Abstract
Background:
Several adalimumab preparations are now available for patients with inflammatory bowel disease (IBD). Comparative satisfaction and tolerability are unknown.
Objectives:
This study investigated IBD patient satisfaction with approved adalimumab biosimilars and their originator.
Design:
In this cross-sectional study, we included 941 consecutive adalimumab-treated patients with IBD across 45 centres affiliated with the Groupe d’Etude Therapeutique des Affections Inflammatoires du tube Digestif who completed a satisfaction questionnaire comprising four items each rated by a 10-point scale.
Methods:
The differences in responses were performed using a one-way analysis of variance followed by Tukey’s honest significant difference test.
Results:
The most commonly used drugs at inclusion were Humira® (436/941, 46.3%), Amgevita® (177/941, 18.8%) and Hulio® (105/941, 11.2%). The mean overall satisfaction rate with adalimumab was 8.5 (standard deviation 1.8). Overall satisfaction was significantly higher in patients treated with Humira (8.6 (1.5)), Hulio (8.6 (1.8)) or Amgevita (8.5 (1.4)) (p < 0.05). Satisfaction with the subcutaneous injection form was higher for patients treated with Yuflyma® (9.0 (1.4)), Humira (8.9 (1.3)) and Hulio (8.9 (1.7)) (p < 0.05). A total of 299 patients (31.8%) described injection site reactions. In all, 223 patients (23.7%) reported being previously treated with another adalimumab of which (32/223, 14.3%) discontinued treatment due to side effects.
Conclusion:
In this real-world setting, patients with IBD had a high level of satisfaction with adalimumab treatment, with some differences in terms of overall satisfaction and satisfaction with the injection device.
Introduction
Adalimumab is indicated for the treatment of moderately to severely active Crohn’s disease (CD) and ulcerative colitis (UC).1,2 Humira® is the adalimumab reference product, and biosimilars have been approved with the same indications of the reference product: Amgevita®, Amsparity®, Hukyndra®, Hulio®, Hyrimoz®, Idacio®, Imraldi® and Yuflyma® (Supplemental Table 1).3–11
Many patients in Europe have been switched from Humira to an adalimumab biosimilar. Several studies investigating the switch from an originator biologic to a biosimilar, from a biosimilar to another biosimilar or multiple switches demonstrated comparable safety and efficacy to the reference biologic.12–16 There remain differences in formulation, device specifications and dosage forms across the biosimilars. The different formulations between adalimumab biosimilars are compared and the reference product is significant, as the presence of citrate or differences in vial volumes may be associated with injection-site reactions. 17 This is an important factor in increasing the confidence of healthcare professionals and patients in using such a formulation. 17
Currently, the use of a particular adalimumab is generally based on non-medical considerations (e.g. costs or additional services for patients). 17 To our knowledge, no data are available for comparisons between biosimilars of adalimumab regarding satisfaction and tolerability, which are unknown. Patient satisfaction with treatment is positively associated with treatment adherence. 18 We need to look beyond efficacy and safety by investigating patient satisfaction to be able to make the right choice. This nationwide study from the Groupe d’Etude Therapeutique des Affections Inflammatoires du tube Digestif (GETAID) is the first to investigate IBD patient preferences for adalimumab.
Methods
Study design
We performed a cross-sectional multicentre survey in 45 centres affiliated with the GETAID. The survey was conducted between 26 September and 7 October 2022, and 5 and 16 December 2022. All consecutive outpatients with inflammatory bowel disease (IBD) who were seen in the outpatient clinic or infusion unit were eligible and were asked to participate in the survey. Inclusion criteria were as follows: (1) age > 18 years; (2) diagnosis of CD or UC based on European Crohn’s and Colitis Organisation (ECCO) criteria; and (3) current treatment with the same adalimumab for at least 3 months. This study was conducted in accordance with French ethics and legal principles through reference methodology MR-004, supervised by and registered in the GETAID. The reporting of this study conforms to the Consensus-Based Checklist for Reporting of Survey Studies statement. 19 All authors reviewed and approved the final manuscript.
Survey instrument
The satisfaction questionnaire was developed and validated by the investigators. The paper questionnaire was distributed consecutively to patients by their gastroenterologist, retrieved during the visit and entered into an anonymous electronic database. The questionnaire comprised 16 questions and was completed by the patients. The assistance of a gastroenterologist was possible if needed.
The questionnaire was designed to explore the following items:
- Patient characteristics, including gender, weight and height, smoking status, age at diagnosis of IBD, type of IBD, duration of IBD, history of intestinal resection, disease activity at inclusion and current treatment.
- Treatment characteristics, including dose, duration, subcutaneous injection form, injection kit and support services.
- Patients’ overall satisfaction with adalimumab, satisfaction with the subcutaneous injection form, satisfaction with the injection kit and satisfaction with support services that were measured using a 10-point visual analogue scale.
- Injection site reactions (including itchy skin, pain, swelling, redness) and need for treatment for injection site reactions.
- Previous treatment with other adalimumab preparations.
Study objectives
The primary outcome was the total satisfaction score. Secondary outcomes were satisfaction with the subcutaneous injection form, satisfaction with the injection kit, and satisfaction with support services and safety.
Statistical analysis
Quantitative variables were expressed as median (interquartile range). Qualitative variables were given as numbers (percentages). Patient satisfaction was expressed for each question as means and standard deviations (SD). Continuous variables were compared by Student’s t-test and categorical variables by Fisher’s exact test or Chi-square test, as appropriate.
Univariate comparison of items in the satisfaction questionnaire was assessed by one-way analysis of variance followed by Tukey’s honest significant difference test.
All statistical analyses were performed using R (version 4.3.1, R Core Team, Vienna, Austria). The statistical significance level was defined as p < 0.05.
Results
Patient characteristics
A total of 941 patients were included in this study. Baseline characteristics are shown in Table 1. There were 463 men (49.2%), the median age at inclusion was 40 (21) years and 187 patients (19.9%) were active smokers. There were 797 patients (84.7%) with CD and 141 patients (15.0%) with UC. The median time since diagnosis was 11 (14) years. Intestinal resections for IBD were performed in 315 patients (33.5%). Most patients were in clinical remission at inclusion (756/941, 80.3%).
Patient characteristics.

IQR, interquartile range.
The drugs used at inclusion were Humira (436/941, 46.3%), Amgevita (177/941, 18.8%), Hulio (105/941, 11.2%), Idacio (67/941, 7.1%), Hyrimoz (54/941, 5.7%), Yuflyma (53/941, 5.6%), Imraldi (40/941, 4.3%), Amsparity (8/941, 0.9%) and Hukyndra (1/941, 0.1%).
Treatment characteristics
The median treatment duration was 25 (48) months (Table 2). Most patients (548/941, 58.2%) received 40 mg of adalimumab every 2 weeks. Among this cohort, 379 patients (40.3%) needed dose escalation: 192 patients (20.4%) received 40 mg every week, 132 (14.0%) received 80 mg every 2 weeks and 55 (5.8%) received 80 mg every week. Among patients who received 80 mg every 2 weeks or 80 mg every week, 79.7% (149/187) were treated with Humira.
Treatment characteristics.

IQR, interquartile range.
The majority of patients (83.4%) used pens for subcutaneous injection of adalimumab and injected themselves (85.7%).
Regarding injection kits, 46.1% of patients received a cooling wallet, 45.9% a leaflet, 31.6% a patient booklet and 26.2% an alcohol pad with treatment. One-third of patients (300/941, 31.9%) did not receive an injection kit at induction.
Most patients (654/941, 69.5%) did not use support services such as assistance calls, website access or video demonstrations, and 17.1% of patients reported that no support service was offered.
Patients’ satisfaction
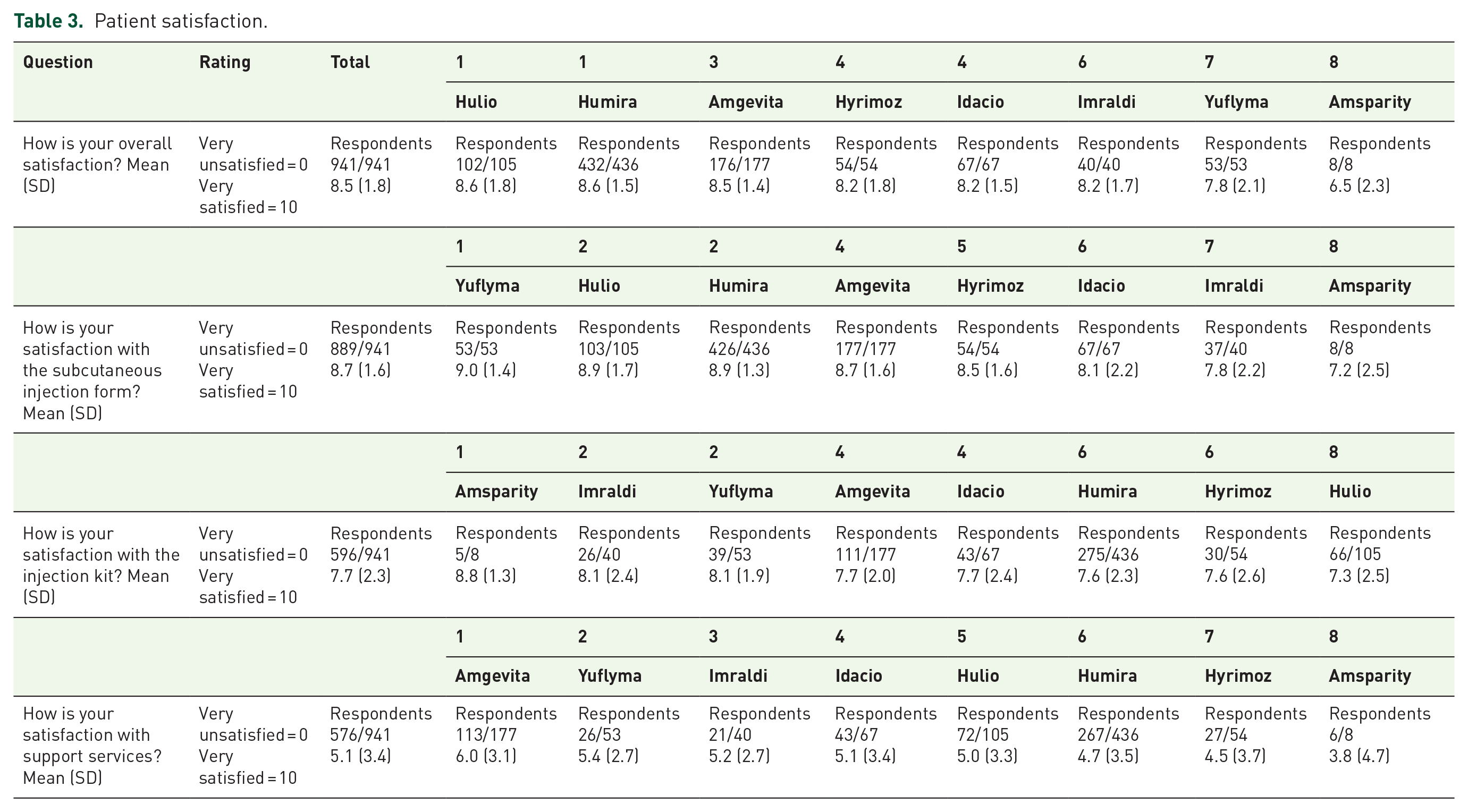
In the global cohort, the mean overall satisfaction rate with adalimumab was 8.5 (1.8) (Table 3). Overall satisfaction rates were 8.6 (1.5) for patients receiving Humira, 8.6 (1.8) for Hulio, 8.5 (1.4) for Amgevita, 8.2 (1.8) for Hyrimoz, 8.2 (1.5) for Idacio, 8.2 (1.7) for Imraldi, 7.8 (2.1) for Yuflyma and 6.5 (2.3) for Amsparity (p < 0.001) (Figure 1(a)). Overall satisfaction was significantly higher in patients treated with Humira than in patients receiving Amsparity (p = 0.007) or Yuflyma (p = 0.027). Overall satisfaction was significantly higher in patients treated with Hulio than Amsparity (p = 0.008). Overall satisfaction was significantly higher in patients treated with Amgevita than Amsparity (p = 0.014).
Patient satisfaction.

Patients’ satisfaction with adalimumab. (a) Overall satisfaction with adalimumab; (b) Satisfaction about subcutaneous injection form; (c) Satisfaction about injection kit; (d) Satisfaction about support services.
In the global cohort, mean satisfaction rates were 8.7 (1.6) with a pen and 8.7 (1.5) with a syringe. Satisfaction with subcutaneous injection form rates were 9.0 (1.4) for patients receiving Yuflyma, 8.9 (1.3) for Humira, 8.9 (1.7) for Hulio, 8.7 (1.6) for Amgevita, 8.5 (1.6) for Hyrimoz, 8.1 (2.2) for Idacio, 7.8 (2.2) for Imraldi and 7.2 (2.5) for Amsparity (p < 0.001) (Figure 1(b)). Satisfaction with the subcutaneous injection form was higher for patients treated with Yuflyma, Humira and Hulio than in patients receiving Idacio (p = 0.048, p = 0.002 and p = 0.044, respectively) or Imraldi (p = 0.019, p = 0.002 and p = 0.02, respectively).
Satisfaction with injection kit rates were 8.8 (1.3) for patients treated with Amsparity, 8.1 (2.4) for Imraldi, 8.1 (1.9) for Yuflyma, 7.7 (2.0) for Amgevita, 7.7 (2.4) for Idacio, 7.6 (2.3) for Humira, 7.6 (2.6) for Hyrimoz and 7.3 (2.5) for Hulio (p = 0.602) (Figure 1(c)).
Satisfaction with support services rates were 6.0 (3.1) for patients treated with Amgevita, 5.4 (2.7) for Yuflyma, 5.2 (2.7) for Imraldi, 5.1 (3.4) for Idacio, 5.0 (3.3) for Hulio, 4.7 (3.5) for Humira, 4.5 (3.7) for Hyrimoz and 3.8 (4.7) for Amsparity (p = 0.111) (Figure 1(d)).
No factors were associated with overall satisfaction with adalimumab.
Safety
A total of 299 patients (31.8%) reported injection site reactions as redness (44.1%), pain (41.5%), swelling (31.8%) and itchy skin (29.8%) (Table 4). Twenty-six (8.7%) patients needed treatment for injection site reactions.
Safety.

Injection site reactions were reported for 50% of patients treated with Amsparity or Imradi, 47.8% for Idacio, 39.6% for Yuflyma, 33.7% for Humira, 27.8% for Hyrimoz, 23.2% for Amgevita and 17.1% for Hulio.
Switching process
In all, 223 patients (23.7%) reported being previously treated with another adalimumab preparation: Humira (171/223, 76.7%), Amgevita (18/223, 8.1%), Imraldi (8/223, 3.6%), Hulio (7/223, 3.1%), Hyrimoz (7/223, 3.1%) and Idacio (5/223, 2.2%) (Table 5).
Switching process.

Among them, 32 patients (14.3%) discontinued treatment because of side effects, most of them (23/28, 82.1%) were treated with biosimilars: Amgevita (8/18, 44.4%), Imraldi (4/8, 50%), Hulio (4/7, 57.1%), Hyrimoz (3/7, 42.8%) and Idacio (4/5, 80%). In all, 14 patients (43.7%) reported persistent injection site reactions after switching adalimumab preparations.
Discussion
Adalimumab biosimilars have previously demonstrated equivalent efficacy, safety and immunogenicity to the reference product. Although some previous studies have investigated the knowledge and perception of adalimumab biosimilars, patient satisfaction remained unclear. For the first time, this observational survey was designed to assess patients’ preferences with adalimumab. Over two 2-week periods, 941 consecutive outpatients from 45 GETAID centres responded to this survey. In this large multicentre study, we observed a high level of satisfaction with adalimumab treatment. We highlighted higher overall satisfaction when patients received Hulio, Humira or Amgevita in comparison to Yuflyma and Amasparity. Satisfaction with the subcutaneous injection form was high, as previously described in other studies on adalimumab biosimilars. 15 We observed a higher satisfaction rate with the subcutaneous injection device when patients received Yuflyma, Hulio or Humira compared to Idacio or Imraldi. Our results suggest that patients with IBD-treated adalimumab did not need additional support like digital platforms, video or assistance calls.
The route of treatment, frequency of administration, related adverse events and the impact on the patient’s quality of life may influence patient satisfaction with treatment. 20 Treatment compliance, persistence and adherence are influenced by patient satisfaction with treatment. 18 Shared decision-making is a process in which both patients and healthcare professionals are involved. This strategy is of considerable interest because it allows patients’ preferences to be integrated into decision-making.20–22 When several treatment options can have different effects on patients’ quality of life, it makes sense to choose the best treatment option together with the patient. Their active participation in the decision-making process could improve the treatment’s effectiveness. 20
Humira was originally formulated with a citrate buffer and administered at 50 mg/ml in a latex-containing device with a 27-gauge needle. Injection site reactions, including pain on injection, were described and led to a modification of the buffer formulation to eliminate certain excipients, notably citrate and citric acid, a reduction in the size of the needle and the elimination of latex, all of which are thought to be responsible for the pain. 23 Larger volumes may cause pain at the injection site. The volume per injection has therefore been revised from 0.8 to 0.4 ml while continuing to deliver 40 mg per injection. 23 By modifying Humira’s formulation and injection device, pain on injection has been resolved. 24 Biosimilars were first introduced in the early 2010s based on the demonstration of pharmacokinetics and clinical biosimilarity with an originator.25,26 In 2022, eight biosimilars were available on the French market. Some of the differences between all of the approved adalimumab biosimilars and reference product stem from their formulations and delivery device parameters. Amgevita, Amsparity, Hukyndra, Hulio, Humira and Yuflyma had all eliminated citrate buffers from their formulations. All adalimumab, except Amgevita, had removed latex in their delivery devices. Hukyndra, Humira and Yuflyma offered lower-volume options. Our results suggest that patients treated with an adalimumab formulation without citrate buffers, without latex and a reduced volume per injection had a higher satisfactory.
Our study has several limitations. First, our findings cannot be extrapolated to other healthcare systems. Second, such a cross-sectional study provides limited changes over time through the management of IBD. A difference was observed in the duration of treatment with Amsparity and Yuflyma (6 months) compared to the treatment duration with the rest. Yuflyma, Amsparity and Hukyndra were the last adalimumab biosimilars available in France, which explains the difference in treatment duration. There may be a memory bias, particularly concerning the reporting of adverse events in patients who have received more than one adalimumab. No information on patient satisfaction with the different adalimumab regimens received was available, but only with the current one. Last, our questionnaire was developed only with physicians and did not involve patients. This first cohort has also some strengths, such as a multicentre national study design and the cohort size. This study is the first to provide data on the patient’s preferences for adalimumab in inflammatory bowel disease.
The healthcare, patient and payer financial landscape in most European countries have been transformed by the availability of adalimumab biosimilars. 23 Indeed, this has led to a substantial reduction in the cost of treatment for IBD, resulting in improved access to biologics.17,27 The main purpose of the current study was to obtain a global perspective on the management of patients receiving adalimumab as IBD treatment from the patient’s point of view. This satisfaction questionnaire provides useful information for improving daily practice with our patients and optimizing all parameters of biosimilars. Recently, Imraldi’s new formulation has been developed to provide an optimized Imraldi injection with a low injection volume and a citrate-free formulation.
Conclusion
In conclusion, this study demonstrated a high level of satisfaction among IBD patients with their adalimumab treatment. Our results suggest a benefit for Hulio, Humira and Amgevita in terms of overall satisfaction, and for Yuflyma, Hulio or Humira in terms of satisfaction with the injection device. Satisfaction measurement is therefore an essential tool for optimal patient care. Patient preferences could possibly have implications, along with other treatment considerations, for future treatment decisions in the management of patients with IBD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241265776 – Supplemental material for Patient preferences for adalimumab in inflammatory bowel disease: a nationwide study from the GETAID
Supplemental material, sj-docx-1-tag-10.1177_17562848241265776 for Patient preferences for adalimumab in inflammatory bowel disease: a nationwide study from the GETAID by Bénédicte Caron, Philippe Seksik, Anthony Buisson, Pauline Wils, Guillaume Savoye, Carmen Stefanescu, David Laharie, Lucas Guillo, Vered Abitbol, Joelle Bonnet, Romain Altwegg, Lucine Vuitton, Driffa Moussata, Arnaud Bourreille, Amélie Biron, Cyrielle Gilletta, Mathurin Fumery, Stephane Nahon, Stephane Nancey, Houda Camara and Laurent Peyrin-Biroulet in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
