Abstract
Background:
Clarithromycin plays an important role in eradicating Helicobacter pylori (H. pylori) through quadruple therapy. However, there is limited research on whether different forms of clarithromycin dosage have similar efficacies against H. pylori.
Objective:
We aimed to evaluate the efficacy of different forms of clarithromycin dosage in bismuth-containing quadruple therapy for eradicating H. pylori.
Design:
A single-center retrospective analysis comparing the efficacy of different forms of clarithromycin dosage in eradicating H. pylori.
Methods:
An analysis was conducted on patients diagnosed with H. pylori infection through the 13C-urea breath test (13C-UBT) at Henan Provincial People’s Hospital, China from 2020 to 2022 who were treated with either a dispersible or sustained-release clarithromycin tablet (500 mg each), alongside amoxicillin (1000 mg), a standard dose of proton pump inhibitors (PPIs), and bismuth citrate (220 mg), administered twice daily as part of bismuth-containing quadruple therapy. Treatment efficacy was assessed using 13C-UBT at least 4 weeks after treatment completion. The H. pylori eradication rate was the primary outcome of this study, and factors influencing it were analyzed.
Results:
Among 2094 screened patients, 307 with H. pylori infection (mean age, 41.8 ± 0.7 years; 43% men) received bismuth-containing quadruple therapy. Univariate analysis of the dispersible and sustained-release tablet groups revealed a lower eradication rate with the sustained-release tablet compared with the dispersible clarithromycin tablet regimen (75.26% (73/97) vs 95.26% (200/210), respectively; p < 0.05). Other factors, such as smoking, age, and PPI type, were not significantly associated with the cure rate. Multivariate analysis identified the form of clarithromycin dosage (dispersible vs sustained-release) to be an independent risk factor for eradication failure using the bismuth-containing quadruple therapy (odds ratio = 0.145, 95% confidence interval: (0.065–0.323); p < 0.05).
Conclusion:
The clarithromycin dispersible tablet demonstrated a higher H. pylori eradication rate, and the sustained-release clarithromycin tablet may be inappropriate for H. pylori eradication.
Plain language summary
The clarithromycin dispersible tablet therapy demonstrated a higher eradication rate for H. pylori infection, and clarithromycin sustained-release tablets may be inappropriate for the eradication of H. pylori.
Keywords
Introduction
Helicobacter pylori (H. pylori) is a gram-negative bacterium that occurs in the gastric mucosa in approximately 50% of the Chinese population.1,2 The majority of bacteria are located deep in the mucus, and they are close to the surface of the epithelium and can induce gastritis, gastric ulcer, pancreatitis, and cancer. 3 Diagnosis of H. pylori infection typically involves the utilization of gastric endoscopy and biopsy for immunohistochemical analysis. However, the complexity of the diagnostic procedures limits their use in some patients. As a noninvasive and rapid screening tool, the 13C-urea breath test (13C-UBT) is increasingly preferred owing to its convenience, leading to an increase in diagnosis. Following diagnosis, treatment typically involves high doses of antibiotics and proton pump inhibitors (PPIs) over extended periods. Over the past few decades, various therapies, including double, triple, and even quadruple regimens, have been extensively reviewed and discussed owing to the challenge of achieving complete eradication of H. pylori in all cases. Numerous studies have confirmed that antibiotic resistance in H. pylori and poor drug adherence on the part of patients could be risk factors for the failure of H. pylori therapy.4–7 Antibiotic resistance is progressively posing greater challenges, particularly in China.8–10 With declining eradication rates, bismuth has been incorporated into traditional therapies. Multiple studies have demonstrated that although the resistance rate to antibiotics has significantly increased, bismuth addition can overcome its resistance to a certain degree and achieve a relatively satisfactory eradication efficacy.11,12 The safety of bismuth-containing regimens has been confirmed, eliminating the need for dose adjustments for individual drugs when combined with bismuth. 13 The Maastricht V Consensus Report recommended bismuth-containing quadruple therapy as the first-line eradication regimen for geographic regions with high clarithromycin resistance. 14 At present, bismuth-containing quadruple therapy is the most commonly used regimen to eradicate H. pylori infection in China.
Clarithromycin is a macrocyclic lipid antibiotic that is widely utilized in the treatment of H. pylori infection as reported previously. 15 However, guidelines do not specify the preferred form of clarithromycin dosage for treating H. pylori infection. At our hospital, both dispersible and sustained-release tablets have been used and both forms of dosage have exhibited similar rates of adverse events, with sustained-release tablets featuring lower drug concentrations than dispersible tablets.16,17 Further research is required to determine whether they have the same efficacy as the regular formulation in the treatment of H. pylori, especially in the context of drug resistance.
This study aimed to assess the efficacy of different forms of clarithromycin dosage for the treatment of H. pylori infection. In this retrospective study, the eradication rate and risk factors such as smoking, alcohol consumption, age, sex, and PPI type that could affect the efficacy of bismuth-containing quadruple therapy were compared among different dosage forms of clarithromycin for eradicating H. pylori infection.
Materials and methods
This single-center retrospective study was conducted between January 2020 and June 2022 at Henan Provincial People’s Hospital, Zhengzhou, China. We enrolled patients with confirmed H. pylori infection via a 13C-UBT, who were subsequently treated with different forms of clarithromycin in the bismuth-containing quadruple regimen. The selection of clarithromycin dosage form to be administered to each patient was assigned at the discretion of the attending physician. Following a 14-day treatment period, post-treatment H. pylori status was evaluated at least 4 weeks after treatment completion. Patients who either did not undergo a 13C-UBT prior to treatment or declined to return for post-treatment assessment were excluded from this study. Amoxicillin was not included in the therapy. All patients included in this study had been diagnosed for the first time and had not used antibiotics for 14 days prior to diagnosis.
The sample size was determined using PASS 16.0 software (NCSS, USA). We estimated the eradication rate in dispersible tablet and sustained-release tablet treated groups to be 95% and 80%,8,12 respectively, with α = 0.05, β = 0.20, and sampling ratio = 1. We aimed to include at least 73 patients in every group.
The patients included in this study were stratified into two groups based on the form of clarithromycin administered. The patient gender, age, smoking habit, alcohol consumption, and treatment outcomes were collected during the study period. This study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 18 All authors had access to the study data and reviewed and approved the final manuscript.
Data analysis
The eradication rate of H. pylori was evaluated using different dosage forms of clarithromycin. Eradication rates were presented as percentages, whereas continuous variables were presented as mean ± standard deviation. Differences between groups were evaluated by Fisher’s exact test. Logistic regression analysis was used to identify the risk factors for eradication failure. A p-value < 0.05 was considered statistically significant. All data were analyzed using SPSS 23.0 software (IBM, Armonk, NY, USA).
Results
Demographic characteristics
The study flowchart is presented in Figure 1. A total of 2094 patients were screened in this study, of whom 1786 were excluded and 308 were enrolled and allocated to groups. In addition, the therapeutic program containing lansoprazole was excluded from the analysis due to sample size-related limitations. Eventually, 307 patients were involved in the analysis.
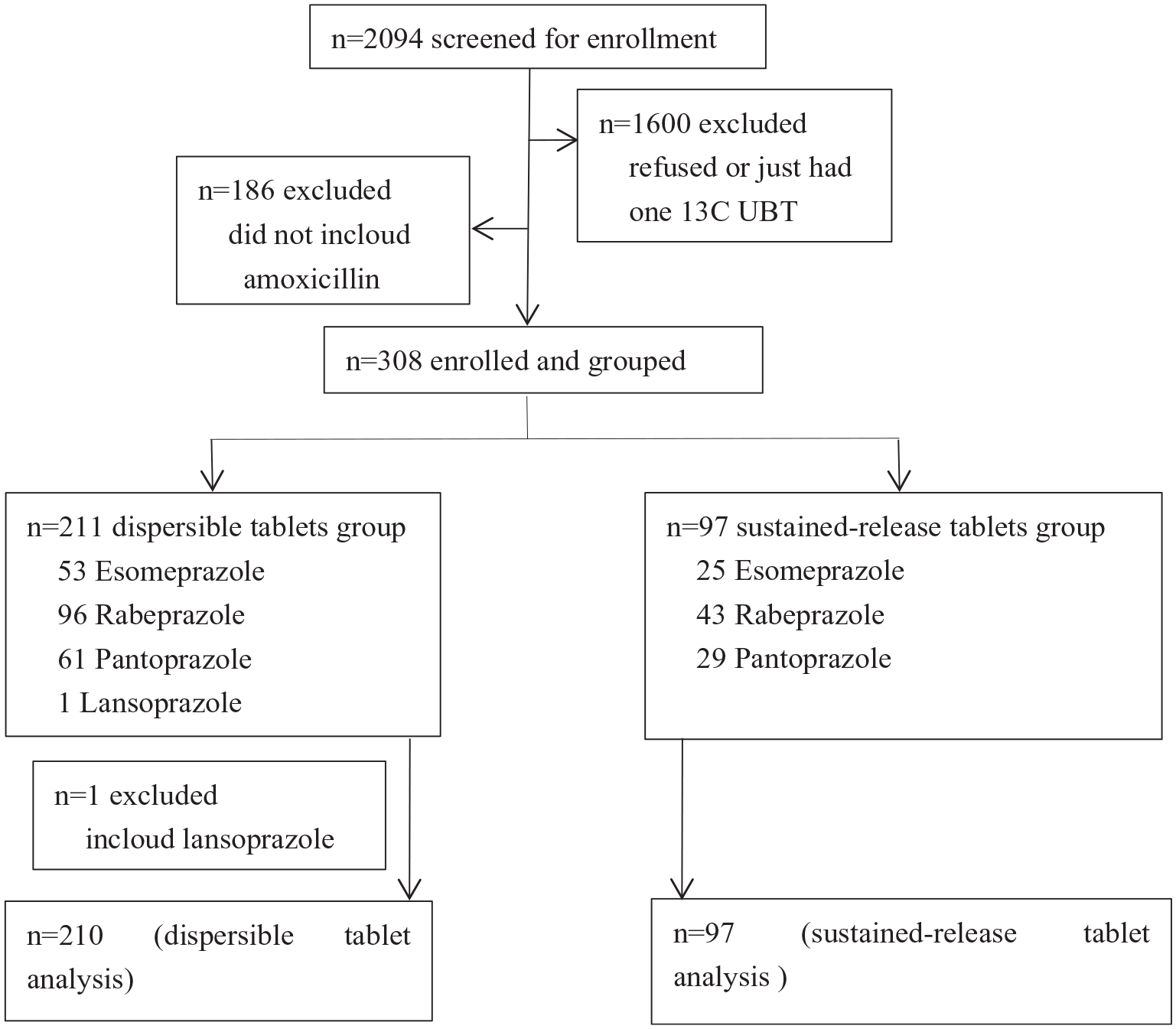
A flow chart of the study.
All patients were treated with bismuth-containing quadruple therapy. There was a discrepancy in the proportion of patients allocated to different dosage forms of clarithromycin: specifically, 210 patients received clarithromycin dispersible tablets, whereas the remaining 97 patients were assigned clarithromycin sustained-release tablets. The baseline and clinical characteristics of patients are listed in Table 1.
Patient demographics and baseline clinical characteristics.

SD, standard deviation.
Eradication outcomes
The outcomes of the bismuth-containing quadruple therapy are presented in Table 2. The overall eradication rate was 88.93% for all patients, with a rate of 75.26% for those treated with sustained-release tablets and 95.24% for those administered dispersible tablets.
Treatment outcomes of patients receiving bismuth-containing quadruple regimen therapy.

Univariate analysis of risk factors associated with H. pylori eradication failure using bismuth-containing quadruple therapy
The results of univariate analysis of the parameters influencing the eradication rate of H. pylori infection using bismuth-containing quadruple therapy are presented in Table 3. The form of clarithromycin dosage was significantly associated with eradication rate: the sustained-release tablet exhibited a significantly lower eradication rate compared with the dispersible tablet (75.26% (73/97) vs 95.24% (200/210), p < 0.05). Other factors, such as smoking, age, alcohol consumption, and type of PPIs, were not significantly associated with the cure rate.
Univariate analysis of factors affecting H. pylori infection eradication rate in bismuth-containing quadruple regimen treatment.

PPI, proton pump inhibitor.
Multivariate analysis of risk factors associated with H. pylori infection eradication failure in bismuth-containing quadruple therapy
The results of multivariate analysis of the parameters influencing the eradication rate of H. pylori infection using bismuth-containing quadruple therapy are presented in Table 4. The type and dose of clarithromycin were independent risk factors for H. pylori eradication failure (odds ratio (OR) = 0.145, 95% confidence interval (CI): 0.065–0.323; p = 0.000).
Multivariate analysis of independent risk factors predicting H. pylori eradication failure in bismuth-containing quadruple regimen treatment.
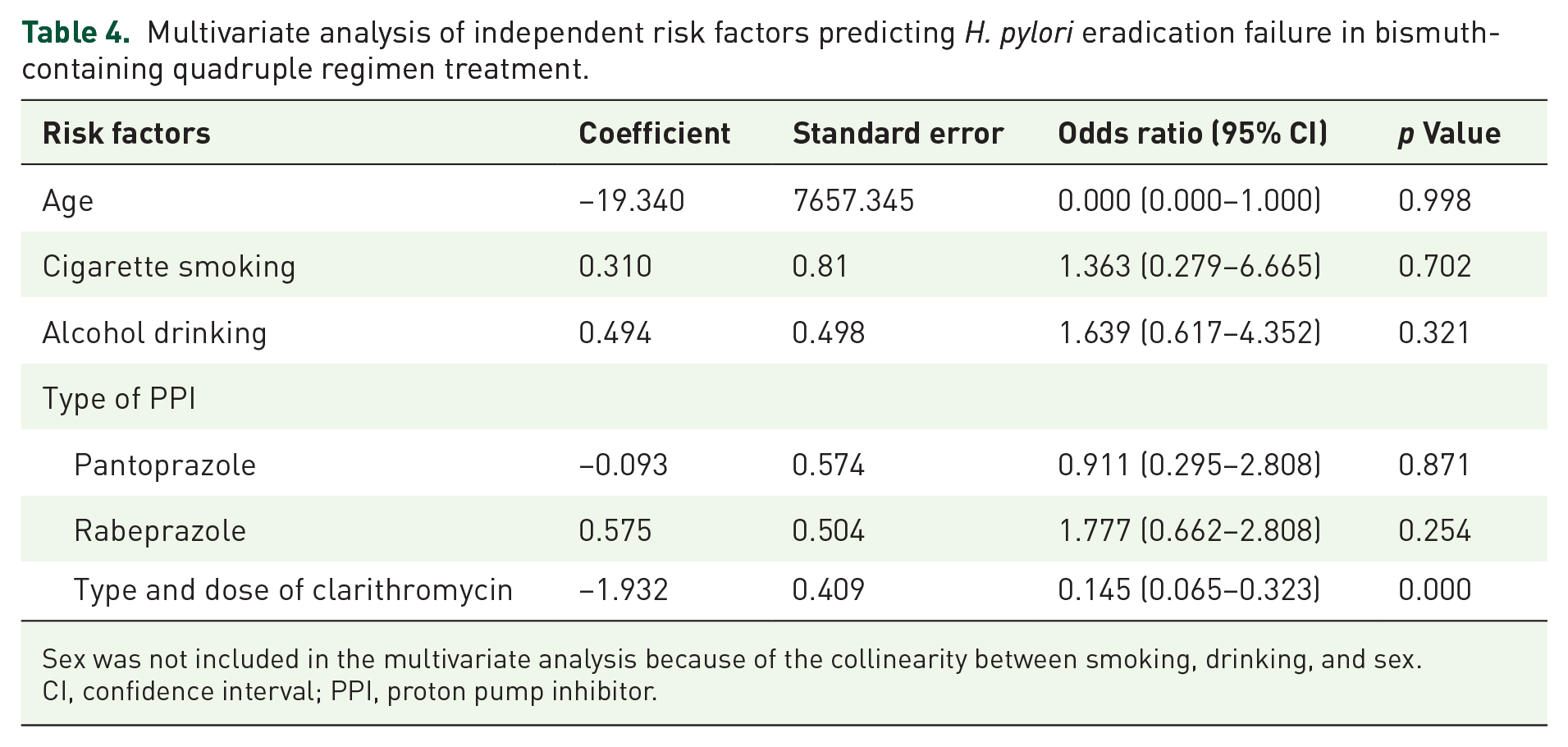
Sex was not included in the multivariate analysis because of the collinearity between smoking, drinking, and sex.
CI, confidence interval; PPI, proton pump inhibitor.
Discussion
This retrospective study included 307 H. pylori-infected patients who received different clarithromycin dosage forms (sustained-release or dispersible tablets) in bismuth-containing quadruple therapy. Univariate analysis indicated that the success rates of sustained-release and dispersible tablet therapies were 75.26% and 95.24%, respectively. Other factors such as age, sex, smoking, alcohol consumption, and PPI type were not significantly associated with the cure rate. The results of the multivariate analysis revealed that the clarithromycin dosage forms could contribute independently to the risk of H. pylori eradication failure when using bismuth-containing quadruple therapy.
The anomaly observed in the method of patient allocation to the dispersible and sustained-release treatment groups may interfere with the aforementioned conclusion. This is because the allocation of a patient to a particular treatment group was based on physician preference; some physicians prefer to use sustained-release tablets and others prefer dispersible tablets. This makes the patient grouping lack randomness. Even if the baseline data showed no difference between the two groups, this still remains a significant potential source of bias. Future randomized controlled trials are warranted to confirm the associations demonstrated in our study.
Recent reports have investigated the risk factors for H. pylori eradication failure, such as smoking, the consumption of tea, coffee, or alcohol, gastric ulcers, PPI type, and antibiotic resistance. Poor medication adherence rates and significant antibiotic resistance levels were significantly associated with cure rates as previously outlined.4,5 In the present study, only patients who underwent both pre- and post-treatment, 13C-UBT assessments were included, and no analysis of medication adherence was conducted. Given the rate of patient return for the post-treatment 13C-UBT, these patients likely demonstrated acceptable adherence comparable to that of the intention-to-treat population. Conversely, patients who declined the post-treatment 13C-UBT may have been less concerned about treatment effectiveness, raising uncertainty about their level of compliance with the treatment protocol.
By contrast, the antibiotic resistance level determines whether H. pylori infection can be successfully eradicated. A meta-analysis conducted in the Asia-Pacific region indicated that the overall prevalence of primary antibiotic resistance in H. pylori between 1990 and 2022 was 22%. 19 As the first antibiotic that was used for the eradication of H. pylori infection, the average resistance rate of metronidazole was 76.5%, and it even reached 95.4% in some areas. 20 Metronidazole may not be the preferred option for H. pylori eradication in China, despite the availability of Pylera capsules in American and European markets. Similar challenges exist with levofloxacin and clarithromycin, with recent primary resistance rates ranging from 68% to 76.93% for clarithromycin and from 55.2% to 61.58% for levofloxacin in China,21,22 which are higher than those reported in Europe. 23 Although the prevalence of primary antibiotic resistance varied widely across different regions, it remained stable for amoxicillin, tetracycline, and furazolidone. Amoxicillin plays a crucial role in the treatment regimen because of the challenges in obtaining tetracycline in clinical settings and the numerous adverse effects associated with furazolidone. Conversely, H. pylori strains have exhibited consistent sensitivity to amoxicillin over the years, with both primary and secondary resistance being rare occurrences. This rarity can be attributed to the need for simultaneous mutations at multiple sites in penicillin-binding protein-related genes to confer significant resistance that may occur infrequently.24,25 This explains why dual therapy combining high-dose amoxicillin with a PPI has been explored as a salvage option. 26 However, the growing concern over amoxicillin-induced kidney injury is noteworthy. Quadruple therapy involving clarithromycin and amoxicillin continues to be the predominant approach.
At our center, the preferred regimen for bismuth-containing quadruple therapy includes amoxicillin and clarithromycin due to the high prevalence of resistance to metronidazole and ofloxacin in the region.1,18,19 Once diagnosed, the patient is put on the preferred treatment regimen for 14 days. Unless the newly diagnosed patient is intolerant to one of the medications, amoxicillin and clarithromycin will be included in the regimen. When eradication fails, furazolidone or tetracycline is empirically substituted for amoxicillin. Considering the complexity of the patient population characteristics and the limitations of sample size, amoxicillin was not included in the therapy for patients included in this study. All the patients included in this study were newly diagnosed. The eradication rates in resistant and penicillin-allergic patients were not analyzed further. Even so, we observed that the different dosage forms of clarithromycin used in the bismuth-containing quadruple therapy had different efficacy rates. The eradication rate of sustained-release clarithromycin tablets was lower than that of dispersible tablets. Our findings are consistent with the results of another clinical study that indicated a lower eradication rate for sustained-release clarithromycin tablets (1000 mg daily). 27 Unfortunately, none of the risk factors such as smoking, alcohol consumption, age, sex, and PPI type were analyzed in that study, and whether the frequency of clarithromycin administration can affect the eradication rate remains elusive. A randomized clinical trial demonstrated that esomeprazole (20 mg) combined with amoxicillin (750 mg four times daily) was superior to triple therapy with bismuth, particularly against antibiotic-resistant strains. This efficacy can be attributed to the time-dependent antibiotic features of amoxicillin, wherein its bactericidal effect relies on the duration for which the plasma concentration exceeds the minimum inhibitory concentration (MIC). Compared with the routine scheme of twice daily dosing, the dosing scheme of four times daily resulted in sustaining the amoxicillin MIC for an extended percentage of time. Similarly, as a concentration-dependent antibiotic, the bactericidal effect depends on the proportion of plasma concentration that exceeds the MIC. Compared with the 500 mg twice daily dosage, the 1000 mg once daily dosage resulted in a higher concentration, potentially enhancing the efficacy against H. pylori, particularly against resistant strains. However, antibiotic resistance was not analyzed in this study. 27
Several recent studies have indicated that an amoxicillin and clarithromycin mucoadhesive delivery system can sustain systemic exposure to antibiotics and enhance their efficacy. This is attributed to the topical effect on the gastric mucosa that is achieved through direct contact of the drugs with the bacteria.28,29 As a concentration-dependent antibiotic, the higher the concentration of clarithromycin, the more conducive it is to eliminating bacteria. The prolonged residence time in the gastric mucosa and intimate contact of the vehicle with the site of action may increase drug absorption, thereby potentially improving treatment efficacy.30,31 Compared with a dispersible tablet, a sustained-release tablet can be absorbed after being released slowly. As the active ingredients are gradually absorbed in the intestine, resulting in lower concentrations, the drug present in the stomach primarily originates from systemic circulation. This may explain why sustained-release tablets are less effective than immediate-release tablets. However, a lower concentration contributes to the emergence of bacterial resistance to all macrocyclic lipid antibiotics. 32 This may lead to an increase in the MIC of clarithromycin against H. pylori infection, which is one of the primary reasons for the eradication failure of H. pylori. After treatment failure, antibiotic resistance may be further elevated.
The rate of resistance to clarithromycin may be relatively high and increases annually in China,9,10,33 while the sensitivity to clarithromycin determines the eradication rate. A previous study has indicated that treatment regimens for H. pylori infection utilizing clarithromycin exhibited eradication rates of 88% and 45% among clarithromycin-sensitive and -resistant cases, respectively. 34 Consequently, bismuth-containing quadruple therapy has become the standard treatment in China. However, the mechanisms underlying drug resistance remain unclear. Ferdaus et al. identified a higher frequency of the A2147G mutation in 23S rRNA among antibiotic-resistant H. pylori strains,35–38 Li et al. 39 found that Hp1181 and Hp1184 mutations may represent the earliest and most persistent responses to clarithromycin resistance. Geng et al. 40 demonstrated that the SpoT locus is involved in H. pylori tolerance to clarithromycin by upregulating the transporters HP0939, HP1017, HP0497, and HP0471. For antibiotic-resistant strains, higher drug concentrations may result in superior bactericidal effects. This may explain why the sustained-release tablets were less effective than the dispersible tablets in this study. However, the included patients were not tested for drug resistance, and drug resistance was not statistically analyzed.
In the present study, we also probed the impact of PPI type on the eradication outcomes of hybrid therapy. The eradication rates of hybrid therapies based on esomeprazole, pantoprazole, and rabeprazole were 91.03%, 91.11%, and 86.33%, respectively, and did not differ significantly amongst themselves. Our data suggested that PPI type did not impact the efficacy of bismuth-containing quadruple therapy. These results are consistent with the conclusion reached by other clinical studies.41,42 Although patient nonadherence to treatment and the occurrence of antibiotic resistance are the most common reasons for H. pylori eradication failure, inadequate acid suppression has also been associated with treatment failure. Vonoprazan (VPZ), a potassium-competitive acid blocker, has the potential to provide potent and sustained acid suppression. 43 Clinical trials have demonstrated the superiority of VPZ-based regimens over PPI-based triple therapy for clarithromycin-resistant strains and in the general study population. 44 A systematic review and meta-analysis suggested that VPZ-based triple therapy outperformed conventional PPI-based triple therapy in eradicating H. pylori. 45 In addition, VPZ efficacy has been validated in the Chinese population 46 and it is expected to become a first-line new therapy for the eradication of H. pylori.
This study has some limitations that may have negatively affected the accuracy of the results. First, this was a small-scale, single-center retrospective study with a relatively small sample size. The retrospective nature of this study may have introduced potential biases, necessitating future verification through large-scale studies. Second, the selection of the form of clarithromycin dosage (sustained-release vs dispersible tablets) given to the patients was based on the preference of the attending physician, and the treatment assignments were not randomized. Future large-scale randomized controlled trials are required to validate these findings. Third, the results suggest that sustained-release clarithromycin tablets may be inappropriate for eradicating H. pylori in regions with high drug resistance. Importantly, patient testing for drug resistance has not yet been conducted, especially among newly diagnosed patients. Lastly, relying solely on 13C-UBT to assess eradication efficacy made it challenging to exclude the possibility of false-positive results, potentially affecting the accuracy of the results.
Conclusion
In conclusion, clarithromycin dispersible tablet therapy demonstrated a higher eradication rate for H. pylori infection (95.24% (200/210) vs 75.26% (73/97), p < 0.05). The form of clarithromycin dosage was an independent risk factor for eradication failure (OR = 0.145, 95% CI: (0.065–0.323); p < 0.05). Clarithromycin sustained-release tablets may be inappropriate for the eradication of H. pylori infection. However, physician preference is a significant source of bias, and future randomized controlled trials are required to confirm the associations demonstrated in our study.
