Abstract
Background:
Barrett’s esophagus (BE) is a precursor of esophageal adenocarcinoma. It is critical to recognize the risk factors associated with BE.
Objectives:
The present meta-analysis aims to systematically estimate the association of hiatal hernia with the risk of BE.
Design:
A meta-analysis with trial sequential analysis.
Data sources and methods:
The PubMed, EMBASE, and Cochrane Library databases were searched. The pooled odds ratios (ORs) and adjusted ORs (aORs) with their 95% confidence intervals (CIs) were calculated for the combined estimation of unadjusted data and data adjusted for confounders, respectively. Heterogeneity was quantified using the Cochrane Q test and I² statistics. Subgroup, meta-regression, and leave-one-out sensitivity analyses were employed to explore the sources of heterogeneity.
Results:
Forty-seven studies with 131,517 participants were included. Based on the unadjusted data from 47 studies, hiatal hernia was significantly associated with an increased risk of any length BE (OR = 3.91, 95% CI = 3.31–4.62, p < 0.001). The heterogeneity was significant (I² = 77%; p < 0.001) and the definition of controls (p = 0.014) might be a potential contributor to heterogeneity. Based on the adjusted data from 14 studies, this positive association remained (aOR = 3.26, 95% CI = 2.44–4.35, p < 0.001). The heterogeneity was also significant (I² = 65%; p < 0.001). Meta-analysis of seven studies demonstrated that hiatal hernia was significantly associated with an increased risk of long-segment BE (LSBE) (OR = 10.01, 95% CI = 4.16–24.06, p < 0.001). The heterogeneity was significant (I² = 78%; p < 0.001). Meta-analysis of seven studies also demonstrated that hiatal hernia was significantly associated with an increased risk of short-segment BE (OR = 2.76, 95% CI = 2.05–3.71, p < 0.001). The heterogeneity was not significant (I² = 30%; p = 0.201).
Conclusion:
Hiatal hernia should be a significant risk factor for BE, especially LSBE.
Registration:
PROSPERO registration number CRD42022367376.
Keywords
Graphical abstract
Introduction
Barrett’s esophagus (BE), a major complication of gastroesophageal reflux disease (GERD), is defined as an abnormal condition in which normal esophageal squamous epithelium has been replaced by columnar-lined epithelium with histological evidence of specialized intestinal metaplasia. 1 BE is the only known precursor lesion for the development of esophageal adenocarcinoma (EAC). It is estimated that about 0.33% of BE individuals progress to EAC annually worldwide 2 and the risk of EAC among BE patients is 10- to 55-fold higher as compared to the general population. 3 BE is usually asymptomatic in nature and can only be diagnosed by endoscopy, therefore, its estimated prevalence in the population remains difficult to assess. 4 Existing risk factors for BE include gastroesophageal reflux symptoms, male, age greater than 50 years, white race, increased body mass index, and/or central adiposity, smoking, and family history.1,5–8
Hiatal hernia is a common digestive disease with acid regurgitation, heartburn, and chest pain as the predominant manifestations, which refers to protrusion of stomach or other abdominal viscera through the esophageal hiatus of the diaphragm into the mediastinum. 9 It has been divided into four types based on the currently recognized anatomical classification. Type 1 hiatal hernia is the most prevalent type, accounting for more than 90% of cases of hiatal hernia, also termed a sliding hiatal hernia; and types 2–4 refer to paraesophageal hernia. 10 Hiatal hernia has been recognized as an important pathophysiology of GERD and strongly correlates with BE. 10 To the best of our knowledge, only one meta-analysis, which was published in 2013, systematically reviewed the effect of hiatal hernia on BE. 11 Nevertheless, since then, numerous individual studies have been published. Furthermore, this previous meta-analysis had a major drawback that patients with endoscopically suspected BE or an irregular Z line as a control group were not explicitly excluded, thereby underestimating the effect of hiatal hernia on BE. Consequently, we have conducted an updated meta-analysis to more comprehensively and precisely determine the clinical significance of hiatal hernia on the risk of BE by combining the most recent evidence.
Methods
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. The PRISMA checklist was shown in Supplemental Material.
Registration
This study was registered on the International Prospective Register of Systematic Reviews (PROSPERO) with a registration number of CRD42022367376.
Literature search
All relevant articles reporting the prevalence of hiatal hernia in patients with and without BE were searched via PubMed, EMBASE, and Cochrane Library databases. The search items were as follows: (‘hiatal hernia’ OR ‘hiatus hernia’) AND (‘Barrett esophagus’ OR ‘Barrett’ OR ‘Barrett metaplasia’ OR ‘Barrett oesophagus’). The last search was conducted on 16 October 2022.
Selection criteria
Inclusion criteria were as follows: (1) observational (case–control or cross-sectional or cohort) studies; (2) BE should be confirmed by endoscopy with biopsies; (3) studies should compare the prevalence of hiatal hernia between participants with and without BE; (4) all participants should be older than 18 years; and (5) studies should be published in English. In case of multiple publications based on the same dataset, one publication with the most updated or inclusive data was given precedence.
Exclusion criteria were as follows: (1) duplicated studies; (2) reviews and meta-analyses; (3) case reports; (4) guidelines, consensus, or reports; (5) editorials, comments, letters, or notes; (6) experimental or animal studies; (7) studies published in the form of abstracts; (8) studies did not explore the association between hiatal hernia and BE; (9) participants included patients with endoscopically suspected BE or an irregular Z line; (10) participants were younger than 18 years old; (11) studies were not published in English; (12) overlapping participants among studies; (13) absence of relevant data; and (14) full texts could not be obtained.
Data extraction
The following data were extracted: first author, publication year, country, study design, diagnostic criteria for BE, diagnostic timing of BE, segment lengths of BE, definition of control groups, number of participants in case and control groups, and prevalence of hiatal hernia in the two groups. In addition, adjusted odds ratios (aORs) with 95% confidence intervals (CIs) and adjusted confounders were also extracted from the studies, in which multivariate regression analyses were performed to further evaluate the association of hiatal hernia with BE. Two investigators (SM and YH) independently extracted the data from the included studies, and any disagreement was resolved by discussion or consultation with a third investigator (XQ).
Study quality assessment
The quality of included case–control and cohort studies was assessed using the Newcastle–Ottawa Scale (NOS), which evaluates the quality of studies in the parts of selection (four points), comparability (two points), and exposure (three points). The maximum NOS score is 9. A score of 0–3, 4–6, and 7–9 represents low, moderate, and high quality, respectively. The quality of included cross-sectional studies was assessed using an 11-item checklist recommended by the Agency for Healthcare Research and Quality (AHRQ). An item is scored ‘1’, if the study being assessed clearly answers the question, which is marked as a ‘yes’; otherwise, it is scored ‘0’. The maximum AHRQ score is 11. A score of 0–3, 4–7, and 8–11 represents low, moderate, and high quality, respectively. Two investigators (SM and YH) independently evaluated the quality of the included studies, and any disagreement was resolved by discussion or consultation with a third investigator (XQ).
Statistical analyses
Meta-analyses were performed via Review Manager software (Version 5.4, Cochrane Collaboration, the Nordic Cochrane Centre, Copenhagen, Denmark), Stata software (Version 12.0, Stata Corp, College Station, TX, USA), and trial sequential analysis (TSA) software (Version 0.9.5.10, CTU, Centre for Clinical Intervention Research, Copenhagen, Denmark). A random-effects model was adopted to calculate the combined results and forest plots were generated for a visual display of outcome of individual studies. 12 Meta-analyses were separately performed among the studies with and without adjustment for confounders. Odds ratios (ORs) with 95% CIs were calculated for the combined estimation of unadjusted data, and aORs with 95% CIs were calculated for the combined estimates of data adjusted for confounders. The Cochrane Q test and I² statistics were conducted to assess the heterogeneity.13,14 I2 > 50% and/or p < 0.1 were considered to have statistically significant heterogeneity. TSA was conducted to assess the reliability and stability of cumulative evidence by minimizing type I error and random error.15,16 The required information size (RIS) was calculated based on a two-sided 5% risk of a type I error, 20% risk of a type II error (power of 80%), and pooled event rates in case and control groups. The trial sequential monitoring boundaries were computed using the O’Brien–Fleming approach. Subgroup analyses were planned according to the definition of controls, study design, publication year, region, sample size, diagnostic criteria for BE, diagnostic timing of BE, and adjustment for confounders to investigate the source of heterogeneity. The interaction between subgroups was assessed. Meta-regression analyses were also grouped based on the aforementioned variables. 17 Leave-one-out sensitivity analyses were employed with the removal of each study once to assess whether any single study could affect the overall result. 14 Egger test was employed to check the publication bias, and p < 0.1 indicated significant publication bias. 18
Results
Study selection
Overall, 2582 studies were searched from the PubMed, EMBASE, and Cochrane Library databases, and one study from hand-searching. Of them, 47 with a total of 131,517 participants were eligible for final review and included in this meta-analysis (Figure 1).

A flowchart of study inclusion.
Study characteristics
The detailed characteristics of included studies are presented in Table 1 and Supplemental Table 1. Among them, 17 studies were case–control studies, 2 were cohort studies, and 28 were cross-sectional studies. They were published between 1985 and 2022. Sixteen studies were performed in Asia,19–34 16 in Europe,35–50 14 in America,51–64 and 1 in Oceania. 65
Summary of study characteristics.

BE, Barrett’s esophagus; CM, columnar metaplasia; GERD, gastroesophageal reflux disease; IM, intestinal metaplasia; LSBE, long-segment BE; NEJ, normal esophagogastric junction; SSBE, short-segment BE; USSBE, ultrashort-segment BE.
Study quality
Among the case–control and cohort studies, 15 and 4 were of moderate and high quality, respectively (Supplemental Table 2). Among the cross-sectional studies, 24 and 4 were of moderate and high quality, respectively (Supplemental Table 3).
Hiatal hernia and any length BE
Based on the unadjusted data from 17 case–control, 2 cohort, and 28 cross-sectional studies, the meta-analysis demonstrated a significantly higher prevalence of hiatal hernia in patients with BE than those without (OR = 3.91, 95% CI = 3.31–4.62, p < 0.001). The heterogeneity was statistically significant (I² = 77%; p < 0.001) (Figure 2). According to TSA, the cumulative Z-curves crossed the conventional test boundary and TSA boundary, and the total sample size surpassed the RIS (n = 1022) as well, indicating that there was enough evidence to conclude a significant association of hiatal hernia with an increased risk of any length BE (Supplemental Figure 1). Results of subgroup analyses were shown in Table 2. Such a statistically significant association between them disappeared in the subgroup analyses of cohort studies (OR = 5.92, 95% CI = 0.58–60.36, p = 0.13), but remained in others. The interaction between subgroups was only significant in the subgroup analysis according to the definition of controls (p = 0.005), but not in others. A high level of heterogeneity was observed between studies in all subgroups. Meta-regression analyses indicated that the definition of controls (p = 0.014) might be a potential contributor to heterogeneity (Supplemental Table 4). Leave-one-out sensitivity analyses showed that no single study influenced the overall result (Supplemental Table 5). Egger test did not show any significant publication bias (p = 0.984).

Forest plots showing the association of hiatal hernia with any length BE based on unadjusted data.
Results of subgroup analyses regarding the association of hiatal hernia with BE in studies unadjusted for confounders.

Note: Bold italic font indicates statistically significant.BE, Barrett’s esophagus; CI, confidence intervals; CM, columnar metaplasia; GERD, gastroesophageal reflux disease; IM, intestinal metaplasia; LSBE, long-segment BE; NEJ, normal esophagogastric junction; OR, odds ratio; SSBE, short-segment BE.
Based on the adjusted data from six case–control and eight cross-sectional studies, the meta-analysis demonstrated a significantly higher prevalence of hiatal hernia in patients with BE than those without (aOR = 3.26, 95% CI = 2.44–4.35, p < 0.001). The heterogeneity was statistically significant (I² = 65%; p < 0.001) (Figure 3). TSA could not be carried out owing to the inability to extract the prevalence rate in multivariate regression models. Results of subgroup analyses were shown in Table 3. Such a statistically significant association between them disappeared in the subgroup analyses of studies performed in America (OR = 1.98, 95% CI = 0.90–4.39, p = 0.09), but remained in others. The interaction between all subgroups was not significant. A high level of heterogeneity was observed between studies in all subgroups. Meta-regression analyses did not identify any source of heterogeneity (Supplemental Table 6). The heterogeneity significantly decreased after omitting the study by Hadi et al. 54 (I² = 0%; p = 0.44), indicating that this study might be a potential contributor to heterogeneity (Supplemental Table 7). Egger test did not show any significant publication bias (p = 0.416).

Forest plots showing the association of hiatal hernia with any length BE based on adjusted data.
Results of subgroup analyses regarding the association of hiatal hernia with BE in studies adjusted for confounders.
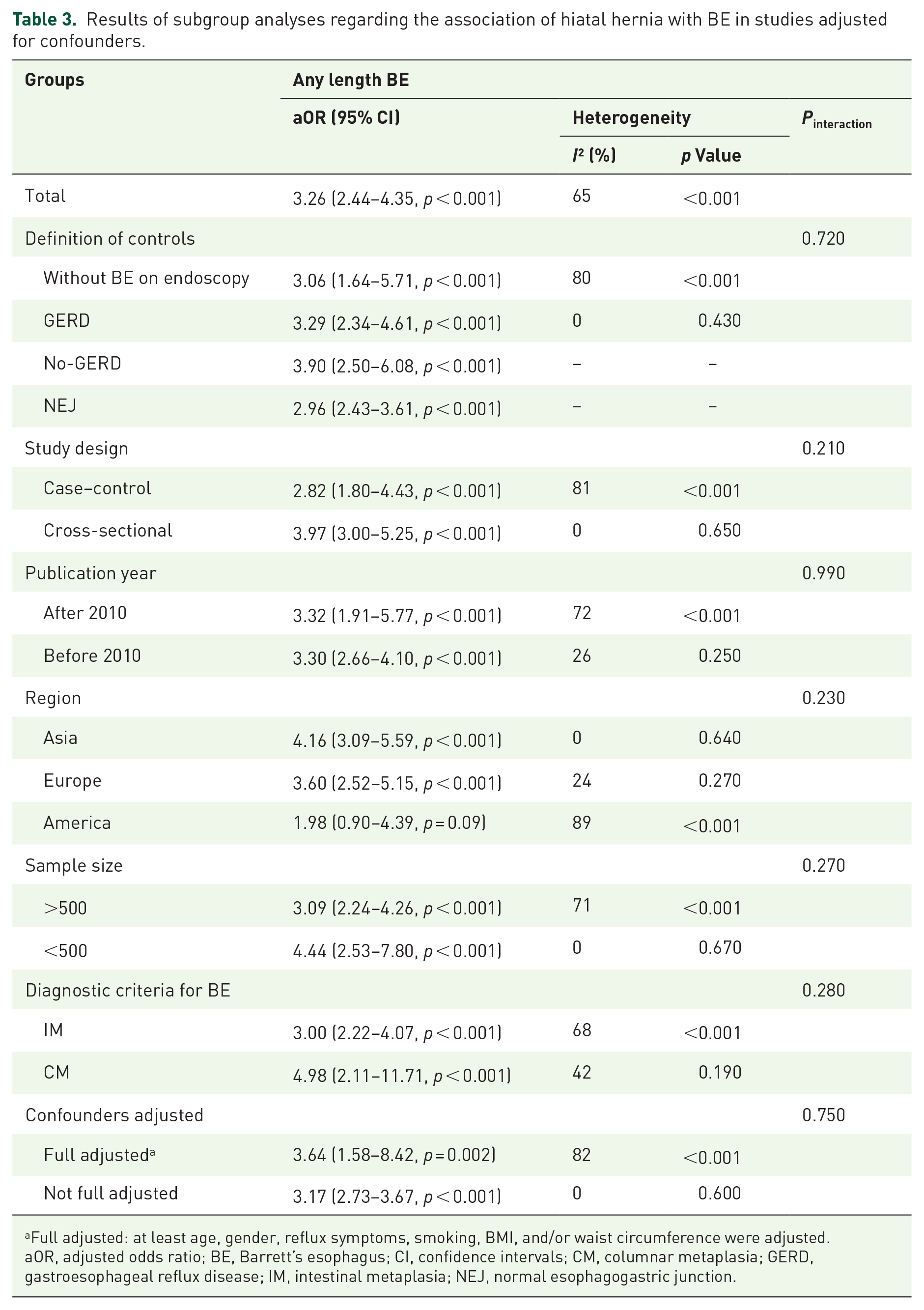
Full adjusted: at least age, gender, reflux symptoms, smoking, BMI, and/or waist circumference were adjusted.
aOR, adjusted odds ratio; BE, Barrett’s esophagus; CI, confidence intervals; CM, columnar metaplasia; GERD, gastroesophageal reflux disease; IM, intestinal metaplasia; NEJ, normal esophagogastric junction.
Hiatal hernia and long-segment BE (LSBE)
Seven studies explored the association between hiatal hernia and LSBE. There was a significantly higher prevalence of hiatal hernia in patients with LSBE than those without BE (OR = 10.01, 95% CI = 4.16–24.06, p < 0.001). The heterogeneity was statistically significant (I² = 78%; p < 0.001) (Figure 4). According to TSA, the cumulative Z-curves crossed the conventional test boundary and TSA boundary, and the total sample size surpassed the RIS (n = 722) as well, indicating that there was enough evidence to conclude a significant association of hiatal hernia with an increased risk of LSBE (Supplemental Figure 2). Such a statistically significant association between them disappeared in the subgroup analyses of cross-sectional studies (OR = 4.39, 95% CI = 0.65–29.80, p = 0.13), those regarding patients without BE on endoscopy as the control group (OR = 9.90, 95% CI = 0.62–158.95, p = 0.11), those published after 2010 (OR = 2.63, 95% CI = 0.16–42.77, p = 0.50), those performed in Europe (OR = 9.90, 95% CI = 0.62–158.95, p = 0.11), and those which included newly diagnosed BE (OR = 1.67, 95% CI = 0.34–8.22, p = 0.53), but remained in others. The interaction between subgroups was statistically significant in the subgroup analyses according to the definition of controls (p < 0.001) and diagnostic timing of BE (p = 0.005), but not in others. Among the subgroup analyses, the heterogeneity remains substantial (Table 2). Meta-regression analyses did not find any source of heterogeneity (Supplemental Table 4). Leave-one-out sensitivity analyses showed that no single study influenced the overall result (Supplemental Table 8). Egger test did not show any significant publication bias (p = 0.210).
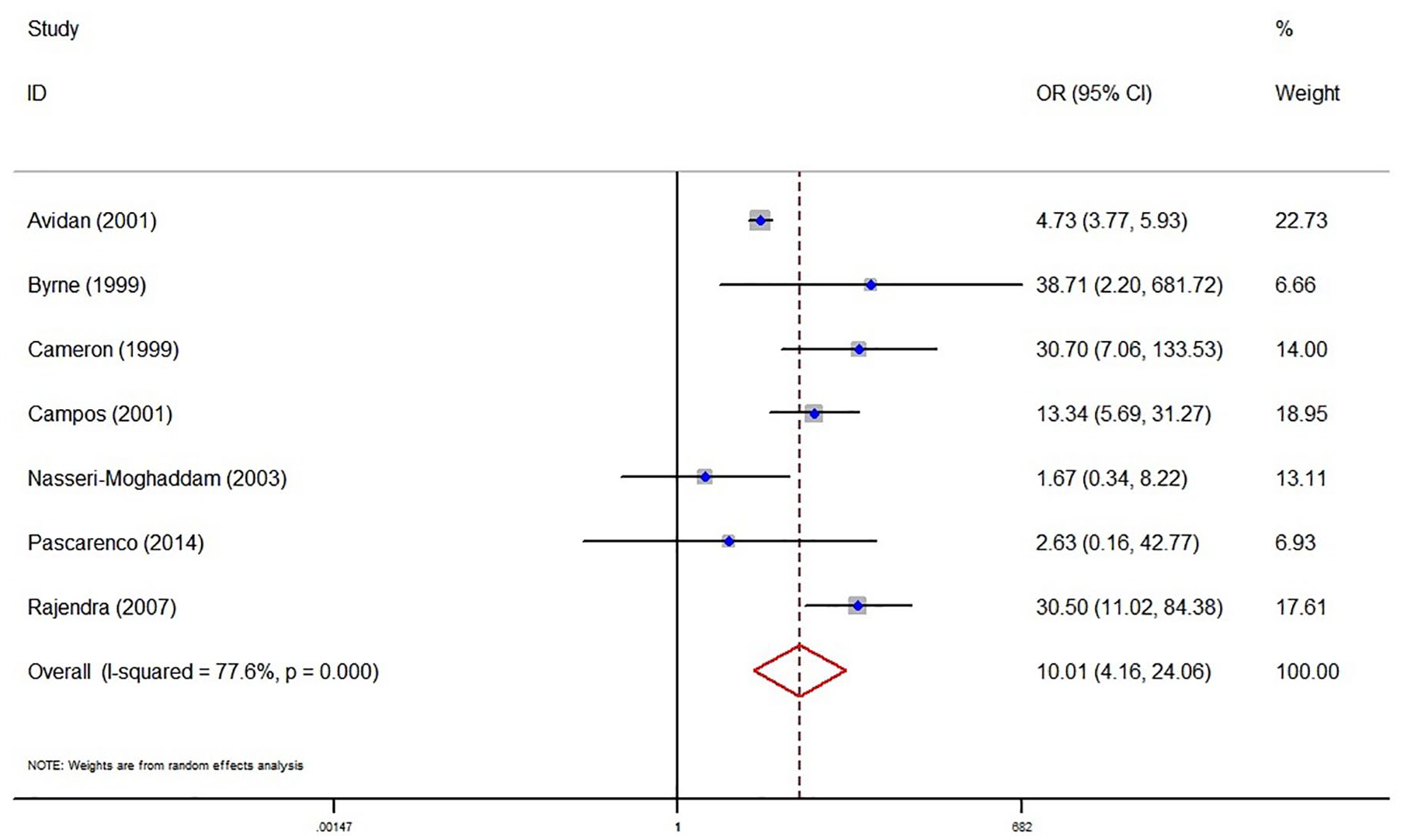
Forest plots showing the association of hiatal hernia with LSBE.
Hiatal hernia and short-segment BE (SSBE)
Seven studies explored the association between hiatal hernia and SSBE. There was a significantly higher prevalence of hiatal hernia in patients with SSBE than those without BE (OR = 2.76, 95% CI = 2.05–3.71, p < 0.001). The heterogeneity was not significant (I² = 30%; p = 0.201) (Figure 5). According to TSA, the cumulative Z-curves crossed the conventional test boundary and TSA boundary, and the total sample size surpassed the RIS (n = 583) as well, indicating that there was enough evidence to conclude a significant association of hiatal hernia with an increased risk of SSBE (Supplemental Figure 3). Such a statistically significant association between them disappeared in the subgroup analyses of studies published after 2010 (OR = 1.84, 95% CI = 0.67–5.06, p = 0.24), those performed in Asia (OR = 2.32, 95% CI = 0.80–6.75, p = 0.12), and those which included newly diagnosed BE (OR = 1.45, 95% CI = 0.71–2.97, p = 0.31), but remained in others. The interaction between subgroups was statistically significant in the subgroup analyses according to the definition of controls (p = 0.008), study design (p = 0.040), and sample size (p = 0.040), but not in others (Table 2). Egger test did not show any significant publication bias (p = 0.261).

Forest plots showing the association of hiatal hernia with SSBE.
Discussion
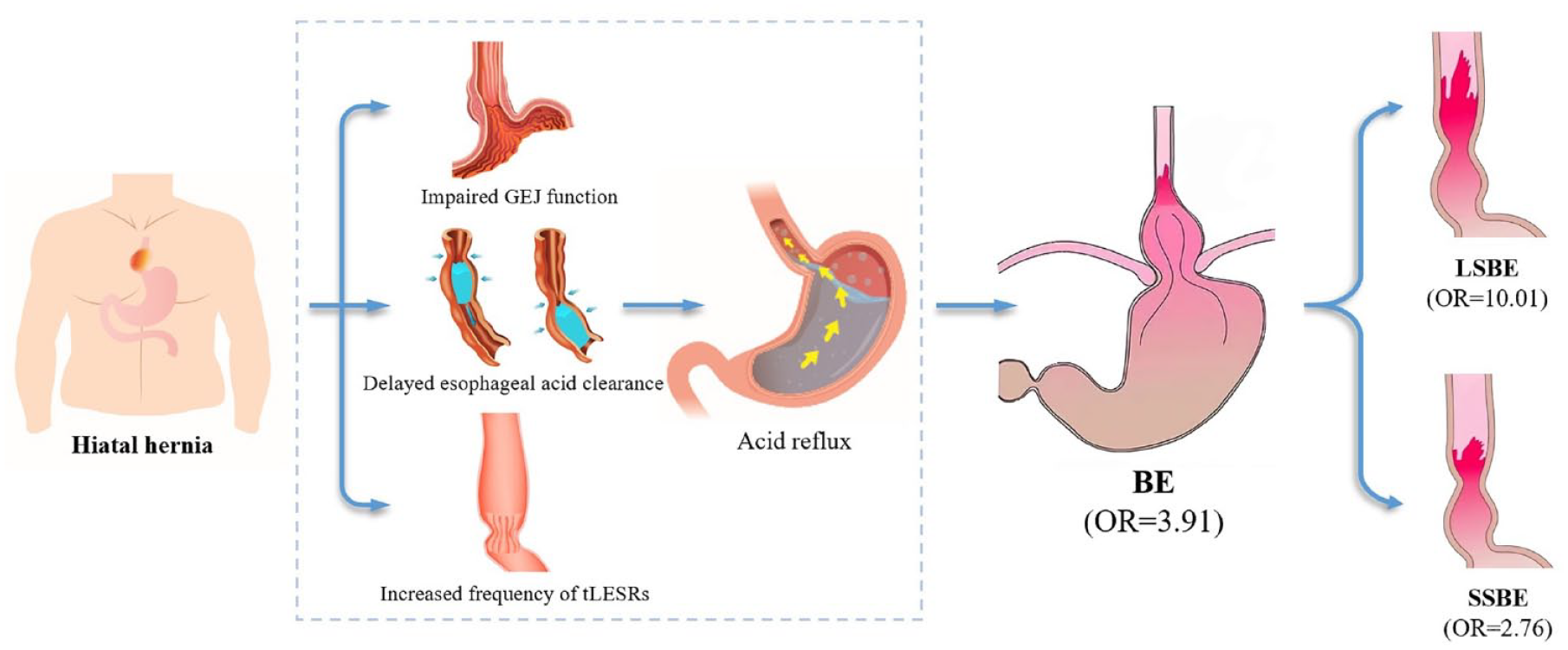
The current practice guidelines do not consider the presence of hiatal hernia as a risk factor for BE1, 5–8. Our meta-analysis comprehensively searched relevant studies to explore the association of hiatal hernia with BE. We found that the presence of hiatal hernia was associated with a 3.91-fold increased risk for any length BE, a 10.01-fold increased risk for LSBE, and a 2.76-fold increased risk for SSBE. This association between hiatal hernia and BE remained significant even after adjustment for potential confounders, suggesting that hiatal hernia should be a significant risk factor for BE.
Currently, only one previous meta-analysis regarding the association between hiatal hernia and BE was published. 11 By comparison, our current meta-analysis had some advantages. First, the final search date was updated to retrieve a more comprehensive collection of eligible studies. Second, TSA was performed by minimizing random errors to evaluate the reliability and conclusiveness of conventional meta-analyses, which has not been conducted in the previous meta-analysis yet. Third, more subgroup analyses were carried out to further explore the association between hiatal hernia and BE according to the seven prespecified baseline subgroups, and tests of interaction were also performed to establish whether the subgroups differed significantly from one another, which has not been performed in the previous meta-analysis yet. Fourth, meta-regression analyses for variables between studies were used to explore the potential causes of heterogeneity in our meta-analysis, but have not been done in the previous meta-analysis yet. Fifth, it is commendable that the selection criteria in our meta-analysis were more rigorous and plausible. Specifically, the participants included in control groups should not have endoscopically suspected BE or an irregular Z line. This consideration was very essential to avert the influences of these potential confounding factors on the reliability of our findings. Such selection criteria have not been employed by the previous meta-analysis, resulting in the inclusion of five ineligible studies.66–70
The pathogenesis of BE is primarily attributed to the impairment of the anti-reflux barrier, which leads to the exposure of esophageal mucosa to gastric and bile acids, resulting in damage to esophageal mucosa.71,72 The causal role of hiatal hernia in an increased risk of BE may be explained by increased acid reflux due to the incompetence of gastroesophageal junction (GEJ), delay of esophageal acid clearance, and increased frequency of transient lower esophageal sphincter relaxations (tLESRs).
The first mechanism should be impaired GEJ function, as follows: (1) Gastroesophageal flap valve (GEFV), an important part of anti-reflux barrier, is a 180° musculomucosal fold formed by the intraluminal extension of the angle of His, which functions as a one-way valve. 73 Loss of the angle of His caused by hiatal hernia can impair the function of GEFV, and then promote reflux. 74 (2) Hiatal hernia may displace the lower esophageal sphincter (LES) proximally, and resting LES pressure can decrease with increasing displacement, thereby promoting reflux. 75 (3) A large hiatal hernia may widen the diaphragmatic hiatus, which weakens the ability of the crural diaphragm to function as an external sphincter, leading to the occurrence of reflux. 76
The second is delayed esophageal acid clearance, as follows: (1) Gastric and bile acids may be trapped in hiatal hernia sac that acts as a reservoir, and then reflux proximally into the esophagus during a swallow-induced LES relaxation.77,78 This sequence can be repeated, resulting in significantly delayed esophageal acid clearance. 76 (2) A large hiatal hernia may decrease the peristaltic wave amplitude and frequency in distal esophagus which is associated with delayed esophageal acid clearance.79,80
The third is tLESRs, a physiological phenomenon during digestion mediated via vagal pathways. 81 The presence of hiatal hernia is associated with a reduced threshold for eliciting tLESRs, increasing the frequency of tLESRs, which finally results in prolongation of esophageal acid exposure. 82
In addition, esophageal injury caused by BE may lead to esophageal shortening and fibrosis, which may increase the size of hiatal hernia and worsen existing anti-reflux dysfunction,83,84 and then further aggravates reflux.
We also found a more significant association of hiatal hernia with LSBE compared with SSBE. It seems obvious that LSBE has a greater esophageal acid exposure than SSBE.85,86 This phenomenon may be attributed to the difference in their pathogenesis where LSBE may have a significantly lower LES pressure and a worse function of esophageal peristalsis primarily caused by hiatal hernia than SSBE.87–89 Hence, LSBE may be more affected by acid reflux attributed to hiatal hernia and demonstrate a stronger association with hiatal hernia.
Our meta-analysis had several limitations. First, a majority of the included studies were retrospective, which inevitably leads to selection bias and recall bias, and a cause–effect relationship between hiatal hernia and BE could not be established due to the inherent weakness of retrospective study design. Second, the heterogeneity among studies regarding the association of hiatal hernia with LSBE was significant, in spite of leave-one-out sensitivity analyses and meta-regression analyses. Third, only some of included studies adjusted the confounders in multivariate regression analyses, and the confounders adjusted were inconsistent among them. Fourth, the association of hiatal hernia with BE was not the primary objective in most of the included studies. The diagnosis of hiatal hernia was based on endoscopy only, which may result in missed diagnoses of some small hiatal hernia or misdiagnosis of hiatal hernia caused by episodes of retching during endoscopy, 10 thereby underestimating or overestimating the effect of hiatal hernia on BE, respectively. Fifth, the information regarding the size of hiatal hernia was limited. In detail, only two studies reported the length of hiatal hernia,58,62 and only three studies reported the number of different sized hiatal hernia grouped by different classification criteria in patients with and without BE.51,55,56 Therefore, the effect of the size of hiatal hernia on BE could not be explored by subgroup analyses. Sixth, the absence of detailed information on the type of hiatal hernia prevented from further analysis regarding the association between subtypes of hiatal hernia and BE. Seventh, our meta-regression analyses indicated that the definition of control group might be a potential contributor to heterogeneity. Specifically, as shown in Table 2, our subgroup analyses demonstrated that the association of hiatal hernia with BE was weaker in the control group of GERD than the control group of non-GERD. These findings suggested that the association of hiatal hernia with BE might be dependent upon the presence of GERD. Indeed, a recent meta-analysis by Eusebi et al. 90 also indicated that hiatal hernia should be the strongest risk factor for the presence of BE in patients with GERD. Therefore, it is unclear about whether hiatal hernia is indirectly associated with BE by increasing the risk of GERD or directly involved in the development of BE in GERD patients.
Conclusion
The current evidence supports a close relationship between hiatal hernia and an increased risk of BE, which remains after adjusting for confounders. Additionally, hiatal hernia seems to correlate more strongly with LSBE compared with SSBE. More large-scale prospective cohort studies are required to confirm our findings in future.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231219234 – Supplemental material for Association between hiatal hernia and Barrett’s esophagus: an updated meta-analysis with trial sequential analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848231219234 for Association between hiatal hernia and Barrett’s esophagus: an updated meta-analysis with trial sequential analysis by Shaoze Ma, Zhenhua Tong, Yong He, Yiyan Zhang, Xiaozhong Guo and Xingshun Qi in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
