Abstract
Acute pancreatitis (AP) is one of the most common acute abdominal conditions, and its incidence has been increasing for years. Approximately 15–20% of patients develop severe AP (SAP), which is complicated by critical inflammatory injury and intestinal dysfunction. AP-associated inflammation can lead to the gut barrier and function damage, causing dysbacteriosis and facilitating intestinal microbiota migration. Pancreatic exocrine deficiency and decreased levels of antimicrobial peptides in AP can also lead to abnormal growth of intestinal bacteria. Meanwhile, intestinal microbiota migration influences the pancreatic microenvironment and affects the severity of AP, which, in turn, exacerbates the systemic inflammatory response. Thus, the interaction between the gut microbiota (GM) and the inflammatory response may be a key pathogenic feature of SAP. Treating either of these factors or breaking their interaction may offer some benefits for SAP treatment. In this review, we discuss the mechanisms of interaction of the GM and inflammation in AP and factors that can deteriorate or even cure both, including some traditional Chinese medicine treatments, to provide new methods for studying AP pathogenesis and developing therapies.
Introduction
Multiple organ dysfunction may occur during the early phase of severe acute pancreatitis (SAP), resulting in a high fatality rate. However, over time, the patient enters a second stage, which accompanies the infection and is another cause of the high mortality rate of SAP. 1 Studies have shown that most pancreatic and extra-pancreatic organ infections are caused by the translocation of intestinal bacteria; such infections result in pancreatic necrosis and sepsis, causing late death in patients with SAP. 2 The gut microbiota (GM) is mutualistic with the human body under certain steady states; some gut bacteria can ferment dietary fiber to form short-chain fatty acids (SCFAs), which are then absorbed by the host. 3 The intestinal mucosa can also maintain the stability of the intestinal environment through its barrier function. Once this stability is disrupted by a persistent inflammatory response in SAP, this can lead to intestinal mucosal damage and a change in the status of the intestinal microbiota. 4 Studies have also indicated that various types of intestinal microbiota participate in different pathological conditions, including pancreatic diseases. 5 The role of the intestinal microbiota in the progression of SAP has gradually been clarified in previous studies.
We have searched articles or other types of manuscripts related to the regulatory mechanism of the intestinal microbiota, inflammation, and pathogenesis of SAP or acute pancreatitis (AP) in PubMed and the China National Knowledge Infrastructure, to describe the interactions between the GM and inflammatory responses in AP. We have identified some new methods of AP pathogenesis and the development of therapies. All of our findings are described in the following chapters.
The influence of intestinal flora changes on the occurrence and development of AP
Intestinal flora migration influences the pancreatic microenvironment in AP
Impairment in microcirculation and blood volume reduction during AP can lead to ischemia and reperfusion damage in the intestinal mucosa, causing loss of intestinal barrier integrity and intestinal bacterial translocation and causing local and systemic infections.6 –8 Fewer antimicrobials secreted by the pancreas in AP can also lead to bacterial overgrowth in the small intestine, which further disrupts the balance of the intestinal microbiota. 9 The imbalance of the intestinal microbiota or mucosal damage can increase intestinal permeability, causing the translocation of bacteria from the gut to the blood or nearby tissues, such as the pancreas, increasing the risk of pancreatic infection and aggravating inflammation. 10 A study has found more than one type of bacterial DNA in the peripheral blood of patients with AP, and these DNA molecules are mainly derived from conditional pathogenic bacteria from the gut, such as Escherichia coli, Shigella flexneri, Acinetobacter lwoffii, Bacillus coagulans, and Enterobacter faecalis. 11 Thus, the transfer of bacteria from the gut to the blood may cause infection of necrotic parts of the pancreas.
Recent studies have revealed that nucleotide-binding oligomerization domain 1 (NOD1), an intracellular innate immune receptor, plays a critical role in host defense functions and inflammation. This is because NOD1 can detect small peptide components derived from bacterial wall peptidoglycan and can be excited by intestinal bacteria. 12 On the other hand, NOD1 has been reported to activate innate responses and produce nuclear factor-kappa B (NF-κB) and type 1 interferon-inducing pancreatitis and contribute to the development of pancreatitis.13,14 Thus, NOD1 may be an intermediate regulatory factor of intestinal microbiota interaction with AP.
Previously, the microbial composition of the infected areas of pancreatic necrosis was mainly gram-negative bacteria from the gastrointestinal tract (GIT), such as Enterobacteriaceae. However, recently, Staphylococcus and Enterococcus have become dominant bacteria owing to the widespread use of prophylactic antibiotics. 15 Meanwhile, the prophylactic use of antibiotics does not reduce the risk of infection, and patients with a higher risk of infection in regional pancreatic necrosis are those who have previously received antibiotics.16,17
Intestinal microbiota attenuates the severity of AP
A normal intestinal microbiota constitutes the intestinal mucosal biological barrier that affects intestinal peristalsis, regulates host immunity, and strengthens the epithelial barrier. 18 Studies have shown that intestinal mucosal barrier damage in patients with AP is closely associated with the imbalance of the intestinal microbiota, for example, increased abundances of the intestinal pathogenic bacteria Shigella and Enterococcus and decreased abundances of the beneficial bacteria Lactobacillus and Blautia.19 –21 Deng showed that the bacterial translocation rates of E. coli and Bifidobacterium and the pathological damage score of intestinal tissue were significantly higher in the intestines of SAP rats than in those of the control group, suggesting that the intestinal barrier function of SAP rats was impaired, resulting in an intestinal microbiota disorder. 22 Moreover, the Acute Physiology and Chronic Health Evaluation (APACHE)-II score, the length of the hospital stay, complications such as infections and the incidence of multiple organ dysfunction syndrome were significantly higher in patients with SAP with a GM imbalance than in individuals with intestinal microbiota ratios similar to those of healthy individuals. 9
It has been reported that inter-intestinal probiotics mitigated AP severity by inhibiting the activation of the NOD-like receptor family 3 (NLRP3) inflammasome in the gut,23,24 which might be the mechanism of regulating the intestinal microbiota to reduce the degree of SAP. E. coli has been reported to induce intestinal mucosal barrier damage and aggravate AP through the activation of the toll-like receptor 4 (TLR4), myeloid differentiation factor 88 (MyD88), and p38 mitogen-activated protein kinase (MAPK) signaling pathways.25,26 Therefore, MAPK inhibitors and TLR4-dependent Phosphoinositide 3-kinase (PI3K), V-akt murine thymoma viral oncogene homolog (AKT), and NF-kB inflammatory signaling pathway inhibitors are important in correcting GM imbalance and mitigating inflammatory responses.27,28 SCFAs, a metabolite of intestinal bacteria, can not only provide growth energy for intestinal mucosal cells but also regulate intestinal pH, maintain the integrity of tight junction proteins between intestinal mucosal epithelial cells, improve intestinal mucosal barrier function, and significantly reduce the severity of SAP.21,24,29 On the other hand, Bacteroides, Escherichia–Shigella and Enterococcus, are the major intestinal microbes in AP, and different levels of AP are associated with different intestinal microbiota disorders. 20 In mild acute pancreatitis (MAP), Finegoldia exhibited the most significant increase, and Brucella was the species of intestinal microbiota that showed the largest decrease. Moderately severe acute pancreatitis (MSAP) patients had the most significant increase in Anaerococcus and the most significant decrease in Eubacterium hallii. The potential biomarkers of MAP are Finegoldia, E. hallii, and Lachnospiraceae. E. hallii and Anaerococcus are potential diagnostic biomarkers for MSAP (Table 1). According to reports, Firmicutes increase while Bacteroidetes decrease in acute patients’ intestines. Enterococcus in Firmicutes can adhere to host cells, invade them, and traverse the epithelial barrier. This can lead to infection and systemic inflammation. Bacteroidetes are capable of producing SCFAs, which have anti-inflammatory effects and help maintain the integrity of the intestinal barrier, thereby protecting it. Certain pathogenic bacteria within Bacteroidetes, such as E. coli and Shigella, can disrupt the intestinal mucosal barrier, resulting in severe colonic inflammation. Therefore, the imbalance between Firmicutes and Bacteroidetes can aggravate the pathogenetic condition of AP. 20 SAP was associated with the most significant increase in the abundance of Enterococcus and the greatest decrease in the abundance of E. hallii. 30 The expression of proinflammatory factors such as interleukin (IL)-1, IL-6, and tumor necrosis factor-α (TNF-α) in the serum of SAP patients was positively correlated with the intestinal aerobic bacteria level but negatively correlated with the level of anaerobic bacteria such as Bifidobacterium. 9 Perhaps modulating the gut flora can reduce the body’s inflammatory response and reduce AP severity.
Effects of alteration of bacteria on AP and GM and their mechanisms.

AP, acute pancreatitis; GM, gut microbiota; IL, interleukin; LPS, lipopolysaccharide; MAP, mild acute pancreatitis; MSAP, moderate severe acute pancreatitis; SAP, severe acute pancreatitis; SCFA, short-chain fatty acid; TNF-α, tumor necrosis factor-α.
The influence of AP on intestinal flora changes
Acute pancreatitis-associated gut barrier and functional damage facilitate intestinal flora migration
Intestinal barrier dysfunction was found in both animal models and clinical patients with AP. 75 The mechanism of intestinal microbiota migration in AP is as follows: intestinal barrier damage and a variety of gastrointestinal polypeptide secretions can destroy Cajal mesenchymal cells, 76 decrease gastrointestinal movement, 77 and impair intestinal motility, 78 resulting in the overgrowth of intestinal bacteria in AP. 79 Early fasting in patients with AP can cause intestinal ischemia–reperfusion injury, which can lead to intestinal mucosal microcirculation disorders and abnormal release of inflammatory factors and reactive oxygen species (ROS). These substances can cause the oxidative stress response in the intestinal mucosa, 80 the apoptosis of intestinal epithelial cells, and increased permeability of intestinal capillaries,81,82 ultimately leading to intestinal barrier function disorders and increased intestinal permeability.83,84 The intestinal immune barrier function is compromised in patients with AP, 85 and the level of secretory immunoglobulin A is decreased, 86 which allows bacteria to pass through the intestinal barrier more easily. In addition to these three effects, long-term fasting and the obstruction of the lower bile duct in patients with SAP can result in a significant decrease in bile secretion or ineffective secretion into the intestine. 75 Deoxycholic acid in the bile can selectively inhibit gram-positive bacilli (Bacillus, Clostridium, Lactobacillus, and Streptococcus pneumoniae). The reduction in bile secretion impairs the normal balance of the intestinal microbiota, resulting in the activation of an oxidative stress response and intestinal epithelial cell apoptosis, thus increasing bacterial migration. 87
Effects of secretion of cell damage factors on intestinal flora in AP
Intestinal barrier dysfunction is the most common complication of SAP. Previous clinical studies have shown that elevated serum levels of many inflammatory cytokines in SAP, including TNF-α, 84 IL-1, 88 IL-6, 89 neutrophil elastase (NE), and myeloperoxidase (MPO), 90 are associated with intestinal barrier dysfunction. One of the main cytokines associated with AP is TNF-α, a proinflammatory cytokine, which is found to have elevated levels both locally, in the intestine, and systemically in patients with intestinal barrier dysfunction.91,92 An increase in the TNF-α levels can lead to inflammation in the intestinal mucosa and to intestinal epithelial cell apoptosis,93,94 which can lead to intestinal epithelial mechanical barrier damage and facilitate bacterial displacement. 95 In addition to direct injury, TNF-α can initiate a positive feedback loop that induces the secretion of other cytokines, such as IL-1 and IL-6, to further injure the intestinal mucosa. 96 An increase in the IL-1 levels in AP and the risk associated with IL-1 and the IL-1 receptor (IL-1R) in the pathogenesis of pancreatitis have been reported. 97 IL-1R-deficient mice pretreated with an IL-1R antagonist recombinant human interleukin-1-receptor antagonist (rhIL-1Ra) experience milder pancreatitis after cerulein induction. The activation of IL-1β can also stimulate the local mucosal immune response and cause mucosal injury by stimulating T-cell proliferation and neutrophil entry to the site of injury or infection through the binding of IL-1β and IL-1R.98,99 Serum IL-6 is another reliable indicator of AP severity that can predict both organ failure and SAP. 100 The production of IL-6 can activate several different pathways in the adaptive immune system, thereby exacerbating inflammation and negatively affecting barrier function. 101 Tan et al. 9 also found that serum IL-6 levels in patients with AP were positively correlated with the abundance of Enterobacter and Enterococcus in the intestinal microbiota and negatively correlated with the abundance of XI groups of Bifidobacterium and Clostridium. In pancreatic tissue from a mouse model of SAP, neutrophil extracellular traps (NETs) decorated with MPO and NE were shown to aggravate tissue damage. 102 Many lethal complications of SAP have been shown to be closely related to NETs. According to previous reports, NETs can disrupt the balance of the intestinal microbiota, cause intestinal epithelial cell damage, and even induce apoptosis, leading to gut barrier damage, increased intestinal mucosal permeability, elevated endotoxin secretion, and imbalances in the GM.90,103 –105
Pancreatic exocrine deficiency affects the composition and diversity of GM in AP
Patients with AP exhibit complications such as pancreatic exocrine impairment (PEI) and acinar cell dysfunction, which significantly impact changes in intestinal microbiota composition, 106 and the secretion of many enzymes, such as lactate and bile acids, declines to a certain level. 107 In animal models of PEI, the intestinal microbes E. coli, Lactobacillus, and Bifidobacterium were increased, and the levels of Fusobacterium and Clostridium hiranonis were decreased, inducing a significant difference in the dysbiosis index between affected animals and healthy individuals. 108 Stool samples from PEI patients were analyzed and showed that pancreatic elastase levels significantly correlated with intestinal flora diversity compared with those of normal individuals, and significant differences were found in the abundances of 22 taxa, such as an increase in Pseudomonas spp. and a decrease in Bacillus spp. 106 These results revealed that changes in pancreatic fluid secretion were also significantly correlated with flora diversity.
Antimicrobial peptide changes in AP-affected intestinal flora
Antimicrobial peptides (AMPs) are oligopeptides that are arranged linearly or circularly and are composed of amino acid residues of different lengths (up to 100). AMPs usually form L-amino acids through secondary structures containing alpha helices, beta sheets, or both. 109 These biomolecules exhibit diverse biological activities against gram-positive and gram-negative bacteria, viruses, fungi, protozoa, and even tumors. 110 The Data Repository of AMPs (DRAMP) database includes over 4800 peptides that are antiproteins 111 and contribute to the maintenance of intestinal bacterial homeostasis and intestinal barrier function. 112 AMPs such as the cathelicidin-related AMP (CRAMP) have been reported to be secreted by pancreatic acinar cells, and reduced secretion of pancreatic AMPs can lead to the abnormal growth of intestinal bacteria and the disruption of the intestinal microbiota balance. Moreover, CRAMP deficiency worsens pancreatic inflammation. 113 Decreased expression of ileal terminal AMPs was found in necrotizing pancreatitis. 114 Hypertriglyceridemia (HTG) affects the expression of AMPs, including α-defensin, lysozyme, phospholipase A2, and regenerating islet-derived protein 3α (Reg3A), 115 in Paneth cells, which may exacerbate HTG-related acute necrotizing pancreatitis in intestinal barrier dysfunction. Pancreatic cells secrete a variety of AMPs to regulate the structure of the intestinal microbiota. 116 Lysozyme and α-defensins have activities against gram-negative and gram-positive bacteria, and some experts believe that fecal levels of α-defensins are a surrogate marker for gut microbial homeostasis.117,118 On the other hand, Reg3A, which has powerful bactericidal activity, can antagonize gram-positive bacteria119,120 by limiting the number of mucosal-adherent bacteria to separate the GM from the epithelium and reduce bacterial translocation 121 (Table 2).
AMP changes in AP-affected intestinal microbiota.
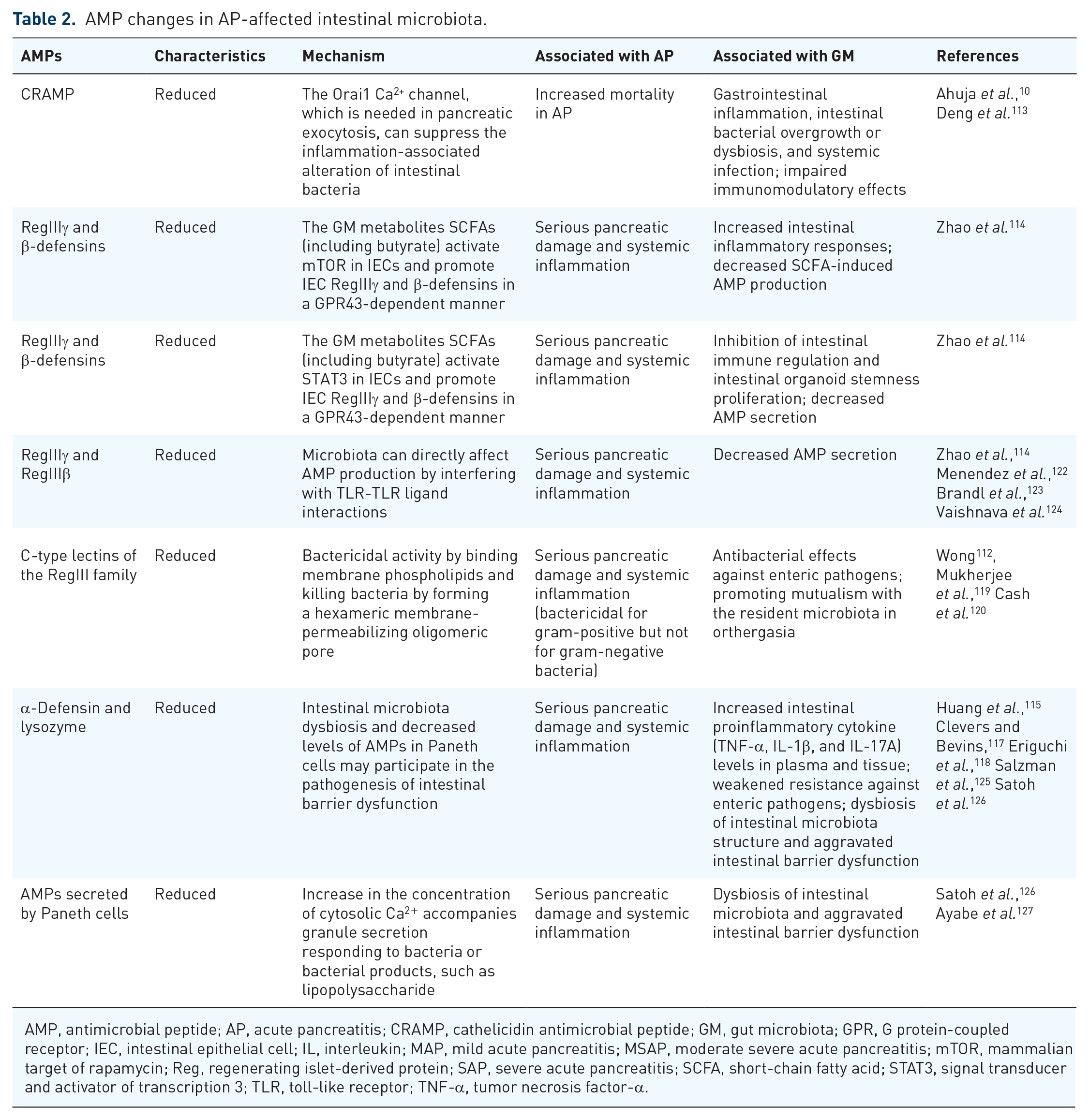
AMP, antimicrobial peptide; AP, acute pancreatitis; CRAMP, cathelicidin antimicrobial peptide; GM, gut microbiota; GPR, G protein-coupled receptor; IEC, intestinal epithelial cell; IL, interleukin; MAP, mild acute pancreatitis; MSAP, moderate severe acute pancreatitis; mTOR, mammalian target of rapamycin; Reg, regenerating islet-derived protein; SAP, severe acute pancreatitis; SCFA, short-chain fatty acid; STAT3, signal transducer and activator of transcription 3; TLR, toll-like receptor; TNF-α, tumor necrosis factor-α.
Risk factors that influence the GM and AP
Trillions of microbes live in the gut, and this community plays a vital role in the regulation of both intestinal and pancreatic functions. The underlying common causes of AP, such as biliary obstruction, alcohol misuse, HTG, and a high-fat/sugar diet, may also cause changes in the intestinal flora. 128 These risk factors affect both AP and the intestinal microflora; thus, interactions between the intestinal microflora and the occurrence of AP can be inferred.
Obesity and hyperlipidemia
Obesity typically presents with low-level systemic inflammation, such as increased leukocyte counts and TNF-α, IL-6, and C-reactive protein levels129,130; furthermore, it is characterized by increased secretion of biomarkers by adipocytes and is associated with AP. Moreover, macrophages in adipose tissue have been reported to participate in inflammation in obesity via the secretion of proinflammatory cytokines, such as TNF-α and IL-6, 131 both of which have been proven to affect AP and GM.
Obesity can also unmask primary HTG due to genetic causes and is a risk factor for secondary HTG, which is associated with pancreatitis.132,133 At present, HTG-induced AP (HTG-AP) has become the second leading cause of AP. 134 Indeed, the proportion of Bacteroides in the intestines of lean mice was found to be higher than that in obese mice after administration of the same diet, while the opposite was true for thick-walled Bacteroides. 135 Rats fed a high-fat diet showed significant increases in serum low-density lipoprotein, total cholesterol, and triacylglycerol, as well as changes in Bifidobacteria, Lactobacilli, Enterococci, Enterobacteria, and Anaphylactic bacteria in the intestinal flora. 136 In an animal model of hyperlipidemic necrotizing pancreatitis, researchers also found intestinal microflora imbalances and decreased AMPs in Paneth cells, further confirming that hyperlipidemia can affect the severity of AP and the intestinal microflora. 115 Unsaturated fatty acids (UFAs) might be an important factor that can affect both AP and GM in obesity and hyperlipidemia, and these factors are mainly transmitted via the lipolysis of circulating triglycerides. 137 The insolubility of UFAs in the aqueous environment of the blood can cause microthrombi formation in the pancreatic vasculature, leading to ischemia and pancreatic infarction. As polar molecules, UFAs usually bind with calcium, resulting in their saponification and inactivation in necrotic fat tissue. 138 Unbound UFAs can increase the serum levels of TNF-α and other inflammatory cytokines, 139 thereby worsening AP and leading to inflammation of the intestinal mucosa and intestinal epithelial cell apoptosis93,94 (Figure 1).

The negative effect of obesity and hypertriglyceridemia both on AP and GM.
In addition to the harmful effects of obesity and hyperlipidemia on the GM, recent studies have shown that the intestinal flora is one of the important environmental factors affecting the occurrence and development of obesity. The intestinal flora can induce adipocytokine gene expression by affecting intestinal epithelial cell fasting, leading to the increased production of triacylglycerols in the body and causing lipid metabolism disorders and the development of obesity. 140 Furthermore, disturbances in the intestinal flora in obese mice may lead to abnormal lipid metabolism, energy metabolism, adipokine synthesis, and cell death, leading to the secretion of a large number of proinflammatory cytokines into the blood and resulting in the exacerbation of pancreatitis. 115 Some studies indicated that intake of probiotic preparations could affect serum cholesterol and high-density lipoprotein levels and indirectly lower blood lipids, suggesting that the establishment of normal intestinal flora can help balance lipid metabolism. 113
Alcohol
In recent years, due to changes in diet and increases in alcohol consumption, heavy drinking has become a risk factor for AP, and this condition easily progresses to SAP. 141 The toxicity of ethanol is mediated by ethanol itself or its oxidative and nonoxidative metabolism. Oxidative ethanol metabolism potentiates cholecystokinin-induced depolarization by sensitizing pancreatic mitochondria to Ca2+-induced mitochondrial permeability transition pore (MPTP) activation, resulting in mitochondrial dysfunction in pancreatic acini and necrosis in the pancreas. 142 Although the main ethanol metabolism in vivo is oxidation, a smaller part undergoes nonoxidative metabolism.143,144 Incubation of isolated pancreatic acinar cells with fatty acid ethyl esters, one of the nonoxidative ethanol metabolites in vivo, induced mitochondrial depolarization, depletion of cellular adenosine triphosphate, 145 and sustained elevations of intracellular Ca2+ levels ultimately associated with cellular dysfunction and cell death. 146 Both alcohol and its metabolites can activate digestive enzymes early in pancreatic acinar tissue, resulting in pancreatic tissue autodigestive injury, and activate pancreatic stellate cells, leading to fibrosis of the pancreas. 143
On the other hand, alcohol has been shown to have a negative impact on the intestinal flora of healthy people, such as decreasing the biodiversity of the intestinal flora and affecting the overall composition of the microbial community. 145 Disturbances in the intestinal flora may cause disorders of glycolipid energy metabolism and other potential functional pathway changes in the body. 147 Alcohol can lead to changes in the composition of the GIT microbiota and metabolic function, contributing to the well-established association between alcohol-induced oxidative stress and intestinal hyperpermeability to luminal bacterial products.146 –148 Exposure to ethanol can increase the release of enterogenous gram-negative bacteria-derived lipopolysaccharide (LPS), leading to macrophage activation and the secretion of cytokines, including TNF-α, IL-1β, and IL-6. 149 IL-22 is mainly involved in maintaining the integrity of the epithelial barrier and linking intestinal immune activation with epithelial repair and barrier protection.150,151 Under inflammatory conditions, IL-22 can be activated through the IL-23-Janus kinase/signal transducer and activator of transcription signaling pathway, resulting in the production of AMPs. 152 Ethanol metabolism in vivo produces acetaldehyde and ROS, which can activate NF-κB and ultimately stimulate the immune response, 153 decrease intestinal expression of IL-22, and alter gut epithelial integrity, causing an increase in intestinal permeability and bacterial translocation 154 (Figure 2).
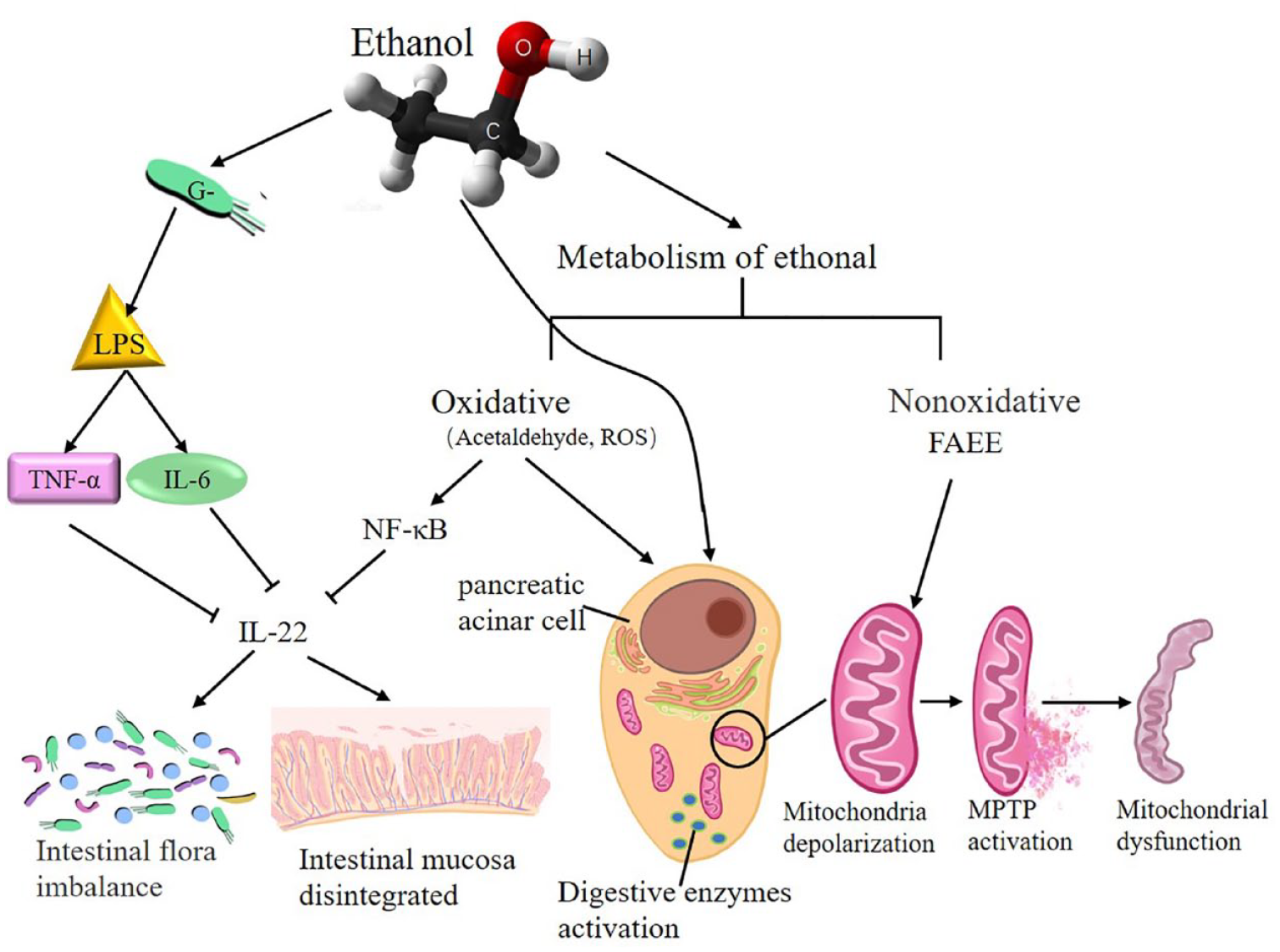
The mechanism and negative effect of ethanol metabolism on AP and GM.
High glucose and insulin resistance
Pancreatic damage, pancreatitis, imbalances in the GM, and blood sugar imbalances may be interrelated. 148 AP exhibits hyperglycemia in the early stage, 155 which can persist as secondary diabetes even after pancreatitis has been resolved. 156 Chronic hyperglycemia may cause oxidative stress, mitochondrial damage, the production of advanced glycation end products (AGEs), and the expression of the receptor for AGEs (RAGE), leading to tissue injury. 157
Researchers have shown that hyperglycemia enhances mitochondrial oxidative stress by increasing ROS production, which is a key step in the pathogenesis of AP, 158 and mediates lipid peroxidation by increasing cytosolic Ca2+.159,160 Furthermore, increased intracellular Ca2+ is also required for premature protease activation, which is an early step in the induction of AP. 158 Elevated glucose levels begin to form covalent conjugates with plasma proteins through a nonenzymatic process called AGE formation. 161 In combination with AGEs, RAGE promotes the development of pancreatitis in part by mediating uninduced nucleosome activation and proinflammatory mediator release via the absence in melanoma 2 (AIM2) inflammasome activation and proinflammatory mediator release in macrophages in an animal model of AP. 162 Under glycoxidative stress, stimulated macrophages can induce oxidative stress and NF-κB activation through activation of the PR21ras and MAPK signaling pathways. 163 Active NF-κB induces the production of TNF-α, which, in turn, leads to enhanced ROS production and more severe damage to tissues. 164 In addition, hyperglycemia was shown to compromise the integrity of the intestinal barrier through glucose transporter 2 (GLUT2)-dependent reprograming of the intestinal epithelial cell transcriptome and disruption of tight and adherence junctions, leading to intestinal flora disorders.165,166 The GM composition of patients and animals with elevated blood glucose was also significantly different from that of normal controls.167,168
Insulin resistance, which is a kind of metabolic dysfunction associated with type 2 diabetes mellitus, is another critical factor that affects both AP and the GM. 169 Observational studies have shown an increased risk of AP among people with diseases linked to insulin resistance.170 –172 Various factors and hormones, such as NF-κB, TNF-α, amylin, leptin, and IL-6, have recently been shown to be increased in patients with insulin resistance, and those factors have been demonstrated to cause AP and intestinal flora disorders.173 –177 In addition, insulin resistance often causes hyperinsulinemia, which can inhibit mucus secretion by promoting fatty acid synthase, to break the integrity of the intestinal barrier, leading to GMs.165,178 Furthermore, insulin resistance has been regarded as a novel risk factor for post-endoscopic retrograde cholangiopancreatography pancreatitis 179 and an independent prognostic factor in patients with AP. 180
Intestinal microbes can also increase insulin resistance by influencing host energy metabolism and the integrity of the intestinal barrier; thus, inflammatory mediators can be transmitted into circulation. 181
Curative substances influence both AP and GM
AP often leads to flora disorder, but some protective cytokines play key roles; for example, IL-22 and IL-23 attenuate intestinal flora disorders.152,182,183 Propolis has recently been reported to reduce the serum levels of proinflammatory cytokines (TNF-α, IL-1β, and IL-6) and increase IL-22 levels, thereby reducing pancreatic neutrophil infiltration and maintaining the intestinal flora in AP rats. 184 Store-operated calcium entry (SOCE) modulators, 185 such as Pyrtriazoles, and the Orai Ca2+ channel inhibitor CM4620, which can reduce endoplasmic reticulum calcium influx, target both parenchymal and immune cells to reduce inflammation in experimental AP. 186 By inhibiting immune cells, SOCE inhibitors can treat imbalances in the GM. Okra pectin could relieve the inflammatory response by inhibiting the expression of proinflammatory mediators, preventing intestinal barrier injury, and regulating the intestinal microbiota by upregulating AMPs and occludin in an AP model. 187 Probiotics have been reported to significantly attenuate pathological injury of the pancreas and reduce the incidence of complications, such as infection, in patients with AP. 188 However, the elevated levels of lactic acid produced by bacterial overgrowth in the small bowel and fermentation of carbohydrates significantly contributed to the high death rate. When considering substituting supplementation for individuals with AP, it is necessary to assess the time, type, appropriate, effective doses of probiotics, and prevent bacterial overgrowth.20,189,190 Some traditional Chinese medicine (TCM) treatments also have effects on both AP and the GM. Saponin A, a monomer of total saikosaponins extracted from Bupleuri Radix, has strong antioxidant properties and can affect the composition of GM by increasing the relative abundance of Lactobacillus and Prevotella species to decrease the development of SAP in rat models. 191 Picroside II is one of the main effective components extracted from Picrorhiza scrophulariiflora Pennell that can improve the intestinal microbiota by inactivating oxidant and inflammatory signals to improve intestinal barrier injury in an SAP rat model. 28 Some studies have reported that berberine can not only repair the gut barrier structure to decrease GM diversity but also reduce blood glucose levels and attenuate insulin resistance; moreover, berberine is regarded as a potential therapeutic agent for AP.192 –194 Meng et al. used acupuncture and moxibustion to stimulate ST36 points to treat SAP based on conventional treatments and found that adjuvant acupuncture treatment could reduce the permeability of intestinal mucosa capillaries, alleviate intestinal dysfunction, and promote recovery in patients 195 (Tables 3 and 4).
Curative substances influencing both AP and GM.

AMP, antimicrobial peptide; AP, acute pancreatitis; GM, gut microbiota; IL, interleukin; SOCE, store-operated calcium entry; JAK, Janus kinase; STAT, signal transducer and activator of transcription; TNF-α, tumor necrosis factor-α.
Promising prebiotic agent for the treatment of SAP.

AP, acute pancreatitis; COS, chitosan oligosaccharides; SCFA, short-chain fatty acid; TLR, toll-like receptor.
Future research prospects
SAP is a severe inflammatory disease of the pancreas and results in a high mortality rate when accompanied by multiple organ dysfunction or secondary infection. 211 Studies have shown that most pancreatic and extra-pancreatic organ infections originate in the intestine and induce inflammatory responses, which are major causes of ‘secondary attack’ and increased late death of patients with SAP. 212 Changes in the GM play an important role in intestinal homeostasis and aggravate the inflammatory response under intestinal flora dysfunction in AP.4,5 The migration and proportion of intestinal flora influence the development and severity of AP. However, the molecular mechanism and signaling pathways associated with changes in the intestinal flora in AP are still unclear. 213 The dominant intestinal microbiota species in MAP, MSAP, and SAP were Bacteroides, Escherichia–Shigella, and Enterococcus, respectively. 20 A majority of diseases are accompanied by changes in the microbiota, and whether there is a way to detect GM species could be helpful in predicting or diagnosing SAP. 214
Obesity and hyperlipidemia are regarded as chronic and systemic inflammatory states induced by adipocytes, which secrete a variety of proinflammatory cytokines and act as reservoirs of inflammatory factors. 23 When obesity and hyperlipidemia cause AP and an intestinal microbiota imbalance, 115 the intestinal microbiota also causes disordered lipid metabolism and the development of obesity by mediating adipocytokine gene expression, leading to a vicious cycle. 140 Pancreatic endocrine cells participate in the regulation of blood glucose metabolism. Hyperglycemia exacerbates mitochondrial oxidative stress, increases intracellular Ca2+ levels, and ultimately promotes the progression of AP.158,160 Patients with AP generally have insulin resistance,80,83,86 and gut microbes have been reported to increase insulin resistance. 181 Insulin resistance also causes AP and intestinal microbiota disorders.85,215,216 Glucose and lipids are sources of energy metabolism and are also factors associated with metabolic diseases. The specific GM species in AP combined with the metabolic disorders associated with glucose and lipids need further study. The effect of probiotics on the treatment of AP combined with metabolic disorders associated with glucose and lipids might be worth studying.31,161
There is currently no specific treatment for AP. The intestinal flora attenuates the severity of AP, and personalized probiotic intervention is considered a future trend. 217 The timepoint, dose, and effectiveness of probiotics used for the treatment of AP are worthwhile of further experiments and clinical studies. In addition, the safety issue of probiotic therapy cannot be ignored. 218 TCM, with multiple approaches including decoctions, powders, acupuncture, and moxibustion, has been reported to improve inflammatory or metabolic disorders.219 –222 Whether treatments combining probiotics and TCM could be beneficial for SAP patients or whether the curative factors mentioned above may be used to prevent pancreatitis are unclear, and few studies have focused on this issue.
In conclusion, the interaction between the GM and inflammatory responses provides a new understanding of AP disease progression and treatment. Further studies on the interaction of GM and inflammatory responses in AP are needed.
Footnotes
Acknowledgements
The authors thank Hui-Juan Chen at Chengdu Qiantu Culture Communication Co., Ltd., for preparing the figures included in this paper.
