Abstract
The coronavirus disease 2019 (COVID-19) pandemic presented unique challenges to patients with decompensated cirrhosis awaiting transplant, with respect to accessing medical facilities for routine clinic visits, imaging, laboratory workup, or endoscopies. There was a delay in organ procurement that led to a decrease in the number of liver transplants (LTs) and an increase in the morality of waitlisted patients at the beginning of the pandemic. LT numbers later equalized to pre-pandemic numbers due to combined efforts and adaptability of transplant centers as well as dynamic guidelines. Due to being immunosuppressed, the demographics of LT patients were at an increased risk of infection. Although there is a higher rate of mortality and morbidity in patients with chronic liver disease, LT itself is not a risk factor for mortality in COVID-19. There was no difference in overall mortality in LT patients compared to non-LT patients, and mortality risk factors were the same: age, hypertension, diabetes, obesity, and chronic kidney disease. The most common causes of death were respiratory complications. Liver-related deaths were reported in 1.6% of patients. The optimal timing of liver transplantation post-infection depends on various factors, such as the severity of liver injury, the presence of comorbidities, and the progression of the underlying liver disease. There is not enough data available on COVID-19 cholangiopathy and the number of cases that will be seen in the future that will require LT. There are some concerns of lower immunogenicity of COVID-19 vaccines in LT patients but available evidence suggests that the vaccines are safe and well-tolerated.
Keywords
Introduction
The coronavirus disease 2019 (COVID-19) has impacted nearly every organ system. Initially, COVID-19 invoked fear and anxiety due to the poor understanding of the virus and its vast burden of disease. While the knowledge surrounding COVID-19 has become less evasive, there remain significant concerns regarding the impact of COVID-19 as it relates to one of the most vulnerable populations, liver transplant (LT) candidates and recipients. In fact, there is an established 3.5-fold increase in mortality in patients with cirrhosis and COVID-19, compared to those without cirrhosis. 1
Unique challenges exist in the field of liver transplantation. Patients with decompensated cirrhosis may require frequent paracenteses. Inevitably, though performed in the outpatient setting, patients could be exposed to nosocomial pathogens. Similarly, variceal screening that requires endoscopy could also increase the risk of and exposure to similar pathogens including COVID-19. However, on the contrary, issues surfaced surrounding access to endoscopy when endoscopy suites were short-staffed and had strict COVID-19 testing policies. Patient concerns regarding accessing medical facilities for routine clinic visits or radiology appointments for hepatocellular carcinoma screening also came to light. Similar to how hospitals adapted to the pandemic with changes to visitation policies, transplant centers adapted by changing listing meetings from in-person to virtual.
In this review article, we will outline the pathophysiology of COVID-19 and hepatic involvement, role of immunosuppression medications, and the impact of COVID-19 on outcomes related to transplantation.
Pathophysiology of interaction between COVID-19 and the liver
The virus causing COVID-19, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), acts via binding to the angiotensin-converting enzyme-2 (ACE2) receptors which are predominantly present in the lungs. These act as a point of viral entry and are also highly expressed in the endothelium, arteries, veins, and type 2 alveolar cells. The other locations where ACE2 is highly expressed include the gastrointestinal tract in the colonic smooth muscles and the enterocytes of small intestine. In the liver, they are found mainly in the cholangiocytes, to a similar level seen in alveolar cells. 2
COVID-19 exerts its effect on the liver in a multi-factorial fashion. These could be due to the direct cytopathic effect of the virus, drug-induced liver injury, uncontrolled systemic inflammation, or sepsis. There is no clear evidence to suggest that patients with underlying chronic liver disease are at an increased risk to acquire COVID-19 infection. However, studies have shown that 2–11% of all COVID-19 patients have an underlying liver pathology. 3
Importantly, studies have shown a higher rate of mortality and morbidity in patients with chronic liver disease. 1 A large meta-analysis pooling 64 studies and 11,245 patients has shown that about 25% of patients with COVID-19 also had elevated bilirubin, aspartate transaminase, and alanine transaminase in patients. Prior hepatic dysfunction was present in about one-third of patients. 4 Cholestasis has been infrequently seen, despite the presence of ACE2 receptors in cholangiocytes, suggesting a direct inflammatory process involving the liver, rather than the virus itself. 5
Role of immunosuppression
SARS-CoV-2 causes a dysregulation of the innate immune system by systemic inflammation via interferon types 1 and 3. This leads to activation of other cytokine signaling pathways comprising of interleukins (IL-6, IL-8), thus resulting in a cytokine storm.6,7 There is overall T-cell activation, lymphocyte dysfunction, granulocyte and monocyte activation, and increased antibody generation. 8
Immunosuppressive therapy mitigates T-cell function and the subsequent cytokine-based inflammation. It is also hypothesized that drugs such as cyclosporine and tacrolimus could inhibit viral replication by suppression of the immunophilin pathway. 9 Mycophenolate mofetil, a key component in the first 6–12 months of post-transplant immunosuppression, has also been shown to inhibit SARS-CoV-2 replication in vitro. 10 This probably leads to a diminished response and a less severe clinical course in these patients. The potential protective effect of immunosuppressants against the host’s immune system may also have been the basis for development of newer drugs like tocilizumab. 11 An important consideration is the potential for these patients to become a source of asymptomatic viral shedding due to their milder course of disease.12,13
The standard of care for treatment of COVID-19 is supportive treatment along with dexamethasone for those with significant hypoxia. 14 Remdesivir was initially used only in hospitalized patients with significant hypoxia. However, at some centers, it is now used routinely for any immunosuppressed patient with COVID-19. 15 Immunosuppressive therapy should be started and continued in all LT patients, even in patients with COVID-19; however, protocols are center specific. Specific consideration should be given to patients with any decrease in cell counts. For example, lymphopenia can represent severe or progressive disease, in which case anti-metabolites like mycophenolate mofetil are held.10,16 The use of mTOR inhibitors such as sirolimus, everolimus, and temsirolimus may cause concomitant drug-induced pneumonitis. In patients who develop ground glass opacities, changing to calcineurin inhibitors should be considered. 17 In patients who develop superimposed bacterial or fungal infections, modification of immunosuppression with reduction in calcineurin inhibitors and withholding anti-metabolites may be beneficial.
Impact of COVID-19 on liver transplantation
In the early 2020 period, there was a decline in the number of LTs performed and the number of patients listed, with a concomitant rise in the number of inactivated patients. 18 This observed decline correlated with the infection rates across the United States. There was an approximate 30% decline in the number of LTs and a subsequent 60% increase in the morality of waitlisted patients. 19 Strauss et al. used the Scientific Registry for Transplant Recipients data to analyze the LT waitlist in the United States between March 2020 and August 2021 and compared it with the trends prior to that from 2016 to 2020. New listings decreased by 11%, living donor LTs (LDLTs) decreased by 49%, and deceased donor LTs decreased by 9%. The outcomes of patients on waitlists were worse in states with higher incidences of COVID-19 with 59% more waitlist deaths. These numbers were a result of various safety measures in place as well as the logistical challenges that the pandemic presented. However, by August 2020, the waitlist outcomes were similar to expected trends in the period prior to the pandemic attributed to the adaptability of various centers. 20
Organ procurement
According to the Center for Medicare Services, solid organ transplantation falls under tier 3b and is considered a high equity surgery which should be continued regardless of indications to postpone other elective surgeries. Decompensated patients with high Model for End-Stage Liver Disease (MELD) scores were disproportionately affected by the pandemic.21,22 There was an overall decline in the rates of organ recovery, with a marked decrease in living donations. During the initial period from February 2020 to April 2020, there was a 25% decrease in the number of deceased donor livers that were recovered.23,24 Reasons for this are unclear; however, we surmise that this could be a result of the rapidity of disease progression along with the strict visitor policies that hindered the organ donation evaluation process, including taking appropriate consent.
One of the major policy changes by the United Network for Organ Sharing (UNOS)/Organ Procurement Transplant Network (OPTN) included three new refusal codes related to COVID-19-related issues in potential recipients, donors, and operational issues. Refusal code 840 included candidate-related reasons like potential exposure, symptomatic patients, patients being tested, and positive test result. Donor-related reasons were included under refusal code 841: donors with high exposure risk, donors where testing is not available, positive, or indeterminate test results, or when a different specimen is preferred. Operational issues included under refusal code 842 were organ recovery scheduling issues, operating room or ICU bed shortages, personnel, or other ancillary services shortages. 25 These codes have been increasingly used and resulted in an increase in rates of waitlist inactivation. COVID-19-related inactivated registrants now account for about 5% of the national waiting list (Figure 1).

Factors affecting organ procurement during COVID-19.
Safety and logistical considerations
An overall shortage of hospital beds, ventilators, operating room schedules, and blood products resulted in unprecedented barriers to potential organ donor evaluations (Figure 1). In addition, the restrictive family visiting policies adopted by most hospitals during the peak of the pandemic posed an obstacle to obtaining consents in a timely manner. Another significant challenge was the travel of surgical teams to perform recovery procedures at donor centers. This was augmented by the interruption in commercial airline travel that disrupted the transportation of organs. 20 This led to new recommendations by the UNOS/OPTN as well as American Society of Transplant Surgeons for more reliance on local donor recovery teams. 26 This requires increased communication between teams and organ procurement organizations (OPOs). The use of technology for digital imaging, and digital microscopy where images are readily and immediately transferred to a computer, can help expedite clinical evaluation. Secure video sharing platforms like DonorNet allow OPOs to add health data from donors, perform a match run, and to respond to organ offers. 24
COVID-19 testing, and history of infection, is now a part of the routine evaluation of donors and recipients. The limitations of testing lie in the variability in false-negative results ranging from 2% to 40% for nasopharyngeal samples, although PCR tests now have increased accuracy with specificities reported to be upwards of 97–99%. 27
Studies have a shown a significantly higher prevalence of fear of acquiring COVID-19 in solid organ transplant recipients and solid organ transplant candidates when compared to their household controls. The fear of infection was higher in females and individuals on higher immunosuppression regimens (steroids or triple agent immunosuppression). There was also a fear of contracting COVID-19 in medical facilities. 28
An important policy change by the UNOS/OPTN was allowing transplant centers to use existing laboratories to extend MELD listing over the period of the pandemic to decrease potential exposure in the form of travel to laboratories and clinics. 29
As we continue to adapt to some of these new barriers, telehealth has played a central role in assessing patients and for coordination between donor teams and transplant centers. Nearly 85% of clinic visits were converted from in-person to virtual. 30 In some centers, patients had telehealth visits 2 weeks post-transplantation with an in-person visit thereafter, for suture removal. Other healthcare workers including pharmacists and transplant coordinators scheduled telehealth visits as well. 30 The Extension for Community Healthcare Outcomes project designed to guide treatment of Hepatitis C via weekly videoconferences with specialists to discuss treatment plans and can be used as a model to develop robust telemedicine multidisciplinary programs for the care of LT patients. 31
LT outcomes
During the initial phase of the pandemic, the effect of COVID-19 on LT patients remained unclear. However, with emerging numbers, there are more data to identify risk factors to predict severe COVID-19 infections among transplant patients.
The studies described below demonstrate that LT itself is not a risk factor for mortality in COVID-19 but age and coexisting morbidities such as hypertension, diabetes, obesity, and chronic kidney disease have been shown to increase the mortality risk in patients awaiting LT (Figure 2).

Factors associated with increased mortality in LT patients with COVID-19.
Several meta-analyses have been conducted to understand the outcomes of LT patients affected with COVID-19. Kulkarni et al. pooled data from 17 studies and 1481 LT patients infected with COVID-19. The cumulative mortality was 17.4% in LT patients but was comparable between LT and non-LT patients. Interestingly, the overall mortality rate in LT patients was lower than that in other solid organ transplantation groups. 32
The most common causes of death were COVID-19-related complications followed by respiratory failure. Liver-related deaths were reported in 1.6% of patients. There was no difference in mortality based on time of infection (within 1-year of transplant versus 1-year post-transplant).
Higher percentage of LT patients with infection was hospitalized than non-LT patients, but the rates of ICU admission were similar. 33 While these studies showed comparable outcomes in mortality between LT and non-LT groups, a study by Colmenero et al. observed contrasting results. This study was carried out in Spain and included 111 LT patients with COVID-19. The incidence was almost double in LT patients, but the mortality rate was 18%, which was lower than the matched general population. 34 This could be attributed to the immunosuppressed state of patients that increases the risk of infection but could reduce the severity of disease. It can be concluded that the overall mortality from COVID-19 was comparable or even lower in LT patients. The higher rate of infections is an important indicator to reinforce preventive measures, especially in this population.
Several studies were conducted across the world in the initial stages of the pandemic to explore the factors associated with mortality in LT patients with COVID-19. A study from Lombardy, Italy in April 2020 of 150 LT patients, six of whom were affected by COVID-19, of which three patients died. Death was more frequently associated with long-term LT patients (defined as LT > 10 years ago) rather than recent transplants, patients greater than the age of 65, male, and overweight patients. 35 In congruence with COVID-19 mortality in the general population, the most common comorbidities associated with mortality in LT patients were hypertension and diabetes34,36,37 (Table 1).
Mortality in LT patients with COVID-19.

COVID-19, coronavirus disease 2019; LT, liver transplant.
Less commonly, chronic kidney disease was also a factor. Given the commonality of these predictors in the non-LT population, LT and immunosuppression might not significantly alter the outcomes of disease in severe COVID-19. Thus, metabolic comorbidities and conditions can increase the mortality risk. These factors are modifiable and can be targeted in waitlisted pre-transplant patients to improve outcomes. As expected, when deaths occurred in LT patients with COVID-19, it was mostly due to respiratory complications.36,37
Acute liver injury was also significantly and independently associated with higher mortality. 35 It is unclear whether these cases were due to underlying comorbidities, or the effect of the virus.
Timing of LT after COVID-19 infection
There has been a huge difference in the severity of patient symptoms with advancement of medical treatment and availability of vaccines. With an overall improvement in therapeutic options and increasing availability of vaccines, the survival rate of hospitalized COVID-19 patients has improved. 38 This has resulted in an overall improvement in outcomes of hospitalized patients and more who were affected with severe disease being eligible for advanced therapies like transplant.
There are limited data available to guide the timing of liver transplantation after COVID-19 infection. The optimal timing of liver transplantation depends on various factors, such as the severity of liver injury, the presence of comorbidities, and the progression of the underlying liver disease.
In general, liver function should be monitored for a period in anticipation of recovery from the acute inflammatory state caused by COVID-19 infection. It is also important to allow sufficient time for the evaluation of any potential long-term effects of COVID-19 on the liver.
Patients who have recovered from mild to moderate COVID-19 can typically undergo transplantation within 3–6 months of their recovery, in the absence of ongoing respiratory or cardiovascular decompensation; however, these policies are center specific. Patients with severe COVID-19 who may have persistent respiratory or cardiovascular dysfunction may require a longer recovery period before transplantation.
With respect to immunosuppression, the use of mycophenolate was associated independently with severe COVID-19. Complete withdrawal of immunosuppression upon hospital admission did not play a role and was not significant. 34 The use of tacrolimus was shown to have a protective effect according to some studies. 39 However, due to a lack of unifying guidelines regarding holding immunosuppression in LT and COVID-19 patients, it is difficult to conduct large-sized studies to validate the above findings.
Future direction: LT for COVID-19-associated cholangiopathy
Some degree of liver injury has been reported in nearly half of the patients affected by COVID-19. 40 Several case reports have portrayed the rapid progression of liver injury leading to COVID-19-related cholangiopathy. It is considered a variant of secondary sclerosing cholangitis in critically ill patients with diffuse intrahepatic biliary strictures. 41
These cases with acute liver injury may need liver transplantation sooner. The presence of pre-existing liver injury may have an exaggerated hepatic injury, which may necessitate early transplant.
It is important to note that the decision to proceed with liver transplantation should be made on a case-by-case basis, considering the individual patient’s clinical circumstances and the recommendations of a multidisciplinary team.
It is difficult to predict with certainty how cases of cholangiopathy due to COVID-19 will affect the number of LTs will increase in the future. This would depend on various factors such as the evolution of the pandemic and advances in medical treatments.
Acute liver injury after vaccination has been reported in some cases as summarized in Table 2.
Cases of acute liver injury after vaccination.
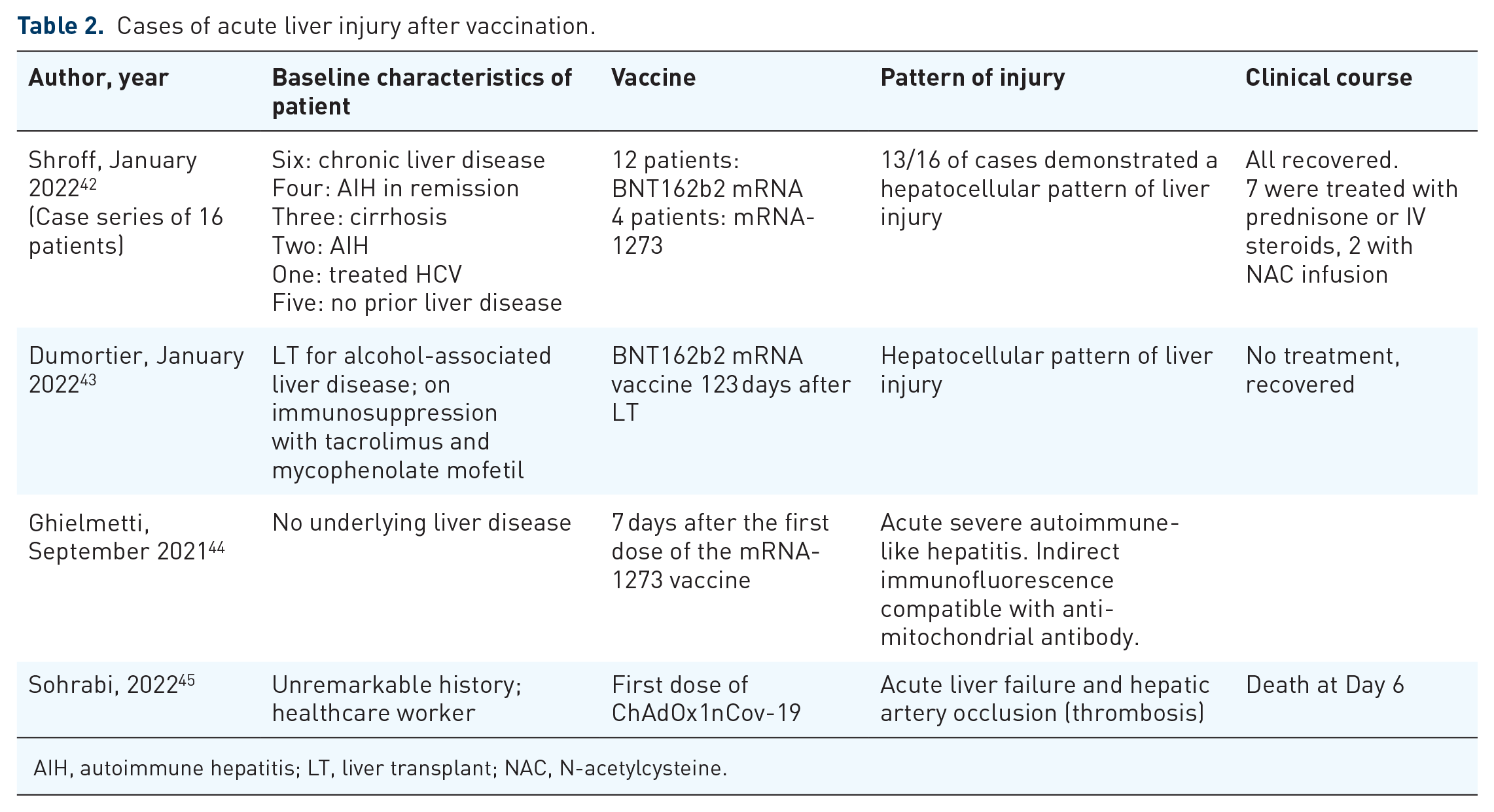
AIH, autoimmune hepatitis; LT, liver transplant; NAC, N-acetylcysteine.
Clinical practice guidelines
The American Association for the Study of Liver Diseases guidelines recommended limiting clinic visits and evaluation of patients with non-severe liver disease. Priority was given to patients with an urgent indication for LT such as patients with hepatocellular carcinoma and high MELD scores. Establishing center-specific policies to triage patients listed for LT that need to be seen in person was recommended. UNOS/OPTN recommended limiting patient visits to laboratories by setting up policy changes. When updated MELD laboratories were not available, candidate MELD scores could be calculated using existing laboratory tests. In-person visits for purposes of updating the MELD score were advised against. Established and stable patients on chronic medications were provided with 90-day supplies with early medication refill and home-delivery options. Patients enrolled in recovery support meetings such as alcoholics anonymous or other transplant-related services such as patient transplant education, dietitian, and financial consultations were encouraged to attend meetings virtually rather than in-person. Other measures like avoiding overcrowding as well as travel were recommended.33,46
Earlier in the pandemic, the Belgian Liver Intestine Transplant Committee (BeLIAC) prohibited the use of COVID-19-positive donors due to the risks to patients and healthcare staff. 47 They also recommended testing of all donors. BeLIAC also recommended LT should be given priority access to non-COVID resources, considering that this is a life-saving surgery. They also adhered to the standard MELD allocation depending on its objectivity and transparency, except for patients with very high MELD for non-oncological etiologies and preserved liver function. Waitlisted patients for LT were to be informed of the possible risks of COVID-19 infection post-LT and the possibility to temporarily opt out from surgery on a case-by-case basis. The risk and benefit ratio of mortality associated with being on the waiting list without transplantation versus potential to acquire COVID-19 infection peri-operatively in the hospital was to be discussed with patients to individualize the decision. 47
Due to limited resources, it was also recommended to consider the expected outcomes while selecting patients for LT. Pre-LT, the evaluation of LT patients was not deferred unless for quality-of-life reasons. LDLT was postponed in stable patients. 47
Vaccination recommendations
Introduction of vaccines turned out to be most cost-effective intervention with the evolution of the pandemic. As the response to vaccine depends on the response of the functioning immune system, this posed several new questions in patients with LT.
The safety of COVID-19 vaccines in LT recipients has been studied extensively. Several studies have shown that COVID-19 vaccines are safe and well-tolerated in this population.
Most initial clinical trials on COVID-19 vaccines excluded immunosuppressed patients, including LT patients, leading to lack of safety and efficacy data. One of the major mRNA-based vaccines Pfizer-BioNTech (BNT162b2) was shown to have minimal and mild adverse events in solid organ transplant recipients. 48 The immunogenicity of COVID-19 vaccines in LT patients has been investigated by measuring the response to vaccination to major mRNA vaccines. The immune response was lower in LT recipients but a positive immune response was seen in most patients, as indicated by a significant increase in antibody levels (Table 3).
Review of studies describing immunogenicity of various vaccines.
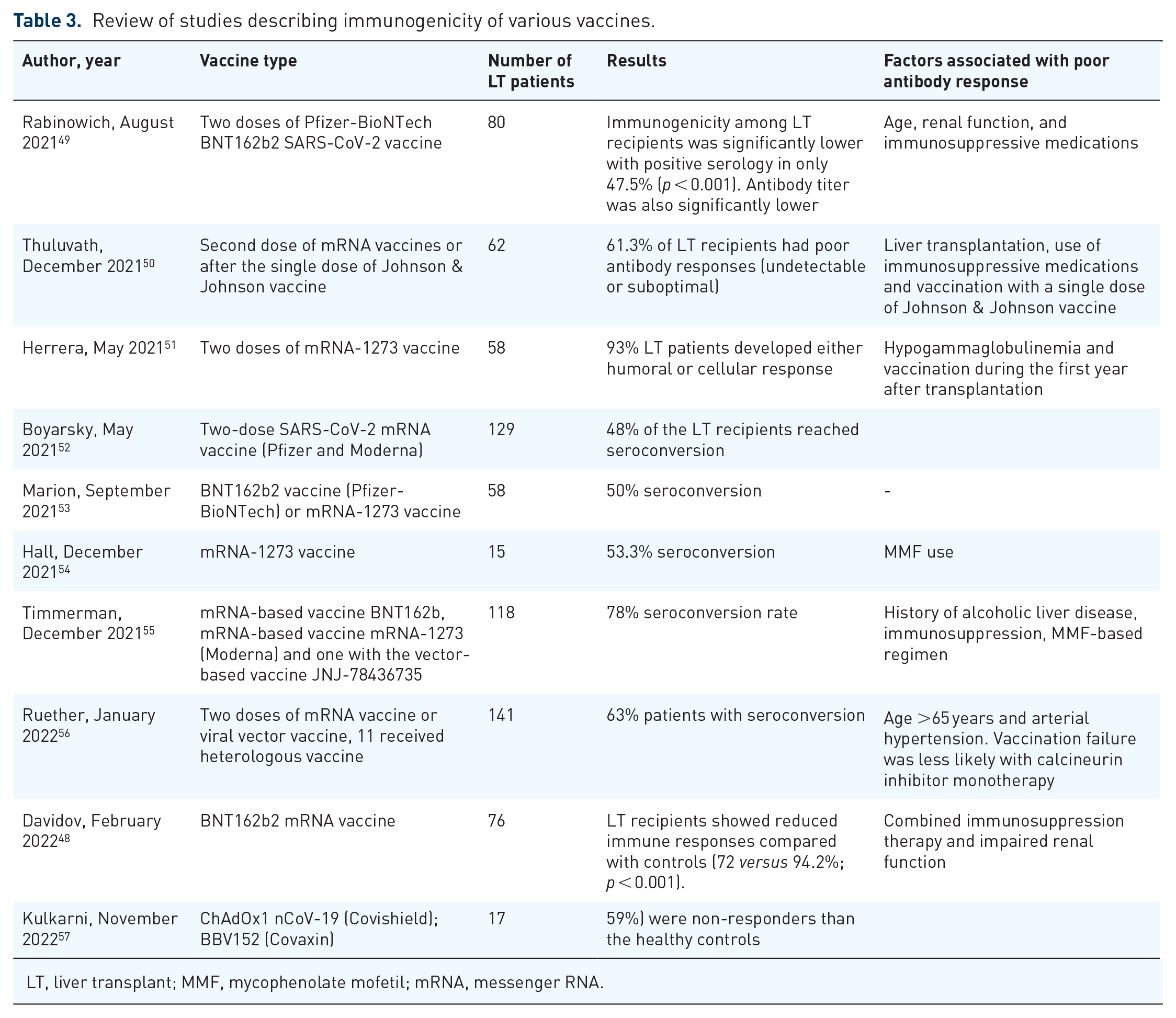
LT, liver transplant; MMF, mycophenolate mofetil; mRNA, messenger RNA.
Rabinowich et al. studied the immunogenicity of the Pfizer-BioNTech BNT162b2 SARS-CoV-2 vaccine in LT recipients. This study showed that only 47.5% of patients developed neutralizing antibody titers, 10–20 days after receiving the second dose. No serious adverse events were reported. Similar findings were recorded in several other studies, with immunogenicity varying between 47.5% and 93%, as described in Table 3. Factors associated with lower immunogenicity in LT recipients to SARS-CoV-2 vaccine BNT162b2 included age, renal dysfunction, and alcohol use disorder prior to transplant. Immunosuppression in general and mycophenolate mofetil was associated significantly with an inferior response.58,59
Most of these studies reported no serious adverse events and in the presence of adverse reactions, they were mild to moderate and not requiring hospitalization (Tables 3 and 4).
Review of vaccine safety data.
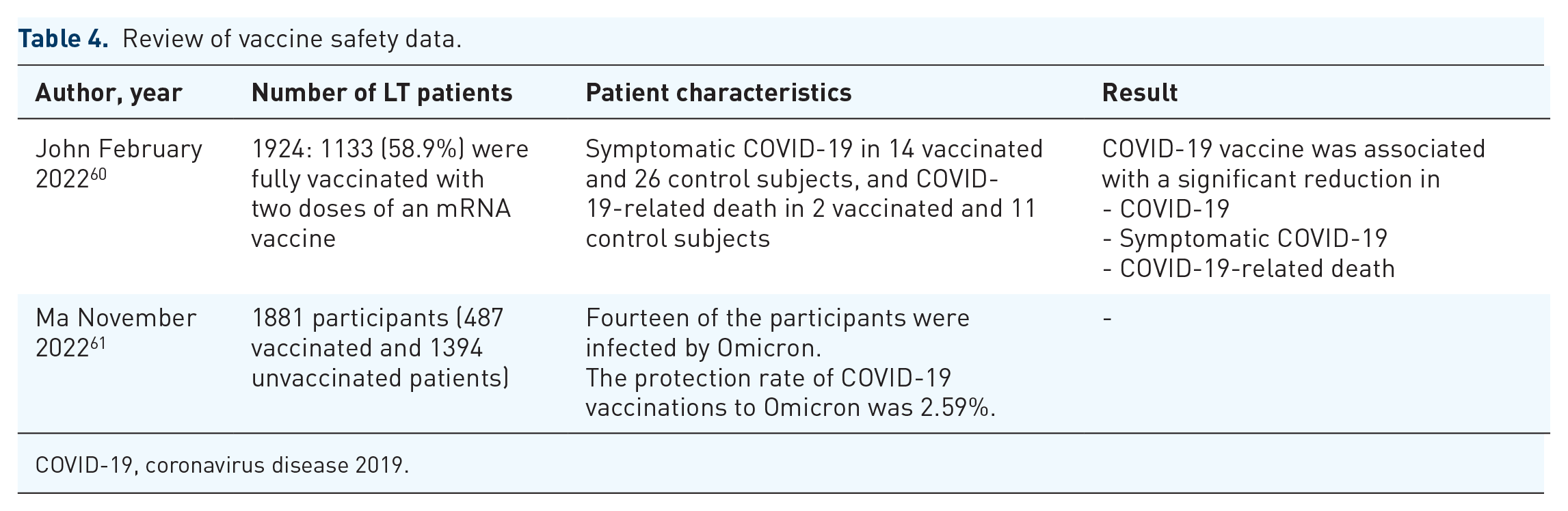
COVID-19, coronavirus disease 2019.
The available evidence suggests that COVID-19 vaccines are safe and well-tolerated in this population. In conclusion, based on the current evidence, LT recipients can receive COVID-19 vaccines with confidence and should follow the recommendations of their healthcare provider.
The use of heterologous vaccines (combination of rAd26/mRNA-1273 (Sputnik/Moderna) was studied by Mendizabal et al. The combination resulted in a higher level of anti-spike IgG in LT patients, when compared to the homologous regimens. 62 Conflicting data were demonstrated in another study that showed a lower humoral response and lower seropositivity in the heterologous group. However, this study including all solid organ transplantation patients. 63
The timing of vaccination is important, and it is recommended that all vaccination should be completed prior to transplantation or at least at the time of listing. Due to the high doses of immunosuppression post-LT, vaccination is usually not recommended in the first 3–6 months.
There is no evidence to suggest contraindication to COVID-19 vaccines in patients with chronic liver disease, hepatobiliary cancer, or immunosuppressed patients with LT. 64
European Association for Study of the Liver (EASL) initially recommended weighing the potential risks and benefits of vaccination in all LT patients. The risks include the detrimental consequences of severe COVID-19 infection and the benefits extended not just to patient’s health, but also the utilization of healthcare resources. EASL also recommended prioritization of vaccination in household members of patients with cirrhosis, hepatobiliary cancer, and LT patients. 65
It was found that the acceptance rates of mRNA-based vaccines (BNT 1622b2, Pfizer-Bio-NTech) were very high at 96.6%, higher than expected in the general population. 66 Among the 283 LT patients that were evaluated, only five patients refused vaccination due to concerns of severe adverse events despite receiving adequate information. Therefore, counseling and dissemination of information regarding vaccines is key in this vulnerable population.
With development of various vaccines with diverse modes of immunogenicity, further studies are required to guide the duration and frequency in LT patients and if this is different from other healthy individuals.
Conclusion
The liver transplantation community was highly impacted during the COVID-19 pandemic by the unique challenges involving organ donation, procurement, immunosuppression, access to elective appointments and procedures. Hepatic manifestations of COVID-19 were seen in the form of elevated liver enzymes, but rarely acute liver failure. It was hypothesized that the systemic inflammatory process of the disease contributed more to the hepatic dysfunction than the cytopathic effect of the virus itself. It was found that liver transplantation patients on immunosuppressive therapy experienced a less severe course of disease due to suppression of T-cell function and the cytokine storm. There was a decrease in the number of LTs performed and the number of patients listed. There was also a concomitant rise in the number of patients inactivated from the waitlist. This was a result of decreased organ procurement, decreased access to organs, policy changes encompassing refusal codes, isolation and visiting policies by hospitals, and shortage of equipment and staff.
There is no overall difference in mortality between LT and non-LT recipients affected with COVID-19. 67 Future efforts should focus on modifiable factors like vaccination that could potentially affect patient-related outcomes and long-term outcomes of patients with COVID-19-related cholangiopathy. The impact of vaccination has been similar to that of general population with less severe infection in vaccinated patients. COVID-19 vaccines have been shown to be safe in LT patients and other solid organ transplant recipients. Challenges remain as more prospective, multi-institutional studies are needed to better understand the unique impact of COVID-19 on LT patients.
