Abstract
Background:
Treatment options for patients with refractory inflammatory bowel disease (IBD) or concomitant IBD and extraintestinal manifestations (EIM) are often limited.
Objective:
This study aimed to examine the efficacy and safety of combining biologics or small molecules in patients with refractory IBD, active EIM, or active immune-mediated inflammatory disease (IMID).
Design:
This was a retrospective and multicenter study.
Methods:
We retrospectively collected demographics and disease characteristics from 47 patients with IBD who received dual-targeted therapy in 3 hospitals from January 2022 to June 2024. The primary endpoint was clinical remission based on the Harvey–Bradshaw index or patient-reported outcome 2 after at least 4 months of combination therapy. The secondary endpoints included clinical response, endoscopic response, and endoscopic remission, as well as all adverse events that occurred within the period of combination therapy.
Results:
In total, 47 IBD patients including 37 with refractory IBD, 5 with active EIM, and 5 with active IMID received dual-targeted therapy, of which 37 achieved clinical response (78.7%) and 27 achieved clinical remission (57.4%) at a median follow-up time of 13.0 months. Among these 47 patients, 29 patients underwent endoscopic follow-up, of which 15 (51.7%) achieved endoscopic response and 8 (27.6%) achieved endoscopic remission at a median follow-up time of 9.0 months. Mild and moderate adverse events were reported in 17 (36.2%) patients within the period of combination therapy, and serious adverse events requiring hospitalization occurred in 1 patient (2.1%).
Conclusion:
The combination therapy of biologics and small molecules for refractory IBD or those with concomitant EIM/IMID is effective and safe.
Plain language summary
This present study examined the efficacy and safety of the combination of biologics and small molecules in patients with refractory inflammatory bowel disease (IBD), active extraintestinal manifestations (EIM) or active immune-mediated inflammatory disease (IMID). Among these 47 patients, the rates of clinical response, clinical remission, endoscopic response, and endoscopic remission were 78.7%, 57.4%, 51.7%, and 27.6%, respectively. The rates of adverse events and serious adverse events were 38.3% and 2.1%, respectively. In conclusion, dual targeted therapy for refractory IBD or those with concomitant EIM/IMID is effective and safe.
Introduction
Inflammatory bowel disease (IBD) is a progressive, immune-mediated inflammatory disease (IMID) including ulcerative colitis (UC), Crohn’s disease (CD), and indeterminate colitis (IC). 1 Various constantly updated biological agents and small molecule drugs have revolutionized the treatment of IBD. However, due to the primary or secondary non-response to the biological agents, only 40% of patients who respond to biological agents can maintain clinical remission after 1 year. 2 In clinical practice, therapeutic drug monitoring, intensive therapy based on inflammatory markers, and drug sequencing have been used to improve the outcomes of biologics for IBD, but the current long-term efficacy remains inadequate. 3
Treatment options for patients with IBD refractory to multiple biological agents are often limited. In recent years, many researchers have proposed the concept of dual-targeted therapy, which is a combination of biological agents and/or small molecule drugs. 4 This is an attractive concept due to multidimensional anti-inflammatory treatment strategies. In patients with partial response to anti-tumor necrosis factor (TNF) therapies and without anti-drug antibodies, the combination therapy of anti-TNF and vedolizumab (VDZ) could serve as a bridge until VDZ reaches the expected efficacy, thereby avoiding the use of additional corticosteroids. 5 Moreover, active extraintestinal manifestations (EIM) may occur while intestinal inflammation is still under control. Considering well-controlled intestinal lesions with existing drugs, it may not be recommended to switch to an alternative agent. Therefore, drugs for controlling EIM can be added, and vice versa. 6 Likewise, combination therapy might achieve benefit in IBD patients with other IMIDs that cannot be controlled with monotherapy. 7 However, the data regarding dual-targeted therapy in refractory IBD remain limited and controversial. The EXPLORER trial showed that patients with high-risk CD receiving the combination of VDZ, adalimumab, and methotrexate therapy achieved an endoscopic response rate of 54.4% and endoscopic remission rate of 34.5% at week 26. 8 VEGA study evaluated the efficacy of a combination induction therapy with guselkumab and golimumab compared to guselkumab or golimumab monotherapy in patients with moderately to severely active UC. However, there was no significant difference between the combination therapy group and both monotherapy groups in clinical response rate at week 12. 9 Herein, we studied the real-world outcomes of dual biologics and small molecules therapy, including the efficacy and safety of combination therapy in patients with refractory IBD or those with concomitant EIM/IMID.
Methods
Study design and population
This was a multicenter retrospective study that included IBD patients who received combination therapy from January 2022 to June 2024 at the First Affiliated Hospital of Zhejiang University, Shangyu People’s Hospital of Shaoxing, and the Second Hospital of Jiaxing. Inclusion criteria were patients over 18 years old diagnosed with CD, UC, or IBD unclassified. The patients were initiated on dual-targeted therapy for any of the following indications: (a) refractory IBD, defined by the failure of biologics and small molecules with at least two different mechanisms of action, postoperative CD recurrence after two or more bowel resections, chronic antibiotic-refractory pouchitis, complex perianal CD, or the presence of psychosocial issues that prevent or impair IBD management4,10; (b) IBD accompanied by active EIM; and (c) IBD accompanied by active IMID. The exclusion criteria were as follows: (a) patients who have received combination therapy for less than 4 months; (b) patients without any follow-up data; and (c) patients noncompliant with therapy. The relevant drugs included infliximab (IFX), adalimumab (ADA), ustekinumab (UST), VDZ, and upadacitinib. Details of medication usage are summarized in Table 1. The study was approved by the Clinical Research Ethics Committee of the First Affiliated Hospital of Zhejiang University (IIT20240371A). Informed consents were obtained from all participants. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 11
Usage instructions for biologics and small molecule drugs.

CD, Crohn’s disease; UC, ulcerative colitis.
Data collection
To minimize selection bias and information bias, we collected data from three tertiary medical centers using a standardized case report form and tried to use objective recorded data as much as possible. Proactive follow-up and financial incentives were used to reduce non-response and loss to follow-up among participants. Baseline data on patient characteristics including gender, age, concomitant diseases, body mass index (BMI), previous surgical history, and previous medication history, and disease characteristics including duration, disease behavior, and extent according to the Montreal classification, complication, extraintestinal symptoms, and past and ongoing treatments were retrospectively collected from electronic medical records and database for the follow-up of IBD patients. Clinical and endoscopic scores including Harvey–Bradshaw index (HBI) and Simple Endoscopic Score for Crohn’s Disease (SES-CD) for CD, patient-reported outcome 2 (PRO2), and Mayo Endoscopic Score (MES) for UC were retrospectively collected at baseline and at least 4 months after combination therapy. Perianal fistulas were assessed by pelvic magnetic resonance imaging at baseline and at least 4 months after combination therapy. Data on adverse events during combination were also collected from electronic medical records and databases. Adverse events were assessed by clinicians based on the Common Terminology Criteria for Adverse Events (CTCAE) V.5.0. Grade 3 and above were defined as serious adverse events (SAEs), including hospitalization, cessation of treatment, or death.
Outcomes
The primary endpoint was clinical remission defined as HBI ⩽4 for CD or PRO2 = 0 for UC after at least 4 months of combination therapy. The secondary endpoints include clinical response, endoscopic response, and endoscopic remission after at least 4 months of combination therapy, as well as any adverse events that occurred during the combination. Clinical response was defined as a reduction of HBI by ⩾3 points from baseline for CD or a reduction of PRO2 by ⩾50% from baseline for UC. Endoscopic remission was defined as SES-CD score ⩽2 for CD and MES = 0 for UC. The endoscopic response was defined as a reduction of SES-CD by ⩾50% from baseline for CD and a reduction of MES by ⩾1 point from baseline for UC. There was no pre-defined time point for clinical and endoscopic evaluation during combination, so the clinical and endoscopic data of the patients were obtained retrospectively from electronic medical records and databases at the latest follow-up appointment.
Statistical analysis
All statistical analyses were conducted using the statistical software SPSS 26.0 (IBM, Armonk, NY, USA). Normally distributed continuous variables were expressed as mean ± standard deviation, non-normally distributed quantitative data were expressed as the median and interquartile range (IQR), and categorical variables were expressed as the frequency and percentage of subjects in each category. Mann–Whitney U test or Kruskal–Wallis test was used for comparison of continuous variables. Fisher’s exact test or Pearson’s Chi-square with continuity correction was used to determine the differences between categorical variables. Logistic regression was used to evaluate the impact of disease duration and treatment duration on the efficacy of dual-targeted therapy. Statistically significant was defined as a p-value <0.05.
Results
Baseline characteristics
After applying the inclusion and exclusion criteria, a total of 47 patients were enrolled in this study, including 42 with CD and 5 with UC (Figure 1). Thirty-seven (78.7%) patients met at least one criterion for refractory IBD. Concomitant EIM or IMID conditions were present in 10 patients, including 3 patients (6.4%) with active IBD and active EIM (3 with oral aphthous ulcers), 2 cases (4.3%) with active EIM but well-controlled intestinal symptoms (1 with peripheral spondyloarthropathy and 1 with CD-related hepatic nodule confirmed by pathological examination), and 5 cases (10.6%) with active IBD and active IMID (3 with psoriasis, 1 with ankylosing spondylitis, and 1 with psoriasis and ankylosing spondylitis). The median age at the diagnosis was 24.0 years (IQR 18.0–43.0), and the current age enrolled was 29.0 years (IQR 23.0–49.5). Among the patients with CD, 59.5% were diagnosed before the age of 40, 14.3% were diagnosed before the age of 16. Before starting dual-targeted therapy, 35 patients (74.5%) received at least 2 biologics, and 12 patients (25.5%) were treated with a combination of single biological and immunomodulatory drugs. Patients received biological monotherapy for an average of 20.8 ± 16.6 months before starting combination therapy. In addition, 13 (27.7%) patients had previous surgery related to IBD. The baseline characteristics of patients who received dual-targeted therapy are shown in Table 2.
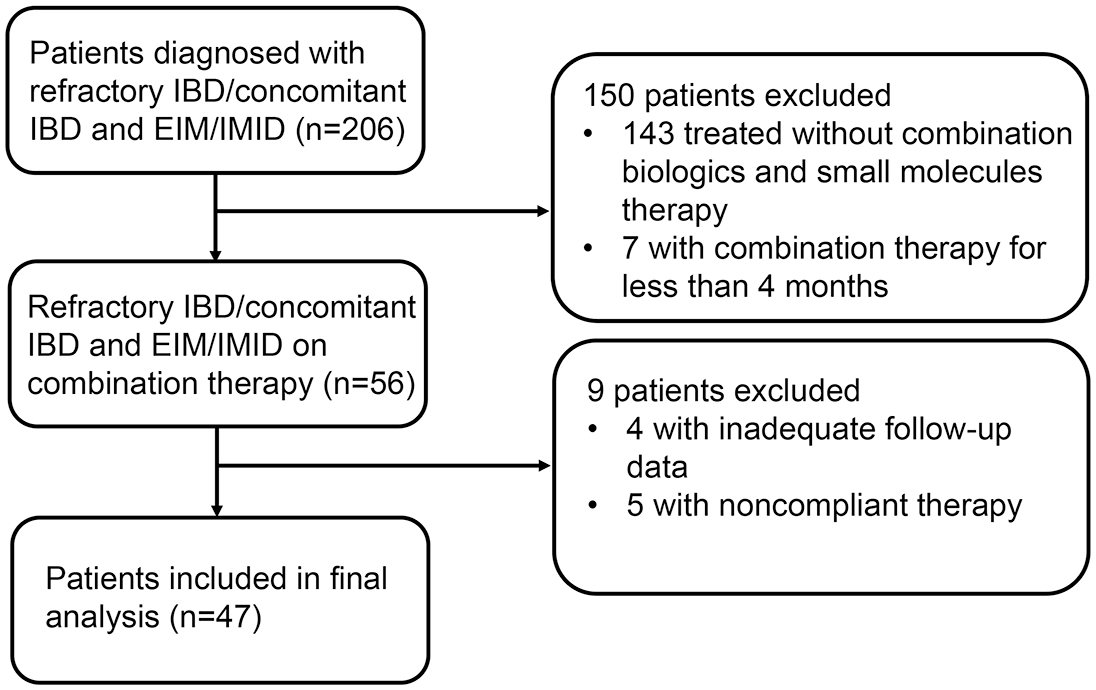
Flow chart of screening and inclusion.
Baseline characteristics of IBD patients with dual-targeted therapy.

One patient did not have an endoscopic examination at the beginning of the combination due to intestinal perforation and enterectomy.
BMI, body mass index; CD, Crohn’s disease; IBD, inflammatory bowel disease; IQR, interquartile range; SES-CD, simple endoscopic score for Crohn’s disease; TNF, tumor necrosis factor; UC, ulcerative colitis; UST, ustekinumab.
Efficacy of dual biological therapy
A total of 47 patients were treated with 6 types of combination therapy, including 25 with anti-TNF + UST (23 ADA and 2 IFX), 8 with anti-TNF + VDZ (5 ADA and 3 IFX), and 9 with UST + VDZ, 3 with anti-TNF + upadacitinib (1 ADA and 2 IFX), 1 with UST + upadacitinib, 1 with VDZ + upadacitinib. The median duration of dual-targeted therapy was 13.0 months (IQR 7.0–18.0), the median duration from starting the combination to endoscopy evaluation was 9.0 months (IQR 7.0–14.0), and the maximum duration until the assessment of the primary endpoint was 23.0 months. At the initiation of the combination, 4 (8.5%) patients were taking systemic corticosteroids at the same time, and corticosteroids were successfully discontinued in all patients during the combination. During the combination period, four CD patients underwent surgical treatment, including one for intestinal obstruction, one for intestinal perforation, one for closure of ileostomy wounds, and one for perianal fistulas.
Among 47 IBD patients receiving dual-targeted therapy, 37 achieved clinical response (78.7%) and 27 achieved clinical remission (57.4%) at a median follow-up time of 13.0 months (IQR 7.0–18.0) of combination therapy. The HBI decreased from 9 points (IQR 8–10) to 2 points (IQR 1–5) (p < 0.001). The levels of ESR and CRP at a median follow-up time of 13.0 months significantly decreased compared to baseline levels (both p < 0.001), while the levels of hemoglobin and albumin significantly increased after the combination therapy (p = 0.02 and p < 0.001). The incidence of perianal fistulas decreased significantly from 55.3% (26/47) at baseline to 17.0% (8/47) after 13.0 months of combination therapy (p = 0.016). The presence of recurrent bleeding declined from 19.1% (9/47) at baseline to 2.1% (1/47) at 13.0 months post-treatment (p = 0.008) (Table 3). Four patients achieved clinical remission and discontinued dual-targeted therapy at 9.5 months (IQR 7.0–11.5), followed by one biologic for maintenance treatment. No disease recurrence was observed among these four patients evaluated by laboratory and radiologic results during the 6-month follow-up period. Seven patients with active IBD ended dual-targeted therapy at 11.0 months (IQR 6.0–19.0) due to lack of clinical response. Clinical response was achieved in all patients with coexisting EIM or IMID. Among them, one patient with a CD-related hepatic nodule showed a complete disappearance of the lesion on abdominal CT after dual-targeted therapy (Figure 2).
Efficacy of dual-targeted therapy in IBD-clinical, endoscopic, and laboratory data.

HBI, Harvey–Bradshaw index; IBD, inflammatory bowel disease; IQR, interquartile range; SES-CD, simple endoscopic score for Crohn’s disease.
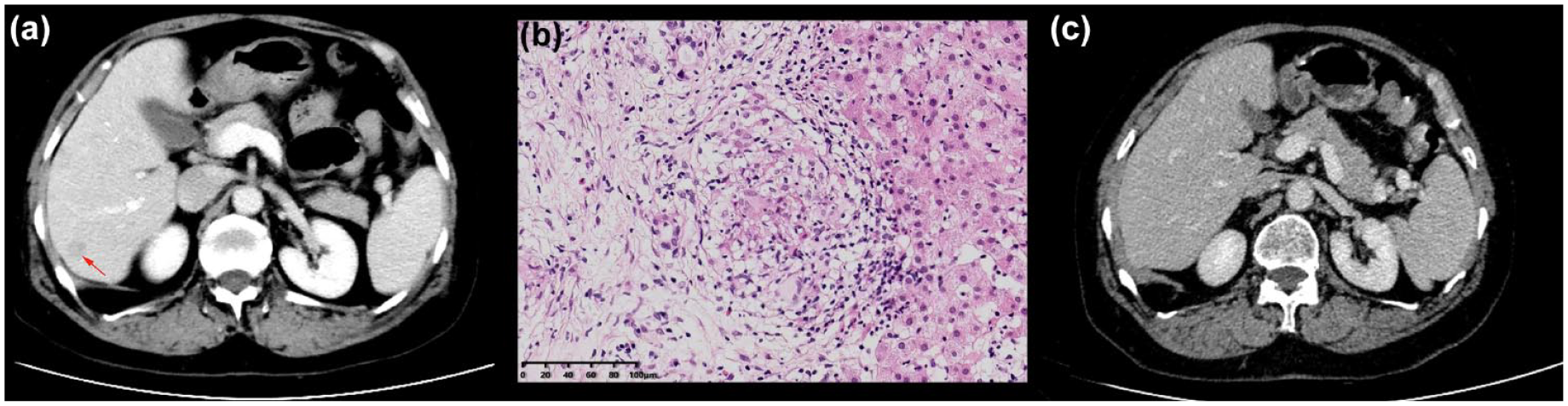
One patient with CD-related hepatic nodule. (a) Hepatic nodule confirmed by CT scan (indicated by the red arrow). (b) Hepatic granuloma confirmed by pathological examination. (c) Disappearance of hepatic nodules after the combination of VDZ and ADA.
Twenty-nine patients receiving dual-targeted therapy underwent endoscopic follow-up, of which 15 (51.7%) achieved endoscopic response and 8 (27.6%) achieved endoscopic remission at a median follow-up time of 9.0 months (IQR 7.0–14.0) of combination therapy. However, all eight patients failed to achieve histological remission (evaluated by Geboes score or Global Histologic Disease Activity Score). Twenty-seven patients had a baseline score of 14 (IQR 11.25–17.75) for SES-CD, which decreased to 6 (IQR 2–10) at 9.0 months (IQR 7.0–14.0) (p < 0.001) (Table 3). In addition, age, BMI, disease duration, treatment duration, disease phenotype, EIM, and perianal fistulas did not influence clinical or endoscopic outcomes of dual-targeted therapy in IBD patients.
Efficacy of different types of combinations
After analyzing the clinical efficacy of three types of combinations, the clinical response and remission rates were 72% (18/25) and 56% (14/25) for UST + anti-TNF at a median follow-up time of 15.0 months (IQR 12.0–18.0), both 77.8% (7/9) for UST + VDZ at 16.0 months (IQR 9.0–21.5), and 87.5% (7/8) and 37.5% (3/8) for VDZ + anti-TNF at 9.5 months (IQR 4.3–12.0) (Figure 3). However, no significant differences were found in clinical improvement among the three types of combinations. Due to the limited number of cases, the combination of small molecules and biologics was not compared with the other three types of combinations. The clinical response and remission rates for these patients were as follows: 100% (3/3) and 66.7% (2/3) for anti-TNF + upadacitinib at a median follow-up time of 6 months (IQR 5.0–7.0), both 100% (1/1) for VDZ + upadacitinib at 6 months, and 100% (1/1) and 0 (0/1) for UST + upadacitinib at 4 months.

Efficacy and safety of different types of combinations.
At the same time, we analyzed the endoscopic manifestations of patients treated with various combinations. The endoscopic response and remission rates for patients receiving dual-targeted therapy were as follows: 43.8% (7/16) and 18.8% (3/16) for UST + anti-TNF at a median follow-up time of 9.0 months (IQR 7.3–13.5), 85.7% (6/7) and 71.4% (5/7) for UST + VDZ at 10.0 months (IQR 6.0–17.0), 50% (2/4) and 0 (0/4) for VDZ + anti-TNF at 12.0 months (IQR 8.3–21.0) (Figure 3). However, no significant differences were found regarding endoscopic improvement among the three types of combinations. Two patients underwent endoscopic re-examination following 4-month treatment with anti-TNF + upadacitinib and UST + upadacitinib, respectively, but they did not achieve an endoscopic response.
Safety analysis
In the present study, 18 of 47 patients (38.3%) experienced a total of 24 adverse events during a median follow-up time of 13.0 months (IQR 7.0–18.0), including 5 patients with pharyngitis, 4 with eczema, 4 with skin erythema, 3 with arthralgia, 2 with headache, 2 with upper respiratory tract infections, 1 with oral herpes simplex, 1 with pneumonia, 1 with eyelid twitch, and 1 with lower limbs edema (Table 4). The incidence of adverse events in various combinations was as follows: 44% (11 of 25 patients) for anti-TNF + UST, 25% (2 of 8 patients) for anti-TNF + VDZ, 33.3% (3 of 9 patients) for UST + VDZ, 66.7% (2 of 3 patients) for anti-TNF + upadacitinib, 0% (0/1) for UST + upadacitinib, 0% (0/1) for VDZ + upadacitinib. Overall, 62.5% (15/24) of adverse events were mild (grade 1 in CTCAE), 33.3% (8/24) were moderate (grade 2 in CTCAE), and 4.17% (1/24) were severe (grade 3 in CTCAE). Two patients discontinued the combination due to adverse events. A patient who had been treated with UST + anti-TNF for 6 months developed eczema (grade 2 in CTCAE) which resolved with topical steroids. Then she discontinued the combination and switched to monotherapy with upadacitinib. Another patient was hospitalized due to pneumonia caused by Talaromyces marneffei (grade 3 in CTCAE) after 4 months of combination with anti-TNF + upadacitinib and subsequently improved with itraconazole. Then he discontinued the combination and switched to monotherapy with VDZ. However, there was no significant difference in the incidence of adverse events among different types of combinations due to the small sample size. Moreover, age, BMI, and IBD phenotype did not show a significant association with the incidence of adverse events.
Adverse events on combination therapy of biologics and small molecules.
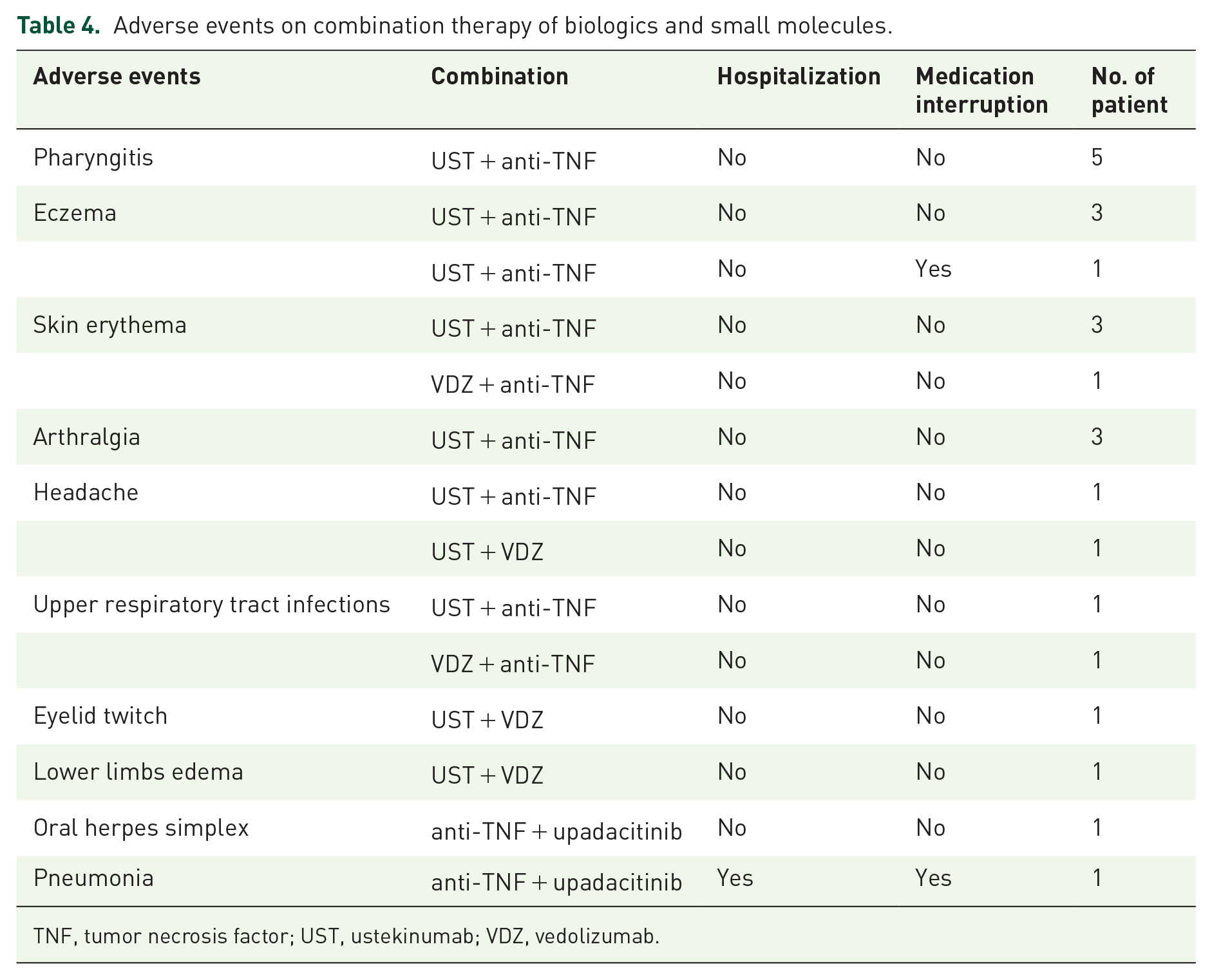
TNF, tumor necrosis factor; UST, ustekinumab; VDZ, vedolizumab.
Discussion
This present study retrospectively analyzed 47 patients with refractory IBD, active EIM, or active IMID treated with dual-targeted agents, and evaluated the effectiveness and safety of dual-targeted therapy. To our knowledge, this is the first report to examine the efficacy and safety of dual-targeted therapy in Chinese patients with refractory IBD or those with concomitant EIM/IMID. Among these 47 patients, the rates of clinical response, clinical remission, endoscopic response, and endoscopic remission were 78.7%, 57.4%, 51.7%, and 27.6%, respectively. All inflammation-related biomarkers showed significant improvements. It is worth noting that 69.2% (18/26) of patients achieved fistula resolution and 88.9% (8/9) of patients with recurrent gastrointestinal bleeding stopped bleeding after combination. A SAE requiring hospitalization occurred in 2.1% (1/47) of patients. Our results were reassuring and encouraging considering that most of the included patients had a severe phenotype with a long disease course, stricturing and penetrating phenotype, steroid refractoriness or dependence, and prior biologics failure.
Currently, the global incidence rate of IBD is gradually increasing. 12 Although biological agents and small molecule drugs are constantly updating, there still remain great challenges for the treatment of patients with refractory IBD because of primary or secondary non-response to the biological agents. The combination of immunosuppressive agents (thiopurines or methotrexate) can significantly improve the pharmacokinetics of anti-TNF, but it does not increase the efficacy of other biological agents such as UST and VDZ. 13 In addition, although studies have shown that the disease outcome of anti-TNF combined with thiopurine was superior to monotherapy, long-term use could lead to safety issues such as increased incidence of infection and tumor. 14 Considering the nonspecific action of immunosuppressive agents without targeting specific targets, the combination of biologics and/or small molecules with different targets has been increasingly used in the treatment of refractory IBD or those with concomitant EIM/IMID.15,16 The currently recommended options are a drug with a favorable safety profile, such as VDZ, UST, or IL-23 antagonists, combined with a second agent with potential adverse effects, such as anti-TNF or JAK inhibitors. This treatment strategy may avoid the established adverse effects associated with the combination of anti-TNF and immunomodulators.17 –19
The latest meta-analysis shows that UST was superior to VDZ and ADA in achieving clinical remission in patients who have previously received anti-TNF treatment. 20 This may be due to the increased expression of IL-23 receptor in the intestinal mucosa of CD patients, leading to an increased resistance of mucosal T cells to anti-TNF-induced apoptosis. 21 In theory, agents targeting IL-23 can restore the body’s response to anti-TNF therapy through synergistic effects. 21 A retrospective study by Kwapisz et al. 22 suggests that anti-TNF or VDZ combined with UST may be an ideal combination for dual-targeted therapy. In our study, the most commonly used dual target strategy was UST + ADA with clinical and endoscopic response occurring in 73.9% and 42.9% of patients, which was the highest number of cases reported to date regarding the combination of these two biologics. Most patients treated with this strategy experienced primary or secondary non-response to IFX and ultimately achieved significant clinical improvement after UST plus ADA therapy. Another interesting observation was that endoscopic response and remission to UST plus VDZ were achieved in 85.7% and 71.4% of patients at a median time of 10 months, respectively. Our finding was comparable with that reported in a previous study. A retrospective study by Yang et al. 16 demonstrated that the combination of UST plus VDZ showed higher rates of endoscopic improvement (68% vs 43%) when compared with anti-TNF and either UST or VDZ, but no difference in endoscopic remission (25% vs 26%) between two groups. Combination therapy with UST plus VDZ has the potential to be a promising and safe option, but there is currently a lack of randomized controlled studies comparing this combination with other combinations, and further research is needed to determine the optimal combination for refractory IBD or those with concomitant EIM/IMID.
In addition, the introduction of a second biologic or small molecule to treat IMID/EIM manifestations was very successful in our cohort, with a clinical response rate of 100%. Similar findings have been observed in a large European multicenter retrospective study. Goessens et al. 23 reported that IMID/EIM activity was clinically improved in 81% of patients. The current findings confirm that this strategy might be an effective option in IBD patients with active IMID/EIM. Although there are no definitive recommendations to determine the optimal combination in patients with IBD and concomitant EIM and/or IMID, it is important to consider the patient’s extra-intestinal diseases or immune-medicated comorbidities for which biological therapy is approved. In the case of concurrent psoriasis or atopic dermatitis, the addition of IL-23 antagonist, IL-12/23 antagonist, TNF antagonist, or JAK inhibitor could be considered. In the case of concurrent rheumatoid arthritis or ankylosing spondylitis, TNF antagonists or upadacitinib are the drugs of choice. Patients with pyoderma gangrenosum would likely benefit from TNF antagonists.17 –19 To date, limited data exist for double indication, additional studies are required to determine the optimal combinations in patients with active EIM or IMID.
Although dual-targeted therapy has shown synergistic effects and high efficiency, safety is still a prime concern. The incidence of adverse events in rheumatologic patients receiving combination therapy was higher than that in patients receiving biological monotherapy. 24 Although available data supporting the safety of UST and VDZ monotherapy,15,25 the rate of serious infection in combination therapy by Glassner et al. 5 was higher than that in biological monotherapy reported in previous clinical trials.26 –28 Two meta-analyses summarized the existing data on the safety of dual biologics or small molecule therapy in patients with IBD. Ahmed et al. 29 included 279 patients in 30 studies and reported pooled adverse event and SAE rates of 31% and 6.5%, respectively. Alayo et al. 30 reported a pooled rate of SAEs of 12.3% for VDZ plus UST and 9.6% for VDZ plus anti-TNF, respectively. The limitations of the pooled analysis were the inconsistent definitions of clinical outcomes for efficacy and safety, and the significant heterogeneity in studies included. The rates of adverse events and SAEs in our study (38.3% and 2.1%) were lower than what Goessens et al. 23 (42% and 20.4%) and Yerushalmy-Feler et al. 31 (46.8% and 27.6%) reported in the real-world studies. In our study, only one case of pneumonia caused by T. marneffei in a patient treated with IFX plus upadacitinib was assessed as a serious drug-related adverse event. Our study also found that adverse events occurred in 44% of patients with combination therapy of UST + anti-TNF, which was higher than those treated with UST + VDZ (33.3%) or VDZ + anti-TNF (25%). However, due to the small sample size, there was no significant difference in adverse events among different combinations. Together, these observations are consistent with the viewpoint that dual biologics or small molecule therapy appears to be a relatively safe option for IBD. 32 Further safety data are needed due to the limited number of participants and the short follow-up period.
This study has several limitations. First, due to the relatively small sample size, we cannot determine whether the type of dual therapy was associated with improved effectiveness or reduced risk of adverse events. Second, due to the retrospective nature of our study, the time points for evaluating clinical and endoscopic responses in this study were not consistent, limiting our ability to accurately determine the effectiveness of dual-targeted therapy in the induction period and remission period. Third, not all included subjects underwent endoscopy before and after combination due to stricturing or penetrating phenotype, which decreased the sample size available for the analysis of endoscopic improvement. In the future, well-designed prospective randomized controlled studies are needed to validate the current results.
Conclusion
In summary, the combination therapy of dual biologics or small molecules for refractory IBD or those with concomitant EIM/IMID appears to be safe and effective. However, the combination use of IL-23 antagonist and TNF antagonist could effectively treat patients without response to anti-TNF, but may still be unable to resolve treatment failure caused by anti-biological antibody. Thus, there still remain significant challenges in the treatment of IBD. The present encouraging results should be followed by a better understanding of the rationale for a combination that may aid clinician’s decision-making and eventually could result in better treatment strategies for these difficult-to-treat populations.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241307598 – Supplemental material for Efficacy and safety of dual-targeted therapy for inflammatory bowel disease: a retrospective multicenter study in China
Supplemental material, sj-docx-1-tag-10.1177_17562848241307598 for Efficacy and safety of dual-targeted therapy for inflammatory bowel disease: a retrospective multicenter study in China by Xiuxiu Jin, Kefang Sun, Liying Wang, Haiyan Shen, Dan Ma, Tejia Shen, Chunxiao Chen and Lan Li in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
