Abstract
Background:
Prospective data about the acceptance and outcomes of switching from the adalimumab (ADA) reference product (RP) to multiple ADA biosimilars in inflammatory bowel disease (IBD) patients are lacking.
Objectives:
To evaluate the acceptance rate of switching from the ADA RP to ADA biosimilars, identify factors associated with non-acceptance, assess persistence at 6 and 12 months, determine reasons for discontinuation, analyze changes in disease activity, and explore patient perceptions at 12 months.
Design:
A prospective study was conducted at a French tertiary center between July 2020 and September 2021, including consecutive IBD outpatients treated with ADA RP for over 6 weeks.
Methods:
Patients were offered a switch to five different ADA biosimilars, with the presence of a trained nurse. Data were collected on acceptance rates, persistence at 6 and 12 months, discontinuation reasons, disease activity, and patient perceptions using a 5-item questionnaire at 12 months.
Results:
Of the 97 patients included, 91.8% accepted the switch. The only factor linked to non-acceptance was a poor opinion of generic drugs. Persistence with the initial ADA biosimilar was 68.6% at 6 months and 60.4% at 12 months. The overall survival rate without biosimilar discontinuation was 76.7% at 6 months and 71% at 12 months. Injection site pain (24.7%) was the leading cause of discontinuation, and 22 patients switched back to the ADA originator. Clinical remission was maintained in 90.4% of patients at 12 months. Among respondents, 65.8% reported a positive experience with the switch.
Conclusion:
The study demonstrates high acceptance and favorable outcomes for switching to ADA biosimilars in IBD patients, including sustained efficacy and good treatment persistence. These findings suggest that switching to ADA biosimilars is a viable option for IBD patients, with positive results in terms of both clinical effectiveness and patient adherence.
Plain language summary
This study explored how patients with inflammatory bowel disease (IBD) respond to switching from the original adalimumab (ADA) medication to its biosimilar versions. Conducted at a French hospital between July 2020 and September 2021, it focused on patient acceptance and outcomes after the switch. The study involved IBD patients who had been on the original ADA for over six weeks. They were offered a switch to one of five ADA biosimilars, with support from a trained nurse. We looked at how many patients accepted the switch, why some refused, how many stayed on the biosimilar at 6 and 12 months, reasons for stopping, and how the disease progressed. Patients also filled out a questionnaire after 12 months to share their experiences. Out of 97 patients, 91.8% accepted the switch. The main reason for refusing was a negative opinion about generic drugs. At 6 months, 68.6% of patients were still using the biosimilar, and 60.4% continued at 12 months. In total, 76.7% of patients stayed on the biosimilar at 6 months, and 71% remained at 12 months. The most common reason for discontinuing was injection site pain, which led 22 patients to return to the original ADA. However, 90.4% of patients remained in clinical remission (their disease stayed under control) at 12 months. Additionally, 65.8% of patients reported a positive experience with the switch. In conclusion, most IBD patients are open to switching to ADA biosimilars and achieve good outcomes.
Introduction
Inflammatory bowel diseases (IBDs), encompassing ulcerative colitis (UC) and Crohn’s disease (CD), are chronic relapsing inflammatory conditions of the gastrointestinal tract, affecting approximately 7 million patients worldwide. 1 These diseases are associated with a significant psychosocial burden and substantial healthcare costs.2,3 IBDs typically require lifelong medical management and treatment. Over the past two decades, anti-TNF therapies, including adalimumab (ADA), have significantly advanced the treatment of IBD patients. 4 In industrialized countries, biological therapies have been the major contributor to direct healthcare costs within the first 5 years after CD and UC diagnosis, accounting for an estimated 75% and 50% of total costs, respectively.3,5
To reduce healthcare costs and improve patient’ access to effective treatment, biosimilars have been developed. 6 The licensing of biosimilars by regulatory bodies such as the US Food and Drug Administration and the European Medicines Agency is based on rigorous standards, ensuring no significant differences in efficacy, safety, or immunogenicity between ADA biosimilars and the ADA reference product (RP), as demonstrated in clinical studies.7–10 Through a process of extrapolation, biosimilars may subsequently be licensed for all indications of the RP. The first biosimilar used in IBD was infliximab.11–13 As of October 2018, and at the time of our study, five biosimilars of ADA had been introduced to the European market: Amgevita®, Hulio®, Hyrimoz®, Idacio®, and Imraldi®. While these ADA biosimilars have shown comparable efficacy and safety to the RP, they may differ in device types, excipients, and volume.
Switching from the RP to a biosimilar in patients with IBD is recommended by the European Crohn’s and Colitis Organisation and local French healthcare authorities. Despite growing clinical evidence supporting that biosimilars are safe and effective in IBD,14,15 patient concerns about biosimilars remain poorly addressed and potentially limit the acceptance of biosimilars in clinical practice and treatment adherence. The process of switching from the RP to a biosimilar or between different biosimilars presents additional challenges. Acceptance of switching by both patients and physicians is crucial for the successful integration of ADA biosimilars into clinical practice.
To date, no study has specifically evaluated the acceptance of switching from the ADA RP to different ADA biosimilars in IBD patients. Therefore, this study aimed to evaluate the acceptance rate and factors associated with the non-acceptance of switching from ADA RP to one of five ADA biosimilars. In addition, we prospectively assessed 1-year biosimilar persistence, rates of switching back to ADA RP, multiple switches during follow-up, clinical outcomes, and patient perceptions of the switch.
Materials and methods
Patient selection and study design
This prospective observational study was conducted between July 2020 and September 2021 at Lille University Hospital (France). All consecutive outpatients meeting the following criteria were included: (1) Adult patients with IBD (CD or UC diagnosed according to standard clinical, morphological, endoscopic, and histological criteria), and (2) currently treated with ADA RP (Humira®) for more than 6 weeks (having completed full induction). Patients were not eligible if they had severe disease (defined by a Harvey Bradshaw Index (HBI) >16 for CD or a partial Mayo score >7 for UC) or if they were pregnant or breastfeeding.
All patients included in the study received standardized, one-by-one verbal information about biosimilars, provided by their regular treating gastroenterologist and a trained IBD nurse specifically assigned to guide the transition (Supplemental Table 1). The information session focused on scientific evidence related to biosimilars, covering their benefits, efficacy, evaluation methods, and safety profile, including tolerance and potential adverse effects, along with details about the study itself. Patients were then offered the option to switch from ADA RP to a biosimilar. Consent for the switch was obtained by their treating gastroenterologist. Patients who accepted the non-medical switch could choose from among the five ADA biosimilars available at the time of the study (Amgevita, Hulio, Hyrimoz, Idacio, and Imraldi, description of formulations are summarized in Supplemental Table 2). These patients were then followed prospectively for 12 months.
At inclusion, each patient was invited to complete a questionnaire during a face-to-face interview with the IBD nurse. The questionnaire was divided into three parts: (1) patient characteristics, including individual opinions related to their gastroenterologist, generics, and the pharmaceutical industry as well as socio-professional data, vaccination adherence, and follow-up adherence with a dermatologist or gynecologist (for women), (2) concerns about biosimilars, and (3) reasons for accepting or declining the switch (Supplemental Materials). At the inclusion visit, patients who accepted the switch were trained on the new injection device by the IBD nurse.
At 12 months, we assessed the patients’ perception of the switch using a 5-item questionnaire (good experience, positive effect, no difference, feeling of obligation, negative effect; definitions available in the Supplemental Materials). This study has been reported in accordance with the guidelines of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 16 (Supplemental Materials).
Data collection
The inclusion date corresponded to the appointment with the gastroenterologist proposing the switch. Data from each patient were anonymized.
The following data were collected for each patient from the electronic patient’s medical record at inclusion: age, age at IBD diagnosis, gender, disease duration, body mass index (BMI), smoking status, type of IBD, disease location, and phenotype were according to the Montreal classification, extra-intestinal manifestations, previous intestinal resection, previous and current IBD therapies, date of ADA RP introduction, and regimen at the inclusion.
At 6 and 12 months, information on concomitant medication use (steroids or immunosuppressants) and clinical disease activity (assessed by the HBI or the partial Mayo score) were collected. In addition, C-reactive protein (CRP) levels, fecal calprotectin levels, serum drug levels, and antidrug antibodies (if available) were collected. This was a non-interventional study, and blood and stool samples were collected as part of routine clinical practice.
Disease-related adverse events, such as hospitalization, surgery, and peri-anal surgery for CD patients, were reported. Medication-related adverse events during the 1-year follow-up were also recorded. Discontinuation and changes in ADA biosimilars (switching back to ADA RP, switching to another ADA biosimilar, or transitioning to another treatment) were at the physician’s discretion, in agreement with the patient. Reasons for discontinuation were documented.
Study outcomes and definitions
The main objective of the study was to assess the acceptance of a switch from the ADA originator to ADA biosimilars in IBD patients and identify factors associated with non-acceptance. Secondary objectives included evaluating, over a 1-year follow-up period: (1) the persistence of ADA biosimilars at 6 and 12 months, (2) the rates of switching back to the ADA originator and multiple switches, (3) the reasons for discontinuation of ADA biosimilars, (4) changes in disease activity, and (5) patients’ perceptions of the switch from ADA RP to a biosimilar.
Clinical remission was defined as an HBI score of ⩽4 for CD or a Partial Mayo Score of ⩽1 for UC. Biochemical remission was defined as a CRP level of ⩽5 mg/L (when available).
Statistical analysis
We did not perform a sample size calculation prior to starting the study, as most IBD patients receiving ADA in the Gastroenterology Department at Lille University Hospital were on the ADA RP, ensuring a sufficiently predetermined sample size.
Descriptive statistics were used to analyze baseline characteristics. Continuous data were described as medians with interquartile ranges (IQR) or means with standard deviations and discrete data were determined as percentages. The Kaplan–Meier method was used to assess the survival rate without ADA biosimilar discontinuation at 12 months (for the biosimilar initially introduced and overall) with censoring applied for loss to follow-up, and treatment discontinuations. The Cox proportional-hazards model was used to investigate the association between the survival rate without ADA biosimilar discontinuation and predictor variables. p-Value < 0.05 was considered statistically significant. Calculations were performed using GraphPad Prism 9.1.0 (GraphPad Software; Dotmatics, Boston, USA).
Results
Characteristics of the study population
A total of 97 patients with IBD (CD, n = 84 (86.6%); UC, n = 13 (13.4%)) treated with ADA RP were consecutively included from July 2020 to September 2021. The median age was 34.8 years (27.4–44.4), 52.6% were male, and the median BMI was 24.2 kg/m² (20.8–27.7). At inclusion, the median disease duration was 12.6 years (8.1–19.6), with a median follow-up with their treating gastroenterologist of 5.7 years (2.5–7.9). Among CD patients, 37 (44%) patients had ileo-colonic involvement, 27 (32.1%) had perianal involvement, and two-thirds had a complicated disease (fistulizing or stricturing). Half of the CD patients had undergone previous intestinal surgery. Two-thirds of UC patients had pancolitis (8/13; 61.5%). Twenty-five (25.8%) patients had extra-intestinal manifestations, and 71 (73.2%) patients were active workers. Fourteen (14.4%) patients had a poor opinion of generics, and 21 (21.9%) patients had a poor opinion of the pharmaceutical industry. Flu vaccination adherence and dermatological follow-up adherence were reported for more than half of the patients. Characteristics of included patients are summarized in Table 1.
Populations’ characteristics.
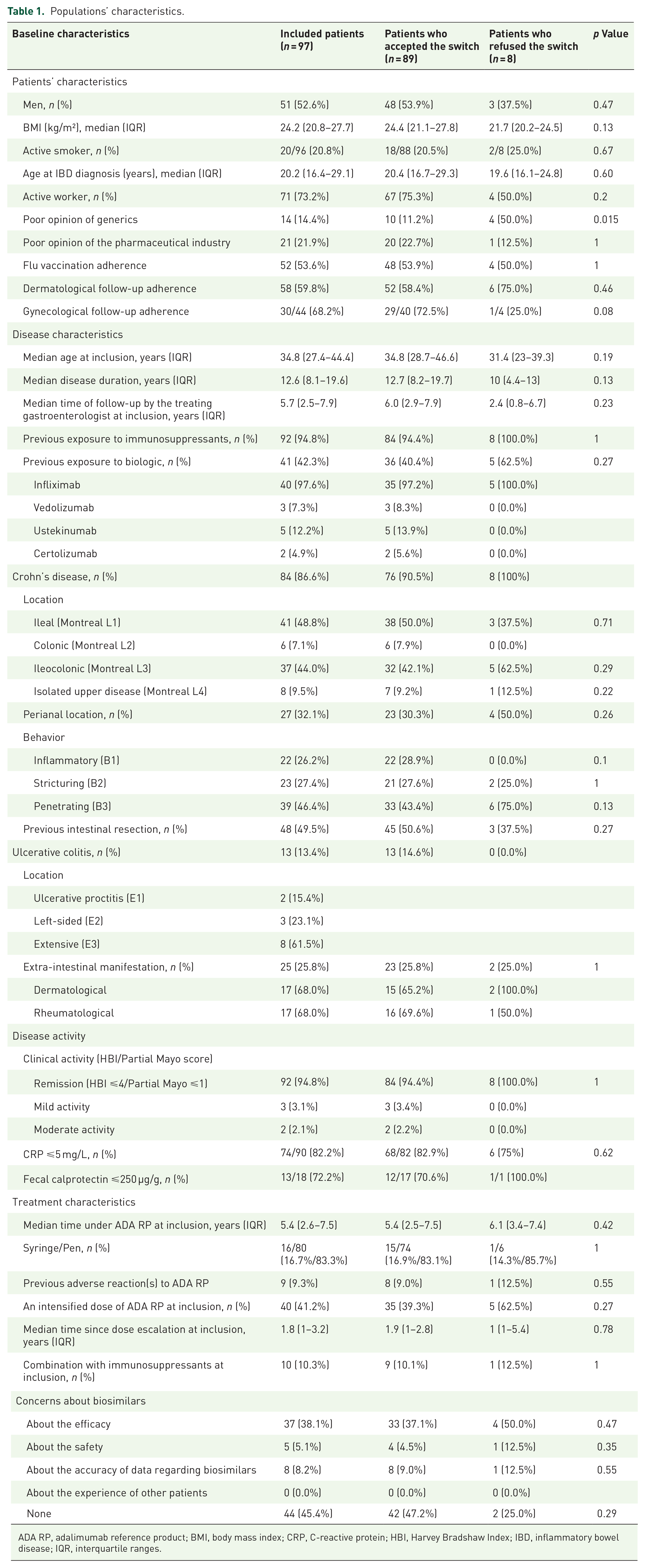
ADA RP, adalimumab reference product; BMI, body mass index; CRP, C-reactive protein; HBI, Harvey Bradshaw Index; IBD, inflammatory bowel disease; IQR, interquartile ranges.
Of the 97 patients, ADA RP was the first-line biologic therapy for 56 (57.7%) patients, and 83.3% of patients used a pen device. Forty patients were previously exposed to infliximab (including three who received an infliximab biosimilar), three to vedolizumab, five to ustekinumab, and two to certolizumab. The median duration of ADA RP was 5.4 (2.6–7.5) years. Forty patients (41.2%) received an intensified dose of ADA RP at the time of the switch (25 patients receiving 40 mg every week, 6 patients 80 mg every 2 weeks, 2 patients 80 mg every week, and 2 patients 40 mg every 10 days). At inclusion, 92 patients (94.8%) were in clinical remission. In addition, 74 out of 90 patients (82.2%) had a CRP level ⩽5 mg/L, and 13/18 (72.2%) patients had fecal calprotectin levels ⩽250 µg/g. ADA levels were available for 66 (68%) patients with a median of 7.75 µg/mL.
Forty-four (45%) patients did not have concerns about biosimilars. Main concerns were related to efficacy (38.1%), the accuracy of data regarding biosimilars (8.2%), and safety (5.5%).
Acceptance rate of the switch
Of the 97 included patients, 89 (CD, n = 76; UC, n = 13; 91.8%) accepted the switch. Characteristics of the patients who accepted and refused the switch are presented in Table 1. A poor opinion of generic drugs was significantly associated with non-acceptance of the switch (odds ratio = 0.02 (0.001–0.5), p = 0.015). Median disease duration, median time of follow-up by the treating gastroenterologist, an intensified dose of ADA RP at inclusion, and median time on ADA RP were not associated with non-acceptance of the switch (Table 2).
Factors associated with switch refusal.
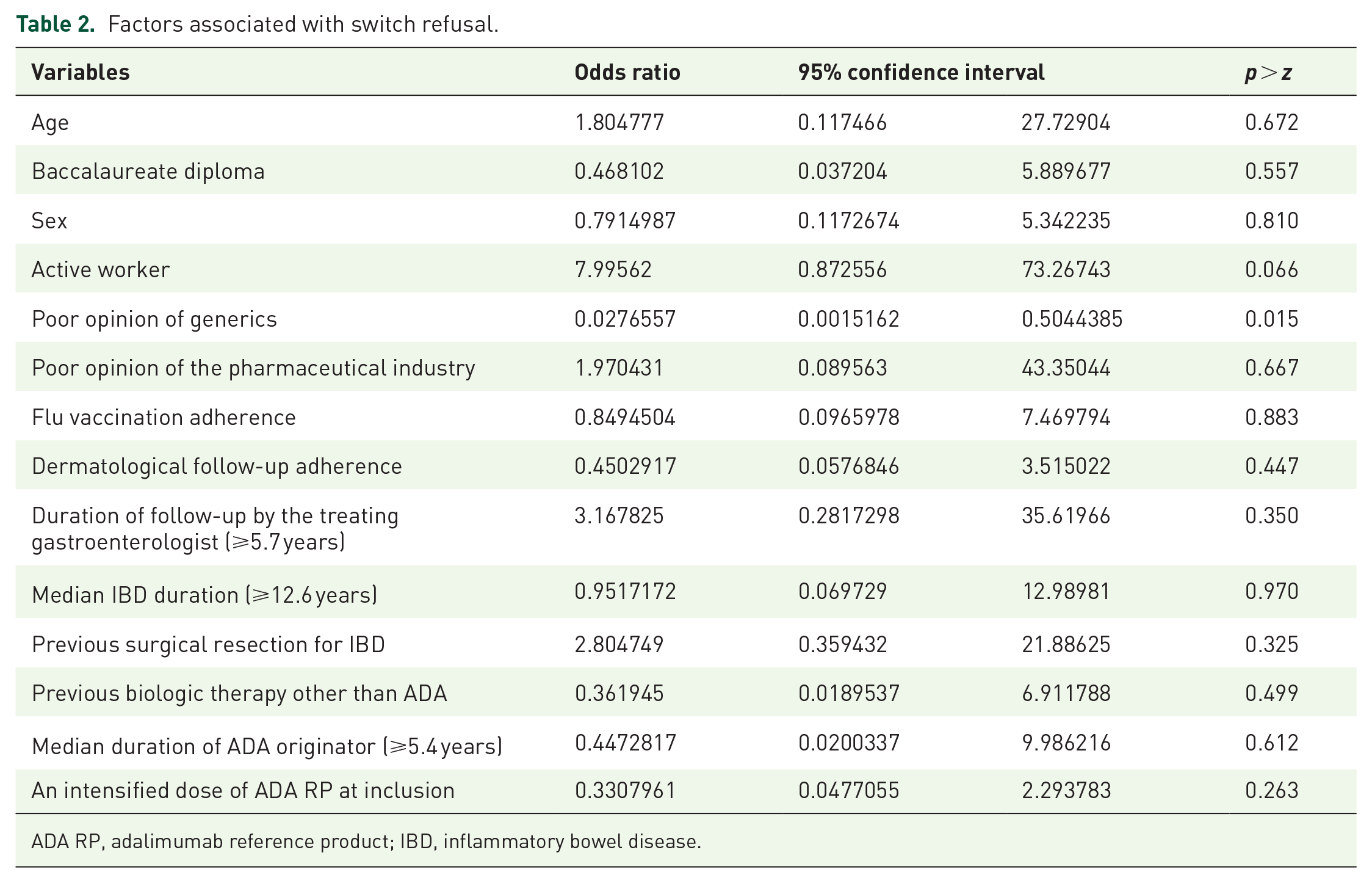
ADA RP, adalimumab reference product; IBD, inflammatory bowel disease.
ADA biosimilar persistence and distribution
Among the 89 patients who accepted the switch, 37 discontinued the ADA biosimilar during the follow-up period: 22 patients switched back to ADA RP (21 within the first 6 months, and 1 additional patient between 6 and 12 months). Three patients discontinued ADA therapy during the follow-up. In addition, eight patients switched to another biosimilar at 6 months, with four more switching by 12 months (including one patient who switched between different biosimilars twice). Two patients (2.2%) were lost to follow-up between 6 and 12 months. Survival analyses of the 86 patients who received more than one ADA biosimilar injection showed that 68.6% of patients continued with the initially introduced ADA biosimilar at 6 months, and 60.4% maintained it at 12 months (Figure 1(a)). Overall, the survival rate without ADA biosimilar discontinuation (including patients who maintained the initially introduced biosimilar and those who switched to another) was 76.7% at 6 months and 71% at 12 months (Figure 1(b)). There were no significant clinical or socio-demographic factors associated with the persistence of the ADA biosimilar whether considering the first-introduced ADA biosimilar or the overall survival (Supplemental Tables 3 and 4).

Survival rates without ADA biosimilar discontinuation whether considering the first-introduced ADA biosimilar (a) or the overall survival (b).
The main reasons for discontinuing ADA biosimilars were injection site pain in 24.7% of patients, loss of efficacy in 6.7% (n = 6 patients, who switched back to ADA RP and regained efficacy), and difficulties with the new injection device in 4.5%. In addition, four patients (13.3%) stopped the biosimilar due to abdominal pain despite the absence of objective disease activity, which their physician considered a nocebo effect. Table 3 provides a summary of the patient’s reasons for biosimilar discontinuation during follow-up (multiple answers were allowed).
Reasons for patients’ biosimilar discontinuation (multiple answers were allowed).

Figure 2 illustrates the distribution of ADA biosimilars chosen by the patients at baseline and throughout the follow-up period. The primary reason for selecting a particular biosimilar over others was its similarity to the ADA RP (Supplemental Figure 1). While the proportion of patients treated with Hulio remained stable at around 30%, the proportion of patients using other biosimilars decreased (from 33.7% to 22.5% for Amgevita, from 20.2% to 5.6% for Idacio, from 8% to 4.5% for Imraldi, and from 10.1% to 5.6% for Hyrimoz).
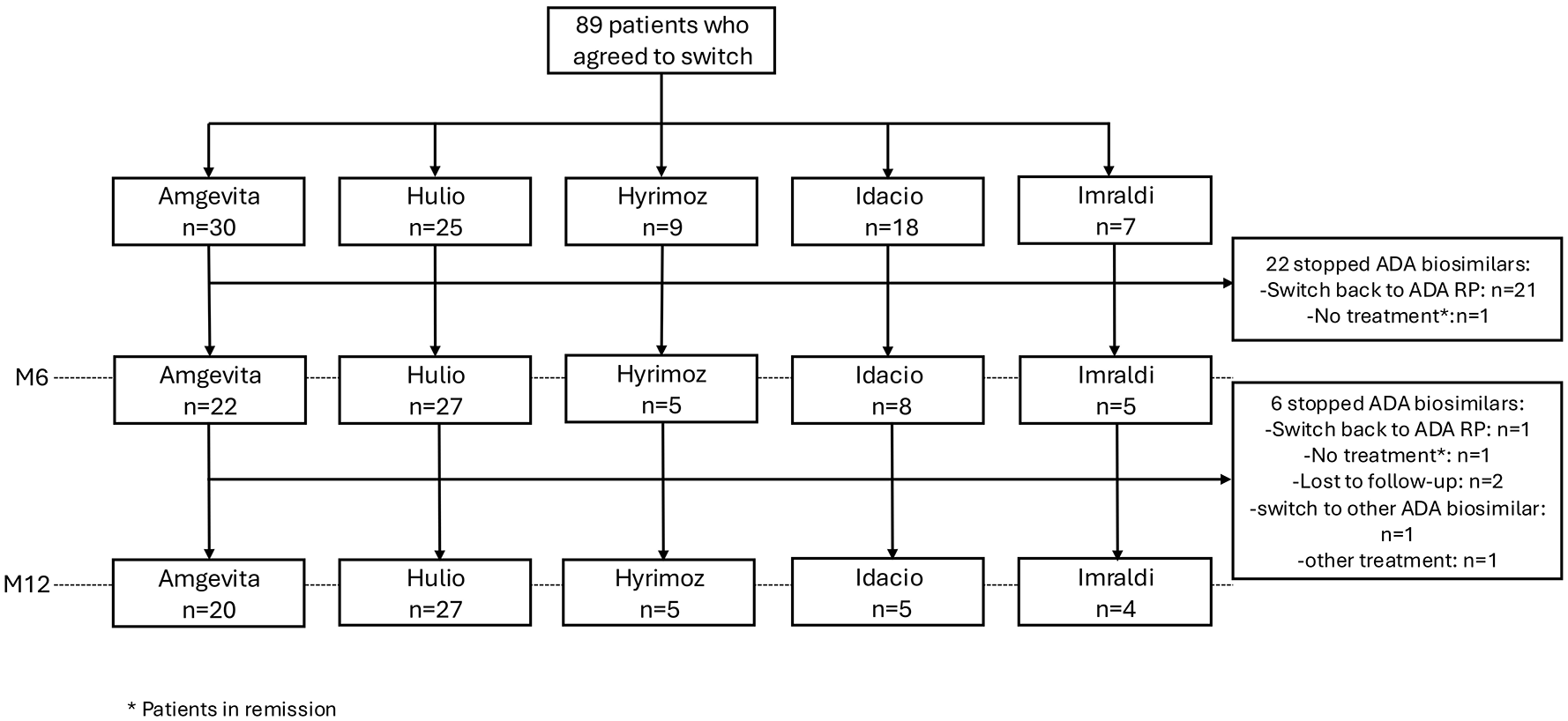
Flow chart depicting the distribution of ADA biosimilars chosen by patients at baseline, 6 (M6), and 12 months (M12).
ADA biosimilar efficacy
Overall, 84 out of 89 patients (94.4%) were in clinical remission at baseline, with 79 (88.8%) maintaining remission at 6 months, and 76 (85.4%) at 12 months. Among the 52 patients who remained on their initial ADA biosimilar, 47 (90.4%) were in clinical remission at 12 months (Supplemental Figure 2). Sixteen patients (18%) experienced a clinical relapse during the follow-up. Thirteen patients required dose intensification of ADA, and five needed steroids. Two patients (3.1%) were hospitalized without discontinuing ADA biosimilar therapy (one for a duodenal stricture and another for a symptomatic ileal stricture). No patient underwent surgery. ADA de-escalation was observed in three patients.
Biochemical remission (defined as a CRP level ⩽5 mg/L) was observed in 68 out of 82 patients (82.9%) at baseline, 48 out of 58 patients (82.7%) at 6 months, and 42 out of 49 patients (85.7%) at 1 year. Fecal calprotectin levels were available for 17 (19%) patients at inclusion and 11 (17%) patients at 12 months, with levels ⩽250 µg/g in 12 patients (70.6%) and 9 patients (81.8%), respectively. Among the 38 patients analyzed, no cases of immunization were reported.
Among the eight patients who declined the switch, one was lost to follow-up. The remaining seven patients, who were in clinical remission with the ADA RP (Humira) at baseline, continued treatment at 12 months of follow-up. Of these, six maintained the same dosage, while one reduced their dosage. All seven evaluated patients remained in clinical and biochemical remission (CRP ⩽5 mg/L). Fecal calprotectin levels were not measured for any of these patients during the follow-up period.
Patient’s perception of the switch
Thirty-eight patients (60%) completed the 5-item questionnaire at the end of the study. Of these, 65.8% reported a positive experience and 2.6% reported a positive impact, while 15.8% indicated a negative impact (Figure 3). In addition, 10% of patients had no opinion.

Patient’s perceptions of the switch of ADA (n = 38).
Discussion
This observational study evaluated, for the first time, the switch from the ADA RP to five different ADA biosimilars among patients with IBD over a 12-month period. The study aimed to assess the acceptance of the switch, persistence with biosimilars, and patient perceptions regarding the switch.
Out of 97 eligible patients with IBD, a high acceptance rate of 91.8% was observed, indicating a positive reception toward the switch from ADA RP to biosimilars. This acceptance rate is consistent with findings from other studies involving patients with IBD 17 or inflammatory rheumatic diseases.18,19 Unlike other studies where patients were asked to transition their treatment from the originator to a single biosimilar, in our study, eligible patients were given the choice among five different ADA biosimilars available at the time of the study. These results suggest that, when patients with inflammatory conditions are properly informed and provided with tailored guidance during the transition (here with standard verbal information about biosimilars from the regular treating gastroenterologist and a trained nurse specifically assigned to guide the transition), high acceptance levels for switching to ADA biosimilars can be achieved.
Our study found a significant association between a poor opinion of generics and the non-acceptance of the switch highlighting the impact of patient attitudes toward drug types on their willingness to switch. Specifically, 50% of patients who refused the switch (4 out of 8) reported a poor opinion of generics, compared to 11% of patients who accepted the switch. This factor has also been identified in other studies as influencing switch acceptance in inflammatory diseases.18,20 Interestingly, neither an intensified dose of ADA RP nor the median ADA duration at the time of the switch was associated with non-acceptance. Therefore, these factors should not influence the physician’s decision to propose a switch from ADA RP to a biosimilar.
Among those who accepted the switch, persistence with the initial ADA biosimilar at 6 months was 68.6%, decreasing to 60.4% at 12 months. These persistence rates are slightly lower than those reported in other studies involving IBD patients transitioning to a single biosimilar,17,21–26 possibly because in our study, patients had the option to continue with the biosimilar or switch back to the original throughout the follow-up period. In our study, the majority of patients who discontinued the ADA biosimilar switched back to ADA RP (22 out of 37 patients). In addition, a small proportion of patients (12 in total) accepted multiple switches among biosimilars during the study period, contributing to a survival rate without ADA biosimilar discontinuation of 76.7% at 6 months and 71% at 12 months. In some countries, prescribers face varying realities regarding the interchangeability of biosimilars which may be required by local regulations. While interchangeability can enhance competition, leading to lower prices and increased accessibility for patients, it also presents challenges in maintaining consistent patient care. Notably, studies have shown the benefits of multiple switching between biosimilars, demonstrating that, when managed correctly, it can be safe and effective without compromising treatment outcomes or adherence.22,27
Consistent with previous studies,17,25 the primary reason for discontinuing ADA biosimilars in our cohort was injection site pain, which affected 24.7% of patients. Several studies have attributed the injection pain to excipients, particularly the citrate buffer.28,29 At the time of our study, three of the five ADA biosimilars contained citrate. In recent years, the tolerance of ADA biosimilars has significantly improved, especially in reducing injection site pain, due to advancements in formulation techniques. These include the development of citrate-free formulations, higher concentration formulations, or thinner needles, which contribute to a more comfortable patient experience.30,31 A recent study on nine ADA formulations (such as ADA RP and eight ADA biosimilars) revealed that IBD patients treated with a citrate-free, latex-free, and reduced-volume ADA formulation reported higher satisfaction with ADA treatment. 32 We observed a low rate of the nocebo effect—defined as patients’ negative expectations toward the therapy change—among our patients. 33 This is likely due to the dedicated time spent on patient education, which aligns with findings from other biosimilar studies. These observations highlight the importance of ongoing patient support and education to effectively address concerns and improve treatment adherence.
The switch did not negatively impact disease control, as evidenced by the high rates of clinical remission after the switch (94.9% at 6 months and 90.4% at 12 months) and biochemical remission. These results are in line with those reported in other studies.22–24,34 The incidence of clinical relapses (18%) and the requirement for dose intensification or steroids were relatively low, further supporting the ADA biosimilars’ effectiveness.
The patient perception data revealed that two-thirds of patients reported a good experience with biosimilars, while 15.8% experienced a negative impact. This overall positive perception reflects well on the acceptance of biosimilars. Continuous patient education throughout follow-up, along with addressing individual concerns by physicians, pharmacists, or specialized IBD nurses, is crucial for improving overall satisfaction with biosimilars and maximizing their benefits.
Our study has certain limitations, including a small sample size and a single-center design. To minimize confounding factors that could impact treatment adherence and continuation, we excluded patients with severe disease activity; however, this may have introduced a selection bias. The consideration of only a subset of ADA biosimilars currently on the market may limit the ability to generalize our findings and the relatively short follow-up period to assess long-term outcomes. In addition, the sample size for the patient perception questionnaire was relatively small (38 patients), which could affect the robustness of the perception data. Moreover, no endoscopic evaluations were performed at the time of the switch or during the follow-up, which could have provided objective markers of disease activity. Another limitation is the absence of a comparator group of patients who underwent the switch without the support of a trained nurse making it difficult to definitively assess the nurse’s role in facilitating the transition. Despite these limitations, our findings support the feasibility and acceptance of switching to biosimilars by physicians managing IBD patients in clinical practice. Future research should consider larger, multicenter trials with extended follow-up to validate these findings and explore the long-term impact of switching to biosimilars in IBD patients.
Conclusion
Our real-word study demonstrates high acceptance and favorable outcomes for switching to ADA biosimilars in IBD patients. After 12 months, 71% of patients maintained an ADA biosimilar, and 60% continued the initially introduced ADA biosimilar. While our findings suggest that addressing patient concerns and enhancing education about biosimilars—potentially with the support of a trained nurse to guide the transition—may improve the overall experience and persistence with these therapies, further studies with comparator groups are needed to better assess the nurse’s role in facilitating these transitions. This is particularly relevant as biosimilars for other biologics, such as ustekinumab, are now approved for IBD and entering the European market.
Supplemental Material
sj-docx-3-tag-10.1177_17562848251332025 – Supplemental material for Switch acceptance and persistence of adalimumab biosimilars in IBD patients: a prospective observational study
Supplemental material, sj-docx-3-tag-10.1177_17562848251332025 for Switch acceptance and persistence of adalimumab biosimilars in IBD patients: a prospective observational study by Gabrielle D’Abbundo, Maria Nachury, Alice Wartski, Aurélie Blondeaux, Sofia Hambli, Romain Gérard, Pierre Desreumaux, Nicolas Duveau and Pauline Wils in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-1-tag-10.1177_17562848251332025 – Supplemental material for Switch acceptance and persistence of adalimumab biosimilars in IBD patients: a prospective observational study
Supplemental material, sj-tif-1-tag-10.1177_17562848251332025 for Switch acceptance and persistence of adalimumab biosimilars in IBD patients: a prospective observational study by Gabrielle D’Abbundo, Maria Nachury, Alice Wartski, Aurélie Blondeaux, Sofia Hambli, Romain Gérard, Pierre Desreumaux, Nicolas Duveau and Pauline Wils in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-2-tag-10.1177_17562848251332025 – Supplemental material for Switch acceptance and persistence of adalimumab biosimilars in IBD patients: a prospective observational study
Supplemental material, sj-tif-2-tag-10.1177_17562848251332025 for Switch acceptance and persistence of adalimumab biosimilars in IBD patients: a prospective observational study by Gabrielle D’Abbundo, Maria Nachury, Alice Wartski, Aurélie Blondeaux, Sofia Hambli, Romain Gérard, Pierre Desreumaux, Nicolas Duveau and Pauline Wils in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
