Abstract
Background:
No head-to-head trial directly compares the effectiveness of vedolizumab (VDZ) and infliximab (IFX) in patients with ulcerative colitis (UC) who were naïve to biologic therapy.
Objectives:
We aimed to compare the clinical and endoscopic effectiveness of VDZ and IFX in biologic-naïve patients with UC in real-world settings.
Design:
It was a multicenter, observational, real-world cohort study conducted at five centers.
Methods:
Patients diagnosed with UC and treated with either IFX or VDZ as their first-line biologic therapy were retrospectively enrolled. Steroid-free remission, clinical response, clinical remission, and endoscopic healing at week 14 and week 52 were compared between the two groups after propensity score weighting.
Results:
A total of 199 patients (117 VDZ and 82 IFX) were included in the study. There were no significant differences in steroid-free remission (64.6% vs 56.1%, p = 0.224), clinical response (83.4% vs 73.4%, p = 0.086), or clinical remission (69.4% vs 60.1%, p = 0.174) at week 14. However, VDZ showed better results in steroid-free remission (67.5% vs 44.4%, p = 0.004), clinical response (69.7% vs 47.1%, p = 0.005), and clinical remission (67.5% vs 44.4%, p = 0.004) at week 52. In terms of endoscopic healing, VDZ was similar to IFX at week 14 (25.7% vs 17.4%, p = 0.185), but VDZ had a significantly higher rate at week 52 (29.5% vs 11.8%, p = 0.027). VDZ was found to be superior to IFX in therapeutic continuation (hazard ratio = 0.339, 95% CI: 0.187–0.614, p < 0.001). The rate of adverse events was similar between the two groups (6.8% vs 8.5%, p = 0.655).
Conclusion:
VDZ demonstrated similar clinical and endoscopic effectiveness to IFX at week 14 in biologic-naïve patients with UC, but appeared to be superior at week 52. The safety outcomes were comparable between the groups.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory intestinal disease characterized by symptoms such as abdominal pain, hematochezia, and tenesmus. 1 The persistent disease activity of UC poses challenges in treatment, leading to significant social and economic burdens. 2 The therapeutic target of UC includes reducing inflammation and symptoms, maintaining remission, and promoting endoscopic healing. Therefore, a more aggressive treatment is required. 3 Conventional therapies such as 5-aminosalicylate (5-ASA), steroids, and azathioprine are commonly used in the treatment of UC. However, there is still a considerable proportion of patients who do not respond or are intolerant to traditional therapies, requiring new drugs acting on different therapeutic targets to address an unmet clinical need. 4
Guidelines for UC recommended the early use of biologics rather than a gradual step-up strategy after the failure of 5-ASA therapy in moderate-to-severe UC, placing biological therapy in a more important position.5,6 Recently, TNF-α inhibitors (TNFi) like infliximab (IFX) and adalimumab, along with anti-integrin α4β7 monoclonal antibodies such as vedolizumab (VDZ), have gained approval in China for the treatment of UC patients.7–10 Among these, IFX and VDZ are the most frequently utilized options in China, and both of them have been proven to be effective in inducing and maintaining remission of UC.11,12 However, there is a lack of head-to-head randomized trials comparing the effectiveness of VDZ and IFX, which has resulted in an ongoing debate regarding the selection and positioning of IFX and VDZ, particularly in biologic-naïve (bio-naïve) patients. Only one head-to-head trial (VARSITY study) has confirmed the superiority of VDZ over adalimumab in clinical remission and endoscopic improvement at week 52, but it is unknown whether the findings from VARSITY can be applied to IFX. 13 Several observational studies and post hoc analyses have indirectly compared the effectiveness of VDZ and IFX, but they have yielded varying conclusions.14–17 Moreover, there is limited data on endoscopic efficacy comparison in existing studies, and the majority of participants were from European and American populations. In this study, we aimed to comprehensively evaluate the clinical and endoscopic effectiveness of VDZ and IFX in both the induction and maintenance stages in the Asian population, providing real-world evidence for clinical decision-making.
Methods
Study design and population
This was a retrospective, observational, real-world cohort study conducted at five centers in China (The Sixth Affiliated Hospital, Sun Yat-sen University; The First Affiliated Hospital of Nanchang University; Peking University Shenzhen Hospital; Second Affiliated Hospital of Guangzhou Medical University, and The First Affiliated Hospital of Hainan Medical University). Consecutive patients diagnosed with UC, who had received at least one dose of VDZ or IFX between January 2020 and September 2023, were considered for inclusion in the study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 18
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) patients with a confirmed diagnosis of UC and were bio-naïve 19 ; (2) patients who received either VDZ or IFX for treatment; (3) patients with moderate-to-severe activity according to the Mayo score or mild activity but refractory to traditional drugs; and (4) patients were no less than 18 years old.
The exclusion criteria were as follows: (1) patients with a definitive or transitory stoma and (2) patients with insufficient clinical data to evaluate efficacy at baseline and week 14 or 52.
Data collection
Data were extracted from the electronic medical records, and information collected included age, sex, active smoking status at baseline, disease duration, symptoms based on patient-reported outcomes, disease location classified by the Montreal Classification, disease activity based on the Mayo score, medication history, endoscopic manifestations, laboratory tests (such as C-reaction protein (CRP) and albumin (ALB)), therapeutic regimens, and adverse events (AEs). Clinical and objective outcome measures at 0 week, 14 ± 4 weeks, and 52 ± 8 weeks were collected for analysis.
Investigated drugs
Both the two drugs were administered in a standard regimen. VDZ was administered at a dose of 300 mg, while IFX was administered at a dose of 5 mg/kg. Both drugs were given intravenously at 0, 2, and 6 weeks for induction and every 8 weeks for maintenance. Treatment escalation during the therapy period was allowed at the physician’s discretion. The selection of the therapeutic optimization regimen was made after shared decision-making between the physician and the patients, such as shortening the medication interval, dose escalation, concomitant steroids or immunomodulators use, and so on.
Outcomes and definitions
The primary outcome was steroid-free remission at week 14 and 52. Secondary outcomes were clinical remission and endoscopic healing at week 14 and 52, therapeutic continuation, and the rate of AEs during the follow-up period.
Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) proposed by the International Organization for the Study of IBD (IOIBD) was adopted in this study, 3 that is, clinical remission was defined as pMayo ⩽ 2 with no single subscore > 1, and steroid-free remission defined as patients in clinical remission without concurrent use of steroids. Clinical response was defined as pMayo score reduction of 3 or more points and 30% or more from baseline, with a rectal bleeding subscore reduction of 1 point or more, or achieving clinical remission after treatment. Endoscopic healing was defined by Mayo endoscopic score (MES) = 0 points. Disease severity at baseline was evaluated using the Mayo score, with mild being defined as a score of 3–5, moderate as a score of 6–10, and severe as a score of 11–12. 20 In determining the proportion of patients achieving clinical and objective response or remission at week 14 and 52, patients who had failed VDZ/IFX and discontinued therapy were categorized as nonresponders, both in clinical and endoscopic assessments. Patients who responded to treatment but with inadequate follow-up time were not included in calculating the overall response rate at specific time points. 21
Statistical analysis
Patient characteristics were described using the mean and standard deviation or median and interquartile range for continuous variables. The χ2 or Fisher’s test was used to compare the proportions of the categorical variables. Student’s t-test or its non-parametric counterparts were used to compare quantitative variables.
To minimize the effect of confounding variables, a propensity score with inverse probability weighting (IPW) was used in the main analysis. First, the propensity score was estimated by logistic regression models as the predicted probability of initiating treatment with VDZ on age, sex, active smokers, disease duration, disease location, 5-ASA and steroids exposed, concomitant 5-ASA or steroids use at baseline, Mayo score at baseline, disease severity, proportion of CRP > 10 mg/L and ALB < 35 g/L at baseline, which based on a comprehensive literature review.22,23 Second, we calculated stabilized weights based on the propensity score. These weights were further used in the analysis models. The level of balance between the VDZ and IFX groups in the unweighted sample (original cohort) and in the weighted sample (pseudo-cohort obtained by weighting) for all variables included in the IPW analysis was verified by computing the standardized mean differences (SMDs), and SMD ⩽ 0.2 has been taken to indicate a negligible difference between treatment groups. Results from the outcomes using IPW analysis were replicated using multivariate logistic regression analysis for validation. We also computed odds ratios (ORs) and their 95% confidence intervals (CI).
The Kaplan–Meier curve was used to describe therapeutic continuation. Cox proportional hazards regression was used to investigate the association between drugs and therapeutic continuation. Results were expressed as hazard ratio (HR) and 95% CI.
Predictors of steroid-free remission at weeks 14 and 52 were identified through univariable and multivariable (including only factors with p < 0.1 at univariable analysis) logistic regression. ORs and 95% CI were also calculated in this analysis.
All statistical tests were two-sided, and statistical significance was defined as p < 0.05. Statistical analyses were performed using R software (developed by the R Foundation for Statistical Computing, version 4.4.2).
Results
Baseline characteristics of the patients
A total of 271 patients with UC were treated with either VDZ or IFX between January 2020 and September 2023. Among them, 199 patients met the inclusion criteria, with 117 receiving VDZ and 82 receiving IFX (Figure 1). The baseline characteristics are presented in Table 1. Before adjustment, patient characteristics were similar between the two groups, except for age (43.0 ± 15.7 vs 38.3 ± 13.2, p = 0.028), previous 5-ASA usage (95.7% vs 82.9%, p = 0.005), previous steroids usage (35.9% vs 57.3%, p = 0.004), Mayo score at baseline (8.0 ± 2.6 vs 9.1 ± 2.4, p = 0.002), the proportion of ALB <35 g/L (29.1% vs 45.1%, p = 0.029), and ALB at baseline (38.1 ± 6.4 vs 35.9 ± 5.3, p = 0.012). After IPW adjustment, no statistically significant difference between the two groups was observed, and the SMDs for all included variables were less than 0.2 (Table 1; Figure 2).

Flow chart illustrating the selection of patients.
Baseline data of patients in 14-week follow-up group.

p < 0.05 indicates statistical significance. ALB, albumin; 5-ASA, 5-aminosalicylate; CRP, C-reaction protein; IFX, infliximab; IPW, inverse probability weighting; SMD, standardized mean difference; VDZ, vedolizumab.
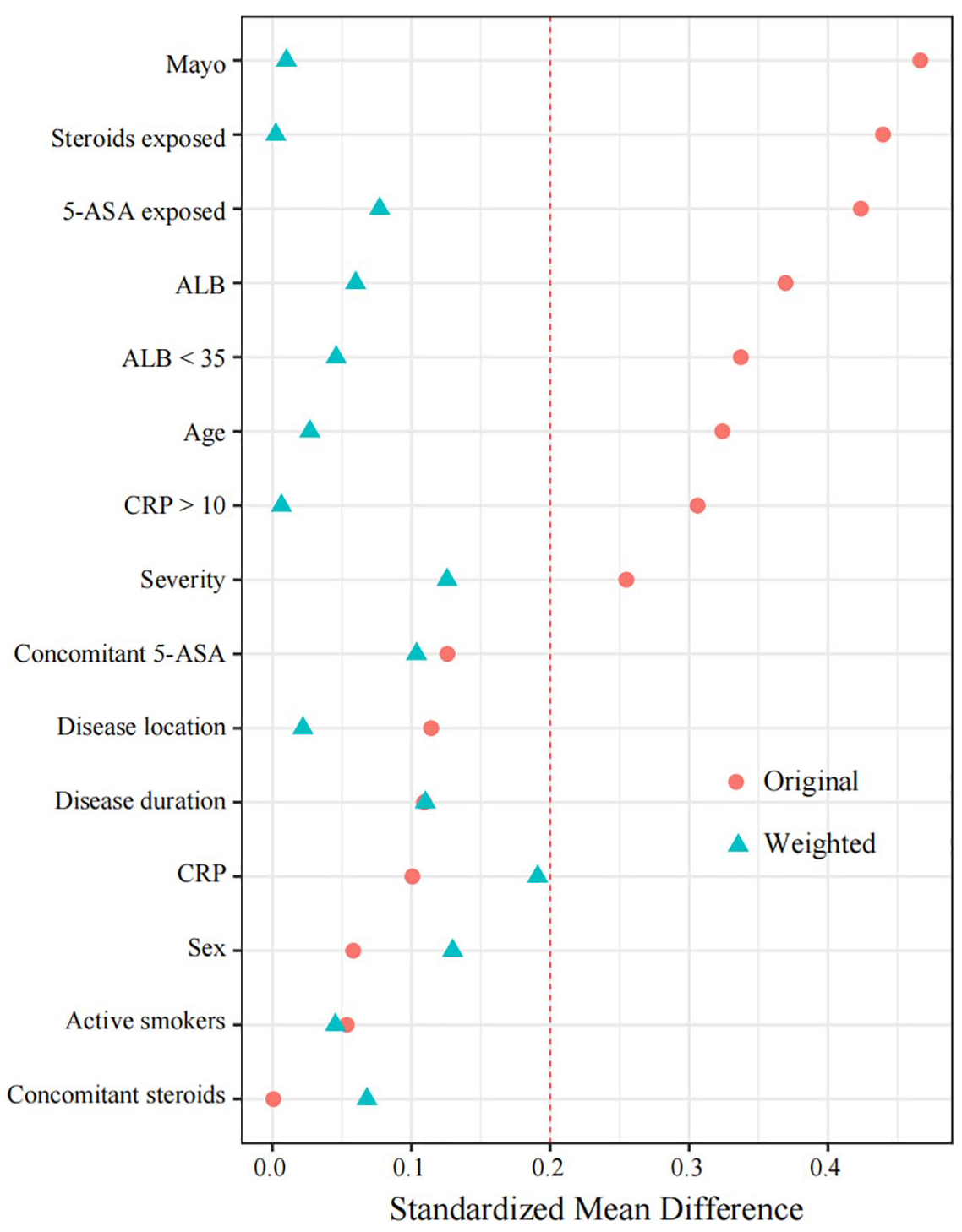
Standardized differences in original and weighted cohort obtained by IPW at week 14.
Clinical effectiveness
At week 14, no significant difference was found between the VDZ and IFX groups in steroid-free remission (64.6% vs 56.1%, p = 0.224, OR = 1.429, 95% CI: 0.803–2.547), clinical response (83.4% vs 73.4%, p = 0.086, OR = 1.829, 95% CI: 0.919–3.672), and clinical remission (69.4% vs 60.1%, p = 0.174, OR = 1.505, 95% CI: 0.834–2.721) after adjustment (Figure 3(a)–(c)).
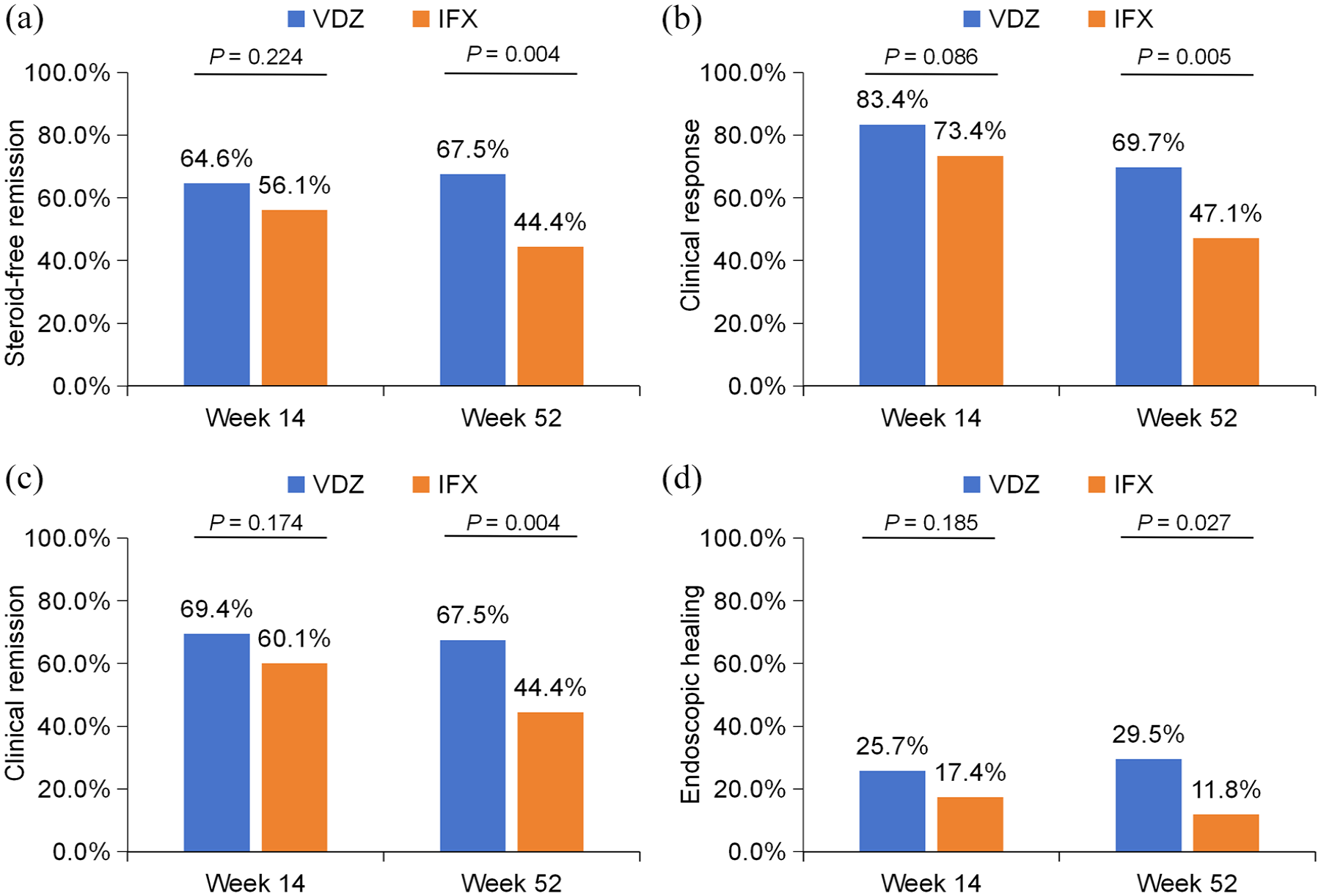
Comparison of clinical and endoscopic effectiveness between VDZ and IFX. (a) Comparison of steroid-free remission at week 14 and week 52; (b) comparison of clinical response at week 14 and week 52 in total patients; (c) comparison of clinical remission at week 14 and week 52; and (d) comparison of endoscopic healing at week 14 and week 52.
At week 52, 41 patients were excluded from the analysis due to insufficient data and 158 patients were finally included for analysis. IPW was conducted again to ensure comparability between the two groups (Supplemental Table 1 and Figure 1). However, VDZ was superior to IFX in achieving steroid-free remission (67.5% vs 44.4%, p = 0.004, OR = 2.608, 95% CI: 1.368–5.050), clinical response (69.7% vs 47.1%, p = 0.005, OR = 2.581, 95% CI: 1.348–5.020), and clinical remission (67.5% vs 44.4%, p = 0.004, OR = 2.608, 95% CI: 1.368–5.050) at week 52 in the adjusted population. The original clinical effectiveness is displayed in Supplemental Table 2. Similar results were obtained using multivariate logistic regression analysis as a sensitivity analysis (Figure 4(a); Supplemental Tables 3 and 4).
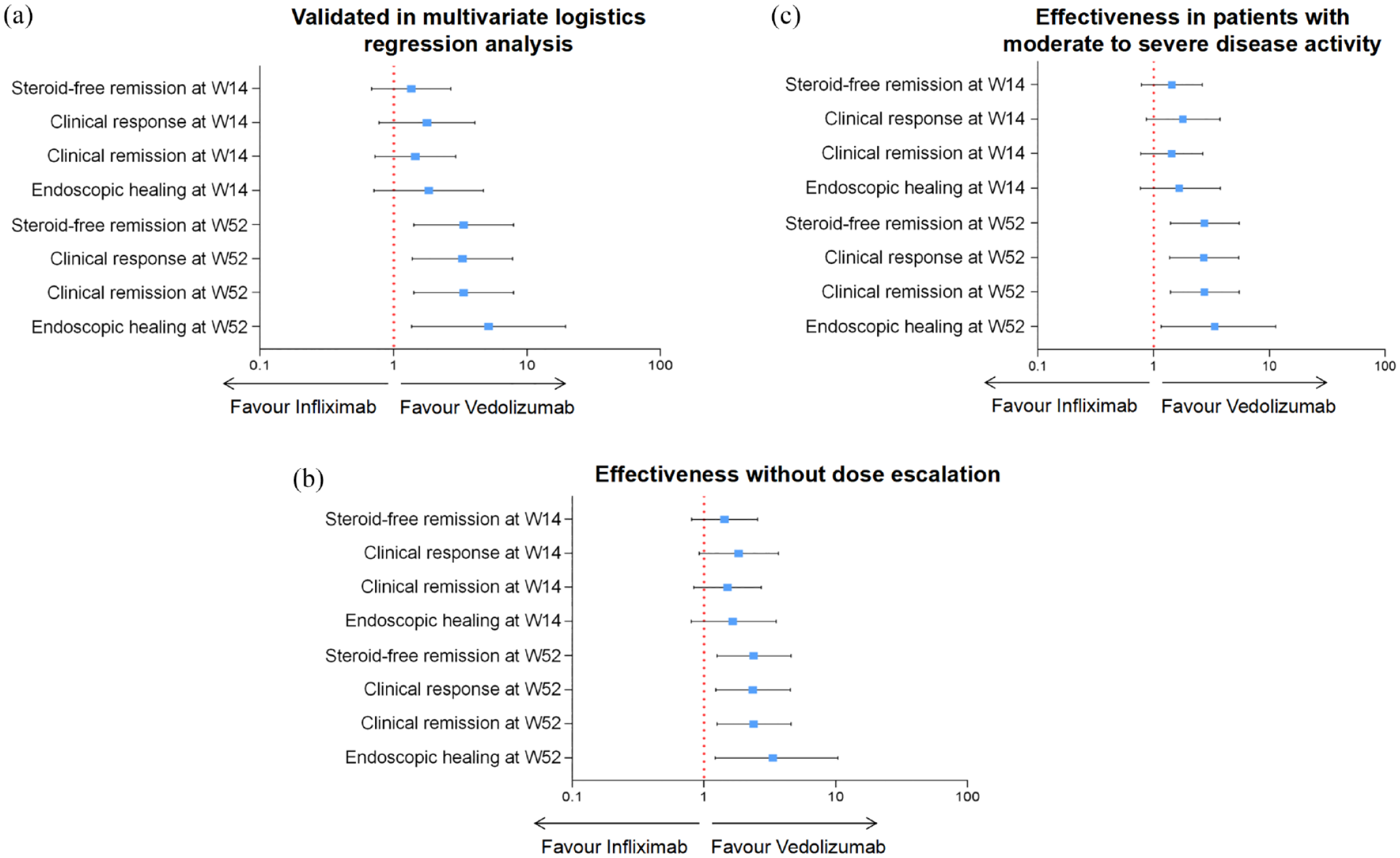
Sensitivity analyses for the comparison of clinical and endoscopic effectiveness between VDZ and IFX. (a) Replicating results from IPW analyses by multivariate logistics regression analyses; (b) comparison of effectiveness if dose escalation was regarded as treatment failure; and (c) comparison of effectiveness in patients with moderate-to-severe disease activity.
We conducted sensitivity analyses for clinical effectiveness comparison focused on patients with moderate and severe disease activity, and the outcomes were consistent with our main findings (Figure 4(c)).
Endoscopic healing
A total of 176 patients (103 VDZ, 73 IFX) underwent endoscopic evaluation from week 0 to week 14, while 112 patients (57 VDZ, 55 IFX) were evaluated from week 0 to week 52. At week 14, the rate of endoscopic healing between the two drugs was comparable (25.7% vs 17.4%, p = 0.185, OR = 1.646, 95% CI: 0.799–3.529) after IPW. However, VDZ showed significantly better results in terms of endoscopic healing (29.5% vs 11.8%, p = 0.027, OR = 3.131, 95% CI: 1.184–9.260) at week 52 compared to IFX (Figure 3(d)). The original endoscopic effectiveness was displayed in Supplemental Table 2. Similar results were obtained using multivariate logistic regression analysis as a sensitivity analysis (Figure 4(a); Supplemental Table 5). Endoscopic effectiveness comparisons in patients with moderate-to-severe disease activity were also conducted in our studies, and the results were similar (Figure 4(c)).
Therapeutic continuation
The Cox regression adjusted for confounders showed that VDZ was superior to IFX in terms of therapeutic continuation (HR = 0.339, 95% CI: 0.187–0.614, p < 0.001). In addition, the Kaplan–Meier curve showing therapeutic continuation in the two groups differed significantly on the basis of the stratified log-rank test (p = 0.008; Figure 5).

Therapeutic continuation of VDZ- and IFX-treated patients with UC after IPW.
Predictors of steroid-free remission at week 14 and week 52
At week 14, the multivariate logistic regression analysis revealed that concomitant 5-ASA at baseline in the IFX-treated group was associated with a higher probability of achieving steroid-free remission (p = 0.042, OR = 0.384, 95% CI: 0.152–0.966). However, no independent risk factor was identified in the VDZ-treated group. Regarding predictors of steroid-free remission at week 52, it was found that achieving steroid-free remission at week 14 (p = 0.021, OR = 0.267, 95% CI: 0.087–0.820) was identified as an independent factor with a higher probability, while previous exposure to steroids (p = 0.012, OR = 4.247, 95% CI: 1.374–13.124) was identified as an independent factor with a lower probability in the VDZ-treated group. No independent risk factor was found in the IFX-treated group (Supplemental Tables 6–9).
Therapeutic optimization
In the VDZ-treated group, treatment optimization was performed on 11 patients (9.4%). This included eight patients who underwent interval reduction every 4 or 6 weeks, one patient who received concomitant steroids due to loss of response, and two patients who received induction therapy again in the maintenance stage. In the IFX-treated group, five patients (6.1%) underwent treatment optimization. Among them, three patients received dose escalation, and two patients received both dose escalation and interval reduction every 6 weeks. Sensitivity analyses were conducted to compare the clinical and endoscopic effectiveness between IFX and VDZ in UC patients regarding dose escalation as treatment failure, and the outcomes were consistent with our main conclusions (Figure 4(b)).
Safety
The incidence of AEs was similar between the two groups (6.8% vs 8.5%, p = 0.655). In the VDZ-treated group, five patients (4.3%) developed symptomatic enteritis associated with Clostridium difficile, two patients (1.7%) had rashes, and one (0.9%) experienced joint pain. No medication discontinuation occurred due to AEs.
In the IFX-treated group, three patients (3.7%) developed symptomatic enteritis associated with C. difficile or cytomegalovirus; three patients (3.7%) experienced anaphylaxis, with two patients having a rash, one experiencing chest tightness, difficulty breathing, and multiple rashes. One patient (1.2%) experienced upper respiratory symptoms. Three patients (3.7%) discontinued IFX treatment due to AEs, with two of them having severe allergic reactions, and one experiencing a severe intestinal infection.
Discussion
In this study, we compared both clinical and endoscopic outcomes of VDZ and IFX in bio-naïve UC patients. Our findings revealed that VDZ and IFX exhibited similarities in terms of steroid-free remission and endoscopic healing during the induction stage. However, VDZ demonstrated superiority in the maintenance stage and therapeutic continuation. Furthermore, the risks of AEs were comparable between the two drugs.
Although the VARSITY trial indicated that VDZ was more effective in clinical and endoscopic efficacy compared to adalimumab, there is not enough evidence to generalize to IFX. A prior meta-analysis suggested that IFX ranked higher than adalimumab, 24 and current guidelines recommended IFX as first-line therapy in UC rather than adalimumab.5,25 Moreover, another network meta-analysis suggested that IFX may be the most effective treatment for bio-naïve patients with UC (compared to VDZ, tofacitinib, and ustekinumab), but no significant difference was found between IFX and VDZ in terms of clinical remission and endoscopic improvement in this indirect comparison. 26 Therefore, more data are needed to compare the efficacy of VDZ and IFX exclusively.
Several observational studies have been conducted to compare the efficacy of VDZ and IFX, but the results have been highly controversial. Three studies have supported our findings. Cohorts from North America (subgroup: 143 VDZ and 165 IFX in bio-naïve patients) and Italy (32 VDZ and 50 IFX) showed that VDZ was superior to IFX in achieving clinical effectiveness.14,27 Another prospective real-world study (VEDOIBD) found that VDZ and IFX were similar in the induction stage, but VDZ had a significantly higher rate of steroid-free remission and therapeutic continuation after 2 years in subgroup analysis. 23 Conversely, a post hoc analysis of three UC clinical trial programs and a systematic meta-analysis demonstrated that IFX showed comparable or even better clinical effectiveness in the induction and maintenance stage.16,28 It is important to note that the patients included in the aforementioned studies were carefully selected based on strict inclusion criteria and enrolled in randomized clinical trials. Therefore, it should be emphasized that the outcomes derived from these highly regulated clinical trial settings may not be directly applicable to routine clinical practice. Furthermore, the controversial results among the mentioned studies highlight the need for endoscopic evaluation to comprehensively compare and validate the findings.
Few studies reported the endoscopic effectiveness comparison between VDZ and IFX. Narula et al. 16 conducted a post hoc analysis of randomized clinical trials and found that IFX was more effective than VDZ (36.0% vs 21.7%, p < 0.001) in achieving 1-year endoscopic remission (MES = 0). 16 The EVOLVE study, on the other hand, did not find a significant difference in mucosal healing (MES ⩽ 1) between the VDZ and TNFi groups (86.6% vs 80.6%, p = 0.66) over 24 months, but no subgroup analysis in terms of IFX. 29 By contrast, our real-world evidence suggested that VDZ was more effective in endoscopic healing (MES = 0) at week 52, further supporting the superiority of VDZ in the long-term stage. These differing results may be attributed to variations in real-world study practices or the focus of our study on a specific TNFi (IFX).
Our study found that concomitant 5-ASA therapy at baseline was associated with steroid-free remission at week 14 in the IFX-treated group, indicating that drug combination in the early stage may help reduce the burden of inflammation more quickly. In addition, we observed that achieving steroid-free remission in the early stage (week 14) was closely related to steroid-free remission at week 52 in the VDZ-treated group, which was consistent with previous reports. 30 This highlights the importance of early response and remission, indicating that early aggressive therapy may be beneficial for patients.
The rate of therapeutic optimization (9.4% for VDZ and 6.1% for IFX) in our study appeared to be lower than what has been reported in previous studies,31–33 which may attributed to the difference of baseline characteristics. Our study focused on bio-naïve patients who were followed up for only 1 year, and 15.4% of VDZ-treated patients and 8.5% of IFX-treated patients were in mild activity, which might explain the reduced need for treatment optimization. In addition, to minimize the impact of dose optimization on the results, we also conducted sensitivity analyses to compare the effectiveness regarding dose escalation as treatment failure.
No significant difference in the rate of AEs between the two groups was observed. However, since our study was retrospective, we may underestimated the incidence of minor AEs that did not require medical intervention. We also observed some cases of enteritis associated with C. difficile in both groups, but further investigation is needed to understand this association. In addition, our study identified some cases of therapeutic discontinuation due to the AEs of IFX, including anaphylaxis and intestinal opportunistic infection, which should arouse attention.
Our study had several strengths that should be highlighted. First, we provided comparative data on the effectiveness of VDZ and IFX in Asian populations, thus filling a gap in relevant data. In addition, we presented both clinical and endoscopic data, allowing for a comprehensive comparison between the two drugs. To minimize the potential bias, we utilized propensity score weighting to handle baseline confounders and reduce the impact of bias. The results were further validated through multivariate logistic regression, enhancing the persuasiveness of our findings. Furthermore, we also conducted sensitivity analyses on different endpoints to confirm our results.
However, our study has several limitations. First, the retrospective observational nature of this study had inherent limitations, so caution is required when interpreting the results of this study. It should be acknowledged that patients included in the IFX-treated group showed more severe activity compared to the VDZ-treated group according to baseline data before adjustment and it may potentially impact the results, even if we have balanced the baseline characteristics by IPW adjustment and conducted sensitivity analyses to further validate the outcomes. Second, some studies suggested that a combination therapy of IFX with azathioprine was superior to IFX monotherapy, and it may impact the results such as narrowing the potential gap between the two drugs.34,35 However, the main objective of our study was to compare the VDZ and IFX as monotherapy, and we believe that it has provided valuable insights in its own right. Third, the fecal calprotectin test was not a routine examination in our clinical practice, and the data on fecal calprotectin were not available. Finally, therapeutic drug monitoring (TDM) may give us some clues about the loss of response to biologics and guide treatment optimization, but it was not routinely conducted during this study. Patients with higher concentrations of IFX may have a higher rate of response, so it may impact the results potentially. However, the primary focus of our study was to compare the effectiveness of the two biological therapies under a standard regimen, and TDM was more often used as a tool to indicate whether treatment optimization was needed.
Conclusion
In conclusion, this real-world study suggested that VDZ was superior to IFX as the first-line biologic therapy for patients with UC in terms of clinical and endoscopic effectiveness in the long-term stage. Prospective, multicenter, large-scale cohort studies are needed to further confirm the results.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241281218 – Supplemental material for Comparison of clinical and endoscopic efficacy between vedolizumab and infliximab in bio-naïve patients with ulcerative colitis: a multicenter, real-world study
Supplemental material, sj-docx-1-tag-10.1177_17562848241281218 for Comparison of clinical and endoscopic efficacy between vedolizumab and infliximab in bio-naïve patients with ulcerative colitis: a multicenter, real-world study by Zhaopeng Huang, Jian Tang, Ruibin Wu, Shunhua Long, Wenke Chen, Tingna Lu, Qiuyue Xia, Yanhui Wu, Hongsheng Yang, Qingfan Yang, Zicheng Huang, Qin Guo, Miao Li, Xiang Gao and Kang Chao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
