Abstract
Background:
The International Consortium for Health Outcomes Measurement has selected the self-administered comorbidity questionnaire (SCQ) to adjust case-mix when comparing outcomes of inflammatory bowel disease (IBD) treatment between healthcare providers. However, the SCQ has not been validated for use in IBD patients.
Objectives:
We assessed the validity of the SCQ for measuring comorbidities in IBD patients.
Design:
Cohort study.
Methods:
We assessed the criterion validity of the SCQ for IBD patients by comparing patient-reported and clinician-reported comorbidities (as noted in the electronic health record) of the 13 diseases of the SCQ using Cohen’s kappa. Construct validity was assessed using the Spearman correlation coefficient between the SCQ and the Charlson Comorbidity Index (CCI), clinician-reported SCQ, quality of life, IBD-related healthcare and productivity costs, prevalence of disability, and IBD disease activity. We assessed responsiveness by correlating changes in the SCQ with changes in healthcare costs, productivity costs, quality of life, and disease activity after 15 months.
Results:
We included 613 patients. At least fair agreement (κ > 0.20) was found for most comorbidities, but the agreement was slight (κ < 0.20) for stomach disease [κ = 0.19, 95% CI (−0.03; 0.41)], blood disease [κ = 0.02, 95% CI (−0.06; 0.11)], and back pain [κ = 0.18, 95% CI (0.11; 0.25)]. Correlations were found between the SCQ and the clinician-reported SCQ [ρ = 0.60, 95% CI (0.55; 0.66)], CCI [ρ = 0.39, 95% CI (0.31; 0.45)], the prevalence of disability [ρ = 0.23, 95% CI (0.15; 0.32)], and quality of life [ρ = −0.30, 95% CI (−0.37; −0.22)], but not between the SCQ and healthcare or productivity costs or disease activity (|ρ| ⩽ 0.2). A change in the SCQ after 15 months was not correlated with a change in any of the outcomes.
Conclusion:
The SCQ is a valid tool for measuring comorbidity in IBD patients, but face and content validity should be improved before being used to correct case-mix differences.
Introduction
Inflammatory bowel disease (IBD) can lead to reduced quality of life and impairment of social life and work productivity. It often requires lifelong drug treatment or major surgery.1–3 Due to both disease burden and increasing use of effective yet costly medication, IBD leads to high societal costs.4–7
Implementation of the International Consortium for Health Outcomes Measurement (ICHOM) IBD outcome set can reveal deficits in care for patients as it enables systematic collection and comparison of outcomes. By identifying and acting upon care deficits, quality of care and health outcomes can be improved, and costs reduced.8,9 The standard set of ICHOM incorporates both patient- and clinician-reported outcome measures across different domains such as quality of life and healthcare use.
To increase validity when comparing healthcare providers using outcomes, a process called benchmarking, case-mix variables need to be identified and adjusted for. In the ICHOM outcome set, a minimum set of case-mix variables has been defined, containing demographics (e.g. year and sex at birth), comorbidities, condition factors (e.g. diagnosis, disease phenotype), and treatment factors (e.g. current medication). 8 Comorbidity is an important case-mix variable as it is known to impact quality of life, healthcare use, and outcomes in other diseases.10–14 There is evidence that this is also the case for IBD, as comorbidities are associated with worse postoperative outcomes and increased healthcare costs.14–16 Identification of comorbidities in IBD is important because comorbidities such as tuberculosis, primary sclerosing cholangitis, concomitant immune-mediated inflammatory diseases, venous thrombosis and osteoporosis influence therapy approach, disease prognosis, or outcome of IBD treatment. 17
To account for the impact of comorbidities on outcomes, the self-administered comorbidity questionnaire (SCQ) was selected by ICHOM.8,18 The general validity and reliability of the SCQ have been established upon inception of the instrument in a sample of inpatients. Although the validity and reliability of the SCQ have been studied in rheumatic and renal disease, no research has been done on the validity of the SCQ in IBD.19–23 This study aims to assess the validity and reliability of the SCQ for measuring the comorbidities of IBD patients.
Materials and methods
Data source
For this study, data from the IBD Value study were used. The design and methodology of this study have been published before. 24 In short, this prospective study evaluated the quality of care of IBD patients treated with a biologic or new small molecule, as well as the effect of a uniform care pathway on quality of care and cost-effectiveness. The project ran from 1 December 2020 until 1 March 2023 and included IBD patients treated with a biologic or new small molecule in the southwest Netherlands at any point during the study period. The current study used data from all patients that were included in the first 3 months of the IBD Value study. The specific instruments and outcomes used in this paper are described below. The consensus-based standards for the selection of health status measurement instrument (COSMIN) recommendations for reporting measurement properties of health status questionnaires were followed (Supplemental Materials). 25
Study instruments
The SCQ is a short and self-administered questionnaire to determine comorbidity. 18 In the IBD Value study, the SCQ was administered on inclusion and at 15 months follow-up as an electronic survey (Supplemental Materials). The questionnaire contains 13 questions on whether a comorbidity is present, if treatment has been received, and whether there are limitations in daily activities due to this comorbidity. In addition, three open-ended questions were included for any other comorbidities. Comorbidities were included in the SCQ based on their prevalence in the general population and inclusion in other instruments used to measure comorbidity. 18 For cancer, the question asks whether a patient has had cancer in the last 5 years. Osteoarthritis and rheumatoid arthritis are scored as one comorbidity, meaning that the maximum score for the questionnaire is 45 for 12 predefined comorbidities and three optional comorbidities, with one point for the presence of a problem, one point for treatment status, and one point for limitation of daily activities. In this study, we used only the close-ended questions, leading to a maximum score of 36.
To assess the validity and responsiveness of the SCQ, we compared it to the healthcare professional (HCP) SCQ, Charlson Comorbidity Index (CCI), healthcare costs, productivity costs, the prevalence of disability, health-related quality of life (HRQoL), and IBD disease activity.26,27
The HCP SCQ was scored from patient charts by trained extractors who assessed whether any of the predefined comorbidities were present. The CCI was calculated based on the revised algorithm using chart data which was collected by the same extractors.26,27 Healthcare use in primary care was determined by the Institute of Medical Technology Assessment (iMTA) Medical Consumption Questionnaire (iMCQ). 28 Data from the electronic healthcare records were used to assess healthcare use related to IBD in secondary care which included outpatient clinic visits, diagnostics, medication, ER visits, admissions, and surgery. Healthcare use was converted to costs with Dutch reference prices or insurer tariffs when reference prices were not available. 29 Productivity losses were defined as absenteeism, presenteeism, and loss of unpaid time due to IBD for patients less than 65 years old and were assessed with the iMTA Productivity Cost Questionnaire (iPCQ). Productivity losses were transformed into costs using Dutch reference prices and the friction cost method.29–31 The iPCQ was also used to determine the prevalence of disability. HRQoL was based on the EQ-5D-5L (both Dutch tariffs and the visual analog scale).32,33 IBD disease activity was measured using the IBD-Control-8 score (IBD-Control) and Manitoba IBD Index (MIBDI).34–36 All questionnaires were sent out to patients at inclusion in the study and after 15 months, while the IBD disease activity questionnaires were sent out at inclusion and after 21 months. 24
The level of education of the patients was assessed via survey, and categorized into low [International Standard Classification of Education (ISCED) < 3], middle (ISCED 3–4), and high (ISCED > 4) according to the United Nations Educational, Scientific and Cultural Organisation (UNESCO) standards. 37 Disease phenotype was extracted from the electronic health records and scored according to the Montreal classification. 38
Statistical analyses
Missing data
Missing data were multiply imputed 100 times to account for data being missing at random. Data were imputed using the Multivariate Imputation by Chained Equations algorithm in R. 39 All variables used in the analyses were incorporated in the imputation procedure to ensure congeniality. Baseline characteristics were added as auxiliary variables to improve imputations. Dichotomous variables were imputed using logistic regression, all other variables with type 1 predictive mean matching. For the baseline table, imputed datasets were pooled to create a mean dataset. All other analyses were ran on the imputed datasets and then pooled using Rubin’s rules.
Criterion validity
To determine criterion validity, the tool to be validated has to be compared with the gold standard. 40 However, for comorbidity, there is no single accepted gold standard. We used the HCP SCQ as a gold standard. The difference and 95% confidence interval of the proportion of patients with each comorbidity between the patient SCQ and HCP SCQ were calculated using the Wald interval with continuity correction. 41 Criterion validity was assessed by calculating Cohen’s kappa between the patient SCQ and HCP SCQ for each of the comorbidities. Cohen’s kappa assesses agreement between the two measures while taking into that agreement can occur by chance. The strength of agreement was classified in line with prior research as poor (κ ⩽ 0.00), slight (0.00 < κ ⩽ 0.20), fair (0.20 < κ ⩽ 0.40), moderate (0.40 < κ ⩽ 0.60), substantial (0.60 < κ ⩽ 0.80), and almost perfect (0.80 < κ ⩽ 1.00). 42 We also further investigated the cause of reduced validity by assessing whether comorbidities were more often reported in the SCQ or the HCP SCQ. As patients might confuse comorbidities and complications of IBD, we assessed the correlation between proximal Crohn’s disease and self-reported stomach disease and between anemia and self-reported blood disease (which also explicitly includes anemia). 43 The presence of floor or ceiling effects was determined by assessing if 15% or more of the respondents had a comorbidity status of 0 or 36, respectively.
Construct validity
Construct validity measures the relation between the studied measure and measures that should theoretically relate. 40 We hypothesized beforehand that the SCQ should at least have a weak positive correlation with the CCI, healthcare costs, productivity costs, and prevalence of disability. 26 Moreover, the SCQ should at least have a weak negative correlation with health-related quality of life (HRQoL) based on the EQ-5D-5L (both Dutch tariffs and the visual analog scale).32,33 The SCQ should not correlate with patient-reported disease activity, as it should measure other diseases than IBD. All correlation coefficients were determined using Spearman’s rank correlation coefficient between the SCQ at baseline and the selected outcome measures. Spearman’s rank correlation coefficient was used as most measures did not follow a normal distribution. The strength of correlation coefficients was defined before the start of the study as none (|ρ| ⩽ 0.2), weak (0.2 < |ρ| ⩽ 0.4), moderate (0.4 < |ρ| ⩽ 0.75), and high (|ρ| > 0.75). 44
Responsiveness
Responsiveness measures the ability to detect clinically important changes over time. For the SCQ and comorbidity in general, no minimal important difference exists. We assessed responsiveness by measuring the correlation between changes in the SCQ and changes in healthcare costs, productivity costs, and quality of life at 15 months and the correlation between changes in SCQ at 15 months and changes in the IBD-control and MIBDI at 21 months. We hypothesized that a change in the SCQ had at least a weak negative correlation with HRQoL, a weak positive correlation with healthcare and productivity costs, and was not correlated with a change in disease activity measures.
Interpretability
To increase the interpretability of the results, we presented the outcomes of the SCQ stratified based on hospital (academic/general), sex (male/female), age group (18–39/40–64/65+), and diagnosis (Crohn/colitis/IBD-unknown).
Results
Study population
We included 613 patients in this study of which 368 (60%) were diagnosed with Crohn’s disease and 234 with ulcerative colitis (38%). The median patient-reported SCQ was 1.67 [interquartile range (IQR): 0.01–3.54] at baseline which increased to 2 (IQR: 0.24–4.00) at 15 months (Table 1). The distribution of comorbidities over the subgroups is presented in Table 2. Patients treated in an academic hospital reported a higher mean [3.20, standard deviation (SD) 3.27] on the SCQ compared to patients from a general hospital (2.28, SD 2.66). The same accounts for patients aged over 65 (3.99, SD 3.18) versus patients in the age group of 18–39 (1.47, SD 2.08) and 40–64 (2.35, SD 2.67). Clinical data were complete, while surveys had a relatively low percentage of missing data at baseline. The percentage of missing data at baseline was between 5% and 30%, dependent on the survey question, and increased to between 20% and 45% at 15 months (Supplemental Table 1).
Characteristics of the study cohort.

Median (IQR); n (%).
IBD-U, IBD-unclassified; IQR, interquartile range; SCQ, self-administered comorbidity questionnaire.
Distribution of the SCQ in subgroups.

IBD-U, IBD-unclassified; IQR, interquartile range; N, number of patients, SD, standard deviation.
Criterion validity
For most comorbidities, there was a fair or better agreement between the SCQ and the HCP SCQ (Table 3). The agreement was almost perfect for diabetes [κ = 0.89, 95% CI (0.82; 0.97)]. However, only slight agreement was found for stomach disease [κ = 0.19, 95% CI (−0.03; 0.41)], blood disease [κ = 0.02, 95% CI (−0.06; 0.11)], and back pain [κ = 0.18, 95% CI (0.11; 0.25)]. These diseases were all more often reported by patients as compared to the HCP SCQ with, respectively, 2.7% versus 0.8% [difference 1.8%, 95% CI (0.2; 3.5)], 5.8% versus 1.8% [4.0%, (1.6; 6.5)], and 29.4% versus 6.2% [23.2%, (18.9; 27.6)]. Arthritis [25.9% versus 9.3%, difference 16.6%, 95% CI (12.7; 20.6)] and depression were also more often reported by patients [8.7% versus 3.9%, 4.8% (2.0; 7.5)] but there was fair agreement between the SCQ and the HCP SCQ for both comorbidities [κ = 0.32, 95% CI (0.24; 0.41) and κ = 0.23, 95% CI (0.10; 0.36)]. There was no evidence for a correlation between anemia and self-reported blood disorders [ρ = 0.06, 95% CI (−0.06; 0.18), p = 0.31] or for proximal Crohn’s disease and self-reported stomach disease [ρ = −0.01, 95% CI (−0.11; 0.10), p = 0.86]. Floor and ceiling effects were, respectively, 36.84% and 0%.
Kappa between patient-reported and electronic health record presence of a disease.

Percentage of patients who did not report a disease while it was found in the EHR.
Percentage of patients who reported a disease while it was not found in the EHR.
CI, confidence interval; EHR, electronic health record; SCQ, self-administered comorbidity questionnaire.
Construct validity
Positive correlations were found between the SCQ and the clinician-reported SCQ [ρ = 0.60, 95% CI (0.55; 0.66), p < 0.0001], CCI [ρ = 0.39, 95% CI (0.31; 0.45), p < 0.0001], and prevalence of disability [ρ = 0.23, 95% CI (0.15; 0.32), p < 0.0001]. Negative correlations were found between the SCQ and the EQ-5D-5L utility score [ρ = −0.38, 95% CI (−0.45; −0.30), p < 0.0001], and EQ-5D-5L visual analog scale score [ρ = −0.30, 95% CI (−0.37; −0.22), p < 0.0001]. There was a significant correlation between the SCQ and both productivity costs (p = 0.0049) and disease activity [both Manitoba IBD index (p = 0.0007) and IBD-control (p = 0.0001)], but the strength of these correlations was negligible (|ρ| ⩽ 0.2). No correlation was found between the SCQ and healthcare costs [ρ = 0.02, 95% CI (−0.06; 0.11), p = 0.58], see also Table 4.
Spearman correlation coefficients between SCQ and selected outcomes.
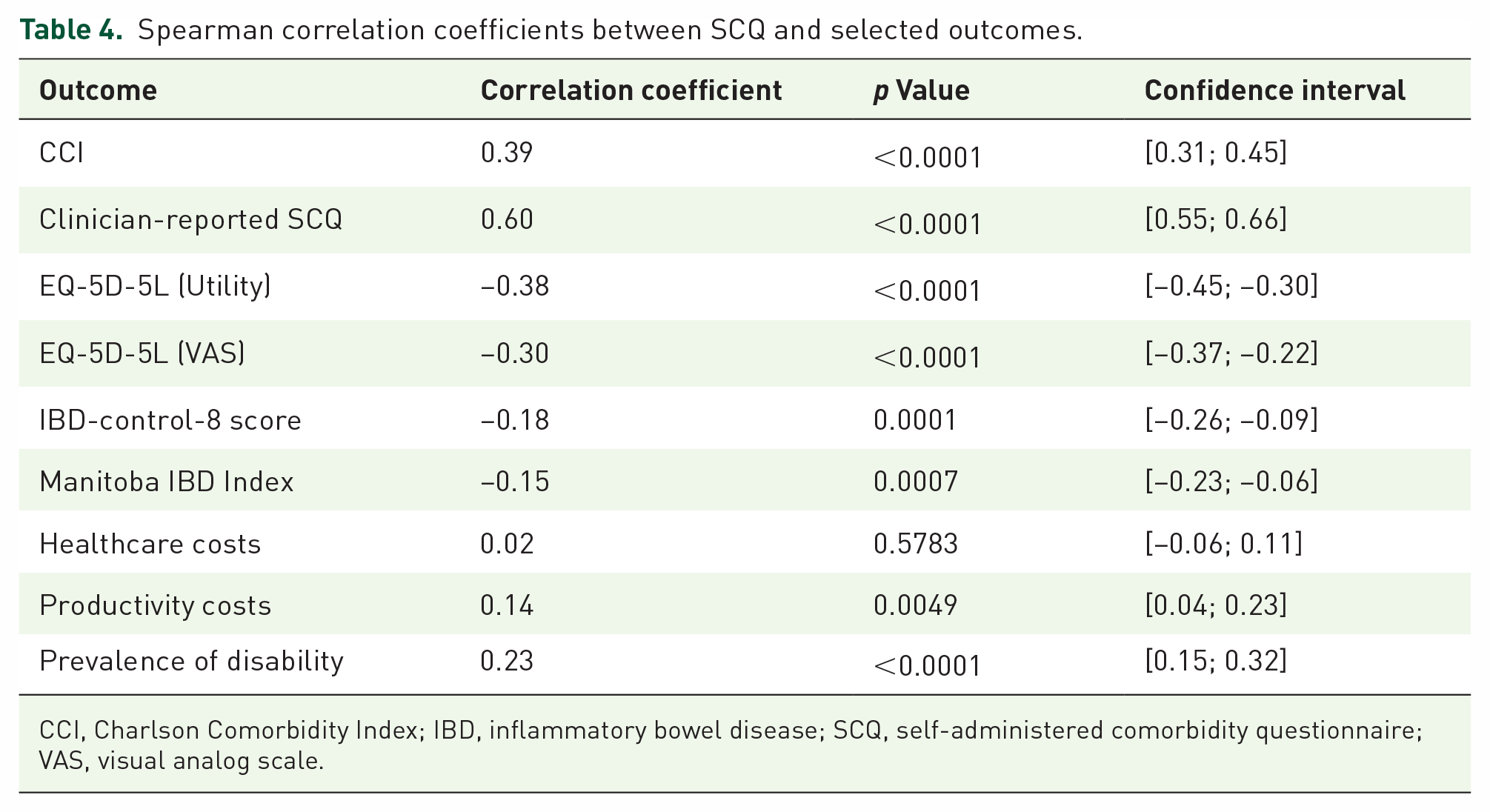
CCI, Charlson Comorbidity Index; IBD, inflammatory bowel disease; SCQ, self-administered comorbidity questionnaire; VAS, visual analog scale.
Responsiveness
There was a correlation between changes in the SCQ and changes in utility as measured by the EQ-5D-5L, but the strength of this correlation was negligible [ρ = −0.16, 95% CI (−0.26; −0.06), p = 0.0025]. There were no other outcomes that correlated with a change in the SCQ (Table 5).
Spearman correlation coefficients between change in SCQ and change in selected outcomes.

IBD, inflammatory bowel disease; SCQ, self-administered comorbidity questionnaire; VAS, visual analog scale.
Discussion
This study has shown that the SCQ seems to be a valid method to measure comorbidities in a population of IBD patients treated with biologics or new small molecules. The agreement between self-reported comorbidities and HCP-reported comorbidities was fair to almost perfect for most diseases except for stomach disease, blood disease, and back pain. These comorbidities together with depression and arthritis were all more frequently reported by patients than HCP. Depression and stomach disease were also more often reported by patients in a study that validated the SCQ in patients with ankylosing spondylitis. 22 A first explanation could be that these diseases are often not registered in the hospital’s electronic healthcare records. Patients may be diagnosed with these comorbidities by a general practitioner, as depression, back pain, hip arthrosis, and knee arthrosis are among the most frequent registered diagnoses in general practice, with a prevalence of, respectively, 31.2/1000, 32.0/1000, 27.7/1000, and 43.5/1000 in the Netherlands. 46 Moreover, in 2021, antacids were the most prescribed drug in the Netherlands by general practitioners, and patients with symptoms of heartburn might have reported that they have a stomach disease. 46 A second explanation for the higher prevalence reported by patients is that patients may not be able to distinguish between different comorbidities and IBD-related complications. This was seen in a cohort of patients with systematic lupus erythematosus. 43 In IBD patients, IBD-related anemia reported as a comorbid blood disease or proximal Crohn’s disease reported as a stomach disease are examples of IBD-related complications that could be reported as comorbidities. However, we found no correlation between stomach disease and proximal Crohn’s disease, nor between anemia and blood disease or self-reported anemia. Overall, the SCQ seems to have good criterion validity and likely captures comorbid problems of IBD patients better than the hospital’s electronic healthcare records.
Construct validity was acceptable with correlations between the SCQ and the HCP SCQ, CCI, the prevalence of disability, and the EQ-5D-5L as expected. The agreement between the CCI and the SCQ in this study is comparable with the agreement reported in the original validation study and validation studies in other diseases.18,21,22 HRQoL demonstrated a weak negative correlation with the SCQ, which is in line with other studies and our hypothesis.21,22 In contrast to our hypothesis, we found no correlation between the SCQ and healthcare or productivity costs. This might be caused by two reasons. First, we only included IBD-related costs in secondary care while all costs were included in primary care. This might reduce the association between the SCQ and healthcare costs, as not all comorbidity-related costs were included. However, we aimed to validate the SCQ in IBD patients, and our results show that there is no evidence that comorbidities impact IBD-related healthcare costs. Second, biologics are the main cost driver for healthcare costs in IBD and the impact of comorbidities on costs is probably relatively small compared to the costs of biologics.3,6 The weak correlation between the SCQ and productivity costs can likely be explained through the same mechanism as healthcare costs, as patients were asked to report productivity losses due to IBD.
A change in the SCQ was correlated to a change in utility as measured by the EQ-5D-5L, but not with any of the other measures. However, the strength of the correlation between a change in the SCQ and a change in the EQ-5D-5L was negligible. It seems likely that in this population of IBD patients, other within-person changes, for example, disease activity, have more impact on the chosen outcomes than comorbidities. The utility of the SCQ as a case-mix variable can therefore be questioned in longitudinal settings.
The main strengths of this study are the sample size, the longitudinal and multicenter approach, and the broad range of outcomes used for testing validity and responsiveness. However, a few limitations should be noted. Only patients on a biologic or small new molecule were included, which might hamper generalization to the overall IBD population. However, it is unlikely that the relation between true comorbidity status and comorbidity as measured by the SCQ is different between the biologic and non-biologic populations. The validity of the SCQ in measuring comorbidity is thus likely generalizable to the overall IBD population. Moreover, missing survey data, mostly at 15 months, might have biased our analyses. By assuming that data were missing at random and by multiplying imputing missing data, we aimed to minimize the impact of missing data. However, if data were missing not at random or the imputation model was misspecified, bias might still have occurred, mostly in the estimates for responsiveness. Last, we did not assess internal consistency and test–retest reliability in this study. With internal consistency, the correlation between the different items in a questionnaire is assessed. This only applies when using multiple questions to measure the same underlying construct. While all the questions in the SCQ relate to the concept of comorbidity, they all relate to a distinct problem or disease that makes up comorbidity. No correlation between diseases has to exist to properly determine comorbidity. As such, we did not test internal consistency. 45 Moreover, we had no indication that test–retest reliability of the SCQ would be different in our population. Test–retest reliability of the SCQ has been studied before, both in the population that was used to develop the SCQ, which consisted of medical and surgical inpatients, as well as in a study on arthritis patients. Both studies showed a high test–retest reliability of 0.94.18,19
While the SCQ seems a valid tool for measuring comorbidity in IBD patients, there are two other important aspects of validity that should be taken into account, face and content validity. Face validity evaluates if a questionnaire appears to measure what it should and content validity assesses whether all important aspects of comorbidity are measured. While the SCQ was chosen to adjust for comorbidity when comparing outcomes between providers, there are some issues with face and content validity. 8 First, the SCQ contains questions on cancer, including colorectal cancer, and anemia. These are important outcomes of IBD treatment and are also included in the ICHOM IBD set. Adjusting for these as comorbidities would mean adjusting for the outcome you aim to measure. Second, some comorbidities that might influence therapy choice and outcome of IBD treatment are not included in the SCQ, for example, tuberculosis, human immunodeficiency virus/acquired immunodeficiency syndrome, primary sclerosing cholangitis, or concomitant immune-mediated inflammatory diseases. While some of these are captured through other patient-reported measures in the ICHOM outcome set, other comorbidities that might be related to IBD such as venous thrombosis, Parkinson’s disease, osteoporosis, and multiple sclerosis are not. As validity and relevance of case-mix adjustment variables differ between outcomes, 47 future research should aim at adjusting the SCQ to improve face and content validity for use in a population of IBD patients. In addition, the lack of responsiveness to the outcomes raises the question of whether the SCQ in its current form is the best measure to adjust for differences in comorbidity. Possibly, when face and content validity are improved, responsiveness increases. Future research should assess if the (improved) SCQ is the best comorbidity measure to adjust case-mix differences when comparing outcomes of IBD care.
Conclusion
The SCQ is a valid tool for assessing comorbidities in IBD patients, but face and content validity should be improved so that the SCQ can be used to correct case-mix differences when comparing outcomes of IBD care.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848231202159 – Supplemental material for Validity of the self-administered comorbidity questionnaire in patients with inflammatory bowel disease
Supplemental material, sj-pdf-1-tag-10.1177_17562848231202159 for Validity of the self-administered comorbidity questionnaire in patients with inflammatory bowel disease by Reinier Cornelis Anthonius van Linschoten, Anouk Sjoukje Huberts, Nikki van Leeuwen, Jan Antonius Hazelzet, Janneke van der Woude, Rachel Louise West, Desirée van Noord, V. de Jonge, F. H. J. Wolfhagen, A. G. L. Bodelier, J. Hoekstra, C. J. van der Woude, A. C. de Vries, R. L. West, D. van Noord, R. C. A. van Linschoten, E. Visser, K. E. Verweij, F. J. G. M. Kubben, I.L. Holster, C. E. Fitzpatrick, K. Robbers, H. G. Vermeulen, S. K. van der Wiel and S. V. Jansen in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
