Abstract
Background:
The existing body of scientific literature offers inconclusive findings on the safety and therapeutic effectiveness of etrolizumab (ETR) for the treatment of ulcerative colitis (UC).
Objectives:
The goal of this meta-analysis is to furnish a comprehensive synthesis of evidence that evaluates the safety and therapeutic effects of ETR in the management of UC.
Design:
Meta-analysis.
Data sources and methods:
PubMed, Embase, and Web of science were searched to collect relevant English studies, and the reference lists of eligible studies were manually searched to avoid missing any eligible studies. Outcome measures encompassed clinical response, incidence of adverse events, histological remission, endoscopic remission, endoscopic improvement, and antidrug antibodies. Relevant data were extracted by two independent investigators.
Results:
The meta-analysis incorporated five eligible studies, involving a total of 1528 patients, with 1015 treated with ETR and 513 with placebo. The pooled analysis indicates that ETR is both effective and safe. The adverse event rates, endoscopic and histological response, as well as overall remission were comparable between the two groups. The monoclonal antibody group had a lower incidence rate of adverse reactions than the placebo group [odds ratio (OR): 0.81; 95% confidence interval (CI): 0.63–1.03; p = 0.09)]. Clinical response was higher in the ETR group than in the placebo group (OR: 1.56; 95% CI: 1.20–2.02; p = 0.0009), and endoscopic improvement was more favorable in the ETR group (OR: 1.88; 95% CI: 1.45–2,45; p < 0.00001). A higher rate of endoscopic remission was found in the ETR group than in the placebo group (OR: 2.48; 95% CI: 1.75–3.50; p < 0.00001); histological remission was significantly higher in the ETR group than in the placebo group (OR: 2.11; 95% CI: 1.55–2.86; p < 0.00001). The placebo group had a lower rate of positive antidrug antibodies (OR: 1.31; 95% CI: 0.79–2.17; p < 0.29), and the incidence of complications was significantly higher in the ETR group compared with the placebo group (OR: 2.05; 95% CI: 1.48–2.83; p < 0.0001).
Conclusion:
Given the heterogeneity and potential biases in the included studies, gastroenterologists should cautiously tailor drug delivery strategies based on their clinical experience and the unique needs of individual patients.
PROSPERO registration:
CRD42023396100
Introduction
Ulcerative colitis (UC) is a chronic gastrointestinal disorder characterized by recurring inflammation and significant morbidity, affecting both physical and psychological well-being of patients, with a high risk of mortality.1,2 The annual incidence of UC ranges from 1.2 to 20.3 cases per 100,000 individuals. 3
Current therapeutic strategies for managing moderate-to-severe UC encompass corticosteroids, immunosuppressive agents, and targeted biologics, like tumor necrosis factor inhibitors (anti-TNFs), vedolizumab, ustekinumab, and Janus Kinase (JAK) inhibitors. 4 Despite these advancements, over 80% of UC patients experience recurrent disease courses, are often complicated by dysplasia, and have an increased risk of colorectal cancer.2,5 Consequently, nearly 20% of these patients undergo colectomy to manage disease complications.
Emerging research posits that UC is a multifactorial disease, influenced by genetic predisposition, gut microbiota, environmental factors, and complex immune pathways.6–12 This heterogeneity precludes a one-size-fits-all treatment approach, making ‘remission’ the more attainable therapeutic goal over ‘cure’.
Targeted therapies, characterized by favorable safety profiles and the potential for achieving clinical remission, may offer a promising treatment option. However, the safety of pharmacological interventions, particularly concerning the elevated risks of infection and malignancy, remains a critical consideration in clinical practice. 13
Infliximab, a pioneering monoclonal antibody-based biologic agent, was approved for UC treatment in 2005. 14 Due to the heavy burden of surgical harm, drug therapies are predominantly employed for the management of UC patients. Meanwhile, surgical interventions like ‘rescue’ ileostomy are considered in cases of acute exacerbations to avert emergency total colectomy. A recent comparative study suggests that etrolizumab (ETR) may have fewer side effects than infliximab, although its long-term efficacy remains under investigation. 15
Etolizumab, a humanized IgG1 monoclonal antibody, selectively acts on the β7 subunit of both α4β7 and αEβ7 integrins to modulate leukocyte migration in the gut. It operates through two separate mechanisms: (i) α4β7 binds to mucosal vascular addressin cell adhesion molecule 1 (MAdCAM-1) to block the migration of leukocytes to the gut; (ii) αEβ7 interacts with E-cadherin to retain specific lymphocyte subpopulations within the intestinal mucosa.4,14,16–23
Despite the promise of targeted therapies, challenges such as high costs and treatment nonresponse persist. 13 Recently, the concept of ‘disease clearance’, encompassing both symptomatic relief (PRO) and mucosal healing (endoscopic and histological healing), has been introduced. This concept suggests a progression from histological to molecular healing in UC management. 3
The aim of this study is to offer a comprehensive analysis of the safety and efficacy of these therapies utilizing randomized controlled trials (RCTs), real-world data, and network meta-analyses, assisting clinicians in tailoring patient-specific treatment strategies. 24
Methods
Literature search
This study was reported in compliance with Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 guidelines 25 and was prospectively registered under PROSPERO (CRD42023396100). The PRISMA 2020 checklist is shown in Supplemental Table S1. A systematic search was conducted in PubMed, Embase, and Web of Science up to March 2023 to collect studies that compared the efficacy and/or safety of etrolizumab with placebo (PLA) in treating UC. The following search terms were utilized: ‘etrolizumab’ and ‘ulcerative colitis’. Supplemental Table S2 provides a detailed search strategy. Reference lists of eligible studies were also manually retrieved. Two independent investigators searched the studies, and any disagreements were resolved through consensus.
Identification of eligible studies
Inclusion criteria for the studies were as follows:
Randomized controlled, cohort, or case–control study designs.
Studies involving adults with moderately to severely active UC.1,4,26–28
Comparative analyses of safety and efficacy between placebo and etrolizumab.
Assessment of at least one of the following outcomes: any adverse event, clinical response, endoscopic improvement, endoscopic remission, histological remission, presence of antidrug antibodies, or remission.
Studies that provided sufficient data for calculating risk ratio (RR) or weighted mean difference (WMD).
Exclusion criteria encompassed letters, corrections, reviews, editorial comments, case reports, conference abstracts, pediatric studies, unpublished works, and non-English articles.
Data extraction
Two investigators independently extracted data, and a third investigator was consulted for consensus in case of disagreements. The data collected included variables such as the first author, publication year, study period, country of study, sample size allocation (Etrolizumab/Placebo), follow-up duration (in weeks), age (in years), gender distribution (male), body mass index (BMI) in kg/m2, duration of disease (in years), Mayo Clinic Total Score (MCS), Nancy Histological Index (NHI), clinical response, incidence of any adverse events, presence of antidrug antibodies, endoscopic improvement, endoscopic remission, histological remission, and overall remission. For continuous variables reported as median with range or interquartile range, the mean ± standard deviation was calculated using validated mathematical methods.29,30
Quality assessment
The quality of eligible RCTs was assessed using the Cochrane Handbook for Systematic Reviews of Interventions (v5.1.0). Seven dimensions were considered: randomized sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, completeness of outcome data, selective reporting, and other potential sources of bias. 31 Each dimension was answered by low, high, or unclear risk. Studies with a predominance of ‘low risk’ results were deemed superior. Any discrepancies in evaluations were resolved through discussion among investigators.
Statistical analysis
Statistical analysis was conducted using Review Manager 5.3 (Cochrane Collaboration, Oxford, UK). Continuous and dichotomous variables were expressed as weighted mean difference (WMD) and risk ratio (RR) with 95% confidence intervals (CIs), respectively. Heterogeneity among studies was evaluated using the χ2 test (Cochran’s Q) and the I2. 32 A χ2 p value below 0.05 or an I2 above 50% signaled significant heterogeneity, and thus, a random-effects model was used to estimate the combined OR.
Results
Systematic literature search and characteristics of included studies
The systematic search and selection process is delineated in Figure 1. A comprehensive literature search yielded 567 relevant articles: PubMed (n = 71), Embase (n = 328), and Web of Science (n = 168). After the elimination of duplicates, 377 titles and abstracts were scrutinized. Ultimately, five full-text articles, involving 1528 patients, were incorporated into the pooled analysis.1,4,26–28 All selected articles were prospective studies. Study characteristics are summarized in Table 1. The details of quality assessment of all eligible studies are presented in Figure 2.

The systematic search and selection process.
Baseline characteristics of include studies and methodological assessment.

The details of quality assessment of all eligible studies. (a) Risk of bias graph. (b) Risk of bias summary.
Demographic characteristics
Statistical analysis revealed no significant disparities between the Etrolizumab and Placebo groups in parameters such as age (WMD: 0.79; 95% CI: −1.39 to 2.96; p = 0.48), gender distribution (male/total, OR: 1.09; 95% CI: 0.88–1.36; p = 0.43), BMI (WMD: 0.13; 95% CI: −0.62 to 0.88; p = 0.73), duration of disease (WMD: −0.70; 95% CI: −1.59 to 0.19; p = 3.05), MCS (WMD: 0.01; 95% CI: −0.16 to 0.17; p = 0.95), and NHI (WMD: −0.06; 95% CI: −0.20 to 0.07; p = 0.36) (Table 2).
Demographics and clinical characteristics of included studies.

BMI, body mass index; CI, confidence interval; MCS, Mayo Clinic total score; OR, odds ratio; WMD, weighted mean difference.
Incidence of adverse events
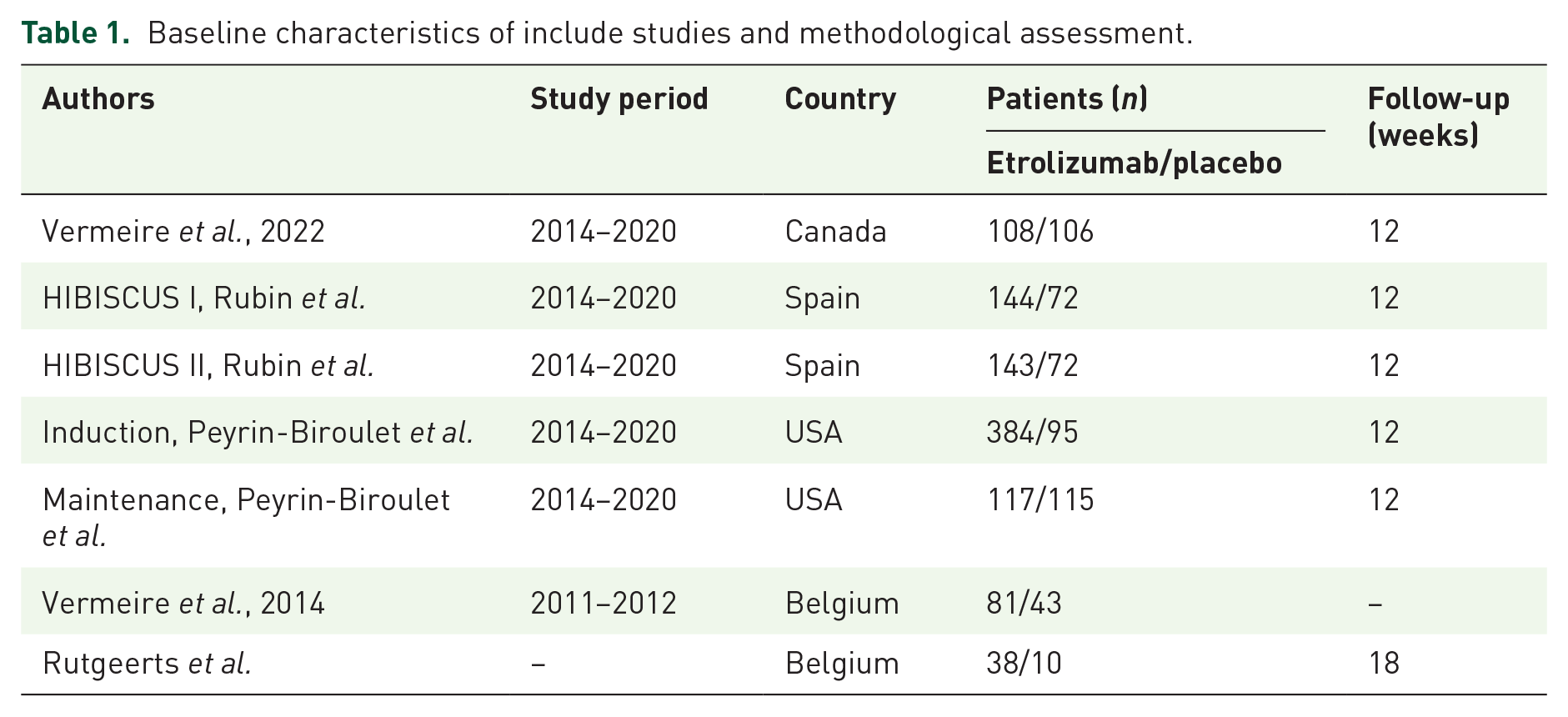
Data from four studies, involving 1470 patients (972 in the ETR group and 498 in the PLC group), were meta-analyzed for adverse events.1,4,26,27 The pooled analysis showed a lower, but not statistically significant, incidence of adverse events in the ETR group (OR: 0.81; 95% CI: 0.63–1.03; p = 0.09) [Figure 3(a)]. No significant heterogeneity was noted (I2 = 34%, p = 0.18), and the funnel plot indicated the presence of a slight publication bias [Figure 4(a)].
The forest plots after the treatment of the drug. (a) Any adverse event. (b) Clinical response. (c) Endoscopic improvement. (d) Endoscopic remission. (e) Histological remission. (f) Positive for antidrug antibodies. (g) Remission.
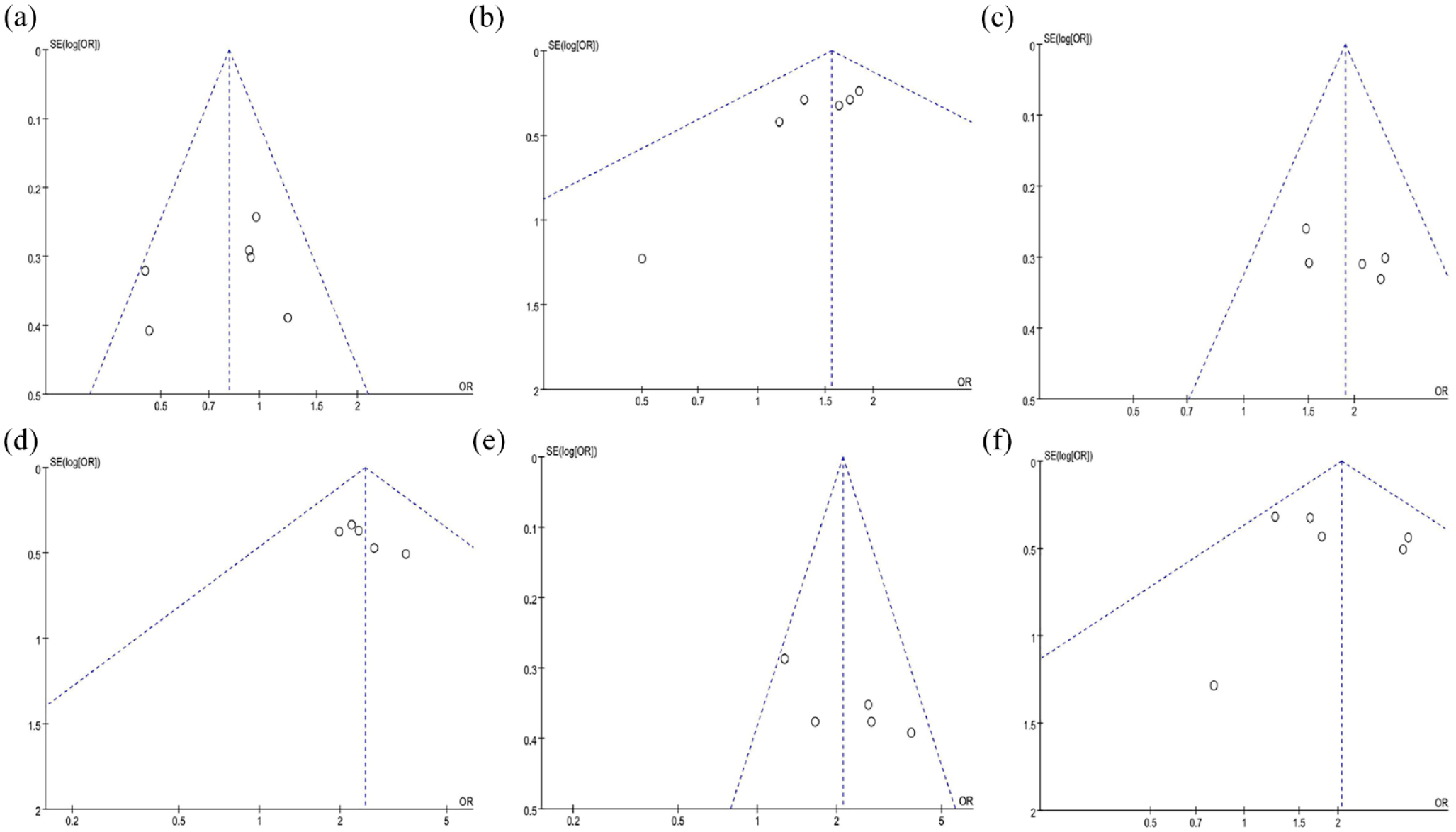
Funnel plots of: (a) Any adverse event, (b) Clinical response, (c) Endoscopic improvement, (d) Endoscopic remission, (e) Histological remission, (f) Remission.
Clinical response
Five studies, involving 1262 patients (875 in the ETR group and 387 in the PLC group), were included in the clinical response analysis.1,4,26–28 A significantly higher clinical response rate was observed in the ETR group (OR: 1.56; 95% CI: 1.20–2.02; p = 0.0009) [Figure 3(b)]. Statistical heterogeneity (I2 = 0%, p = 0.08) and potential publication bias were detected [Figure 4(b)].
Endoscopic improvement
Three studies, comprising 1346 patients (891 in the ETR group and 387 in the PLC group), contributed to the analysis of endoscopic improvement.1,4,26 The ETR group showed significant endoscopic improvement (OR: 1.88; 95% CI: 1.45–2.45; p < 0.00001) [Figure 3(c)], with no significant heterogeneity (I2 = 0%, p = 0.61), and no evidence of publication bias [Figure 4(c)].
Endoscopic remission
Data from five studies, involving 1465 patients (969 in the ETR group and 496 in the PLC group) were meta-analyzed. The results indicated a higher rate of endoscopic remission in the ETR group1,4,26,27 (OR: 2.48; 95% CI: 1.75–3.50; p < 0.00001) [Figure 3(d)], with no significant heterogeneity (I2 = 0%, p = 0.88), and no evidence of publication bias [Figure 4(d)].
Histological remission
Three studies, involving 1088 patients (714 in the ETR group and 374 in the PLC group) reported histological remission. The results revealed a significantly higher rate of histological remission in the ETR group1,4,26 (OR: 2.11; 95% CI: 1.55–2.86; p < 0.00001) [Figure 3(e)]. Heterogeneity was not significant (I2 = 41%, p = 0.15), although a slight publication bias was noted in the funnel plot [Figure 4(e)].
Antidrug antibodies
Data on antidrug antibodies from four articles, covering 553 patients (337 in the ETR group and 216 in the PLC group) were meta-analyzed.1,26–28 The analysis results indicated a lower rate of positive antidrug antibodies in the PLC group (OR: 1.31; 95% CI: 0.79–2.17; p < 0.29), with no significant heterogeneity (I2 = 38%, p = 0.20) [Figure 3(f)].
Incidence of complications
Remission rates were reported in five studies, involving 1488 patients (987 in the ETR group and 216 in the PLC group).1,4,26–28 The pooled results revealed a significantly higher incidence of complications in the ETR group (OR: 2.05; 95% CI: 1.48–2.83; p < 0.0001) [Figure 3(g)]. No significant heterogeneity (I2 = 20%, p = 0.28) or evidence of publication bias was observed [Figure 4(f)].
Discussion
This systematic review synthesizes evidence from five placebo-controlled RCTs evaluating etrolizumab as induction therapy for moderate-to-severe UC. Etrolizumab, a unique dual mechanism of action (MOA) inhibitor, effectively inhibits immune pathways responsible for chronic mucosal inflammation, with minimal adverse effects. 18
The randomized phase I study by Rutgeerts et al. 28 in 2013 initially evaluated the safety and pharmacological effects of etrolizumab in moderate-to-severe UC patients. Subsequently, multiple RCTs that assess the safety and efficacy of etrolizumab against placebo in this patient population have been published.1,4,26,27
Hence, we conducted an updated systematic review and pooled analysis of five RCTs, which included 1528 patients (1015 receiving etrolizumab and 513 receiving a placebo). Several important findings emerged from this analysis.
First, the heterogeneity in dosing and administration routes across different RCTs is summarized in Supplemental Table S3. Rutgeerts’ 2013 study, a phase I trial, divided patients into single ascending dose (SAD) and multiple dose (MD) groups. It reported the clinical remission rate at week 10 for the MD group, finding no statistically significant difference in efficacy between etrolizumab and placebo. 28 Following this, Vermeire’s 2014 study, the first prospective, randomized, placebo-controlled trial, reported that etrolizumab significantly improved clinical remission at week 10 in patients with moderate-to-severe UC, compared to placebo. 27 HICKORY2022, a multicenter, double-blind, placebo-controlled phase III study, showed the results that etrolizumab increased the proportion of patients in remission at week 14 when compared to the placebo. Yet, by week 66, the enhanced remission among those responding to etrolizumab was not sustained. Nevertheless, etrolizumab was generally well-tolerated throughout the treatment course. 1 LAUREL2022 is a randomized, double-blind, placebo-controlled phase III trial. Compared to the placebo group, a higher proportion of patients receiving etrolizumab maintenance therapy achieved remission by week 62, although this difference was not statistically significant. Notably, almost twice as many patients completed maintenance therapy with etrolizumab compared to placebo. 26 HIBISCUS2022 consisted of two identically designed, multicenter, randomized, double-blind, placebo-controlled, and active-controlled phase III studies, aiming to compare the safety and efficacy of etrolizumab with adalimumab and placebo in inducing remission in patients with moderately to severely active UC. 4 Although etrolizumab was not universally superior to placebo in inducing remission across all studies, it was well-tolerated in both therapies and yielded similar numerical results across multiple clinical and endoscopic endpoints.
Second, the pooled data from HIBISCUS I and II indicated that etrolizumab outperformed placebo in 10-week remission rates, yet it was not superior to adalimumab. This has sparked a broad controversy: why is etrolizumab not a well-established and successful cornerstone in the treatment of moderate-to-severe UC compared to vedolizumab? Although etrolizumab has a superior effect on innate immune cell subsets and nonclassical monocytes compared to vedolizumab,33–36 wound healing is further delayed after treatment with etrolizumab compared to vedolizumab, as is the perilesional presence of macrophages with a wound-healing phenotype. Etrolizumab has shown encouraging results in both phase II and large-scale phase III clinical trials for UC.1,4,26,27,37,38
Third, in the Rutgeerts 2013 study, 28 no significant change was observed in the total lymphocyte count in the blood of patients treated with etrolizumab compared to baseline, suggesting that the proportion of leukocytes affected by etrolizumab is relatively small. However, their study could not detect differences in efficacy due to limited power. Subsequently, Binder et al. employed staining of integrin α4β7 on peripheral blood mononuclear cells and granulocytes, followed by flow cytometry analysis on its expression. The results revealed considerable variability in integrin expression across different cell populations, indicating a correlation between differential expression and function. 39 The limited sample size of the Vermeire 2014 27 phase II study (81 patients treated with etrolizumab, two-thirds of whom had prior TNF treatment experience, and 21% were nonresponders to multiple anti-TNF therapies) could impact the assessment results of exposure–response relationship of the drug. The study leaves open the possibility that the reduced efficacy observed in the high-dose group might be merely coincidental. Analysis across various dosage groups showed inconsistent effects on mucosal healing scores (MCS), suggesting that etrolizumab treatment may exert a dose-dependent influence on the migration of various cell types, such as regulatory T cells. Given the lack of clear clinical efficacy at maximum β7 occupancy, this study posits two hypotheses: either pre-existing immune cells in the gut have proinflammatory activity, or there are potential β7-dependent mechanisms for leukocyte migration to the intestinal mucosa. Dai et al. 40 confirmed the critical role of the interaction between E-cadherin and αEβ7 in T cell aggregation within intestinal epithelium, achieved by inhibiting both α4β7 and αEβ7, thereby targeting lymphocyte homing and retention.
This research suggests that blocking the function of α4β7 and αEβ7 may offer an innovative therapeutic strategy for treating moderate-to-severe UC. Nonetheless, additional prospective research is imperative for a more comprehensive understanding of the influence of etrolizumab on the trafficking and functional dynamics of immune cells expressing α4β7 and αEβ7.
Fourth, to gain further insights into alternative mechanisms of inflammation, an analysis was conducted on patients who have been under long-term treatment with etrolizumab. Informed by the phase II EUCALYPTUS study, the phase III study selected a 105 mg dose of etrolizumab for evaluation. 27 However, this single-dose focus could potentially skew the correlation between disease risk factors and drug exposure, possibly leading to an overrated perception of the exposure–response relationship. Although measurements of etrolizumab exposure confirmed that serum trough concentrations were several-fold higher than the concentrations required for at least 90% β7 receptor occupancy in various studies (seven-fold in LAUREL2022 and HICKORY2022, and ten-fold in HIBISCUS2022), the HIBISCUS2022 study had inherent limitations. Specifically, the HIBISCUS study did not allow for dose modifications and thus did not evaluate adalimumab exposure. However, other studies indicated that increasing the dose in the peripheral circulation beyond complete receptor occupancy could potentially offer additional therapeutic advantages for this kind of anti-integrin therapy.41,42 Due to the disparate patient populations and study designs between HICKORY2022 and HIBISCUS2022, as well as the use of different methods for drug resistance detection, it becomes challenging to directly compare the incidence of antidrug antibodies across phase III and earlier studies. Interestingly, the LAUREL2022 study reported a 32% incidence of antidrug antibodies in both treatment groups, a rate higher than those observed in phase I and phase II studies of etrolizumab. Although no definitive link was established between the presence of etrolizumab antidrug antibodies and pharmacodynamic markers in these phase III trials, the subtle influence of such antibodies on both therapeutic efficacy and safety cannot be ruled out.
While Sommer et al. first illuminate the role of αEβ7 on nonclassical monocytes as a therapeutic target for etrolizumab, the MOA and efficacy of the drug remain elusive. Although significant outcomes were observed in objective endpoints, namely endoscopic and histological manifestations, the driving forces behind the study results remained unidentified when examining the MCS metrics. This appears to explain the challenge observed in phase III clinical trials of etrolizumab, where only a few primary endpoints were met.18,43 Although baseline expression of αE in the intestine is considered as a potential biomarker for predicting etrolizumab response, 44 and some conclusions about its biological activity have been drawn from phase III trials,1,4,26,37 some factors that limit the remission-promoting activity of etrolizumab have been overlooked. It is important to clarify that our in vivo data establish a correlative link rather than a causal relationship between the reduction in classic monocyte recruitment and wound healing. 43
Etolizumab emerges as a significant new treatment for moderate-to-severe active UC. The analysis incorporates data from five RCTs, demonstrating that etolizumab is effective and generally well-tolerated in treating moderate-to-severe active UC compared to a placebo. Nevertheless, the risk of patients testing positive for antidrug antibodies needs to be further investigated. Despite tofacitinib having certain advantageous features, etolizumab holds a superior edge, particularly in the frequency of adverse reactions. Moreover, there is a statistically significant difference between etolizumab and placebo in terms of clinical, endoscopic, and histological remission rates. As clinical experience with this novel therapeutic agent accumulates, its definitive role relative to more established biological therapies is expected to become increasingly clear. 17
While this meta-analysis provides valuable insights into the efficacy and safety of etolizumab for treating UC, it has several limitations that warrant further investigation. First, the number of included RCTs is limited, which may compromise the conclusion that etolizumab may serve as a viable induction therapy for moderate-to-severe UC patients who have failed conventional treatments. The risk of testing positive for antidrug antibodies remains unclear due to the small sample size of patients in dosage subgroups. Second, the sparse recruitment data leaves us uncertain about the likelihood of patients experiencing adverse events, severe adverse events, or discontinuation due to these events. Expanding the sample size in future studies could potentially unveil both statistically and clinically meaningful differences between etolizumab and placebo. Thirdly, the included studies primarily enrolled patients from North America, South America, Europe, Asia, Oceania, and the Middle East. Conversely, there are a small number of participants from Asian countries, which may cause biased results, because dietary habits vary among countries and might influence the progression of the disease. Nevertheless, this is the first meta-analysis to comprehensively evaluate the efficacy and safety of etolizumab, filling a notable gap in the existing literature.
Conclusion
In summary, etrolizumab is a viable and safe therapeutic alternative for UC. It outperforms placebo in clinical response, endoscopic improvement, endoscopic remission, histological remission, and overall remission.
However, given the heterogeneity and potential biases in the included studies, gastroenterologists should cautiously tailor drug delivery strategies based on their clinical experience and the unique needs of individual patients.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241253685 – Supplemental material for Meta-analysis of etrolizumab versus placebo in ulcerative colitis: safety and efficacy outcomes
Supplemental material, sj-docx-1-tag-10.1177_17562848241253685 for Meta-analysis of etrolizumab versus placebo in ulcerative colitis: safety and efficacy outcomes by Rui Zhang, Ziran Jia and Yingshi Piao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848241253685 – Supplemental material for Meta-analysis of etrolizumab versus placebo in ulcerative colitis: safety and efficacy outcomes
Supplemental material, sj-docx-2-tag-10.1177_17562848241253685 for Meta-analysis of etrolizumab versus placebo in ulcerative colitis: safety and efficacy outcomes by Rui Zhang, Ziran Jia and Yingshi Piao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
