Abstract
Background:
The application of vedolizumab (VDZ) subcutaneous (SC) formulation has brought more convenience and hope to patients with moderate-to-severe inflammatory bowel diseases (IBDs) in the coronavirus disease 2019 context.
Objective:
This study aimed to systematically evaluate all previous studies that used VDZ SC formulation for maintenance therapy in patients with IBD.
Design:
Systematic review and meta-analysis.
Data Sources and Methods:
The search was conducted using the subject and free terms related to ‘Vedolizumab’, ‘Subcutaneous’, and ‘IBD’, in Embase, PubMed, Web of Science, Cochrane, and at ClinicalTrials.gov databases between 2008 and 2022. The methodological quality of randomized controlled trials (RCTs) and cohort studies was assessed using the Cochrane Handbook of Systematic Reviews and the Newcastle–Ottawa Scale, respectively. The endpoints included efficacy, safety, and immunogenicity.
Results:
A total of 60 studies and 2 completed clinical registry trials were retrieved, of which 3 RCTs with high methodological quality, and 3 cohort studies with large heterogeneity were included in the meta-analysis. In the RCT study design, patients with ulcerative colitis (UC) under different conditions after treated with VDZ SC were significantly distinct than those for placebo (PBO) in clinical remission, endoscopic remission, and biochemical remission. In Crohn’s disease (CD), the aforementioned parameters were slightly higher than those for PBO, but there was not statistically significant in endoscopic remission and the efficacy of anti-tumor necrosis factor-naive patients. The clinical remission, endoscopic remission, and biochemical remission in patients with UC after VDZ SC treatment were similar to those after intravenous (IV) treatment. The risk ratios in patients experiencing adverse events (AEs) and serious AEs after VDZ SC and PBO treatments were 86% and 89% in UC, and 96% and 80% in CD, respectively. Compared with IV, safety was not statistically different. The risk of developing anti-VDZ antibody after VDZ SC treatment was only 20% of that after PBO in patients with UC, but it was 9.38 times in CD.
Conclusion:
VDZ SC treatment maintained the clinical efficacy of IV induction in patients with IBD without increasing the safety risk, and the efficacy was more pronounced in patients with UC. Immunogenicity might be a potential factor for the decrease in efficacy rate in patients with IBD.
Registration:
INPLASY 2022120115
Introduction
Vedolizumab (VDZ) is a fully humanized monoclonal antibody that targets the migration and adhesion of leukocytes in the inflamed intestine by specifically antagonizing α4β7 integrins and blocking their binding to the intestinal mucosal addressin cell adhesion molecule 1.1,2 VDZ was first investigated for the treatment of active Crohn’s disease (CD) in 2008 under the name MLN0002. 3 As a novel, gut-specific biologics, VDZ was highly anticipated by gastroenterologists and patients with inflammatory bowel diseases (IBDs). After several preliminary clinical trials, VDZ was approved by the U.S. Food and Drug Administration in May 2014 and has since been marketed worldwide. 4 It is used to treat adult patients with moderately to severely active ulcerative colitis (UC) or CD who are intolerant, no longer responsive, or have an inadequate response to conventional therapy or tumor necrosis factor (TNF)-alpha antagonists.
A large number of high-quality studies suggest that VDZ is superior to placebo (PBO) as induction and maintenance therapy for IBD.5–7 VDZ therapy for patients with UC is more efficient and longer term than other therapies. 8 Likewise, it has a significant effect and no increased safety risk in the elderly, children, pregnant women, and fetuses.9–14 GEMINI and other clinical trials demonstrated that the efficacy of VDZ was similar to that of other TNF inhibitors in clinical remission and endoscopic mucosal healing in patients with moderate-to-severe IBD and could still stimulate a clinical response in patients with IBD when other drugs failed.15–17 VDZ was found to be safe and effective for treating ileal pouchitis that developed after anal anastomosis during the treatment of patients with UC. 18 As a first-line biological agent in moderate-to-severe UC, VDZ may be superior to infliximab in terms of the durability of efficacy. 19 The safety profile of VDZ is excellent in both short-term and long-term treatments. 20 Moreover, VDZ is the safest compared with other biologics and small-molecule drugs.21,22 Early intervention with VDZ may be associated with lower surgical rates; surprisingly, the preoperative use of VDZ in patients with IBD is relatively safer and does not increase the risk of overall postoperative complications compared with non-VDZ therapy.23,24
In recent years, the prevalence of IBD has stabilized at approximately 0.3–1% in western countries. 25 However, the prevalence of IBD continues to increase in the ever highly socialized eastern countries, and patients’ symptoms are more complex in different regions.26,27 The high prevalence rate of IBD increases the burden of healthcare costs and other negative socioeconomic impacts, with annual healthcare economic costs of approximately €5400 (CD) and €3900 (UC) per case of IBD in Europe. 28 VDZ can significantly enhance the efficacy and cost savings compared with golimumab. 29 However, significant cost savings can be derived when VDZ is used as a first-line treatment option for UC and CD rather than just as a second-line therapy. 30
VDZ is administered primarily by intravenous (IV) drip. However, among drugs such as infliximab, adalimumab, golimumab, and VDZ, VDZ IV treatment cannot be cost-effective in patients with moderate-to-severe UC. 31 Even patients have to spend a long time on treatment, usually 3 h or more. Patients with IBD must make regular hospital or clinic visits over a long period of time, but patients with IBD have restricted travel since the onset of coronavirus disease 2019 (COVID-19). Fortunately, Takeda has developed a subcutaneous (SC) formulation of VDZ that can be self-administered, which can be an excellent option for treating patients with moderate-to-severe IBD. Preliminary pharmacokinetic studies have concluded that the switching from IV to SC administration requires an adjustment of the dose and frequency of administration from 300 mg once 8 weeks (Q8W) to 108 mg once 2 weeks (Q2W) to achieve similar serum drug concentrations. 32
VDZ SC therapy is low-level cost-effective than conventional therapy but has shown high-level cost-effectiveness in patients who are not effective with anti-TNF. 33 Physicians are convinced that SC injections are the better choice if they are effective and safe. Patients, moreover, showed a preference for convenient and time-saving SC therapy after obtaining a clinical response to IV induction. 34 Many clinical efficacy studies and patient intention surveys on VDZ SC have been conducted to date. High-quality studies that summarize the clinical data on VDZ SC maintenance therapy in patients with IBD are still lacking. This study aimed to provide a more convenient, effective, and safe treatment option for patients with IBD by systematically analyzing all current randomized controlled studies reporting VDZ SC maintenance therapy for IBD and other trials with complete outcomes.
Materials and methods
Registration
The study protocol was registered in International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY), with registration number: INPLASY2022120115.
Data sources and searches
This system evaluation followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses statement (PRISMA).
35
Targets included studies completed and published in the Embase, PubMed, Web of Science, and Cochrane databases and at ClinicalTrials.gov. Two key search terms, ‘P’ and ‘I’, were selected in the target study PICO-TS (population, intervention, comparators, outcomes, timing, and setting) features. The keywords included subject and free terms related to ‘Vedolizumab’, ‘Subcutaneous Injections’, and ‘Inflammatory Bowel Diseases’ and were obtained
Selection criteria and data extraction
The inclusion criteria were as follows: (1) P: patients diagnosed with IBD or IBD treated with VDZ as directed by their physician; (2) I: intervention: VDZ SC; (3) C: if a control group, then VDZ IV and/or PBO intervention; and (4) O: the outcomes of the included studies in line with the review value. The parameters included efficacy (clinical remission, endoscopic remission, and biochemical remission), safety/tolerability [adverse events (AEs), serious adverse events (SAEs)], pharmacokinetics (serum VDZ concentrations), and immunogenicity [anti-VDZ antibody (AVA)]; (5) S: randomized controlled trials (RCTs), non-RCTs, observational cohort studies, retrospective studies, case series, reviews, meta-analyses, and so on. The exclusion criteria were as follows: repeated reports, animal experiments, clinical studies in non-VDZ SC treatment groups, and related reports unrelated to clinical practice. The aforementioned criteria were jointly formulated by the two researchers and implemented independently. In the case of disagreement, a third researcher was involved in decision-making.
The basic data were extracted using Excel, including authors, time of publication, type of study, participants, and results. The inclusion criteria were jointly determined and independently extracted by the two researchers, and the extracted details were cross-checked to prevent errors. Eligible studies were considered in the meta-analysis.
Study outcomes
The outcomes needed to be defined separately with reference to the included studies.36–41 The primary efficacy endpoint was clinical remission (defined as a total Mayo score of ⩽2 and no individual subscore of >1). The following scores were considered: Harvey–Bradshaw index (HBI) score ⩽4 or patient-reported outcomes-CD (PRO2-CD) score ⩽11 in patients with CD and simple clinical colitis activity index (SCCAI) score ⩽2 or PRO2-UC score = 0 in patients with UC. The secondary efficacy endpoints included endoscopic improvement and biochemical remission, defined as a Mayo endoscopic subscore ⩽1 and fecal calprotectin level (FCL) <250 mg/mL, respectively. Corticosteroid-free remission (defined as discontinuation of oral corticosteroids in patients receiving oral corticosteroids at baseline and subsequent clinical remission at endpoints) and the clinical remission of anti-TNF-naive patients as well as the clinical remission of anti-TNF failure patients were exploratory endpoints.
The safety assessment included all AEs and SAEs. These events might result from the changes or exacerbations in the patient’s clinical presentation and from blood, urine, or electrochemical grinding findings. Hence, all AEs, regardless of their causality, were included.
Pharmacokinetic and immunogenicity studies, such as serum VDZ concentration and AVA positivity, were conducted using verified sandwich enzyme-linked immunosorbent assay and electrochemiluminescence, respectively. AVA-positive status was defined as the patient having at least one positive AVA from premedication to endpoint.
Quality assessment of the included studies
We assessed the methodological quality of RCTs and cohort studies using the Cochrane Handbook of Systematic Reviews 42 and the Newcastle–Ottawa Scale, 43 respectively.
Statistical analysis
The Cochran software Review Manager 5.4 (Cochrane Collaboration, London, UK) was used for the statistical analysis of data from the included studies. Efficacy endpoints were expressed as odds ratio (OR) [95% confidential interval (CI)] and safety outcomes and positive serum AVA as risk ratio (95% CI). If the pharmacokinetic data were sufficient and with low heterogeneity, it was indicated using weighted mean difference/standard mean difference (95% CI).
44
All target outcomes were combined using the Mantel–Haenszel method. The CochranQ test results (
0–40%: might not be important;
30–60%: may represent moderate heterogeneity;
50–90%: may represent substantial heterogeneity;
75–100%: considerable heterogeneity.
Fixed-effects models were used when heterogeneity was absent or small, and random-effects models were used when heterogeneity was large; the meta-analysis was discarded if heterogeneity was substantial. A
Results
Included studies: A total of 62 results were obtained from the search, including 60 studies and 2 clinical trials. In all, 60 retrieved studies were imported into Zotero. After automatically eliminating duplicates and manually excluding corresponding clinical trials already reported, 38 publications were defined as eligible and required to be browsed. After a detailed reading of titles, abstracts, and full texts, 25 studies were analyzed as ineligible and hence excluded from this analysis. Finally, 13 studies were included in this systematic review (Figure 1). The authors, year of publication, study type, participants, and outcomes of included studies were extracted and summarized (Supplemental Table S2).

Flow chart illustrating the identification, screening, and selection of the eligible clinical trials.
All studies were published between 2019 and 2022 and included 3 RCT (2 studies on UC and 1 on CD) and 10 non-RCT reports. Three RCTs and three prospective cohort studies were of relatively high methodological quality and had complete results, and hence were included in this meta-analysis (Supplemental Figures S1 and 2 and Supplemental Table S3).
Efficacy
Different effect models were selected based on heterogeneity for the meta-analysis of the three RCTs and the three cohort studies. The efficacy of UC and CD was evaluated in two subgroups due to the differences between the diseases. Similarly, the control group of studies included PBO SC and VDZ IV, and hence the outcomes needed to be analyzed separately. This was to rigorously discriminate the true outcomes of VDZ SC across different study methods, different diseases, and different reference standards, including clinical, endoscopic, and biochemical remissions.
Clinical remission
Among the RCT subgroups, the clinical remission rate in patients with UC treated with VDZ SC was 4.72 times (
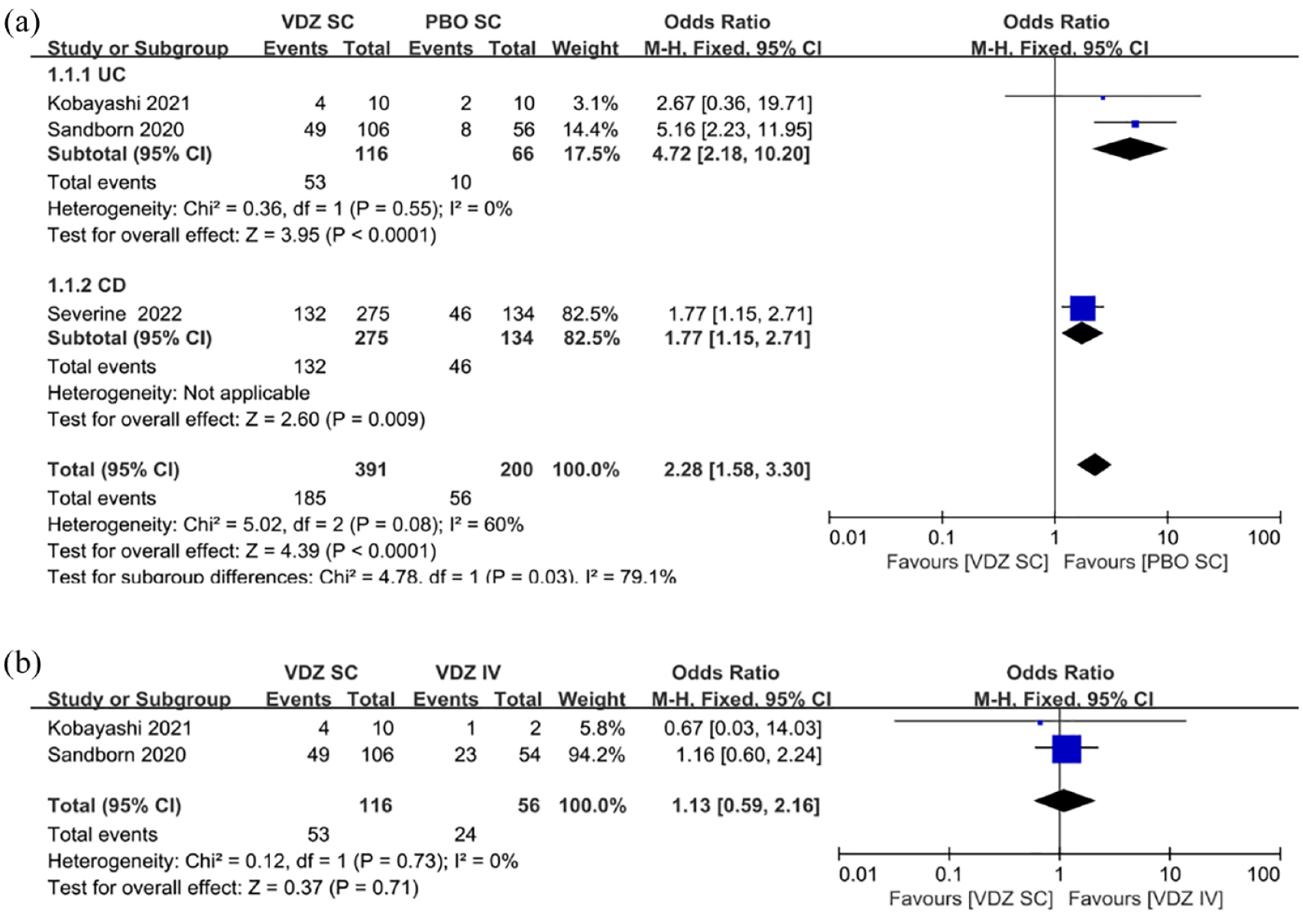
(a) Forest plots of RCTs using the fixed-effects model showing a significant contribution of VDZ SC to clinical remission in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the fixed-effects model showing no significant reduction of VDZ SC in clinical remission in patients with UC compared with VDZ IV.
This subgroup of the cohort study described the changes in clinical remission rates in patients with IBD after switching VDZ from IV to SC. The heterogeneity of the cohort study was high, especially in the CD subgroup (

Forest plots of cohort studies using the random-effects model showing no significant contribution of VDZ SC to clinical remission in patients with IBD compared with that of VDZ IV.
Moreover, the clinical remission rates in specific patients were analyzed separately, including corticosteroid-free clinical remission, anti-TNF-naive patients’ clinical remission, and anti-TNF failure patients’ clinical remission. In the UC subgroup, the clinical remission rate in corticosteroid-free patients treated with VDZ SC was 3.27 times higher than that with PBO SC, but lacked statistical significance (

(a) Forest plots of RCTs using the fixed-effects model showing a significant contribution of VDZ SC to corticosteroid-free remission in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the random-effects model showing no significant reduction of VDZ SC in corticosteroid-free remission in patients with IBD compared with that of VDZ IV.

Forest plots of RCTs using the fixed-effects model showing a significant contribution of VDZ SC to clinical remission rate in anti-TNF-naive patients with IBD compared with that of PBO SC.

Forest plots of RCTs using the fixed-effects model showing a significant contribution of VDZ SC to clinical remission in anti-TNF failure patients with IBD compared with that of PBO SC.
Endoscopic remission
Three RCTs reported endoscopic remission in patients in the maintenance arm in different dosing patterns. Compared with PBO SC, VDZ SC significantly promoted intestinal mucosal healing in patients with UC (
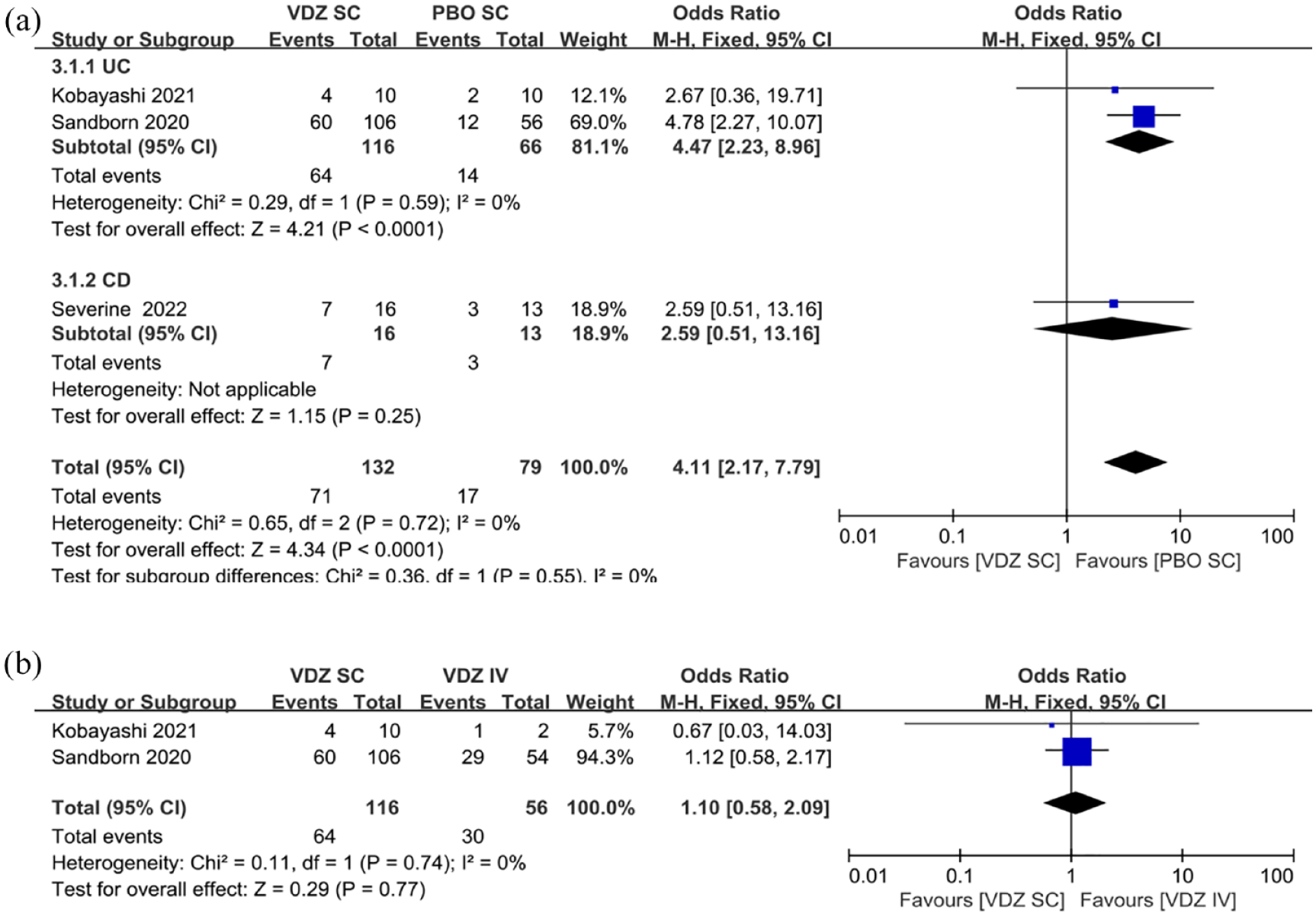
(a) Forest plots of RCTs using the fixed-effects model showing a significant contribution of VDZ SC to endoscopic remission in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the fixed-effects model showing no significant reduction of VDZ SC to endoscopic remission in patients with UC compared with that of VDZ IV.
Biochemical remission
Besides the three RCTs, two cohort studies reported biochemical outcomes in patients. In terms of reduction in FCL level in patients with UC, VDZ SC was 4.06 times more efficient than PBO (

(a) Forest plots of RCTs using the fixed-effects model showing a significant contribution of VDZ SC to FCL remission in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the fixed-effects model showing no significant reduction of VDZ SC in FCL remission in patients with IBD compared with that of VDZ IV.
The data from the cohort study showed that the switch from VDZ IV to VDZ SC regimen yielded more significant efficacy in UC cases (

Forest plots of prospective cohort studies using the fixed-effects model showing no significant contribution of VDZ SC to biochemical remission in patients with IBD compared with that of VDZ IV.
Safety
The probability of AEs and SAEs in patients with IBD was reported in all three RCTs. In the UC subgroup, the risk ratio of AEs in VDZ SC was only 86% of that in PBO, and no statistically significant difference was observed between the two treatments (

(a) Forest plots of RCTs using the fixed-effects model showing no significant contribution of VDZ SC to AEs in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the fixed-effects model showing no significant contribution of VDZ SC to AEs in patients with UC compared with that of VDZ IV.
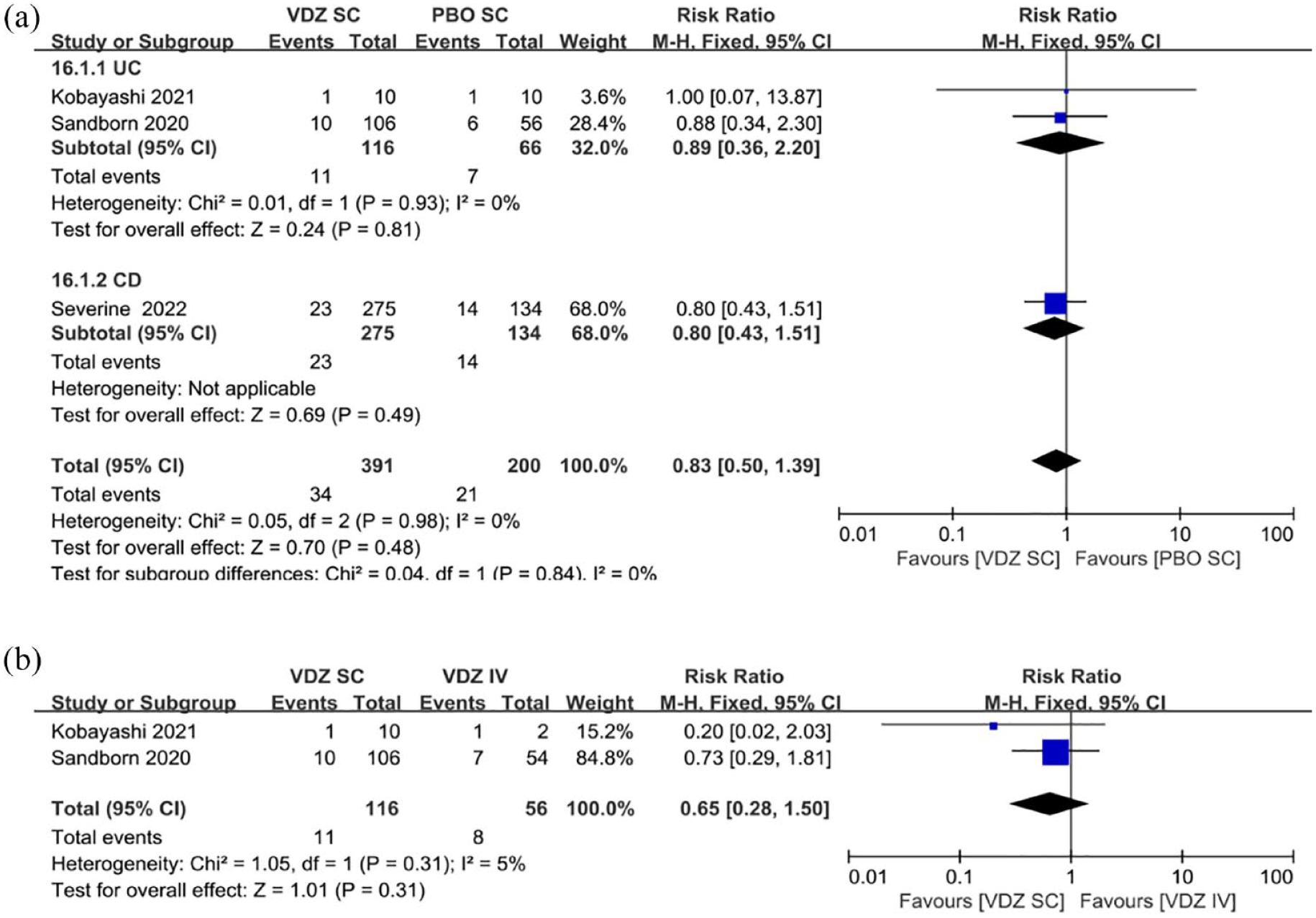
(a) Forest plots of RCTs using the fixed-effects model showing no significant contribution of VDZ SC to SAEs in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the fixed-effects model showing no significant contribution of VDZ SC to SAEs in patients with UC compared with that of VDZ IV.
Pharmacokinetics and immunogenicity
Few studies reported serum VDZ concentrations in patients, including one each in the UC and CD subgroups. Therefore, we did not conduct the meta-analysis of pharmacokinetics. VDZ SC treatment promoted a completely opposite trend in the incidence of serum AVA in patients with UC and CD compared with PBO. In the UC group, the risk of AVA positivity in patients after VDZ SC treatment was only 15% of that of PBO (

(a) Forest plots of RCTs using the fixed-effects model showing an erratic effect of VDZ SC in serum AVA positivity in patients with IBD compared with that of PBO SC. (b) Forest plots of RCTs using the fixed-effects model showing no significant contribution of VDZ SC to serum AVA positivity in patients with UC compared with that of VDZ IV.
Discussion
Most patients with IBD are treated with IV biologics, but the IV approach increases both the time and economic costs for patients. Most biologic SC dosage forms are now being invented and used in the clinic, such as infliximab biosimilar CT-P13 SC formulation (CT-P13 SC) and ustekinumab.47–50 VDZ is the only intestinally selective targeted drug that has been developed and applied for treating IBD. Although it has not been available for more than two decades, it has already been found to be extremely effective. VDZ SC agents were discovered at the time of the initial phase of the COVID-19 pandemic, when many IV biologics were delayed or postponed due to concerns about in-hospital viral transmission and patients urgently needed to be able to self-administer therapy. 51 A relatively large number of trials on VDZ SC reported satisfactory outcomes.34,36–41,52–60 Therefore, we sought to systematically review studies on VDZ SC for treating patients with IBD and to meta-analyze studies with relatively complete outcomes.
The VISIBLE study team is dedicated to evaluating the efficacy and safety of VDZ SC maintenance treatment in patients with IBD, with VISIBLE 1 being a clinical study in patients with UC and VISIBLE 2 being a trial in patients with CD. Two VISIBLE 136,37 and one VISIBLE 2 38 trials were included in this systematic study, both completed randomized, double-blind, placebo-controlled phase III trials. Furthermore, three eligible cohort studies39–41 were included in the meta-analysis. We used the UC and CD subgroup analyses for all data included in the meta-analysis to better visualize the respective efficacy and safety in patients with UC and CD.
The pooled data from RCTs and cohort studies on efficacy suggested that the clinical remission rates in patients with UC did not decrease in either VDZ SC and VDZ IV comparisons or treatment conversion from IV to SC. High-quality studies comparing SC and IV were not available in the CD subgroup, and the meta-analysis results showed great heterogeneity. Volkers’ results 40 were satisfactory, with a lower risk of bias and more complete published data. Wiken 41 found a slight decrease in clinical remission rates after switching to SC treatment in patients with CD. However, this result was less clear because the total number of patients in both groups at the endpoint was not provided in these studies, and the data in the meta-analysis were our calculations based on remission rates. Furthermore, the duration of treatment in patients with CD differed between the two studies, with the former lasting 3 months and the latter having a median follow-up of 27 weeks (interquartile range: 19–37). Therefore, we preferred Volkers’ results; alternatively, it could be argued that SC did not reduce the clinical efficacy of VDZ in patients with CD. SC treatment demonstrated an approximately threefold advantage in clinical remission compared with PBO in both subgroups of patients, indicating that VDZ was clearly effective. Two other conference abstracts also showed that patients with IBD who switched from VDZ IV induction therapy to SC maintenance therapy could continue their previous clinical outcomes. Norgaard 53 observed that after conversion, 2 of 15 patients achieved complete clinical remission at 24 months of maintenance therapy.
We separately pooled corticosteroid-free clinical remission, anti-TNF-naive patients’ clinical remission, and anti-TNF failure patients’ clinical remission. In all three cases, VDZ SC uniformly demonstrated much better efficacy than PBO in treating patients with UC. Regarding clinical remission rates in patients with CD without oral corticosteroids and in anti-TNF-naive patients, VDZ SC was significantly better than PBO, but SC had only a small advantage in improving anti-TNF failure patients’ clinical remission. Furthermore, SC was less effective than VDZ IV in patients with UC without oral corticosteroids.
Patients with IBD are generally diagnosed simply when they exhibit clinical symptoms, and endoscopic mucosal changes are often considered the gold standard in diagnosing IBD.61,62 The clinical symptom assessment is cumbersome to use and relies heavily on subjective factors, such as the SCCAI and HBI. Colonoscopy is probably the most standard reference for the diagnosis and evaluation of IBD, but it is invasive and time-consuming, and has an interval between examinations and a risk of complications during the procedure. Serological indicators such as C-reactive protein (CRP) are often used to assess the severity of inflammation in the incoming bowel.63,64 However, FCL seems to be more sensitive and specific and can compensate for other tests as a convenient and noninvasive test.63,65 Vermeire’s study 38 examined serum CRP concentrations before and after VDZ SC treatment in selected patients with CD and found that VDZ SC could reduce serum CRP concentrations in patients to some extent. Three RCTs and cohort studies focused on the improvement in patients’ FCLs. More than twofold better FCL remission was observed in patients with UC and CD after VDZ SC treatment compared with PBO (FCL < 250 μg/g). Patients with UC had almost identical improvement in their FCL after different injection treatments. Two RCT studies existed on VDZ SC compared with PBO in patients with UC with greater heterogeneity, but the sample size of Kobayashi’s study was small. Therefore, we believe that Sandborn’s data better reflect the real situation.
Also, three RCTs reported in detail all AEs in patients during maintenance treatment. No deaths occurred during all treatments, and most of the AEs were mild or moderate, with SAEs occurring in only a few patients. Overall, the probability of AEs and SAEs in patients with IBD treated with VDZ SC was lower than that in both the PBO and VDZ IV groups after pooled analysis, but lacked significance. The use of a placebo alone exposed patients to greater safety risks possibly attributable to the absence of clinical response or further disease progression. The most common AEs that occurred after VDZ SC treatment were skin and tissue lesions and gastrointestinal reactions. Skin and tissue lesions usually manifest as injection site reaction (ISR), which are rashes, swelling, erythema, and pruritus at the injection site. Gastrointestinal reactions are characterized by exacerbation of original symptoms or the development of new intestinal manifestations, including abdominal pain, nausea, and vomiting. Infections and invasions, musculoskeletal and connective tissue disorders, respiratory, thoracic, and mediastinal disorders, and neurological disorders are the next most common AEs. Nasopharyngitis was considered the most common associated AEs in several studies.36–38 Severe AEs include perianal abscesses, intestinal abscesses, and other severe infectious diseases of the intestinal tract, as well as tumor-related diseases.37,38 Vermeire 38 reported two and three patients with malignancy in the PBO and VDZ SC groups during maintenance treatment. However, no correlation was found between VDZ SC treatment and malignancy.
The serum drug concentration in patients reflects the pharmacokinetics of the drug. After analyzing several studies, we found that the maintenance serum VDZ concentrations in patients usually reached 30–40 μg/mL regardless of SC or IV administration.39,52 However, the median steady-state VDZ Ctrough in the placebo group was 0 μg/mL (lowest to highest, 0–31.9 μg/mL) (week 46). 38 We hypothesized that a close association might exist between increased serum VDZ exposure and patients with IBD who achieved clinical remission and clinical response. Chen 66 found that the patients’ clinical symptoms improved more significantly as serum VDZ concentrations increased and were maintained at higher levels. Sandborn 67 arrived at a similar conclusion: after shortening the dosing interval to once a week (QW) in patients who failed the standard dose of VDZ SC (108 mg Q2W), 27.1% (13/48) of patients achieved clinical remission in week 16 as their serum VDZ concentrations increased. Similarly, after Wolf 56 switched the injection schedule to VDZ SC 108 mg QW treatment in 35 patients with UC who had failed to induce clinical response with 8 IV doses of VDZ in the VISIBLE 1 trial, the clinical remission and clinical response rates of patients improved to 76.9% and 76.9%, respectively. These data indicated that higher serum drug concentrations appeared to reflect high efficacy.
Bergqvist and Wiken,39,41 however, found no relief of symptoms after IV conversion to SC, despite a onefold to twofold increase in serum VDZ concentrations in patients. Rosario 68 also found that a serum drug concentration of 1 μg/mL in patients was sufficient to saturate the α4β7 binding expressed on the intravascular cell surface. Thus, in his review of Bergqvist’s study, Nadesalingam 51 believed that the elevated serum VDZ levels in patients might simply reflect the steady-state drug distribution of the SC formulation rather than better bioavailability. He suggested that the almost complete occupancy of VDZ in peripheral blood memory T cells was an important reason why increased VDZ serum levels did not necessarily translate into better clinical outcomes. Bergqvist’s latest response 69 also made clear his belief that the lack of better clinical outcomes might be related to immunogenicity, as the incidence of AVA was found to be higher in patients with IBD in the VISIBLE study than in the GEMINI study, and none of the patients with persistent positive AVA in the GEMINI study achieved clinical remission, along with increased clearance of VDZ. Therefore, we pooled AVA generation data from selected patients in three RCTs. However, VDZ SC was found to show completely heterogeneous results in the UC and CD subgroups compared with PBO. The serum AVA levels were much lower in patients with UC than in PBO, while the opposite was true in patients with CD. Immunogenicity consistent with IV treatment was demonstrated in the SC treatment in the UC subgroup. This result might be consistent with previous differences in remission rates between different subgroups of patients, with lower immunogenicity and higher clinical remission rates in patients with UC and minimal improved and unstable remission rates in patients with CD. Nevertheless, the results of the comparison with IV treatment differed from those of the previous VISIBLE and GEMINI studies. The differences in testing modalities cannot be ruled out, and perhaps patients’ neutralizing AVA may be more important than overall AVA, which should be taken into account. Therefore, dose optimization may not be the best option for restoring clinical remission, or at least not the only option. Repeated or intermittent dosing is more likely to induce immunogenicity than single use of monoclonal antibodies. 70 At this point, adding immunomodulators, high doses, and short intervals of dosing may help restore efficacy.
This study had some limitations. As a systematic review and meta-analysis, although the quality of RCTs was high, the number of high-quality studies included in the meta-analysis was far from adequate, and the cohort studies showed a more pronounced heterogeneity. Moreover, we identified other potentially eligible cohort studies during the search. However, most lacked complete data and were published only as conference abstracts and therefore had to be excluded from the meta-analysis. Finally, only one high-quality RCT on patients with CD was available, and a control group for VDZ IV was lacking.
Conclusions
In the COVID-19 setting, VDZ SC was an effective, safe, and cost-effective option that could be used as an alternative therapy to VDZ IV. The efficacy was clearer in patients with UC, and the risk of AE was lower. We also tried to prevent the occurrence of ISR. However, the efficacy in patients with CD was generally inferior to that in patients with UC, although it might be related to a smaller number of meta-analyses. Also, the decision on the post-conversion treatment regimen needed to take factors such as immunogenicity into account.
Supplemental Material
sj-docx-1-tag-10.1177_17562848231166227 – Supplemental material for Vedolizumab subcutaneous formulation maintenance therapy for patients with IBD: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tag-10.1177_17562848231166227 for Vedolizumab subcutaneous formulation maintenance therapy for patients with IBD: a systematic review and meta-analysis by Qiong Hu, Xing-zhou Tang, Fang Liu, De-wu Liu and Bo Cao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
