between these imaging methods and enteroscopy was based on clinical guidelines and logistical considerations. This timeframe minimizes the influence of treatment response variability, as inflammatory activity in CD typically does not show significant variation within such a short period. This interval was therefore deemed appropriate to ensure comparability across assessments.
Enteroscopy was indicated for all patients due to findings from gastroscopy that suggested duodenal inflammation, coupled with imaging and other examinations that could not exclude the possibility of small bowel lesions. This was particularly important given the potential diagnosis of L4-type or small bowel CD.
Exclusion criteria
Exclusion criteria were as follows: (1) The presence of other immune-related diseases; (2) serious cardiopulmonary or other conditions; (3) and did not receive all three examinations (enteroscopy, CTE, and intestinal ultrasound).
Examination methods and observations
Double-balloon enteroscopy
After the patient was anesthetized, oral and anal enteroscopy was performed by the same experienced endoscopist to evaluate the whole intestine. The depth of entry was estimated based on the depth of entry in endoscopy, 11 and the small intestine was divided into seven segments: duodenum, upper jejunum, lower jejunum, upper ileum, median ileum, lower ileum, and distal ileum. The endoscopic observation indices included the following: (1) Mucosal inflammation: congestion, edema, erosion, ulceration; (2) morphology of intestinal lumen villi and folds; (3) distributions and characteristics of blood vessels in the intestinal lumen; and (4) CD-related complications: fistula, stenosis, hemorrhage. 12 Stenosis was defined as a narrowing that prevents passage of the endoscope through the intestinal lumen. For the endoscopic assessment of small bowel mucosal inflammation, reference was made to the colonic Simple Endoscopic Score (SES-CD) scoring standard. Due to the absence of a standardized scoring system for assessing small intestine activity during enteroscopy, the Intestinal Enteroscopy Activity Score (IEAS-CD) was developed specifically for this study, modeled after the SES-CD scoring system for colonic CD. 13 In this system, the presence of ulcers serves as the primary indicator of disease activity in each intestinal section. Within endoscopic scoring frameworks, ulcers are consistently recognized as a key marker of mucosal inflammation. The IEAS-CD score was based on the presence and size of ulcers: 0 points, no ulcers; 1 point, disseminated aphthous ulcers; 2 points, larger ulcers with a diameter of 0.5–2.0 cm; 3 points, large ulcers >2.0 cm in diameter. An IEAS-CD score ⩾1 was defined as endoscopic inflammatory activity, and a score = 0 was defined as endoscopic mucosal healing. For double-balloon enteroscopy, insertion depth was roughly estimated by multiplying the insertion length by 5–8 times. Based on this, segment distances from the ligament of Treitz were referenced: proximal jejunum (100–120 cm), mid-jejunum (180–200 cm), distal jejunum (260–280 cm), proximal ileum (280–380 cm), mid-ileum (380–480 cm), distal ileum (480–580 cm), and the terminal ileum near the ileocecal valve (approximately 30 cm).
Computed tomography enterography
The observation indices of CTE were as follows: (1) Signs of small intestine mucosal inflammation: intestinal wall thickening, mucosal enhancement; (2) signs of mesenteric inflammation: comb sign, mesenteric fat crawling sign; and (3) evidence of CD-related complications: perianal abscess, abdominal and pelvic abscesses, fistula formation, and stenosis. 14 Stenosis was defined as a luminal diameter reduction of <50% of the adjacent normal bowel, accompanied by wall thickening exceeding 20% and proximal bowel dilatation greater than 3 cm. Intestinal wall thickening, mucosal enhancement, mesenteric vascular proliferation (comb sign), and mesenteric fat accumulation (fat crawling sign) were all indicative of active inflammation. 15 The reference value of the CTE Activity Score (Computed Tomography Enterography Activity Score, CTAS-CD) was set as: 1 point for abnormal enhancement of the intestinal wall; 1 point for thickening of the intestinal wall (thickness of the intestinal wall >3 mm); 1 point for the comb sign and the fat crawling sign; 1 point for transmural inflammatory signs, such as fistula-grade abscesses. 16 A CTAS-CD score ⩾1 was defined as CT inflammatory activity, and a score = 0 was defined as CT transmural healing. The CTAS-CD scoring system is not a standardized, validated scoring method, but was constructed based on individual imaging indicators widely recognized as markers of disease activity in CD. This system was developed to facilitate consistent and quantifiable assessment of disease activity in our analysis.
For CTE and intestinal ultrasound, the small bowel was divided into seven parts, including six groups and distal ileum according to Cole’s Precision Location Method. 17 The groups are defined as follows: group 1, duodenum; group 2, small intestine in the left upper abdomen (proximal jejunum); group 3, small intestine in the left lower abdomen (distal jejunum); group 4, mid-abdominal small bowel (proximal ileum); group 5, right mid- and lower-abdominal small bowel (mid-ileum); and group 6, pelvic portion of the small bowel (distal ileum) and distal ileum.
Intestinal ultrasound
Intestinal ultrasounds were performed and assessed by the same sonographer experienced in inflammatory bowel disease. Intestinal ultrasound observations included the following: (1) Intestinal wall structure: intestinal wall thickness, intestinal wall layers, intestinal wall blood flow, and intestinal peristalsis; (2) intestinal lumen internal: intestinal lumen stenosis, intestinal lumen dilatation, ulceration, inflammatory polyps. Stenosis was defined as bowel wall thickening with congestion, a rigid and narrowed lumen of <10 mm in diameter, a fixed gaseous high echogenic area, and proximal bowel dilatation with increased peristalsis; (3) external intestinal lumen: perimembranous fat hyperplasia, mesenteric lymph nodes, abdominopelvic effusion; and (4) intestinal complications: abscess, intestinal fistula, sinus tract. 18 Thickening of the intestinal wall, disruption and loss of intestinal wall layers, increased blood flow signals, and perimembranous fat hyperplasia may indicate disease activity. In this study, the reference value of the International Bowel Ultrasound Segmental Activity Score (IBUS-SAS) was set as: 1 point for intestinal wall thickening (intestinal wall thickness >3 mm); 1 point for intestinal wall layers lack of clarity; 1 point for intestinal wall blood flow signal abnormality; and 1 point for perimembranous adipose hyperplasia. 19 An IBUS-SAS score ⩾1 was defined as ultrasound inflammatory activity, and IBUS-SAS = 0 was defined as ultrasound transmural healing.
Statistical analysis
Count data were expressed as numbers and percentages. The χ2 test or Fisher’s exact test was used to compare categorical variables. Agreement between the evaluations of quantitative indicators was assessed using the intra-class correlation coefficient (ICC). A two-sided value of p < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS (Statistical Package for the Social Sciences) version 25.0 software.
Results
General information
The baseline data of the patients are shown in Table 1.
Baseline characteristics of patients (n = 198).

Comparative analysis of the detection of lesion location in different intestinal segments by the three examination methods
The data and comparative analysis of enteroscopy, CTE, and intestinal ultrasound for the detection of lesions in different intestinal segments are summarized in Table 2.
Detection rate of lesions in different parts of bowel segments by enteroscopy, CTE, and intestinal ultrasound (n (%)).

The more advantageous of the three checks.
Comparative analysis of the detection of complications of enteroscopy, CTE, and intestinal ultrasound
The data and comparative analysis of enteroscopy, CTE, and intestinal ultrasound on the detection of complications are summarized in Table 3.
Detection rate of the three tests for complications (n (%)).

Comparative analysis of enteroscopy, CTE, and intestinal ultrasound on identification of inflammatory activity
To analyze the differences between enteroscopy, CTE, and intestinal ultrasound in identifying inflammatory activity, the IES-CD was used to classify endoscopic findings into inflammatory activity (IES-CD ⩾ 1) and inflammatory remission (IES-CD = 0); the CTS-CD was used to classify CTE findings into inflammatory activity (CTS-CD ⩾ 1) and inflammatory remission (CTS-CD = 0); the IUS-CD was used to classify ultrasound findings into inflammatory activity (IUS-CD ⩾ 1) and inflammatory remission (IUS-CD = 0).
The differences in the ability to detect inflammatory activity between enteroscopy, CTE, and intestinal ultrasound were statistically significant (p = 0.017, p < 0.05). The detection of inflammatory activity by enteroscopy, CTE, and intestinal ultrasound was 146 (74%), 167 (84%), and 164 (83%) cases, respectively. Paired testing suggested that CTE was better than enteroscopy for the detection of inflammatory activity (p = 0.023, p < 0.05), while there was no difference between intestinal ultrasound and CTE (p > 0.05).
The ICC was used to analyze the consistency of the three methods for determining inflammatory activity. The results showed a poor correlation between IES-CD and CTS-CD, with ICC = 0.23 (95% confidence interval (CI): 0.06–0.37, p < 0.001), a poor correlation between IES-CD and IUS-CD, with ICC = 0.26 (95% CI: 0.02–0.45, p < 0.001), and a moderate correlation CTS-CD and IUS-CD, with ICC = 0.44 (95% CI: 0.30–0.56, p < 0.001).
Representative images of CD by enteroscopy, CTE, and intestinal ultrasound are shown in Figures 1–3, respectively.
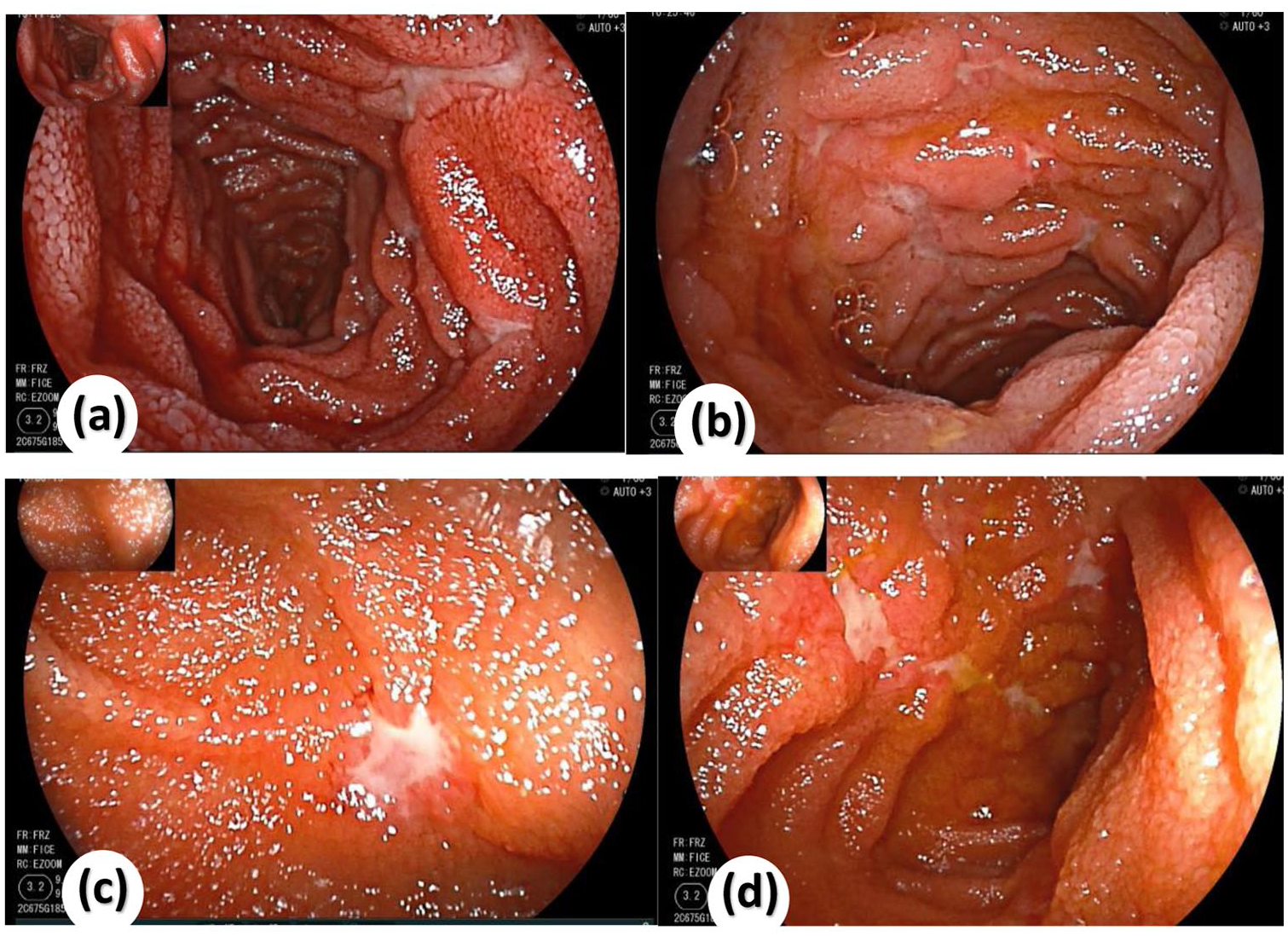
Endoscopic presentation of small bowel Crohn’s disease. (a) Multiple longitudinal ulcers with white moss covering the ulcer surface, surrounded by congested and edematous mucosa. The ulcers exhibit a “star-shaped” or “rat-bite” appearance. (b) Scattered, longitudinally distributed ulcers with similar “rat-bite” or “star-shaped” morphology, white moss covering the surface, and surrounding mucosal congestion and edema. (c) Close-up view of a “star-shaped” ulcer with a white moss-covered surface and peri-ulcer congestion. (d) A single longitudinal ulcer with white moss on its surface and peri-ulcer mucosal congestion. All lesions were located in the jejunum but were not detected by CTE or intestinal ultrasound.

CTE manifestations of CD. (a, b) CTE images of the same level. Segmental thickening of parts of the intestinal wall in groups 5 and 6 of the small bowel (small bowel of the right middle and lower abdomen, small bowel of the pelvic region), ileocecal region, and at the end of the ileum can be seen. Obvious enhancement of the mucosa is noted. (c) Uneven thickening of the intestinal wall at the point indicated by the black and white arrows, with obvious enhancement of the mucosal layer, showing the “target sign.” (d) Peri-intestinal blood vessel aggregation can be seen in the area indicated by the yellow arrow, showing the “comb sign.”
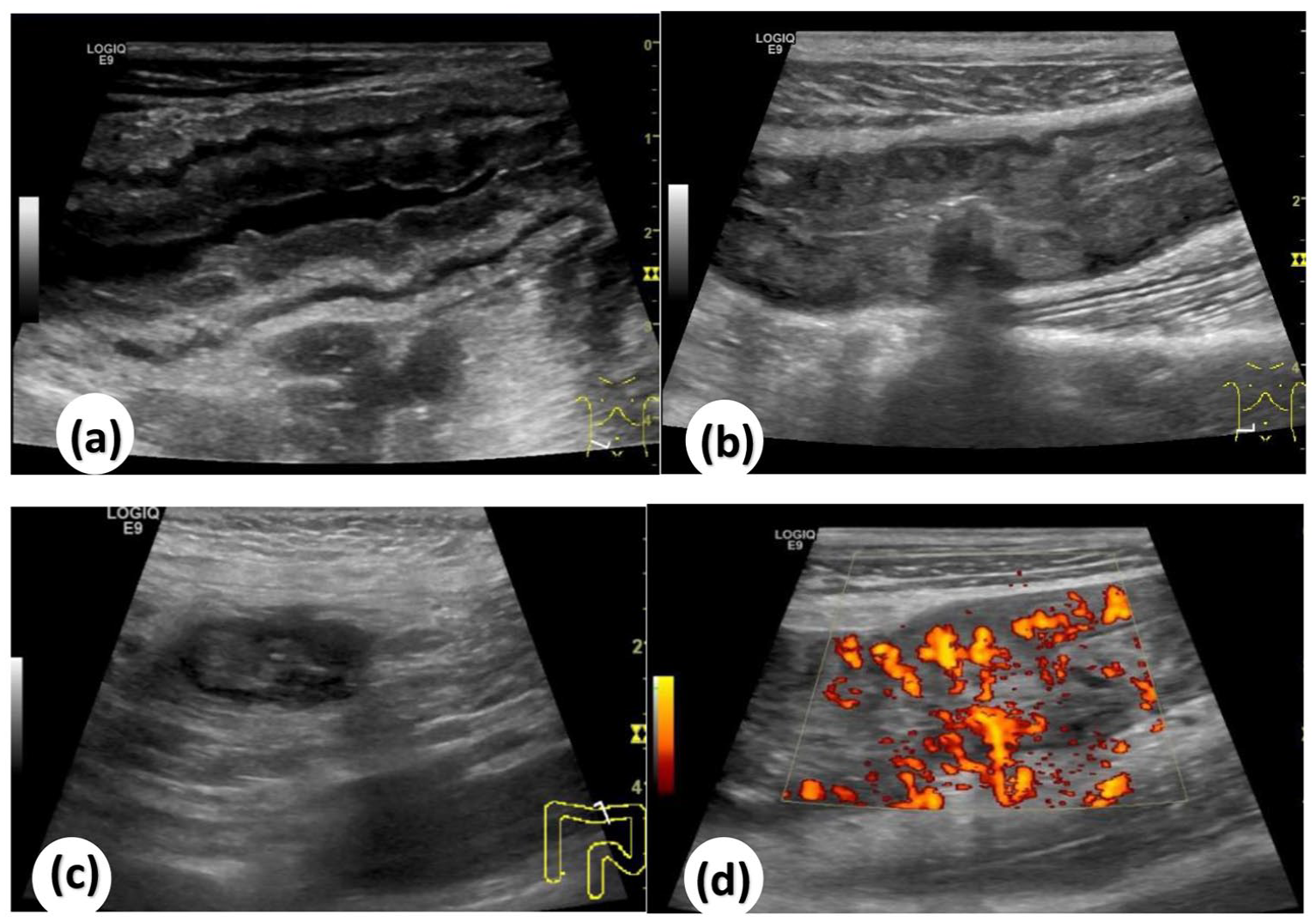
Intestinal ultrasound manifestations of CD. (a) Increased thickness of the intestinal wall is noted. (b) Abnormal layers of the intestinal wall can be seen. (c) Peri-intestinal fat crawling and hypertrophy. (d) Increased blood flow signal.
Discussion
Enteroscopy allows for direct observation of superficial mucosal changes, villi morphology, and vascular distribution within the intestinal lumen. However, as an invasive procedure, it carries risks such as bleeding and perforation and is technically limited in evaluating the outer intestinal wall and transmural inflammation, which are crucial in assessing CD. 20 In addition, procedural complexity and high technical demands may make enteroscopy challenging, especially in cases involving unique anatomical variations. 21 By contrast, noninvasive imaging techniques are advantageous for evaluating transmural inflammation and aiding in the differential diagnosis of complex CD cases.
Previous studies have typically assessed each imaging method independently, lacking a direct comparative analysis focused on proximal small intestinal lesions and the specific detection strengths for complications like fistulas and stenosis. Our study bridged this gap by providing a comprehensive, side-by-side evaluation, offering clinicians clearer guidance on method selection based on lesion location and complication type.
In this study, the detection rates of enteroscopy, CTE, and intestinal ultrasound for lesions in different intestinal segments were evaluated to further assess the diagnostic value of the three methods. The intestinal lesions evaluated for detection by the different methods included mucosal inflammation (congestion, edema, erosion, and ulceration), changes in intestinal lumen morphology (villi and fold structure), vascular characteristics (distribution and nature of blood vessels), and CD-related complications (fistulas, strictures, and hemorrhages). Enteroscopy exhibited superiority in detecting proximal small intestinal lesions, identifying 19 duodenal lesions (9.6%) and 62 upper jejunal lesions (31.3%), compared to just 1 lesion identified by CTE and 4 lesions identified by intestinal ultrasound (p < 0.001). This finding underscores the need to prioritize enteroscopy for suspected proximal lesions; this is consistent with the report by Jensen et al. 22 who observed that capsule endoscopy had higher sensitivity for detecting jejunal lesions compared to CTE and magnetic resonance enterography, supporting the role of enteroscopy in identifying upper intestinal lesions in CD patients. A study by Esaki et al. 23 also concluded that enteroscopy was more accurate in identifying upper small bowel lesions in CD. The lower detection rates of CTE and intestinal ultrasound for upper small bowel lesions may be due to inadequate dilation of these areas during imaging, limiting accurate visualization of small lesions. In addition, the small size of upper intestinal lesions makes them harder to detect with noninvasive imaging. Consequently, detection rates for these lesions are lower with other methods compared to enteroscopy. Studies also indicate that patients with the L4 phenotype face a higher risk of complications, surgery, postoperative recurrence, and hospitalization. 24 Our results showed that noninvasive imaging is less effective for detecting upper small bowel lesions, which may lead to missed diagnoses and delayed treatment. Therefore, upper small bowel evaluation should be prioritized in the assessment of CD, and enteroscopy is recommended for patients with suspected lesions in this area, with CTE as a complementary option if enteroscopy is not feasible. While each method has strengths across different bowel segments, noninvasive options like CTE and ultrasound offer better patient comfort and may be combined with enteroscopy when needed. Notably, CTE was more effective in detecting mid-ileum lesions, likely due to the anatomical limitations of enteroscopy at this site.
Stenosis is the most common complication of CD, and the primary reason for surgical intervention in 70%–90% of patients. 25 Chronic intestinal inflammation often leads to fibrous scarring of the intestinal wall, increasing the risk of luminal stenosis and resulting in partial or complete intestinal obstruction. 26 In this study, enteroscopy showed significant superiority over CTE and intestinal ultrasound for detecting stenosis (p < 0.001), indicating its absolute advantage in assessing stenosis. However, for patients with long-segment stenosis or obstruction, enteroscopy may not be feasible. Therefore, noninvasive examinations such as CTE and intestinal ultrasound are more beneficial. In addition to stenosis, fistulas and abscesses are important complications of CD. CTE excels at detecting fistulas, with a detection rate of 13 cases (6.6%), significantly greater than enteroscopy (3 cases, 1.5%) and intestinal ultrasound (11 cases, 5.6%; p = 0.038), making CTE the recommended method for fistula evaluation. For abscess detection, there was no statistical difference among the three methods (p > 0.05). This may be due to an insufficient sample size. Since enteroscopy does not visualize extraluminal manifestations, noninvasive imaging methods such as CTE and intestinal ultrasound provide better assessments of the intestinal wall, mesentery, and transmural inflammation.27,28
Our results showed differences between the three methods for assessing inflammatory activity: a detection rate of 146 cases (74%) for enteroscopy, 167 cases (84%) for CTE, and 164 cases (83%) for ultrasound (p < 0.05). In addition, the ICC analysis showed poor correlations among the three methods. This suggests that enteroscopy’s inability to assess lesions in the intestinal wall, such as fibrosis and transmural lesions, limits its effectiveness compared to CTE and ultrasound, which identified more inflammatory cases.
Furthermore, the absence of significant differences in abscess detection (0 for enteroscopy, 4 for CTE, and 2 for ultrasound, p = 0.174) indicates that imaging methods are preferable for evaluating abscesses. Based on the characteristics and strengths of each modality, we propose the following recommendations: for suspected or confirmed proximal small bowel lesions and for assessing small bowel strictures, enteroscopy should be the first choice due to its direct visualization capabilities. For trans-wall complications such as fistulas or abscesses, CTE or intestinal ultrasound is preferred. To assess overall inflammatory activity, the three modalities are complementary and should be integrated to provide a comprehensive evaluation. In patients with asymptomatic remission, follow-up with intestinal ultrasound is recommended as a convenient and economical option. However, it is important to note that none of these modalities are established as a definitive gold standard, limiting the ability to develop predictive or diagnostic models or decision curves. While intestinal ultrasound shows promise for monitoring asymptomatic patients, further validation is required to fully establish its role in clinical practice. This descriptive evaluation highlights the need for tailored approaches to integrate these methods into clinical decision-making to optimize diagnostic accuracy and patient management.
This study has several limitations. First, the retrospective design may introduce selection bias, as patient selection was restricted to available records and may not fully represent the broader patient population. Future prospective studies employing randomized sampling could enhance the validation and generalizability of these findings. In addition, due to the lack of a definitive gold-standard diagnostic method for precise lesion localization, conducting sensitivity analyses was not feasible in this study.
Conclusion
Enteroscopy was significantly superior to CTE and intestinal ultrasound for assessing lesions in the proximal small intestine in patients with CD. For evaluating transmural inflammation, fistulas, and abscesses, CTE and ultrasound are more advantageous. While all three methods need to be used in combination for a comprehensive assessment of CD, we recognize that practical considerations such as availability, cost, and patient preference should guide their combined use in clinical practice. Thus, we recommend prioritizing enteroscopy for suspected proximal small intestinal lesions, followed by CTE or ultrasound as needed. For asymptomatic patients in remission, intestinal ultrasound may offer a cost-effective option for long-term monitoring.
