Abstract
Background:
Risk factors for colectomy following an episode of acute severe ulcerative colitis (ASUC) have been well studied, but data examining the early complications following an episode is limited.
Objectives:
We aimed to evaluate the prevalence and risk factors for medical and surgical complications within 90 days of an ASUC admission and determine if a high-intensity induction infliximab dose is associated with these complications.
Design:
Retrospective analysis.
Methods:
We conducted a retrospective study of ASUC admissions between January 2015 and July 2021 at a tertiary hospital. The primary outcome was the prevalence of total, medical and surgical complications within 90 days following an ASUC admission. Multivariate linear regression analysis assessed for factors associated with the prevalence of complications.
Results:
A total of 150 patients had 186 hospital admissions for ASUC. In total, 101/186 (54.3%) admissions required rescue medical therapy. Standard infliximab induction occurred in 65/100 admissions, accelerated infliximab induction in 35/100 and cyclosporine in 1/100 of admissions. In total, 117 complications, including 74/117 (63.2%) medical and 43/117 (36.8%) surgical complications, arose. Low serum albumin was independently associated with a higher incidence of total [β = −0.08 (95% confidence interval (CI): −0.15, −0.01), p = 0.03] and surgical complications [β = −0.1 (95% CI: −0.18, −0.001), p = 0.047], while an increased age was associated with increased incidence of surgical complications [β = 0.06 (95% CI: 0.01, 0.12), p = 0.02]. A higher Charlson score was associated with increased medical complications [β = 0.12 (95% CI: 0.01, 0.24), p = 0.03]. Infliximab induction dose intensity was not associated with an increased incidence of any complications.
Conclusion:
Early complications following an ASUC admission is prevalent although the majority are not serious. Risk factors associated with complications include low serum albumin, older age and a higher comorbidity score. Induction infliximab dose intensity, however, is not a risk factor.
Keywords
Introduction
Acute severe ulcerative colitis (ASUC) is a medical emergency, most frequently defined by the fulfilment of the Truelove and Witt’s Criteria. 1 Up to 20% of UC patients have a late onset diagnosis after 60 years of age, a figure that is likely to increase with the ageing population. 2 Mortality rates from acute severe colitis are four times higher in patients aged over 50, 3 and morbidity is also higher in the elderly.4,5 In recent years, there has been a shift toward using intensified infliximab induction dosing (IIID) regimens for treating ASUC, with some studies suggesting a possible short-term benefit in decreasing colectomy rates, while other studies have not shown any meaningful improvement.6,7 In clinical practice, some physicians express anxiety when considering IIID in the older, more ‘vulnerable’ population, who may experience an increased incidence of complications, especially following a colectomy.7–12 Risk factors that predict for a colectomy from ASUC have been well explored, but there is a paucity of data examining the predictors for early surgical and particularly medical complications following a hospital admission for ASUC.8,13
Multiple risk factors have been determined to increase post-colectomy morbidity for outpatients with severe ulcerative colitis. These risk factors include an increased age,3,4,8,14 high comorbidity level,5,15,16 low serum albumin,17,18 cumulative corticosteroid dose, 19 preoperative infliximab use in those requiring a colectomy20,21 and intensity of preadmission immunosuppression.15,22,23 It is unclear, however, whether these risk factors also apply to the same extent as patients hospitalized with ASUC, who are considerably more unwell and for ASUC patients that do not undergo a colectomy, that have only received IIID. Moreover, the body of evidence for the impact of IIID on morbidity outcomes is inconclusive.24,25 While some studies have shown that infliximab may confer an increased risk of post-operative complications,20,21 other more recent studies have not confirmed this,24–26 and there is a major gap in literature on the impact of IIID on the incidence of acute medical complications following an admission for ASUC. To that end, we aimed to study the prevalence of medical and surgical complications up to 90 days following a hospital admission for ASUC and to identify the risk factors associated with these complications, including the use of IIID.
Methods
Study design and patients
We performed a single centre retrospective study of all patients admitted for ASUC between 1 January 2015 and 31 July 2021 at Fiona Stanley Hospital, a major tertiary inflammatory bowel disease (IBD) referral centre, which services one million patients across the southern portion of metropolitan Perth and greater Western Australia. Eligible patients were identified by ICD-10-AM tenth edition codes for ‘ulcerative (chronic) pancolitis’, ‘ulcerative (chronic) proctitis’, ‘ulcerative (chronic) proctosigmoiditis’, ‘left sided colitis’, ‘other ulcerative colitis’ and ‘ulcerative colitis, unspecified’, 27 on their discharge summaries. Patient data were then extracted by chart review of hospital electronic medical records. The gastroenterology fellow and resident medical officer, AL and MC, respectively performed the chart review. All data was reviewed by the senior author LT, for accuracy and discrepancies. Patients were included if they (a) had a confirmed endoscopic and histopathological diagnosis of ulcerative colitis; (b) fulfilled a diagnosis of ASUC based on the Truelove and Witt’s criteria; (c) required an admission at the tertiary centre for ASUC and (d) had at least 3 months of clinical follow-up data available after hospital discharge from clinic outpatient records.
The Fiona Stanley Hospital Ethics review board assessed the study and low-risk ethics approval was obtained (no: 39543). The study was performed and reported in concordance with the STROBE guidelines. Patients’ identifying information were separated from the clinical details in the data set and analysed in a de-identified manner with a robust process of keeping identifying information and research data separate and secure. Thus, patient consent was not required as per ethics approval.
Clinical practice
The standard clinical practice for patients presenting with ASUC was to commence 100 mg QID of hydrocortisone and have their clinical response assessed at day 3 using the Oxford criteria, 28 before receiving rescue therapy with infliximab, if they qualified. In the study cohort, some patients did not receive any hydrocortisone as they had already been on high doses of oral corticosteroids as an outpatient (e.g. 40 mg for >2 weeks prescribed by their general practitioner) prior to their admission, and it was felt that another 3 days of IV hydrocortisone was futile. Other patients re-presented with ASUC after a recent admission with ASUC when they had received at least 3 days of hydrocortisone, thus went straight on to receive rescue therapy with infliximab.
All patients also had a flexible sigmoidoscopy performed within 72 h of admission to determine the severity of inflammation and to take biopsies for exclusion of cytomegalovirus infection on histology and immunohistochemistry. Stools were also collected on admission to exclude any concomitant gastrointestinal (GI) infection. Rescue medical therapy commenced at day 3 to 5. The choice of rescue therapy, including the decision to administer a standard (5 mg/kg dose at week 0, 2 and 6), or an accelerated infliximab regimen (three doses over a shorter interval than 6 weeks and/or >5 mg/kg per dose, or both), was at the discretion of the treating gastroenterologist. ASUC patients were routinely reviewed at the acute IBD clinic 2 weeks following discharge and at completion of their infliximab induction (if they required rescue therapy) to ensure thiopurines were commenced and compliance maintained.
In Australia prior to 2022, all patients with ulcerative colitis were required to complete a 6-week prednisolone tapering course commencing from 40 mg and have commenced at least 3 months of a 5-aminosalicylate therapy and 3 months of thiopurine therapy, unless limited by contraindications or intolerances, to meet eligibility criteria for accessing long-term biologic therapy on the Pharmaceutical Benefits Scheme. For this reason, immunomodulator-naïve patients were commenced on thiopurine therapy shortly after presentation with ASUC, which was also the standard clinical practice to minimize immunogenicity to infliximab. Patients on corticosteroids, thiopurines and infliximab all received cotrimoxazole for prophylaxis against pneumocystis jiroveci pneumonia. Following induction therapy with infliximab, patients were continued on this biologic at a dose and frequency at the discretion of the treating gastroenterologist.
Outcomes
The primary outcome measured was the prevalence of total, medical and surgical complications within 90 days following a hospital admission for ASUC expressed as a proportion of total admissions for ASUC in the study period. An interval of 90 days was chosen to capture the period just prior to when patients would be due to receive their first maintenance dose of infliximab. On average, those who received infliximab rescue therapy would receive three doses during this period. The secondary outcomes evaluated were the clinical factors associated with an increased incidence of medical or surgical complications expressed as an odds ratio and, the time to colectomy in weeks, in those who underwent accelerated (IIID), compared to standard infliximab induction.
Definitions
The Charlson comorbidity index (CCI) score was calculated for each patient as a measure of their comorbidity level. The index incorporates 16 predefined comorbidities and predicts the 10-year survival in patients with multiple comorbidities. 29 The total hydrocortisone dose was defined as the total intravenous hydrocortisone dose received by a patient during their ASUC admission in milligrams (mg). Those who went straight on to receiving rescue therapy with infliximab as previously discussed would therefore have a total hydrocortisone dose in that admission of 0 mg. The nadir serum albumin during the admission was used as the measure for serum albumin, expressed as g/L. An accelerated infliximab induction (IIID) regimen was defined as any dosing schedule greater than the standard induction regimen of 5 mg/kg given at week 0, 2 and 6. To standardize the overall dose administered for infliximab induction, accounting for the patient’s body weight and duration over which it was administered, we calculated the ‘infliximab dose intensity’. This figure was calculated as the total infliximab induction dose within a 6-week period, divided by the number of induction weeks over which it was administered, expressed as mg/week. Thus, patients who only received hydrocortisone and not rescue infliximab therapy, would have an infliximab dose intensity value of 0 mg/week.
A surgical complication was defined as any complication relating to a colectomy for ASUC, including any complications requiring pharmacological treatment, blood transfusion, parenteral nutrition, surgical or radiological intervention. The Clavien–Dindo classification, a validated and reproducible classification system for surgical complications (Table 3), was used to grade the severity of surgical complications. 30
A medical complication was defined as any complication that occurred which was not directly related to surgery. These complications included respiratory complications, thromboembolic events (including myocardial infarction), infections unrelated to surgery, neurologic and/or psychiatric complications, drug reactions related to any of the medications administered during the ASUC admission and death.
The use of preadmission immunosuppression was defined as the active use of any infliximab, adalimumab, vedolizumab, tofacitinib, tacrolimus, mercaptopurine, azathioprine, methotrexate and/or combination of biologic and immunomodulators at the time of admission without accounting for dose or duration. Likewise, preadmission corticosteroid use included the use of any budesonide (Cortiment) of 9mg or prednisolone of at least 20 mg at the time of admission, without accounting for the duration of therapy prior to admission, as this information was not always available.
Statistical analysis
Baseline characteristics are summarized using descriptive statistics with non-normally distributed variables expressed as a median and corresponding interquartile range (IQR). Normally distributed variables are expressed as a mean (±standard deviation). The prevalence of complications was expressed as a percentage of admissions involved. Linear univariate and multivariate regression analyses were conducted to determine the associations of risk factors of interest, with the total number of medical and surgical complications. Chi-square/Fisher’s exact test was used to determine any significant associations between categorical variables on sub-analyses.
Time to colectomy was first conducted using the Kaplan–Meir survival analysis and comparisons between groups (standard dose versus IIID) using the log-rank test. Patients were censored at the date of colectomy, date of last follow-up, date of death or the 31st of July 2021 whichever came earlier. The multivariate model was created from known risk factors for complications determined a priori (low serum albumin, preadmission use of corticosteroids/immunosuppressants and/or biologics, age, CCI and the variable of interest: infliximab dose intensity) or any factors with a p-value < 0.2 on the univariate analysis. The backward stepwise regression method was used to determine significant covariates that remained in the model. Covariates were removed stepwise if the p value was >0.1. A two-sided p value of ⩽0.05 was considered statistically significant. The results are reported with β regression coefficients and their 95% confidence interval [95% confidence interval (CI)]. Calculations were performed using SPSS version 20 software.
Results
Baseline characteristics
A total of 152 patients had 188 hospital admissions for ASUC during the study period. Two patients were excluded from the study due to incomplete data, resulting in a final cohort of 150 patients that had 186 hospital admissions for ASUC. The baseline characteristics of each patient admission for ASUC is presented in Table 1. The median duration of disease at each admission was less than one year (0 years, IQR [0–1]), indicating many presented with ASUC as their first diagnosis of UC. The median duration of follow-up per patient was 94.9 weeks [IQR: 24.9–195.2].
Baseline clinical and biological characteristics of patient admissions for ASUC.

AZT in those who had colectomy within 90 days from admission.
ASUC, acute severe ulcerative colitis; AZT, azathioprine; CCI, Charlson comorbidity index; DVT, deep vein thrombosis; 6MP, 6-mercaptopurine.
The median duration of intravenous hydrocortisone was 5 (IQR [3.5–6.3]) days and the median total hydrocortisone dose during the admission was 1650 mg (IQR [1100–2200]. In 24 (12.9%) admissions, fewer than 3 days intravenous hydrocortisone were administered in total. In 12 (6.5%) admissions, patients did not receive IV hydrocortisone for reasons explained above under section 2.2 ‘clinical practice’. In 82 admissions (44.1%), patients presented with a corticosteroid dose of ⩾20 mg on admission. Rescue therapy with infliximab or cyclosporine was required in 101 of 186 admissions (54.3%). A standard dose of infliximab induction was prescribed in 65/186 (34.9%) of admissions, while 35/186 admissions (18.8%) required IIID. The median dose intensity was 225 mg/week (IQR [200–250]) for those treated with standard infliximab induction and 400 mg/week (IQR [250–525]) for those who had IIID, p < 0.0001. Thirty-five of 150 (23.3%) patients with ASUC required a colectomy within 90 days of admission. Of these 35 patients, the median duration to colectomy was 10 days (IQR [6–14]). Twenty of 35 (57.1%) of colectomies were performed laparoscopically, whereas 15/35 (42.9%) of colectomies were open surgeries. The median duration of hospital stay for an admission with ASUC was 7 days (IQR [5–10]).
Prevalence of complications following an admission for acute severe colitis
Of 186 admissions, 93 admissions were associated with a medical or surgical complication within 90 days of the admission date, resulting in an overall complication rate of 50%. Some admissions encountered multiple complications, with a total of 117 complications arising from 186 admissions. Of the 117 complications, 74 (63.2%) were classified as medical complications and 43 (36.3%) were surgical complications (Table 2). The median duration of follow-up of the 150 included patients in the study was 1.8 years (IQR [0.5–3.7]).
Breakdown of total, medical and surgical complications within 90 days of admission for acute severe colitis.
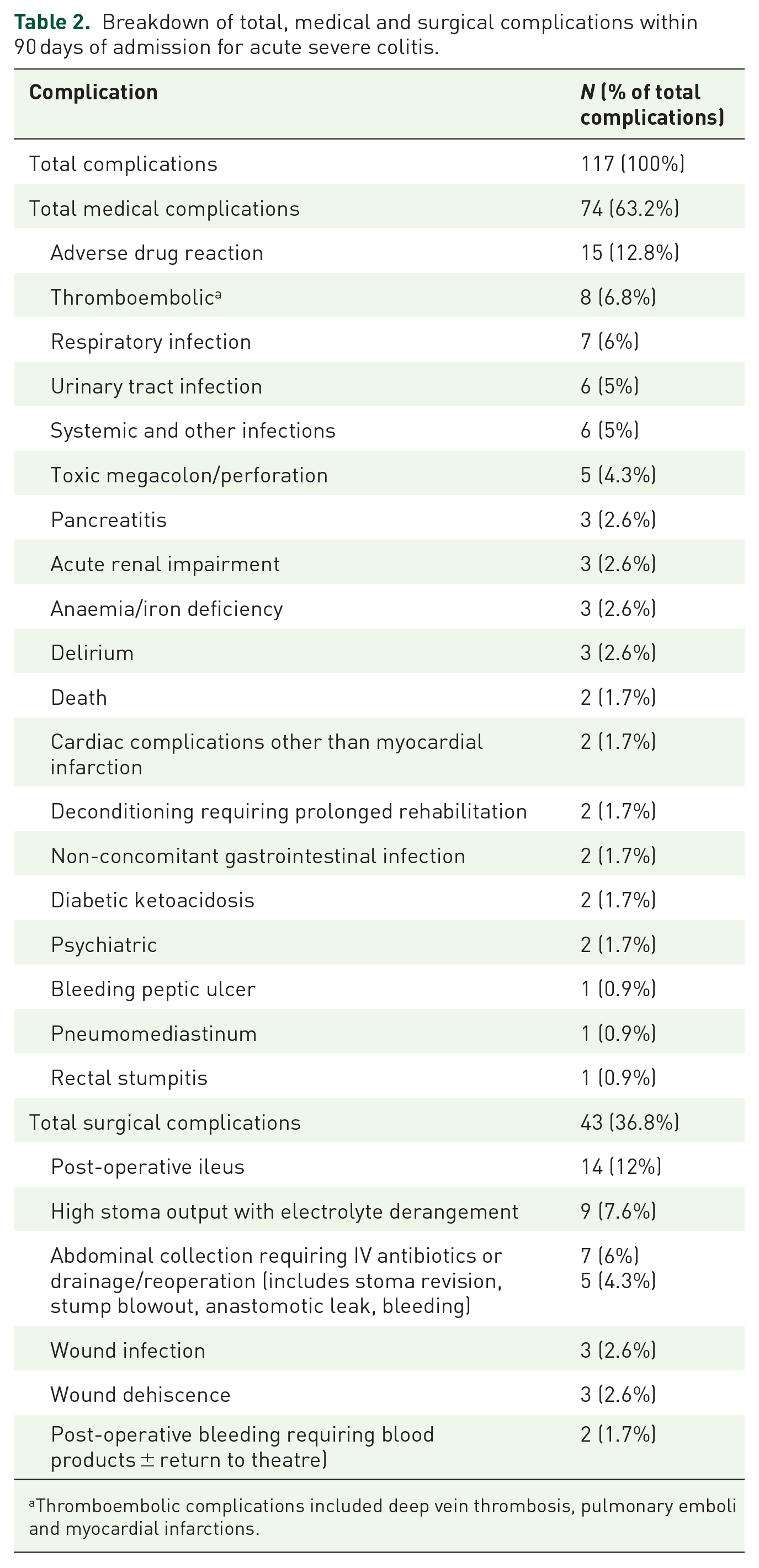
Thromboembolic complications included deep vein thrombosis, pulmonary emboli and myocardial infarctions.
The most frequently documented medical complication was an adverse drug reaction to an administered medication during the ASUC admission (15/117, 12.8%), most commonly from thiopurine exposure causing nausea and/or vomiting followed by rash. The second most frequently reported medical complication was myocardial infarction and thromboembolism (8/117, 6.8%). DVT prophylaxis was administered in most admissions (179, 96.2%). When stratified by severity, 16/117 (13.7%) medical complications were considered serious or life-threatening, including myocardial infarction and thromboembolism in 8/117 (6.8%), toxic megacolon and perforation in 5/117 (4.3%), pneumomediastinum in 1/117 (0.9%) and death in 2/117 (1.7%). One death was due to pulmonary embolism and a second death was from respiratory failure due to pre-existing advanced pulmonary fibrosis. Twenty of 117 (17%) of complications were non-surgical infections including respiratory infections (7/117, 6%), urinary tract infections (6/117, 5%), GI infections acquired after the ASUC episode (2/117, 1.7%) and other systemic infections such as thrombophlebitis with cellulitis (6/117, 5%).
Surgical complications occurred in 19 of 35 patients who underwent a colectomy within 90 days of ASUC admission, giving a total 90-day surgical complication rate of 54.3%. Forty-three surgical complications arose from the 19 patients within 90 days of admission for ASUC. The most common surgical complication was post-operative ileus (14/43, 32.6%) followed by high stoma output with electrolyte disturbance (9/43, 20.9%). Eleven of 43 (25.6%) of surgical complications were post-operative wound infections. According to the Clavien–Dindo classification, 30 22/43 (51.2%) were grade I, 9/43 (20.9%) were grade II, 2/43 (4.7%) were grade IIIa, 9/43 (20.9%) were grade IIIb and only 1/43 (2.3%) was grade IVa severity (Table 3). There were no grade IVb or V surgical complications. There was no difference in the proportion of colectomy patients that experienced any surgical complications who underwent laparoscopic (11/20, 55%) versus open (9/15, 60%) surgery, p = 0.52.
Ninety-day surgical complications classified according to Clavien–Dindo classification.

Source: Clavien et al. 30
ICU, intensive care unit.
Risk factors for 90-day complications following an admission for ASUC
In the univariate analysis, no single covariate was associated with an increased incidence of 90-day total, medical or surgical complications following an admission for ASUC; however, there was a non- statistically significant trend for a higher CCI being associated with a higher incidence of medical complications [β = 0.11 (95% CI: −0.01, 0.22), p = 0.06] (Table 4). The time from admission to colectomy was not associated with an increased risk of total, medical or surgical complications on the univariate analysis (all p > 0.05).
Univariate and multivariate linear regression analyses for factors associated with total, medical and surgical complications.

p Value ⩽0.05 denotes statistical significance; multivariate linear regression using eight covariates [age, CCI, infliximab induction intensity (mg/week), total hydrocortisone dose (mg), serum albumin (g/L), preadmission CS, preadmission IM or biologic therapy, preadmission IM and biologic therapy].
Preadmission CS use was defined as taking budesonide (Cortiment) or prednisolone ⩾20 mg at time of admission (yes/no).
Preadmission IM use was defined as the use of any mercaptopurine, azathioprine, tacrolimus or methotrexate at time of admission.
Preadmission biologic use was defined as the use of any biologic therapy at time of admission including infliximab, adalimumab, vedolizumab, ustekinumab, golimumab and tofacitinib.
β, regression coefficient; CCI, Charlson comorbidity index; CI, confidence interval; CS, corticosteroids; IM, immunomodulator.
The final multivariate model included the following co-variates: age, the CCI score, infliximab induction intensity (mg/week), total hydrocortisone dose in (mg), serum albumin (g/L), use of any of the following: preadmission corticosteroids, preadmission immunomodulator or biologic/advanced therapy or the combination of preadmission immunomodulator AND a biologic/advanced therapy.
In the multivariate regression analysis, a significant inverse relationship between serum albumin and frequency of 90-day total complications [β = −0.08 (95% CI: −0.15, −0.01), p = 0.03] was found. An increased age [β = 0.06 (95% CI: 0.01, 0.12), p = 0.02] and low serum albumin [β = −0.1 (95% CI: −0.18, −0.001), p = 0.047] were also associated with a greater frequency of 90-day surgical complications. Interestingly the CCI was found to be inversely associated with the frequency of surgical complications [β = −0.62 (95% CI: −1.13, −0.11), p = 0.02]. CCI was the only factor that positively correlated with an increased incidence of 90-day medical complications [β = 0.12 (95% CI: 0.01, 0.24), p = 0.03]. Notably, induction infliximab dose intensity, total hydrocortisone dose and preadmission immunosuppression were not associated with an increased incidence of total, medical or surgical complications after 90 days of hospital admission for ASUC.
Time to colectomy: Standard induction versus accelerated induction of infliximab
Figure 1 shows the cumulative colectomy-free survival probability over time of the 52 patients who received standard infliximab induction therapy compared to the 32 patients who received accelerated induction therapy from their index ASUC admissions. Patients who received standard induction therapy had a mean colectomy-free survival estimate of 282.9 weeks compared to accelerated induction patients who had a mean colectomy free survival estimate of 188.1 weeks (p = 0.016). Table 5 summarizes the comparison in clinical characteristics between the standard and accelerated induction groups. There were no clinical factors that differentiated either group (p > 0.05 for all factors).

Kaplan–Meir curves for colectomy free survival in weeks, (a) in patients undergoing standard and (b) accelerated (intensified) IIID. Differences in colectomy free survival measured with log rank test and two-sided p value.
Patient characteristics of those given standard and accelerated induction doses of infliximab.

GI, gastrointestinal; IM, immunomodulator; IQR, interquartile range.
Sensitivity analysis
There was no difference in the proportion of patients with a concomitant GI infection that received standard infliximab induction rescue therapy [7/52 (13.5%); six with Clostridium difficile, one with CMV infection] compared to those with a concomitant GI infection that received accelerated infliximab induction [n = 6/32 (18.8%); two with C. difficile, two with CMV and one with Campylobacter infection], p = 0.36. Of the 150 patients, five patients (3%) who had a colectomy also had a concomitant GI infection (three with C. difficile and two with CMV). When patients with a concomitant GI infection were excluded from the time to colectomy analysis, there was still a numerically longer colectomy free survival in those who received standard induction therapy compared to patients who received accelerated induction (mean colectomy free survival in weeks = 281.7 versus 169.6, p = 0.07), but the difference did not reach statistical significance.
Discussion
The present study shows that early complications following a hospital admission for ASUC is prevalent with 50% of admissions involving a medical or surgical complication. The rate of serious complications in our study however was low with 13.7% of complications considered a serious medical complication and 27.9% of surgical complications classified as Clavien–Dindo grade III or higher. There were two deaths (1.7%) during the 90-day follow-up period. To our knowledge, our study is the first to (i) thoroughly explore the frequency of medical as well as surgical complications, (ii) characterize the complications following a hospitalization for ASUC and (iii) assess the impact of IIID, among other risk factors, on the frequency of complications. Our 90-day colectomy rate of 23% is comparable to reported rates in the literature.31,32
We found age, comorbidity level and serum albumin to be significant risk factors for the incidence of complications. Importantly, the total hydrocortisone dose, induction infliximab dose intensity and preadmission immunosuppression use were not associated with an increased frequency of 90-day complications. Several studies have shown an inverse relationship between serum albumin and clinical outcomes in patients with ulcerative colitis. 17 Albumin is a robust marker of inflammatory burden with evidence that when adjusted for CRP is a good predictor of clinical severity in ulcerative colitis. 33 Lower albumin levels are associated with infective complications, 34 anastomotic leaks 35 and increased duration of hospital stay. 36 Our results affirm that serum albumin is an important risk factor for early total and surgical complications following an admission for ASUC. Thus, prolonged futile medical therapy leading to severe protein losing states should be avoided.
The emerging concept of biologic age in IBD, which is largely determined by comorbidities and frailty, is now a recognized predictor of adverse outcomes in IBD.4,37 Multicentre observational studies have shown that elderly IBD patients are more likely to suffer treatment failure, serious adverse events, serious infections, malignancy and death.14,37 We found that older age was a significant risk factor for experiencing a 90-day surgical complication following a colectomy for ASUC, but was not a significant risk factor for experiencing a medical complication during this period. Clinicians may be apprehensive about immunosuppression, particularly with high doses of infliximab in older patients for fear of infective complications. Our results show that with increasing age, the increased risk of surgical complications seems to outweigh the risk of any immediate medical complications arising from the use of short-term high-intensity infliximab dosing, and that the latter should be given rather than avoided in elderly patients, assuming they have no other contra-indications.
There are currently no IBD-specific clinical scores to describe frailty. 38 The CCI has been widely used in epidemiological studies to quantify comorbidity level, but has only been recently studied in IBD patients.29,39 Recognizing this limitation, we proceeded to use the CCI instead of the Rockwood Frailty Index as domains from the latter index (consisting of physical activity, independence with activities of daily living and severity of medical comorbidities) 40 were not possible to determine in a retrospective analysis. Our study observed a significant positive association between a higher CCI and the frequency of medical complications arising within 90 days of admission [β = 0.12 (95%: 0.01, 0.24), p = 0.03]. On the other hand, we found a significant inverse relationship between CCI and surgical complications [β = −0.62 (95% CI: −1.13, −0.11, p = 0.02] on the multivariate analysis. A possible explanation for this finding is a potentially more meticulous pre-operative workup and intra-operative diligence in the patients with a higher CCI, given their higher surgical and anaesthetic risks. Such selection biases intrinsic to retrospective analyses may limit the interpretation of our findings.
The risk of serious infections is a concern for IBD patients receiving immunosuppressive therapy. Ferrante et al. 19 found that a moderate to high dose of corticosteroids was a predictor of short-term post-operative infectious complications in patients with ulcerative colitis albeit not specific to ASUC. Bregnbak et al. 11 found that the use of infliximab was not associated an increased risk of 30-day post-operative complications in ulcerative colitis, but corticosteroid use was. Minimization of corticosteroid exposure before major abdominal surgery has thus been an established priority in IBD management. We did not find any association between total admission hydrocortisone dose, the use of preadmission immunosuppression or preadmission corticosteroid with the incidence of medical and/or surgical complication rates.
Although most patients received at least 3 days of intravenous hydrocortisone, there were 24 (12.9%) admissions in which less than 3 days of intravenous hydrocortisone was given for reasons including: direct readmission for colectomy due to prior failure of biologic therapy, early readmission after recent failure of high-dose IV corticosteroid therapy or a prolonged high-dose oral corticosteroid therapy prescribed by GPs as an outpatient. To standardize the heterogeneity in administered IV hydrocortisone dose, the total dose given during the admission was analysed as a linear covariate. Our analysis of the impact of corticosteroids on the prevalence of complications was limited by the fact we could not account for the total corticosteroid burden as a single covariate. Given the limitations of a retrospective study, the total duration and therefore total oral corticosteroid dose taken, was not always recorded, if at all. Thus, oral corticosteroid doses on admission of ⩾20 mg (expressed as a binary (yes/no) covariate) and total IV hydrocortisone dose administered (expressed as a linear covariate) were analysed separately. The impact of total corticosteroid exposure on the risk of developing medical and/or surgical complications may have been significantly underestimated as a result.
In recent years, there has been increased use of high-intensity infliximab induction as rescue therapy for ASUC, although the true benefit in decreasing early colectomy rates compared to a standard induction is still to be determined. 6 Moreover, there is apprehension regarding the potential increased risk of serious infections, particularly in older patients, when giving IIID. Govani et al., 24 reviewed outcomes from 66 patients with ASUC, of which 50% received accelerated induction and detected no difference in the 30-day post-operative readmission or surgical complication rates between accelerated and single-dose rescue induction. However the study was small and primarily examined the risk of colectomy rather than the direct complications from IIID. Our results showed that a high-intensity infliximab induction was not independently associated with an increased risk of total, medical or surgical complications at 90 days. The lack of signal, however, may have been due to the relatively small sample size of our cohort, which may have been underpowered to detect any differences.
The rate of emergent colectomy for ASUC has declined over recent years, with a parallel increase in the rate of elective surgery for medically refractory disease, 41 a testament to the success of timely and effective medical therapy. We found that patients who received IIID had a shorter colectomy free survival compared to their counterparts who received standard infliximab induction, despite being well matched in all relevant clinical characteristics (p = 0.016). Our findings echo those of Shah et al., 42 who found that ASUC patients who underwent accelerated dosing progressed more quickly to colectomy than patients who had a standard induction. These findings may be due to treatment biases where sicker patients were more likely to receive IIID although we did not find any meaningful differences in disease or clinical characteristics of patients who had standard induction versus IIID. When we performed a sensitivity analysis to remove patients who had a concomitant GI infection, the difference in colectomy free survival was no longer statistically significant (p = 0.07). Our results and those of Shah et al., 43 highlight the uncertainty surrounding the true benefit of high-intensity infliximab dosing in ASUC in preventing early colectomy. In the absence of good quality prospective data (e.g., from the highly anticipated PREDICT-UC study), recommendations for prescribing high-intensity infliximab induction should be limited to ‘expert opinions’.
Our study had several strengths. Despite being only a single centre study, our centre is one of three major tertiary hospitals which services a large population of Western Australia (southern and southwest region of Western Australia), thereby leading to a high referral rate for new IBD patients. As Western Australia is a vast state, access to health care in regional centres is relatively poor. Thus, many of our ASUC cases were new presentations with first diagnoses of UC, since many of these patients were not able to access a gastroenterology or colonoscopy service early enough to establish their diagnosis and prevent a presentation with ASUC.
Despite the retrospective nature of the study, only two patients were excluded due to incomplete data. We derived our data from hospital coding, which was considered up to date and complete. Further data extraction was comprehensive from electronic medical records with all three authors being involved in verifying the accuracy of the extracted data. There was also relatively low heterogeneity in patterns of clinical practice between the five IBD physicians that managed ASUC at this centre.
Our study, however, had limitations including the retrospective methodology. Some complications, albeit likely to be less serious, may not have been captured in the medical records. In addition, the duration of intravenous hydrocortisone therapy and the selection of infliximab dose intensity were at the discretion of the treating gastroenterologist. This treatment bias as alluded earlier, may preselect for patients to receive IIID who are more likely to have a severe episode, and therefore perhaps at greater risk of experiencing a complication and/or a colectomy. Despite this selection bias, however, no association was found between infliximab dose intensity and frequency of complications. We did not collect infliximab levels due to the long turn-around time for results to be made available. Thus, we could not reliably verify if a patient was being underdosed. Dashboard algorithms using infliximab levels to predict dose intensity/ frequency show promise for future studies in ASUC. We also excluded the mode of surgery (laparoscopic versus open) from the univariate analysis due to low numbers (15 open and 20 laparoscopic). The impact on the prevalence of surgical complications directly, if not indirectly, from the potential confounding relationship that choice of surgical approach had with the pre-operative morbidity level of the patient would not have been insignificant. Larger studies, however, would further clarify this question.
Finally, the study’s primary outcome was the prevalence of complications at 90 days. Longer-term data on complications were not obtained due to the heterogeneity of the treatment approaches post induction and dose optimization was not always pursued by all treating gastroenterologists. Treatment heterogeneity is one of the main limitations in retrospective studies on ASUC and is likely the reason why a professional consensus on the optimal infliximab induction dose regimen for ASUC has not been reached.
In conclusion, our data show that early complications within 90 days following a hospital admission for ASUC occur in 50% of admissions. A low serum albumin is associated with a higher frequency of total and surgical complications, an increased age with a higher frequency of surgical complications following a colectomy for ASUC, and an increased CCI with a higher frequency of medical complications at 90 days. Immunosuppressive therapy, in particular high-intensity infliximab rescue therapy, is not associated with an increased incidence of 90-day total, medical or surgical complications but is also not associated with a superior colectomy free survival compared to standard infliximab induction. Early recognition of these risk factors will assist physicians to optimize therapy for ASUC patients while mitigating the risk of complications, resulting in better quality care.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231215148 – Supplemental material for Prevalence and risk factors for early medical and surgical complications following an admission for acute severe ulcerative colitis
Supplemental material, sj-docx-1-tag-10.1177_17562848231215148 for Prevalence and risk factors for early medical and surgical complications following an admission for acute severe ulcerative colitis by Angel Li, Matthew Coote and Lena Thin in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
STROBE statement
This study has been performed in accordance with the STROBE guidelines. 44
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
