Abstract
Background:
Faecal microbiota transplantation (FMT) is a highly effective treatment for Clostridioides difficile infection. Its use is backed by solid evidence, but application methods differ. Encapsulated FMT is a non-invasive, patient-friendly and scalable application method that may be preferred over colonoscopy or nasoduodenal tube application.
Objectives:
We describe a detailed protocol, the Glyprotect protocol, for producing glycerol-based capsules to increase FMT accessibility.
Design:
Using iterative quality improvement methods, we developed and validated the Glyprotect protocol as a reproducible protocol for cryopreserving minimally processed donor faeces in a standard hospital laboratory setting.
Methods:
We describe detailed standard operating procedures for producing glycerol-based capsules, including all necessary materials and troubleshooting guidelines. Capsule integrity was tested at various temperatures and pH levels. Flow cytometry was used to measure microbiota counts and dose accuracy.
Results:
The Glyprotect protocol has been used for more than 2500 capsule-based FMT treatments and complies with European tissue and cell standards. The protocol is optimised to preserve microbes and minimise modulation of the donated microbiota by removing debris and water, which also reduces the number of capsules needed per FMT treatment. The intestinal microbiota is preserved in glycerol for cryoprotection and to prevent capsule leakage. Each capsule contains 650 µL microbe–glycerol mass, estimated to contain an average of 2.5 × 108 non-specified bacteria.
Conclusion:
The Glyprotect protocol enables hospitals and tissue establishments to set up capsule production in a standard laboratory, improving patients’ access to FMT. The protocol facilitates the scalability of FMT services because capsule FMT is less time-consuming and less expensive than liquid-suspension FMT applied by colonoscopy or nasojejunal tube.
Trial registration:
Not applicable.
Plain language summary
We provide a detailed description of how to produce encapsulated donor faeces for faecal microbiota transplantation (FMT). FMT is a highly effective treatment for diseases where the intestinal microbiota is compromised, in particular Clostridioides difficile infection. Encapsulated application is patient-friendly and non-invasive, but protocols for the production of FMT capsules have not been published or are not reproducible. We here provide detailed guidance for standard-equipped hospital laboratories to enable capsule production under the current regulatory framework for substances of human origin in Europe. The dissemination of this protocol will increase patients’ access to a life-saving therapy.
Keywords
Introduction
Faecal microbiota transplantation (FMT) is the transfer of intestinal microbiota from a healthy donor to a patient to correct a disrupted intestinal microbiome. 1 Substantial evidence demonstrates above 90% effectiveness and has established FMT as a crucial treatment in managing recurrent Clostridioides difficile infections (rCDI).2–4 Despite its success in clinical use for CDI and promising results in emerging experimental indications, 1 only 10% of Europeans with rCDI receive FMT, primarily due to logistical challenges in providing the treatment. 5
As a substance of human origin (SoHO), donor faeces for FMT are governed by rigorous ethical and practical regulatory standards.6,7 This requires careful handling of the donated faeces sourced from healthy donors, which typically is best done in blood and tissue centres 8 under appropriate medical oversight. Administration of FMT requires a prescription by physicians and should be carried out by healthcare professionals. 9 Since its first use in modern medicine, 10 FMT has evolved drastically. Currently, FMT is managed in well-organised stool banks providing FMT components for administration with refined methods such as colonoscopy, 11 nasoduodenal tube 12 and capsules.13,14
Encapsulated FMT represents a scalable, non-invasive treatment option that requires fewer hands-on personnel 15 than other application methods while demonstrating effect rates comparable to colonoscopy-based FMT for rCDI.2,16 Despite the advantages related to encapsulated FMT, many FMT centres continue to favour endoscopic procedures with a lower potential for scalability.17,18 This preference may be explained by the absence of a universally accepted protocol for capsule production, along with the inherent complexities in developing and validating such protocols. While existing reports do provide protocol outlines, they lack exhaustive details to be fully operational. 16 Moreover, no currently published capsule protocol meets the legislative requirements for, that is, procurement, processing, validation, quality requirements and implementation.
This paper aims to describe the Glyprotect protocol for preparing glycerol-conserved, encapsulated FMT and the experiences gained from preparing more than 2500 encapsulated FMT components with high clinical effect rates. 19 Tailored for standard-equipped laboratories, the protocol is a validated and reproducible protocol that adheres to stringent quality standards and legislative requirements. Conducted as a quality improvement study, we report it in accordance with the SQUIRE 2.0 guideline 20 (Supplemental Table 7).
Protocol development
The Glyprotect capsule was developed at the Centre for Faecal Microbiota Transplantation (CEFTA) at Aarhus University Hospital, Denmark, 9 in response to a growing clinical demand for FMT that could not be met by solely using endoscopic procedures. Initiated in 2018, the early capsule production and validation phases were conducted at a small scale in a research laboratory by trained technical staff. By 2020, the production protocol had been improved and tested sufficiently to be upscaled. New staff was trained to independently establish routine stool bank laboratory production within the framework of the public blood centre, integrated into the hospital facilities’ standard operations. 8 This increased the capsule production for clinical use (Figure 1).

Annual use of FMT components for clinical application since the CEFTA was initiated in 2014: (a) separated by treatment place, and (b) by application method. Capsule production was introduced in 2018 and enabled the scalability of the tissue establishment.
In our current setup, the Glyprotect FMT capsules are integrated into the daily routine clinical practice of treating patients with rCDI at public hospitals in Denmark. In response to the growing demand for FMT, capsule production has been further expanded and the production rate has increased exponentially (Figure 1). Today, around 90% of FMTs provided by CEFTA are applied using capsules. In 2023, CEFTA produced and conserved 1865 FMT components, predominantly Glyprotect capsules (n = 1766) and a limited number of liquid-based suspension cryobags (n = 99) (Figure 2). Of these, a total of 1094 components were used for patient treatment in 2023 (Figure 1).

(a) Encapsulated donor faeces for oral ingestion. (b) Liquid donor faeces suspension for FMT through nasojejunal tube or colonoscopy.
Clinical trials 21 and cohort studies 19 have validated the clinical effect and safety of the Glyprotect protocol, demonstrating cure rates exceeding 90% in trial-independent clinical practice. 19 As CEFTA remains a non-profit initiative based on a public hospital setting, we maintain transparent cost management and uphold equal pricing for both Glyprotect capsules and liquid-suspension FMT in cryobags. In 2022, the cost of each FMT component was €881. 22
Preconditions
Faeces donors
We rely on voluntary, unpaid blood donors to serve as faeces donors. 8 All donors are recruited and screened at the Blood Bank at Aarhus University Hospital, Denmark, as previously described, 23 in accordance with European tissue and cell standards. 7 In a standard donation round, the donors provide faeces donations up to five times within 30 days. At each donation, the donors confirm that they are healthy (questionnaire screening). Faeces and blood are screened at the first and the last donation in the donation round. To ensure optimal sample integrity, we adhere to a strict timeframe from collection to processing: the duration from the donor’s toilet visit to the freezing of the capsules is capped at 8 h, and this applies to both capsules and liquid-suspension cryobags. This period includes a maximum of 2 h for transportation to the laboratory and up to 6 h for laboratory processing. 9
Materials
The Glyprotect capsule protocol fits a standard laboratory with basic equipment requirements, including a fume cupboard. The whole procedure is performed in a fume cupboard and requires a homogeniser and a capsule-filling system, which must be acquired. For homogenisation, we used a Smasher® (BioMérieux, Marcy L’Etoile, France); for capsule-filling, the 0 ProFiller 1100 and 00 ProFiller 1100 (100-Hole Capsule Machine, by Capsule Connection, LLC, Prescott, AZ). Figure 3 summarises the necessary equipment for the Glyprotect protocol, and Supplemental Table 1 lists all utensils used.

Selected equipment for a protocol for encapsulated FMT.
The microbiota concentrate is double encapsulated in size 0 acid-stable ENprotect capsules and size 00 Vcaps® Plus capsules from Lonza (Capsugel/Lonza, Colmar, France).
Capsule storage is in 100 mL size buckets (Duma®; Gerresheimer, Düsseldorf, Germany) with matching lids (Duma® Twist-Off Closure). The lid contains an integrated desiccant designed to withstand humidity both during storage at −80°C and throughout thawing.
Protocol for Glyprotect capsule preparation
Standard operating procedure
This protocol was designed to concentrate intestinal microbiota by removing granules and liquid while avoiding intentional manipulation of the microbial composition. A detailed step-by-step protocol for Glyprotect capsule production is outlined in Table 1, and Supplemental Table 2 is a printer-friendly version for use in the laboratory and Supplemental Table 3 demonstrates a dose calculation example. Figure 4 illustrates the critical steps of the protocol.
Standard operation procedure for processing glycerol-based encapsulated suspension of donor faeces for FMT.

,‡,$Variables for determining capsule number and dose per component. The calculation example is shown in Supplemental Table 3.
FMT, faecal microbiota transplantation.

Processing of donor faeces for FMT.
After delivery to the laboratory, the faeces donation is kept at 2°C–8°C until processing. The donation is weighed, and the stool consistency is assessed and graded according to the Bristol Stool Scale (BSS). 24 For a standard preparation, 45–50 g of crude faeces grade 3–4 are used per FMT component. Faeces donations with BSS outside this range or a wet weight below 45 g are discarded. Donations with wet weights above 355 g are inspected visually for integrity by at least two trained technicians.
Safety samples are collected from all donations, which enables re-screening and research while also ensuring traceability at a microbial level. Crude stool safety samples are collected in 2 mL tubes for each donation and stored at −80°C.
For processing, the crude faeces are first diluted in sterile saline and homogenised in a plain blender bag using a Smasher (BioMérieux). The homogenised faeces are centrifuged in three centrifugation steps to separate faeces into an upper phase I, an intermediate phase II and a lower phase III (Figure 4). Each phase represents different density properties with different liquid and microbial masses. The first centrifugation step serves to remove any large granules and fibres. Steps 2 and 3 serve to remove liquid and concentrate microbiota, thereby ensuring a microbiota-rich suspension with a low water content. Glycerol is added before and after the third centrifugation.
Capsule filling is performed using stainless steel fillers and automated pipettes with 650 µL in each capsule. For encapsulation, we utilise a double encapsulation technique. The microbiota concentrate is first encapsulated in size 0 acid-resistant capsules, followed by a secondary encapsulation in size 00.
After manual capsule filling and double encapsulation, the Glyprotect capsules are counted, registered and transferred into labelled desiccant buckets ready to be stored at −80°C. The approved range of capsules (one FMT capsule component) for one FMT treatment is 15–30 capsules, which are swallowed within 15–30 min. Each FMT capsule component originates from 45 to 50 g wet-weight faeces.
Cleaning and disinfection are crucial parts of the standard operating procedure and aim to avoid cross-contamination. Cleaning is a two-step process performed before and after each preparation, regardless of whether the preparation is from the same donor or two different donors. The first step is to clean off visible dirt. The second step is disinfection using a suitable method, such as a qualified dishwasher or wet wipe chlorine cloth followed by a 70% ethanol application.
Documentation, release and traceability
Registration and collection of multiple data throughout each FMT component preparation are useful when tracing and validating the production and the capsule product. This allows for data-driven learning and facilitates potential troubleshooting. Sample weights and BSS grades are collected for each donation, and measures at each critical step are documented (Supplemental Table 2). The faeces content in each capsule (noted as wet weight) and the number of capsules per FMT component can be calculated using these measures.
All components are quarantined in a separate freezer until all screening tests have been approved.
FMT component preparation, their release upon approval and data for full traceability, allowing for potential look-back in case of adverse reactions, are documented in the same system as used for blood components, that is, a nationwide blood bank registration system. This system is compliant with regulatory tissue and cell standards. All components are labelled with a unique donation code and component number based on the ISBT 128 coding system. The component number links to the donor’s identification, donation date and number of components from the stool donation in question (Table 1).
Capsule storage
The Glyprotect capsules are packed based on the number of capsules calculated for a component and distributed in buckets (one bucket is one FMT capsule component) with desiccant lids. Bucket labelling with corresponding component numbers is essential to secure traceability. Buckets containing FMT capsules are quarantined in a separate −80°C freezer until screening results determine whether to proceed with the release or discard of the components. Released buckets are moved to a release storage freezer where they can be stored at −80°C for up to 2 years from the donation date without compromising the clinical effect.
On the day before an FMT, we recommend transferring the FMT component(s) from −80°C to a −20°C freezer to reduce thawing time and prevent the potential risk that the outer capsules crack. Once capsules are stored at −20°C, they are stable for at least 4 weeks. 18 Although we observed capsule cracks in some components after thawing directly from −80°C, we did not perform formal studies to quantify the number of cracked capsules when thawing directly from −80°C versus gradual thawing via −20°C. Also, we did not test if long-term storage at −20°C is in fact feasible.
Fully thawed capsules should be used within 4 h to prevent capsule disintegration.
Adherence to regulatory standards
Technical guidance from international expert groups facilitates and standardises quality improvement through experience exchange. 7 Adherence to regulatory standards is constantly improved in a dialogue with national competent authorities for blood, tissues and cells.
Technical guidance for obtaining and processing intestinal microbiota was established in the 4th edition of the guide to the quality and safety of tissues and cells for human application, issued by the European Directorate for the Quality of Medicines & HealthCare of the Council of Europe (EDQM) in 2019 and further elaborated in the recently published 5th edition. 7 The latest edition includes monographs for both liquid suspension and encapsulated FMT. Additionally, the proposed integration of donor faeces as a SoHO in the Blood, Tissues and Cells regulation of the European Union (EU) 6 will further clarify the regulatory domains of donor faeces and faeces-derived products for human application. Engaging in European networks for the use of FMT, we aim to further improve the quality and safety of microbiome therapies. 25
Validation studies
Capsule integrity
The outer and inner capsules used in the Glyprotect protocol are ENprotect (inner) and VCap Plus (outer; Lonza/Capsugel, Colmar, France). We validated these capsules against prespecified criteria to ensure their compatibility with the Glyprotect protocol for oral delivery of glycol-conserved faecal preparations. The inner capsule, ENprotect, was tested for its ability to securely contain the glycerol–stool mixture in acidic conditions without leaking. The outer capsule, Vcap Plus, was evaluated for its durability through freeze–thaw cycles, surface cleanliness before ingestion and initial resistance to stomach acid.
The ENprotect capsules were evaluated at room temperature and after thawing from −80°C, with specification requirements for no visible leakage and minimally insignificant denting after 4 h (Supplemental Table 4 and Supplemental Figure 1). Acid resistance tests for both ENprotect #0 and Vcap Plus #00 capsules involved parallel single-capsule immersion in various pH solutions: acidic (pH 1.5), stomach fluid (pH 1.5 or pH 1.0) and water (pH 7) or stomach fluid (pH 6). The capsules were expected to withstand acid and stomach fluid for up to 4 h without leakage and dissolve in neutral pH after 2 h, simulating the transit through the stomach and dissolution in the small intestines (Supplemental Tables 5 and 6 and Supplemental Figure 2). All tested capsules met the required specifications.
Microbiota counts and dose measurements
Flow cytometry-measured microbiota counts served as validation markers during protocol development, providing insights into the alterations of the microbial concentration. Samples of non-processed wet-faeces and final Glyprotect capsules from four different donors were stained for DNA by SYTO9 (only with a SYTO™ 9 Green Fluorescent Nucleic Acid Stain) green fluorescent nucleic acid stain (SYTO9) from the LIVE/DEAD™ BacLight™ Bacterial Viability and Counting Kit for flow cytometry (Invitrogen, Thermo Fisher Scientific, Waltham, MA). Each sample was analysed in duplicate measurements and adjusted consistently with diverse densities. Subsequently, samples were pre-diluted 1:10,000 before being stained with SYTO9 according to the protocol. The stained samples were incubated for 15 min and immediately analysed using a 488 nm blue laser on a MACSQuant Analyzer 10 (Miltenyi Biotec, Bergisch Gladbach, Germany). The counts primarily reflect microbial counts based on size evaluation and flow cytometry sensitivity. Still, SYTO9 stains all viral, fungal and vesicle DNA. Due to sample diversity and complexity, the counts were based only on the SYTO9 staining and did not reflect viability or a specific microbe live/dead ratio.
Because the wet weight and consistency of raw donor faeces vary considerably with water content and debris, we standardised microbiota counts to defined volume units reflecting microbial density relative to the weight in raw faeces before processing and per capsule after final processing. We found a broad microbe count variation in wet, non-processed faeces that became less variable after processing. This indicates that microbe counts are more comparable and less donor-dependent after the processing (Figure 5). Variations in duplicates were minimal and insignificant. Assuming that all capsules may be filled equally to 650 µL, one capsule is estimated to contain 2.48 × 108 microbes per standardised unit (Table 2).

Boxplot showing the median measurements (counts/mL) of microbiota counts from four donors before (crude faeces) and after processing (end product) of donor faeces for FMT.
Microbiota counts at different steps during capsule processing for FMT and loss of bacteria during processing.
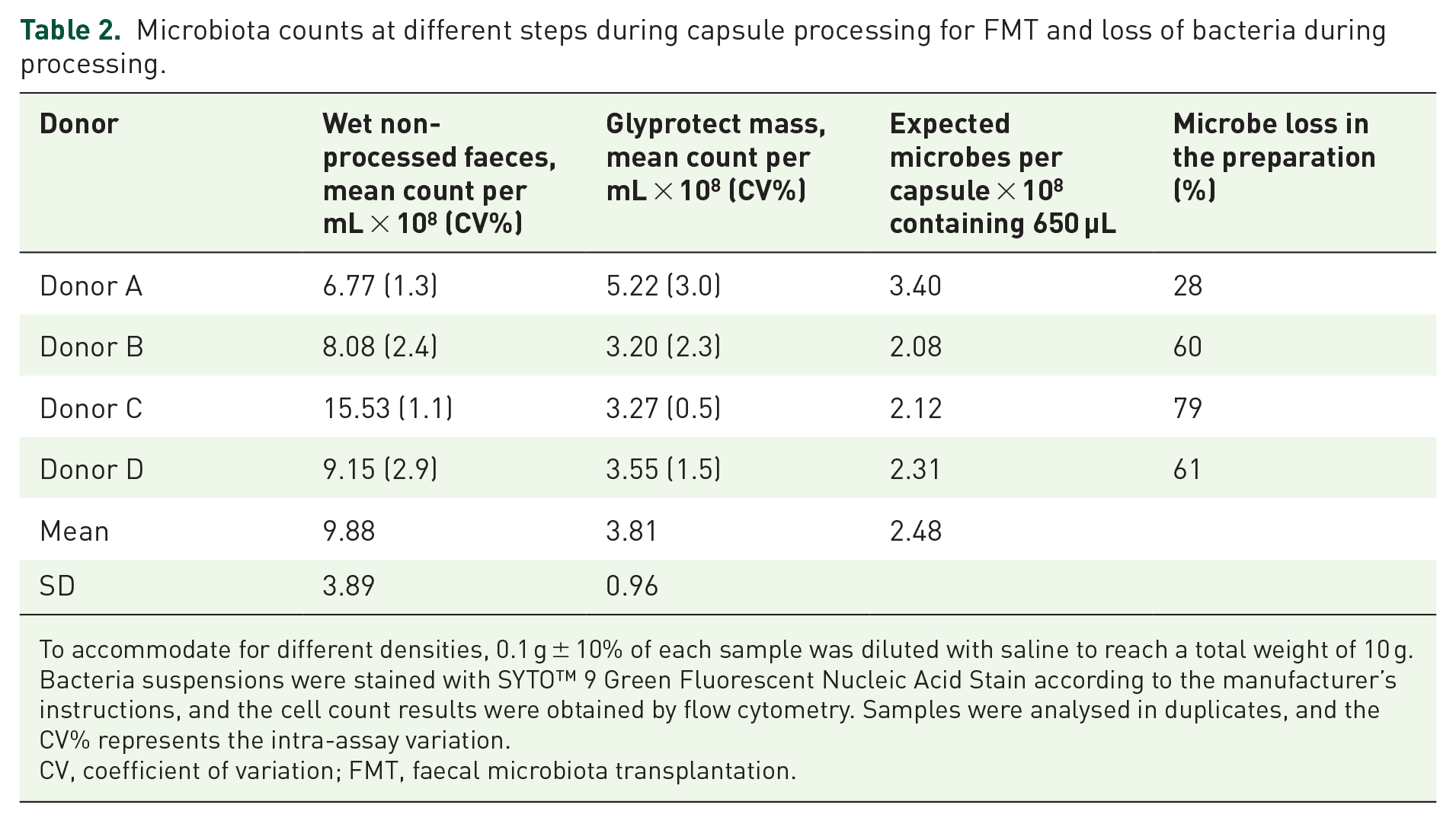
To accommodate for different densities, 0.1 g ± 10% of each sample was diluted with saline to reach a total weight of 10 g. Bacteria suspensions were stained with SYTO™ 9 Green Fluorescent Nucleic Acid Stain according to the manufacturer’s instructions, and the cell count results were obtained by flow cytometry. Samples were analysed in duplicates, and the CV% represents the intra-assay variation.
CV, coefficient of variation; FMT, faecal microbiota transplantation.
Standardisation throughout the process aims to optimise the robustness and reproducibility of capsule production. Still, variation exists. The number of capsules obtained from 50 g of crude donor faeces, equivalent to one FMT capsule component, typically varies between 15 and 30 capsules but may exceed this number. Deviations from this range do not automatically lead to the discarding of a donation. In a close collaboration between clinicians and technical staff, out-of-range-sized FMT components may be used for FMT. For instance, an FMT capsule component with fewer capsules may be used for patients who tolerate a low dose or can swallow only very few capsules. The maximum limit of 30 capsules for an FMT capsule component is arbitrary and was selected following a pilot trial where the clinical effect of 30 capsules was observed. Analyses from our CDI patient cohort show that variations in the number of capsules per FMT component and the use of different protocol versions within the applied range did not affect the clinical outcomes. 19
Troubleshooting
Drawing from our accumulated processing experience, we propose several measures to handle unexpected outcomes, tackle challenges and offer suggestions for handling minor deviations. Table 3 summarises these suggestions for troubleshooting and problem-solving.
Troubleshooting during capsule processing for FMT.

FMT, faecal microbiota transplantation.
Discussion
We here describe a detailed, operational and reproducible protocol for processing glycerol-protected donor intestinal microbiota capsules for FMT used at large scale in the routine clinic. The use and clinical efficacy of the capsules have been validated in routine and experimental clinical use 19 and a recently published clinical trial. 21
Dosing of intestinal microbiota for human application remains a challenge. In this protocol, each FMT component contains concentrated intestinal microbiota from 50 g of crude donor faeces, yielding 15–30 capsules per component after mainly removing water and debris. Despite having the same starting point, we observed notable variations as the number of capsules varied from 15 to 30, leading to differing volumes of discarded material and subsequent loss of microbiota. Upon comparison with wet, non-processed faeces, it appeared that the donations with the highest microbiota counts were also the donations that suffered the greatest microbiota loss during processing. Although variations in crude faeces and the ensuing phases affect capsule content, we observed that the microbe loss evens out from the initially donated wet-weight faeces to the final product, that is, the glycerol–microbe mass for application. While this may result in the alignment of microbial counts in the applied capsules from separate donations, microbial counts should still be interpreted with caution, and they do not reflect microbe composition. A broader investigation is needed to increase our understanding of which microbes remain and which are discarded during processing and how this may affect the clinical efficacy of FMT.
We made several attempts to compensate for different microbe densities when sampling for microbe counting. Dietary fibres and undigested food may contribute to variation in wet faeces, and the dynamic addition of glycerol, essential to obtain optimal viscosity for pipetting, contributes to variation in suspension volume for capsule filling, hence affecting the number of capsules. Because glycerol is added before the final centrifugation stage, it is not possible to determine the precise glycerol concentration in the capsules. Eventually, to minimise variation in the product and establish a standard FMT dosing unit, it may be expedient to determine a dose for FMT based on microbial counts following the first removal of liquid and debris rather than relying on the wet-weight measurement of crude faeces. This approach would enable defining a dosage based on the number of microbes per capsule volume, offering a more precise dose measurement for FMT administration.
This study assessed bacterial losses during processing by determining bacterial counts from fresh faeces and FMT capsules. This provided new insights to improve capsule validation. Future studies should provide more data on capsule quantity and composition, including counting viable bacteria in FMT capsules, fresh faeces and liquid suspensions for infusion. This could contribute to the further development and standardisation of capsule processing and relate it to the clinical effect of encapsulated FMT and liquid suspension FMT for different indications.
Minimising water content in the applied capsules is pivotal for reducing the number of capsules required per treatment and increasing capsule stability. We describe the use of glycerol-conserved donor faeces and water removal through centrifugation. During further protocol optimisation, lyophilisation may further minimise water content. Studies have reported comparable efficacy between using lyophilised donor faeces for FMT and employing glycerol-conserved donor faeces.13,26 Moreover, lyophilisation may offer less restrictive storage conditions, albeit the equipment required for lyophilisation is costly. Additional improvement steps, particularly those aimed at conserving microbes during processing, should be investigated to set benchmarks for improvements in clinical efficacy.
A scalable system for donor recruitment, donor screening, laboratory processing and clinical application is pivotal to meeting the growing demand for FMT in routine and experimental indications. In our experience, it is necessary to implement a protocol for encapsulated FMT to achieve this goal. At the same time, while maintaining this protocol, the availability of liquid suspension FMT remains essential for patients with swallowing difficulties or compromised gut access, such as patients with ileus secondary to fulminant C. difficile colitis. Also, it could be necessary to consider different doses or application forms according to the clinical or experimental indication for FMT. Comparing capsules and liquid suspension cryobags, an increased faeces wet weight dose could potentially compensate for a lower microbial count in capsules. Regardless, a fully operational FMT service should include a close collaboration between the tissue establishment and clinical services to cater to diverse patient needs. 3
In conclusion, the present paper provides a thorough and operational protocol that enables standard-equipped laboratories to transform crude faeces into encapsulated FMT in conformity with established standards. Implementing this protocol has the potential to enhance patient access to this treatment option.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848241289065 – Supplemental material for Encapsulated donor faeces for faecal microbiota transplantation: the Glyprotect protocol
Supplemental material, sj-pdf-1-tag-10.1177_17562848241289065 for Encapsulated donor faeces for faecal microbiota transplantation: the Glyprotect protocol by Mette Mejlby Hansen, Nina Rågård, Pia Winther Andreasen, Sara Ellegaard Paaske, Jens Frederik Dahlerup, Susan Mikkelsen, Christian Erikstrup, Simon Mark Dahl Baunwall and Christian Lodberg Hvas in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors thank all voluntary donors who provided faeces for patient treatment and protocol development at the Centre for Faecal Microbiota Transplantation (CEFTA). The authors also thank all technical personnel in the development laboratory and the production laboratory at CEFTA, along with clinical staff involved in FMT application in CEFTA.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
