Abstract
Background:
Anti-tumor necrosis factor (TNF) monoclonal antibodies, especially infliximab (IFX) and adalimumab (ADA), are considered the first-line treatment for active Crohn’s disease (CD). However, the predictive role of therapeutic drug monitoring (TDM) of serum anti-TNF in monitoring the treatment of inflammatory bowel disease (IBD) remains controversial.
Objectives:
To explore the correlation between serum anti-TNF levels and early endoscopic response in active CD using a TDM-based nomogram.
Design:
Cross-sectional study.
Methods:
The simplified endoscopic activity score for CD (SES-CD), Crohn’s disease activity index (CDAI), laboratory parameters, and the serum trough levels of IFX and ADA were assessed.
Results:
The trough levels of IFX or ADA were significantly higher in patients with endoscopic response compared to non-responders in the development cohort (p < 0.001). The IFX and ADA levels showed a weak but significantly negative correlation with SES-CD (p < 0.001), CDAI (p < 0.001), and C-reactive protein (CRP) (p < 0.001) at week 14 post-IFX therapy in the development cohort. Furthermore, the receiver operating characteristic curve revealed that an optimal level of IFX (4.80 μg/mL) and ADA (8.80 μg/mL) exhibited the best performance in predicting endoscopic response. Concomitantly, we developed a novel nomogram prediction model based on the results of multivariate logistic regression analysis, which consisted of CRP, albumin (Alb), and anti-TNF trough levels at week 14. The nomogram showed significant discrimination and calibration for both IFX and ADA in the development cohort and performed well in the external validation cohort.
Conclusion:
This study demonstrates a robust association between serum concentrations of IFX, ADA, Alb, and CRP and primary endoscopic response in active CD patients. Importantly, the TDM- and laboratory marker-based nomogram may be used to evaluate the primary endoscopic response to anti-TNF therapy, especially for optimizing treatment strategies and switching therapy in CD patients.
Plain language summary
The present study established a therapeutic drug monitoring-based nomogram, which exhibits an exceptional predictive value, remarkable accuracy, and discrimination. This algorithmic nomogram holds the potential to enhance clinicians’ comprehension of the underlying mechanisms contributing to individual patients’ failure in achieving expected efficacy. Such approach is crucial for optimizing therapy options and facilitating biologic switching in refractory Crohn’s disease.
Introduction
The utilization of monoclonal antibody (mAb) therapy targeting tumor necrosis factor (anti-TNF), such as infliximab (IFX) and adalimumab (ADA), has become a crucial requirement for treating active Crohn’s disease (CD). Controlled clinical trials have demonstrated the efficacy and safety of anti-TNF mAbs in the treatment of moderate to severe CD for the induction and maintenance of clinical and endoscopic remission, reducing hospitalizations, relapse rates, and surgeries and improving the quality of life.1–6 However, approximately one-third of the patients fail to respond to initial anti-TNF therapies by the end of an induction period. Furthermore, 20–40% of patients show an initial response but lose response over time.7–10 Thus, strategies to optimize anti-TNF therapy are necessary.
Several lines of evidence have demonstrated that therapeutic drug monitoring (TDM) is emerging as a standardized treatment approach for optimizing anti-TNF treatment in inflammatory bowel disease (IBD). Some retrospective cohort studies have proposed TDM as a strategy to maximize the safety, efficacy, and cost-effectiveness of anti-TNF therapy.11–13 It has been well established to effectively guide treatment de-escalation for etanercept 14 and ADA15,16 in patients with rheumatoid arthritis. However, the NORwegian DRUg Monitoring study trial has shown that the routine use of TDM during induction therapy with IFX did not improve the rates of disease remission. 17 A meta-analysis has also demonstrated that the proactive TDM regimen showed no significant difference in the maintenance of clinical remission compared with conventional management [RR, 0.96; 95% confidence interval (CI), 0.81–1.13]. 18 However, the role of TDM in predicting the efficacy of anti-TNF therapy in IBD patients remains controversial, particularly, the predictive values of proactive TDM and reactive TDM. Two induction trials with ADA for CD and UC (SERENE-CD and SERENE-UC) have indicated that proactive TDM strategy is not superior to the reactive TDM for prediction of endoscopic and clinical response.19,20 Nevertheless, a pediatric study has indicated that proactive monitoring and ADA dose intensification (serum concentrations >5 μg/mL) could contribute to significantly higher rates of corticosteroid-free clinical remission than reactive monitoring in cases of loss of response (LOR). 21
Despite being controversial, emerging evidence has indicated that high serum anti-TNF drug concentrations appear to be associated with better clinical outcomes in IBD.22–25 Furthermore, TDM can help to identify the underlying mechanisms of poor clinical outcomes, such as insufficient drug exposure or mechanistic failure due to non-adherence or pharmacokinetic issues. 26 It is worth mentioning that the majority of TDM-related studies have mainly focused on the evaluation of maintenance therapy instead of the induction phase. More importantly, there are only limited data mentioning TDM during the induction phase in CD and even less data regarding the therapeutic trough threshold of anti-TNF as a target for an induction endpoint. 27
To date, there is limited evidence to evaluate the predictive value of the TDM strategy for inducing the remission of active CD. Hence, in this retrospective clinical study, we aimed to explore the correlation between pharmacokinetic parameters and primary clinical efficacy and to predict early endoscopic response of patients with active CD using a novel TDM-based nomogram.
Materials and methods
Patients
Patients diagnosed with active CD were treated with anti-TNF mAbs between March 2017 and February 2022 at the Center for IBD Research and Department of Gastroenterology, Shanghai Tenth People’s Hospital of Tongji University as a development cohort (n = 279). From May 2022 to August 2023, consecutive patients with active CD were prospectively enrolled from other nine tertiary centers as the validation cohort (n = 137). All patients with CD were diagnosed based on the European Crohn’s and Colitis Organization guidelines. 28 The inclusion criteria for the present study were as follows: (1) adult patients (18 ⩽ age ⩽ 75); (2) a confirmed diagnosis of CD for at least 3 months; (3) simplified endoscopic activity score for CD (SES-CD) ⩾ 4 and Crohn’s disease activity index (CDAI) > 15029–31; (4) without receiving any anti-TNF mAbs and other biologics within the previous 3 months; and (5) colonoscopy and TDM data for IFX and ADA available. Concomitant treatment with immunosuppressants [e.g. azathioprine (AZA), methotrexate], oral 5-aminosalicylic acid, oral corticosteroids (tapering regimens), or antibiotics was allowed. The exclusion criteria were as follows: (1) history of bowel surgery; (2) lack of colonoscopy procedures, clinical information, and laboratory parameters; and (3) inability to assess disease extent.
Therapeutic drug monitoring
The serum samples were obtained in all CD patients at week 14 after receiving IFX or ADA therapy following a protocol as described below and sent to the Qingdao Bodhi Huisheng Medical Laboratory (Qingdao, China) for further analysis. The serum trough levels of IFX and ADA were then measured on serum samples using a commercially available ELISA kit (Tarcine BioMed Inc., Beijing, China) according to the manufacturer’s recommendations. All samples were run in singlicate and expressed as μg/mL. Free anti-IFX or anti-ADA antibodies were measured in parallel to drug levels using the ELISA kit. As the kit is sensitive to drugs, it only detects anti-drug antibodies.
Study design and assessments
The reporting of this study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 32 Two hundred and seventy-nine patients with active CD were included in the development cohort. Among them, 158 patients received intravenous infusion of IFX (REMICADE; CilagAG, Schaffhausen, Switzerland) (5 mg/kg) over 30 min on day 1 (namely week 0) and at weeks 2, 6, and 14, while 121 patients received subcutaneous administration of ADA (Humira; AbbVie Inc, Chicago, IL, USA) at doses of 160 mg at week 0, 80 mg at week 2, and thereafter 40 mg every 2 weeks for up to 14 weeks. The serum levels of IFX and ADA were measured just prior to injection at week 14.
Ileo-colonoscopy was performed before treatment and at week 14 post-injection. The endoscopic images were evaluated by experienced central readers (DK and LC) who received training in scoring the SES-CD (score range 0–56). In cases of discrepancy in the assessment between the two investigators, a third investigator (ZL, with 32 years of experience in colonoscopy) reviewed the images to make the final decision. Clinical assessments were performed using CDAI scores and laboratory biomarkers, including serum C-reactive protein (CRP), blood platelet (Plt), albumin (Alb), and erythrocyte sedimentation rate (ESR).
Study endpoints
The primary endpoint of this study was to assess the association between serum anti-TNF levels (ADA and IFX) and primary endoscopic response at week 14. The primary endoscopic response was defined as a decrease in the SES-CD score by >50% compared with the baseline. 30 Exploratory endpoints included the associations between endoscopic response and the disease severity (i.e. CDAI) and laboratory parameters (i.e. CRP, ESR, Alb, and Plt).
Development and validation of a prediction nomogram
Clinical variables were selected using univariate and multivariate logistic regression analyses. Selected variables were integrated into a prediction model and presented as a nomogram. The nomogram translated complex mathematical models into a simple graph of scaled variables, facilitating a quick approximation of the event probability. A receiver operating characteristic (ROC) curve was used to assess the discrimination of the nomogram. A calibration plot was applied to assess the calibration of the nomogram by comparing the actual and predicted probabilities. The nomogram stability was verified by ten-fold cross-validation.
Prospective validation cohort
We developed a prospective external validation cohort from eight tertiary centers between May 2022 and August 2023 (n = 137). Seventy-nine patients received intravenous infusion of IFX (REMICADE), and 58 patients received subcutaneous administration of ADA (Humira). The serum levels of IFX and ADA were measured just prior to injection at week 14. Clinical and laboratory parameters of the patients were collected, as mentioned above.
Statistical analysis
Continuous variables were presented as interquartile ranges (IQRs). The Chi-square test was performed to compare categorical data such as patient sex and other clinical features. The Mann–Whitney U test was performed to compare non-parametric variables such as CDAI and SES-CD. The correlations between the anti-TNF levels and CDAI, SES-CD, CRP, Plt, Alb, and ESR were tested using Spearman’s rank correlation coefficient. ROC curve analysis was used to obtain the cutoff value with optimal sensitivity and specificity. Multivariate regression analysis was used to identify clinical predictors of endoscopic response. A p value less than 0.05 was considered statistically significant. Statistical analyses were performed using Statistical Package for the Social Sciences software (version 26.0; SPSS Inc., New York, NY, USA), Prism software (version 8.0), and R software (version 3.6.3).
Results
Patient characteristics
In total, 416 patients with active CD were enrolled in this study. The clinical characteristics of 279 patients in the development cohort are listed in Table 1, and 137 patients in the validation cohort are exhibited in Supplementary Table 1. Of these patients, 237 were treated with IFX, and 179 were treated with ADA. Most individuals (98.79%) were naïve to biological agents. The median age at baseline was 32 years (22.75–42 years). The median course duration of these patients was 27 months (6.0–65.0 months). Among these patients, 232 (55.77%), 114 (27.40%), and 150 (36.06%) were taking oral 5-aminosalicylates, AZA, and corticosteroids, respectively. The majority of these patients exhibited a moderate active disease with a mean CDAI of 291.91 (236.20–372.22) and SES-CD of 11 (6–19). The baseline clinical characteristics were well balanced between the two cohorts (Supplementary Table 1).
Baseline clinical characteristics.
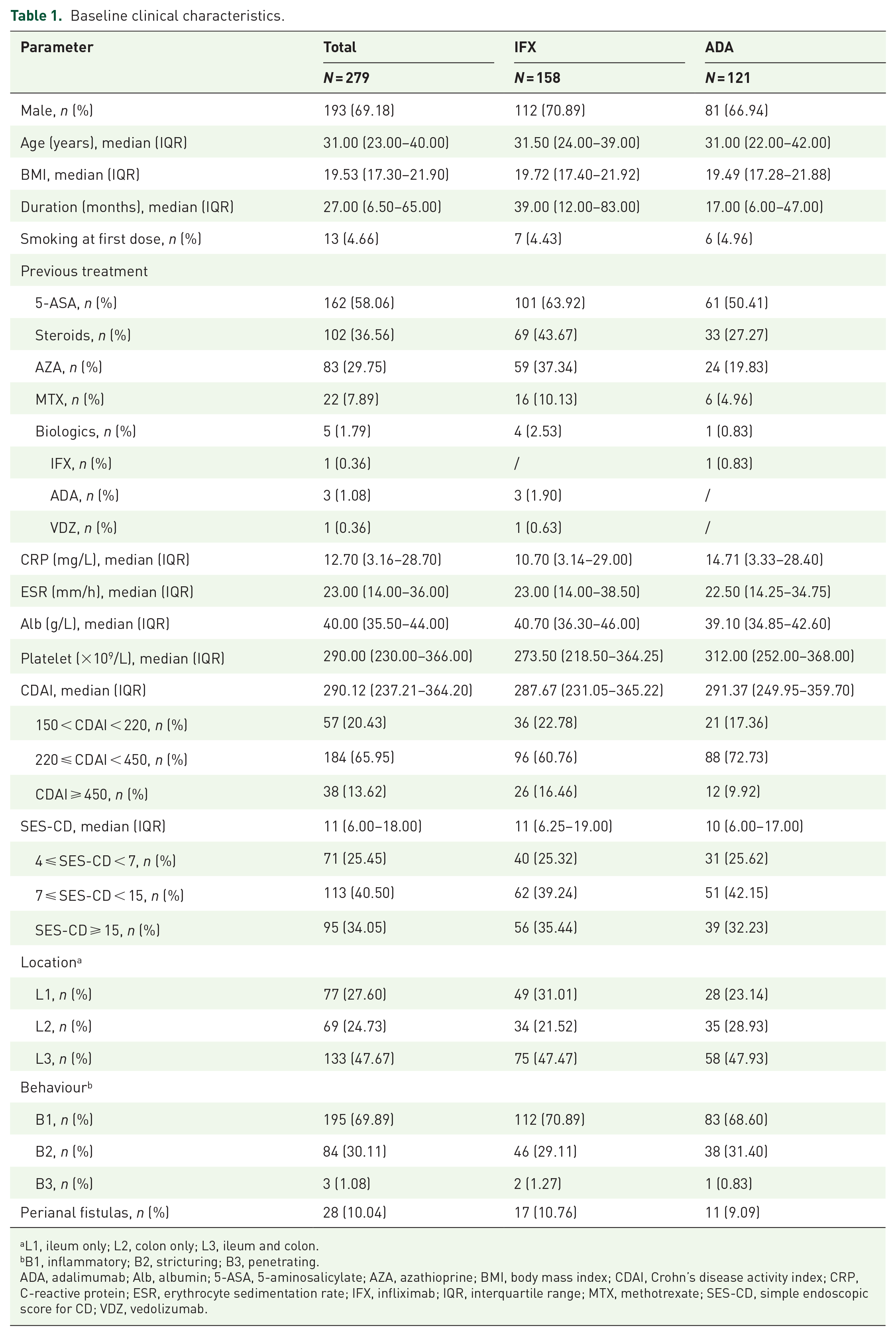
L1, ileum only; L2, colon only; L3, ileum and colon.
B1, inflammatory; B2, stricturing; B3, penetrating.
ADA, adalimumab; Alb, albumin; 5-ASA, 5-aminosalicylate; AZA, azathioprine; BMI, body mass index; CDAI, Crohn’s disease activity index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IFX, infliximab; IQR, interquartile range; MTX, methotrexate; SES-CD, simple endoscopic score for CD; VDZ, vedolizumab.
Clinical predictors of primary endoscopic response
An intention-to-treat analysis revealed that 70 of 158 patients (44.30%) in the IFX group and 66 of 121 patients (54.55%) in the ADA group had an endoscopic response at week 14 in the development cohort. Univariate logistic analysis was performed to further explore the available factors that could predict the primary endoscopic response. Notably, the serum trough levels of IFX and ADA, but not anti-drug antibodies, were significantly higher in patients with endoscopic response rather than in non-responders (p < 0.001) (Figure 1 and Table 2). Moreover, lower levels of SES-CD, CDAI scores, CRP, ESR, and Plt, and higher levels of Alb all contributed to an endoscopic response in both IFX- and ADA-treated groups (p < 0.05) (Table 2). In a univariate analysis, the characteristics of CD patient, including sex [OR (odds ratio) 0.84, 95% CI 0.42–1.69, p = 0.627], age (OR 1.02, 95% CI 0.99–1.04, p = 0.293), body mass index (OR 0.99, 95% CI 0.90–1.08, p = 0.750), disease duration (OR 1.01, 95% CI 1.00–1.01, p = 0.114), and concomitant immunosuppressants (OR 1.51, 95% CI 0.80–2.84, p = 0.201) showed no statistically significant differences in the development cohort receiving IFX therapy (Table 2). Moreover, the same pattern of changes was also observed in the development cohort receiving ADA treatment, except for age (OR 1.04, 95% CI 1.01–1.07, p = 0.013) and disease duration (OR 1.01, 95% CI 1.00–1.02, p = 0.030).
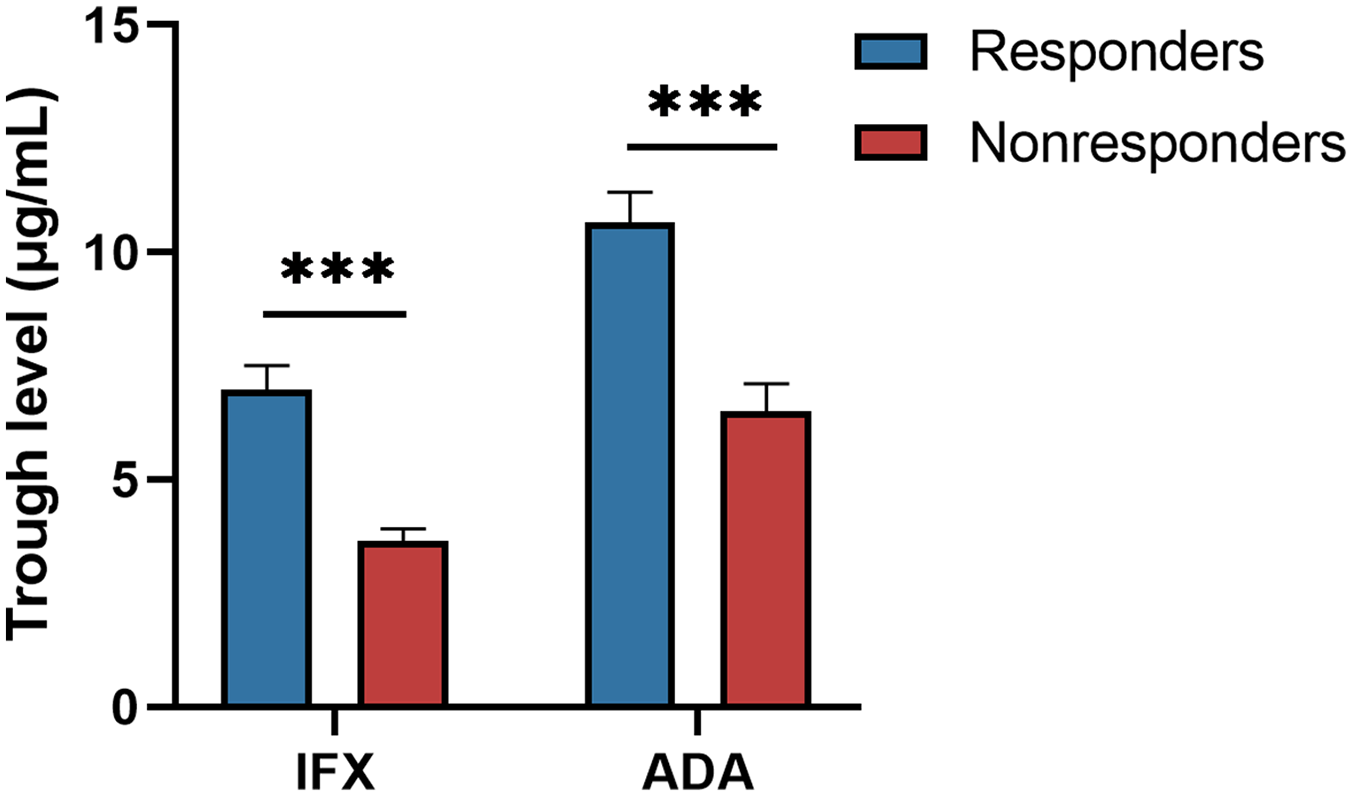
Trough levels of IFX and ADA between responders and non-responders at week 14 post-therapy. The trough levels of IFX and ADA were significantly lower among non-responders compared with those in responders (p < 0.001; Mann–Whitney U test).
Univariate analysis of the variables determining endoscopic non-response to IFX and ADA therapy.

ADA, adalimumab; BMI, body mass index; CDAI, Crohn’s disease activity index; CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IFX, infliximab; IQR, interquartile range; OR, odds ratio; Plt, platelet; SES-CD, simple endoscopic score for CD.
Correlations among anti-TNF level, CDAI, SES-CD, and laboratory parameters
The correlation between endoscopic response and the concentrations of IFX and ADA was tested in the development cohort. The trough levels of IFX demonstrated a weak but significant negative correlation with SES-CD (r = −0.374, p < 0.001), CDAI (r = −0.383, p < 0.001), and CRP (r = −0.335, p < 0.001), respectively [Figure 2(a)–(c)]. The trough levels of IFX also exhibited a weak but significant correlation with ESR (r = −0.297, p = 0.002) and Alb (r = 0.409, p < 0.001), respectively [Supplementary Figure 1(A) and (B)]. Moreover, the levels of ADA exhibited a modest but significant negative correlation with SES-CD (r = −0.332, p < 0.001), CDAI (r = −0.404, p < 0.001), and CRP (r = −0.305, p < 0.001) at week 14 post-ADA therapy in the development cohort [Figure 2(d)–(f), Supplementary Figure 1(D)–(F)].
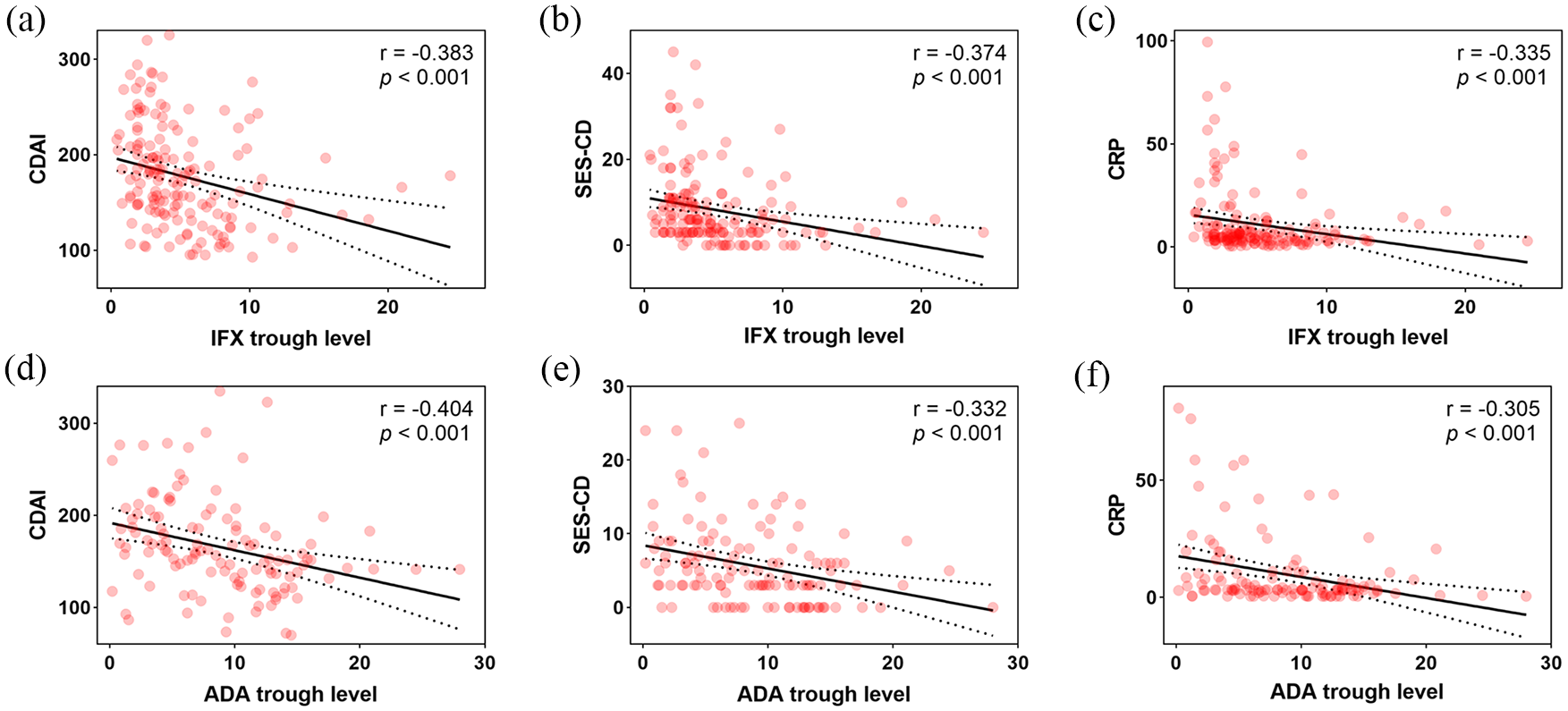
Correlations among anti-TNF trough levels and CDAI, SES-CD, and serum CRP. (a–c) Correlations among IFX trough levels and CDAI (a), SES-CD (b), and CRP (c), respectively. (d–f) Correlations among ADA trough levels and CDAI (d), SES-CD (e), and CRP (f), respectively (Spearman’s correlation coefficient).
The cutoff values of IFX and ADA trough levels and diagnostic performances
Endoscopic non-response was considered the primary outcome. Endoscopic response was defined as a decrease in SES-CD of >50%. 30 ROC curves were constructed to determine the optimal cutoff levels of IFX or ADA to predict treatment failure in the development cohort. IFX level of 4.80 μg/mL showed the best performance in predicting endoscopic non-response [area under the curve (AUC) = 0.770, p < 0.001; sensitivity = 81.8%, specificity = 64.3%] [Figure 3(a)], with a positive predictive value (PPV) of 74.2% and a negative predictive value (NPV) of 73.8% (Table 3). Similarly, ADA level ⩽8.80 μg/mL appeared the best model in predicting endoscopic non-response (AUC = 0.732, p < 0.001; sensitivity 72.7%, specificity 65.2%) [Figure 3(b) and Table 3], with a PPV of 63.5% and NPV of 74.1% (Table 3).
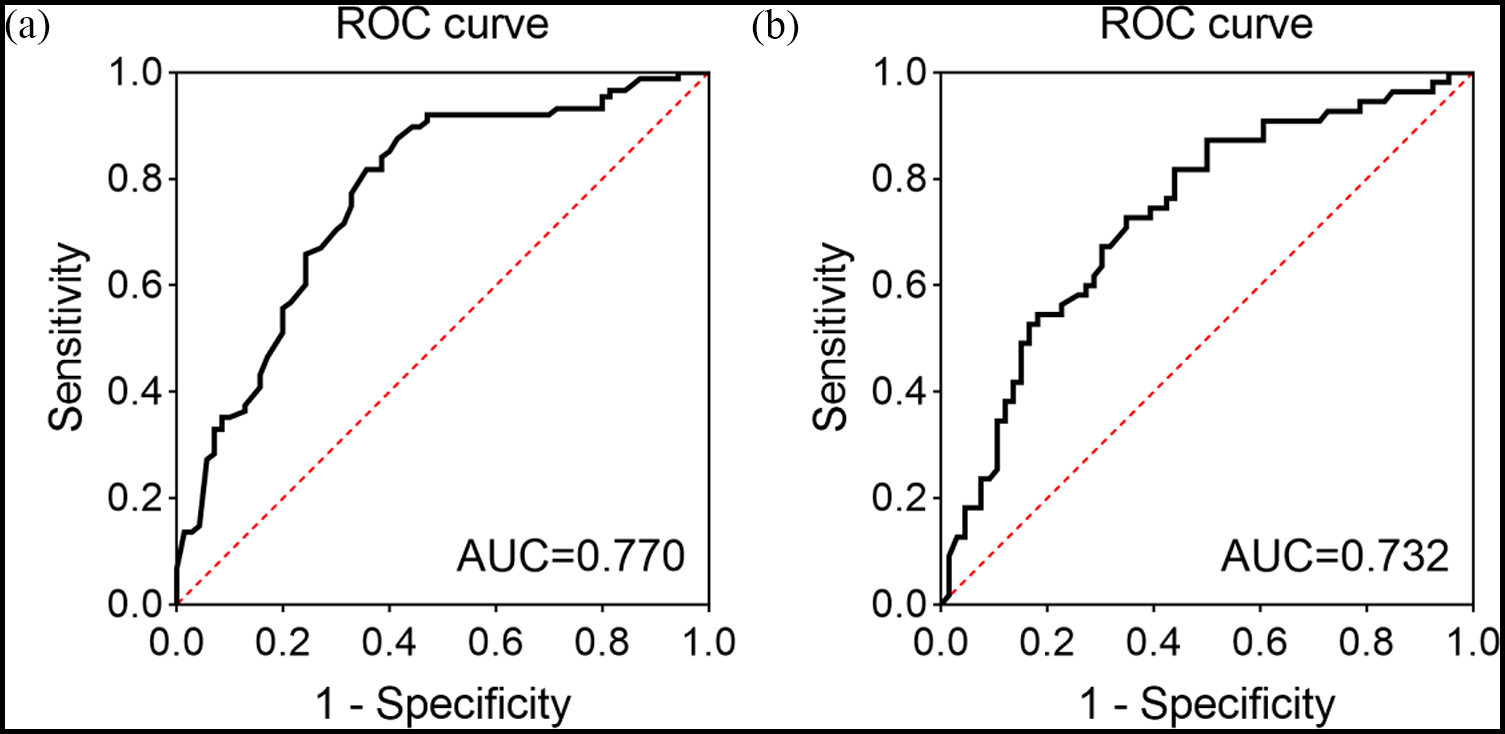
ROC curves of the trough levels of IFX and ADA in predicting the endoscopic non-response of the patients with CD at week 14 post-therapy. (a) The AUC of the trough levels of IFX (AUC = 0.770). (b) The AUC of the trough levels of ADA (AUC = 0.732).
The final performances of trough levels of IFX and ADA for diagnosis of CD.

The endoscopic response was defined as that SES-CD score decreases by >50% compared with the baseline.
Sensitivity, 0.818 (95% CI: 0.738–0.899); specificity, 0.643 (95% CI: 0.531–0.755).
Sensitivity, 0.727 (95% CI: 0.590–0.839); specificity, 0.652 (95% CI: 0.524–0.765).
ADA, adalimumab; CD, Crohn’s disease; CI, confidence interval; IFX, infliximab; SES-CD, simple endoscopic score for CD.
Multivariate outcome analysis
Multivariate analysis was performed to investigate the clinical factors in predicting endoscopic non-response using multivariate logistic regression in the development cohort. The variables were selected by comprehensively considering the results of the univariate analysis and clinical significance. Multivariate logistic regression analysis demonstrated that IFX level (OR 0.81, 95% CI 0.70–0.93, p = 0.002), CRP (OR 1.08, 95% CI 1.02–1.14, p = 0.014), and Alb (OR 0.81, 95% CI 0.72–0.90, p < 0.001) were potent predictors of endoscopic non-response to IFX therapy (Table 4). In addition, ADA level (OR 0.90, 95% CI 0.81–0.99, p = 0.030) and Alb value (OR 0.88, 95% CI 0.80–0.97, p = 0.010) were observed to be associated with endoscopic non-response to ADA treatment (Table 4).
Multivariate analysis of the variables associated with endoscopic response to IFX and ADA induction.

ADA, adalimumab; Alb, Albumin; BMI, body mass index; CI, confidence interval; CRP, C-reactive protein; IFX, infliximab; OR, odds ratio.
Development and validation of nomogram prediction models
We subsequently developed a final prediction model based on the results of the multivariate logistic regression analysis, which consisted of CRP, Alb, and anti-TNF levels at week 14 in the development cohort. A prognostic nomogram that integrated these three important variables was created to calculate the risk probability of endoscopic non-response to anti-TNF treatment in patients with active CD (Figure 4). The nomogram was calculated using one scale corresponding to each clinical variable: a score scale, total score scale of each variable, and probability scale. The final probability of endoscopic response was calculated by adding the scores for all variables. ROC curves were established to evaluate the predictive ability of the nomogram in the development and external validation groups, respectively. ROC analysis indicated that an AUC of IFX and ADA was 0.874 (95% CI, 0.812–0.921) and 0.828 (95% CI, 0.748–0.890), respectively, in the development model [Supplementary Figure 2(A) and (B)], while an AUC of IFX and ADA was 0.816 (95% CI, 0.812–0.921) and 0.817 (95% CI, 0.748–0.890), respectively, in the external validation cohort [Figure 5(a) and (b)]. The calibration plots demonstrated that the development model had average errors of 0.048 and 0.040 for IFX and ADA, respectively [Supplementary Figure 2(C) and (D)], while the external validation cohort showed errors of 0.037 and 0.031, respectively [Figure 5(c) and (d)]. These results indicate no significant difference (p > 0.05) between the model’s calculated probability and endoscopic non-response based on the data. Comparisons between the nomogram and three independent factors indicated that the nomogram had excellent predictive value and discrimination in both the development (Supplementary Table 2) and external validation cohorts (Table 5).

Nomogram based on anti-TNF trough levels, CRP and Alb predicts the risk of endoscopic non-response at week 14. (a) Nomogram for CD patients treated with IFX. (b) Nomogram for CD patients treated with ADA. For example, a 25-year-old male CD patient treated with IFX for 14 weeks; the laboratory findings were as follows: IFX trough levels 1.4 μg/mL, CRP 44.82 mg/L, and Alb 35 g/L. By adopting IFX nomogram to this case, a total of 178.8 points (IFX trough level 71.3 points, CRP 45 points, and Alb 62.5 points) was calculated, indicating that the probability of endoscopic non-response was above 90%.

External validation of nomogram prediction models. (a) ROC curves of the IFX nomogram. (b) ROC curves of the ADA nomogram. Calibration plots of the nomogram of IFX (c) and ADA (d).
The predictive ability of validation cohorts of IFX and ADA.

ADA, adalimumab; AUC, area under the curve; CI, confidence interval; IFX, infliximab; NPV, negative predict value; PPV, positive predict value; SE, sensitivity; SP, specificity.
Discussion
In the present study, we described a robust association of the trough levels of IFX and ADA, Alb, and CRP with endoscopic response in active CD patients receiving treatment with anti-TNF mAbs. Accordingly, we constructed a novel nomogram based on anti-TNF serum trough levels, Alb, and CRP to predict the LOR when initiating anti-TNF therapy. The performance of the nomogram prediction model was demonstrated by ROC analysis and calibration curve in both development and validation cohorts. Clinical and endoscopic responses were more likely to occur in patients with CD with high trough levels of IFX and ADA, high concentrations of Alb levels, and low levels of CRP. Overall, our findings revealed an association between the concentrations of IFX and ADA and early endoscopic response in active CD. More importantly, this novel nomogram enables us to optimize individual treatment strategies and promote clinical response in patients with active CD.
Recent years, several lines of evidence have evaluated an association of early induction of IFX with clinical outcome, while most studies included a limited number of patients and focused on maintenance therapy. The cut-offs for IFX levels over an early induction period and observation endpoints for clinical response to IFX induction have not been well defined.9,18,24,25,27,33,34 Therefore, the primary aim of this study was to evaluate the short-term effects of anti-TNF therapy on the induction of endoscopic response in active CD. In our study, we found that both IFX and ADA levels were higher in primary responders than those in non-responders at week 14 post-therapy. Furthermore, the serum levels of IFX or ADA differed according to the disease activity status, which was consistent with previous reports.35,36 Previously, Ward et al. 35 identified target thresholds for IFX of 1.5, 3.4, and 5.7 μg/mL that were in accordance with clinical and biochemical remission and calprotectin normalization, respectively. However, they found no relationship between ADA levels and any of the indices of disease activity. 35 Nevertheless, other reports have shown that ADA target thresholds of 7.1 μg/mL have the optimal value to predict mucosal healing. 36 It should be pointed out that both studies mentioned above focused on anti-TNF maintenance therapy for IBD patients. In our study, we identified target thresholds for IFX (4.80 μg/mL) and ADA (8.80 μg/mL), which were significantly associated with the endoscopic response over an induction period. Concerning the variant concentrations of IFX or ADA in predicting the clinical outcome during therapy, different bioavailability, pharmacokinetics, and administration methods may be all associated with a change in the levels of IFX and ADA. The timing of drug level measurement is also not consistent in ADA and IFX. Patients receiving IFX always have trough levels measured, while the drug levels of ADA appear to be relatively stable across fortnightly treatment cycles. Thus, the above factors may cause a different biological threshold for IFX and ADA.
Currently, predictions of the primary outcome of anti-TNF therapy are mostly based on endoscopy, radiography, and laboratory examination monitoring, and few studies have focused on the scoring system to determine patients’ risk of LOR. In this study, we constructed a nomogram based on a combination of parameters (serum concentrations of IFX, ADA, Alb, and CRP) to predict an early endoscopic response to anti-TNF therapy. The nomogram showed an excellent prediction with good calibration and was also well-performed using a ROC analysis. The nomogram analysis indicated that lower concentrations of IFX, ADA, and Alb as well as higher levels of CRP were linked to a greater risk of primary endoscopic non-response to anti-TNF therapy. For the training and external validation groups of this nomogram, 10-fold cross-validation was employed, which showed good stability. Both CRP and Alb levels showed significant statistical differences in the univariate and multivariate analyses. Previous studies have indicated that Alb level is associated with serum IFX levels, possibly contributing to its predictive power for good response to IFX. 37 There were also studies demonstrating that CRP, nutritional status, and a combination of parameters (Alb, CRP, and endoscopy) could serve as predictive indicators for clinical outcomes in patients with IBD.33,38,39 Therefore, the aforementioned studies demonstrated the importance of CRP and Alb levels in assessing the disease severity of IBD. Few studies have investigated the significance of serum levels of IFX and anti-IFX antibody (ATI) in predicting the clinical outcomes for patients with IBD. Bar-Yoseph et al. took advantage of the serum levels of IFX and ATI to predict the primary non-response in patients with active CD. The AUC of the IFX ROC curve at weeks 2 and 6 and the ATI ROC curve at week 6 were 0.68, 0.78, and 0.78, respectively. 27 In our study, the AUC of the nomogram prediction model for IFX treatment was 0.87 and 0.816 in the development and external validation groups, respectively, which was significantly higher than that of IFX or ATI concentrations alone. Additionally, Baert et al. 40 utilized the ADA concentration in UC patients at week 4 to predict the clinical response at week 12 with a sensitivity of 80%, specificity of 56%, PPV of 85%, and NPV of 47%. In our study, the AUC of the nomogram predicting the primary non-response to ADA at week 14 was 0.83, with a sensitivity of 82%, specificity of 76%, PPV of 74%, and NPV of 83% in the development cohort. The predictive value of ADA was significantly higher than that of ADA alone. The same results were also observed in the external validation cohort. Therefore, we suppose that our TDM-based nomogram may offer a more precise prediction of early endoscopic response in patients with active CD treated with anti-TNF mAbs as compared to other parameters such as TDM or biochemical biomarker alone.
The present study has several limitations. First, although the relationships between the serum concentrations of IFX and ADA, Alb, CRP, and the efficacy outcomes are both robust and consistent, these data are retrospective and may have been influenced by other factors in addition to both clinical outcomes and drug concentrations. Furthermore, as a retrospective study, the grading of SES-CD was based solely on previous colonoscopy images in the developmental cohort. Third, only 416 patients were included in this study, including 237 for IFX and 179 for ADA, and more prospective cohort multicenter studies are needed to further explore the predictive value of the TDM-based nomogram.
In summary, this study has established a strong association between serum concentrations of IFX and ADA, Alb, CRP, and primary clinical outcomes in CD patients. Importantly, we established a TDM-based nomogram showing an excellent predictive value with remarkable accuracy and discrimination. The nomogram algorithm may enable clinicians to better understand the underlying mechanisms by which an individual patient fails to achieve the expected efficacy, which is important for optimizing therapy options and switching biologics in refractory CD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241256237 – Supplemental material for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease
Supplemental material, sj-docx-1-tag-10.1177_17562848241256237 for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease by Liang Chen, Dengfeng Kang, Leilei Fang, Mingming Sun, Mingsong Li, Guangxi Zhou, Chunjin Xu, Zhi Pang, Yulan Ye, Baisui Feng, Huili Wu, Jian Lin, Baijing Ding, Changqin Liu, Yanhong Shi and Zhanju Liu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848241256237 – Supplemental material for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease
Supplemental material, sj-docx-2-tag-10.1177_17562848241256237 for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease by Liang Chen, Dengfeng Kang, Leilei Fang, Mingming Sun, Mingsong Li, Guangxi Zhou, Chunjin Xu, Zhi Pang, Yulan Ye, Baisui Feng, Huili Wu, Jian Lin, Baijing Ding, Changqin Liu, Yanhong Shi and Zhanju Liu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-3-tag-10.1177_17562848241256237 – Supplemental material for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease
Supplemental material, sj-tif-3-tag-10.1177_17562848241256237 for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease by Liang Chen, Dengfeng Kang, Leilei Fang, Mingming Sun, Mingsong Li, Guangxi Zhou, Chunjin Xu, Zhi Pang, Yulan Ye, Baisui Feng, Huili Wu, Jian Lin, Baijing Ding, Changqin Liu, Yanhong Shi and Zhanju Liu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-4-tag-10.1177_17562848241256237 – Supplemental material for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease
Supplemental material, sj-tif-4-tag-10.1177_17562848241256237 for Development and validation of a novel therapeutic drug monitoring-based nomogram for prediction of primary endoscopic response to anti-TNF therapy in active Crohn’s disease by Liang Chen, Dengfeng Kang, Leilei Fang, Mingming Sun, Mingsong Li, Guangxi Zhou, Chunjin Xu, Zhi Pang, Yulan Ye, Baisui Feng, Huili Wu, Jian Lin, Baijing Ding, Changqin Liu, Yanhong Shi and Zhanju Liu in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
