Abstract
Background:
Crohn’s disease (CD) and ulcerative colitis (UC) arise from a dysregulation of the balance between commensal microbiota and mucosal-associated immune system, in patients with genetic and environmental predisposition. Different pathophysiological mechanisms have been reported to influence disease history, with impact on disease phenotype and risk of complications.
Objectives:
This review aims to summarize the definitions of early CD and UC, analyze the underlying immunological mechanisms, and evaluate the impact of recognizing and treating early inflammatory bowel disease (IBD) on patients’ prognosis (short- and long-term outcomes).
Design:
To address this issue, we have performed a scoping review.
Data sources and methods:
Three online databases (MEDLINE, Web of Science, and ScienceDirect) were searched and the results were independently screened by two reviewers.
Results:
From 683 records identified, 42 manuscripts evaluating early IBD in adult patients were included. The ‘early CD’ concept was first described in 2008. Four years later, an international consensus proposed the definition of diagnosis up to 18 months, in patients without previous or current need for disease-modifying therapies. Several other definitions have been proposed; the ‘2 years since diagnosis’ is the most used, regardless of disease characteristics or medication. The amount of evidence on early UC is lower and more recent. Regarding early disease pathogenesis, most theories emphasize the prominent role of innate immunity, followed by early-Th1 adaptive response.
Conclusion:
The treatment of early CD seems to be crucial for the management of CD patients, impacting short-, medium-, and long-term outcomes. On the other hand, the early treatment of UC appears to be less advantageous, yet evidence comes from only a few retrospective studies.
Introduction
Inflammatory bowel diseases (IBDs), including Crohn’s disease (CD) and ulcerative colitis (UC), result from the dysregulated crosstalk between commensal microflora and the mucosal-associated immune system, in patients with a genetic predisposition and environmental exposure to risk factors. CD and UC pathogenesis are believed to have different yet interrelated immunological phases. 1 From a pathophysiological point of view, several cytokines have been reported to be differentially expressed in distinct disease phases; particular attention shall be devoted to interleukin (IL)-12 and IL-17 during early and late stages, respectively. 2 IBD-related inflammation may go unnoticed for prolonged periods delaying clinical presentation. Therefore, at the time of diagnosis, disease may be advanced, both immunologically and phenotypically. 3 CD diagnosis is often delayed 5–9 months due to the variability of the initial manifestations, as opposed to UC, which is usually diagnosed earlier, due to the consistent and alarming initial symptoms and manifestations. 4
Recognizing early disease as a clinically distinct entity may change the objectives of the treatment strategy and impact clinical outcomes, as it has been suggested for rheumatoid arthritis. 3 The relevance of the transition from early to late disease in the management of IBD relies on the association of these states with disease phenotype, extension, severity, and prognosis. 1 In fact, IBD progressively induced damage results from a continued phenomenon. Early CD phases have been reported to be more dependent on the breakdown of epithelial barrier function and on the impairment of innate immunity, triggering subacute inflammation. The subsequent compromise of bacterial clearance and the dysregulated adaptive immune responses perpetuate the inflammation state. 1 The interest on UC early phases is more recent and, as in CD, innate immunity is also a key player. It has been described that early intervention with biologics may slow disease progression and improve long-term outcomes in IBD, reducing irreversible damage. 2 This supports the existence of a ‘window opportunity’ for intervention, before severe inflammation and bowel damage become established.2,5
In this scoping review, we aimed to analyze the evidence regarding early CD and UC definition and the immunological mechanisms associated with early disease stages, as well as to summarize the available information on the impact of prompt treatment on prognosis, including its strengths and limitations.
Materials and methods
A scoping review was performed, following the PRISMA checklist for scoping reviews. 6 Three databases [MEDLINE (through PubMed), ScienceDirect, and Web of Science] were searched, from inception to 12th August 2022, using the following terms: [Crohn Disease (MeSH terms) OR Crohn’s disease OR ulcerative colitis OR Inflammatory Bowel Diseases (MeSH terms) AND early disease].
The inclusion criteria were as follows: (i) including adults diagnosed with CD or UC using clinical, endoscopic, and/or pathological features and (ii) mentioning early disease and/or its immunopathogenesis and/or the impact of early treatment on clinical outcomes. Exclusion criteria were as follows: (i) including only patients without CD or UC and (ii) not referring to early disease.
The reference lists of the included studies and of important reviews on the topic were hand-searched to identify further relevant publications. The studies were independently screened by three reviewers (MM Estevinho, P Leão Moreira, and I Silva). Any study, whose title and abstract clearly indicated that it failed to meet the selection criteria, was excluded; for all others, the full text was carefully analyzed to decide for inclusion or exclusion. The following information was collected by three independent reviewers: authors’ name, publication year, study type, patients’ characteristics, intervention/exposure, comparator (posology and treatment duration), follow-up, assessed outcomes, and main results (regarding early disease).
Studies selection and data extraction
Overall, 683 results were identified (88 in PubMed, 502 in ScienceDirect, and 93 in Web of Science); from these, 39 studies were included in the scoping review: 28 regarding CD, 8 on UC, and one including both CD and UC. In addition, three reports on UC were identified through manual search, totalizing 42 included studies. Figure 1 depicts the studies’ selection process.

Flow diagram of studies selection.
Definition of early disease
Crohn’s disease
The success of the early treatment of rheumatologic conditions, along with the diversification of CD treatment options, has brought the spotlight to the concept of ‘early disease’. Although the development of a clear definition has been challenging. The concept of ‘early CD’ emerged in 2008 and was first defined as a diagnosis within 4 years, in patients without exposure to corticosteroids, antimetabolites, or biological agents. 7 The diagnosis-based time was selected to avoid recall bias associated with symptoms and to bypass the poor association between symptoms and inflammation. Since then, multiple definitions have been proposed, including not only time and medication, but also other characteristics like disease phenotype (Figure 2, Table 1). For example, in the SONIC trial, published in 2010, the 3 years cutoff was applied to stratify short/long disease duration. 8 In the same year,4,9 the definition was upgraded to include parameters reflecting active disease (CD activity index >220, plus C-reactive protein >10 mg/L, ulceration of more than 10% in at least one bowel segment on endoscopy, bowel enhancement on computerized tomography or magnetic resonance imaging, or ‘positive’ fecal calprotectin), in patients in whom the diagnosis was performed less than 2 years before. In this definition, 9 patients with early CD should not have fistula, abscess, stricture, prior CD-related surgery or endoscopic dilation, altered continence or need for enteral or parenteral supplementation or nutrition, exposure to immunomodulators, biologics, been steroid dependent or have ever received intravenous steroids. In 2012, the Paris definition was published by international opinion leaders. In this document, early CD was defined as a diagnosis up to 18 months, 10 in patients without previous or current need for disease-modifying therapies (immunomodulators or biologics). 1 Unlike the previous 2010 definition, corticosteroids were allowed, regardless of the posology. The Paris definition was independent of symptoms, presence of bowel damage (fistula, abscess, or stricture; prior CD-related surgery, perianal fistulas, or presence of anorectal strictures), and disease activity, yet such characteristics were recognized to have prognostic value. Due to the absence of objective analytical, endoscopic, and/or imagological markers, the application of the Paris definition was limited and the concept of ‘early disease’ remained mutable. 11 The ‘2-years from diagnostic cutoff’ remained the most used definition throughout the years, due to its undeniable easiness of application.12–25 The fact that the same teams have used different definitions throughout time reinforces the need of adjusting translational research to the demanding circumstances of real-life clinical practice. A recent meta-analysis, including individual-patient data from 16 randomized controlled trials (RCT), used the cutoff of 18 months to define short-duration disease, regardless of other characteristics like phenotype or prior treatment. 26 Besides that, some studies have focused on the concept of ‘very early’ CD, again with variable definitions (up to 6 27 or 1213,28,29 months since diagnosis). The current definitions of early CD have some main limitations: (i) they mostly rely on the time to a timely diagnosis, what is problematic considering the diagnostic delay that is frequently seen; (ii) inflammation may occur without symptoms; (iii) up to 20% of CD patients have stenosing and penetrating phenotypes without recalling prior symptomatology; and (iv) acute and chronic lesions may coexist, but as soon as irreversible bowel damage is detected, it appears legitimate to consider that the disease is no longer in an early stage. Indeed, patients with stenosing and penetrating phenotypes may present immunologically more advanced CD, and their labeling as ‘early’ may underestimate disease progression and postpone treatment introduction.
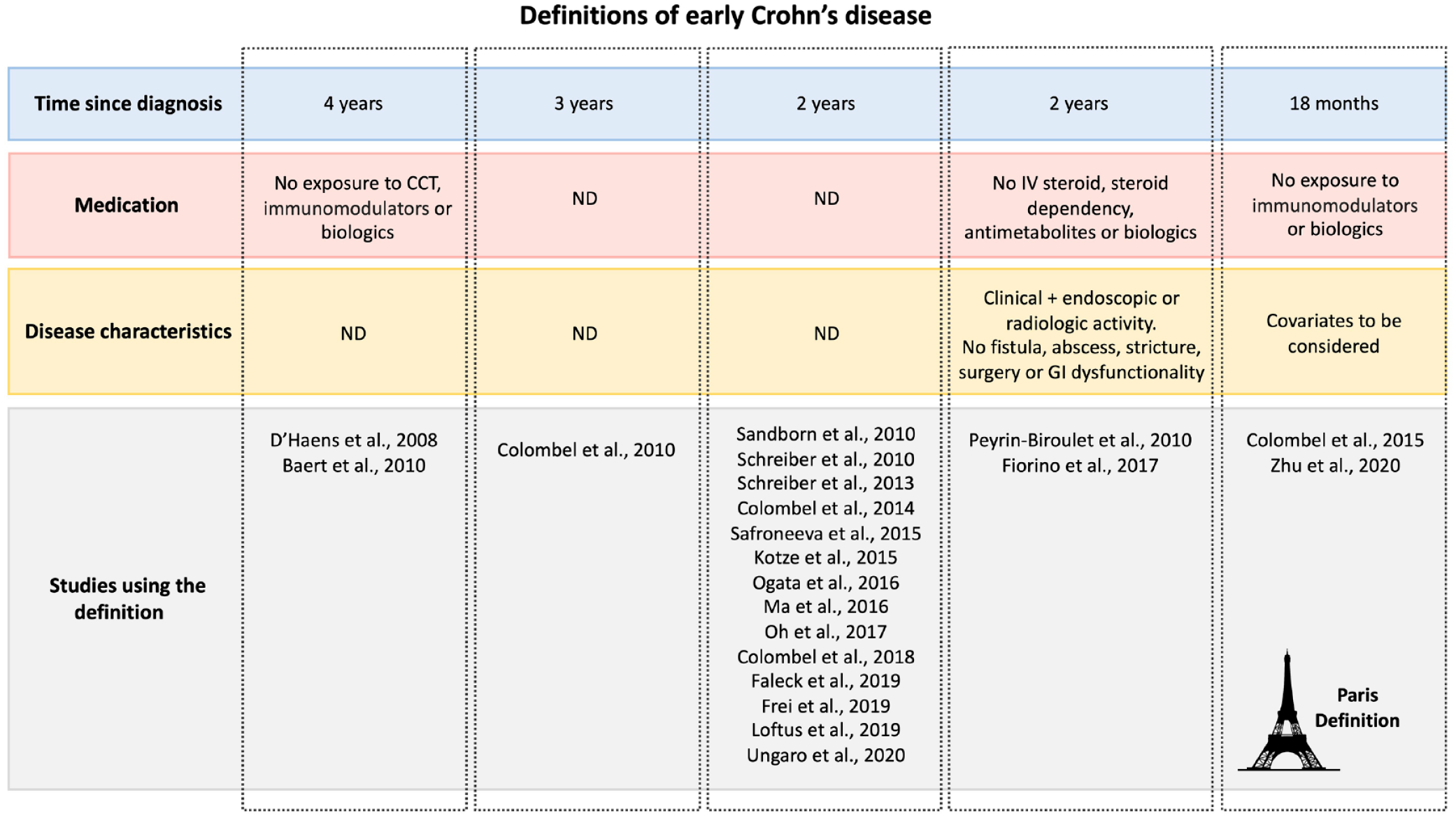
Summary of early CD definitions (parameters included and studies using such definition).
Evidence regarding therapeutic intervention on early CD.
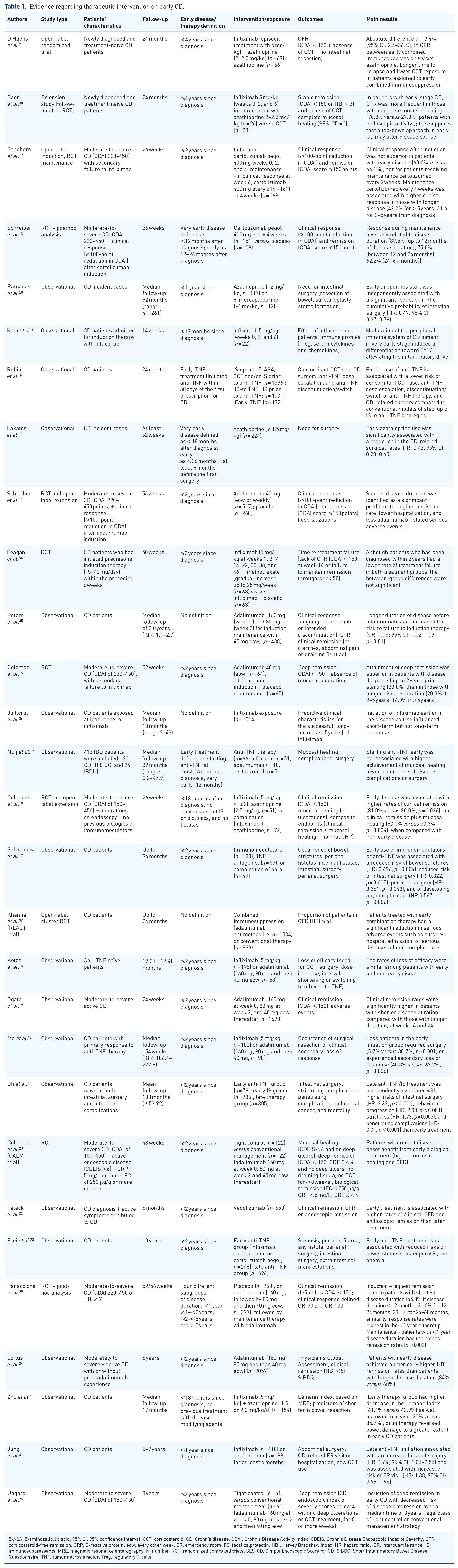
5-ASA, 5-aminosalicylic acid; 95% CI, 95% confidence interval; CCT, corticosteroid; CD, Crohn’s disease; CDAI, Crohn’s Disease Activity Index; CDEIS, Crohn’s Disease Endoscopic Index of Severity; CFR, corticosteroid-free remission; CRP, C-reactive protein; eow, every other week; ER, emergency room; FC, fecal calprotectin; HBI, Harvey Bradshaw Index; HR, hazard ratio; IQR, interquartile range; IS, immunosuppressants; MRE, magnetic resonance enterography; N, number; RCT, randomized controlled trials; SES-CD, Simple Endoscopic Score for CD; SIBDQ, Short Inflammatory Bowel Disease Questionnaire; TNF, tumor necrosis factor; Treg, regulatory T-cells.
Ulcerative colitis
The main differences between UC and CD are related to bowel involvement, disease progression, and response to therapeutic intervention. 42 However, as CD, UC is also a progressive disease, and its standard therapeutic approach has been questioned. It has been hypothesized that controlling the disease in earlier stages would be the most adequate approach to manage its progression. Identifying the disease in early stages allows taking advantage of the pathophysiological ‘window of opportunity’, in which distinct treatment interventions may potentially modify the natural history of the disease and reach better outcomes. Yet, unlike CD, the concept of ‘early UC’ is still to be clarified. According to the available literature, summarized in Table 2, the early disease definition for UC is variable. In four published studies22,43–45 and one ongoing trial, 46 the cutoff to define early disease was 2 years or less since diagnosis. However, other studies have stretched the time range to 318,47 or even 548,49 years. A Canadian study 45 evaluating early anti-tumor necrosis factor (TNF) treatment verified that the impact of the timing of anti-TNF therapy, on long-term outcomes of UC patients, was not significant. This trend was also confirmed in a Korean study. 44 Information on the immune profile, including cytokine patterns, could provide a more solid basis for a clearer definition of ‘early disease’ in UC.
Evidence regarding therapeutic intervention on early UC.

95% CI, 95% confidence interval; CCT, corticosteroid; CFR, corticosteroid-free remission; ER, emergency room; HR, hazard ratio; N, number; RCT, randomized controlled trials; TNF, tumor necrosis factor; UC, ulcerative colitis.
Immunological mechanisms underlying early IBD pathogenesis
Crohn’s disease
Accumulating evidence supports that early CD pathogenesis relies on a ‘permeability-to-inflammation’ pathway, with a ‘defective-to-overactive’ imbalanced immune response to stimuli. In early disease phases, following the activation of innate immunity, mucosal T cells mount a Th1 response, with higher production of IL-12, 52 interferon-gamma (IFN γ), TNF-α,1,53 as well as higher expression of IL-12 receptor b2 (IL-12Rb2) chain, resembling an acute infectious process. The reverse seems to occur in late CD, in which IL-12Rb2 chain expression and IL-12-induced IFN-γ production are decreased. Indeed, later in the disease course, the immunological background changes, with a shift toward a strong Th2 response 1 and higher expression of IL-23, IL-33, IL-13, IL-5, and IL-17 (whose production is mediated by IL-23). 53 Besides this ‘T-cell signature’, the dysregulation of microRNAs has also been reported to play a role in early disease phases. 54 A study on gene and microRNA expression profiles in ileal mucosal biopsies from CD patients showed that microRNA dysregulation was more relevant in post-operative recurrent CD and newly diagnosed patients, suggesting an important role in the early stage of CD. Folate Hydrolase 1 emerged as the most dysregulated gene either in newly diagnosed CD or postoperative recurrent CD. 54
Ulcerative colitis
It has been proposed that the immunopathogenesis of UC changes during the disease course, with a transition from a Th1- in early UC into a Th2-driven disease later in disease course.55,56,57. Also, higher mucosal mRNA expression of IL-23 has been detected in newly diagnosed patients, in comparison with those with longstanding disease. 55 As the disease evolves, other cells such as Th-17, Th-9 (possibly due to the polarization of naïve T cells in the presence of IL-4 and transforming growth factor beta), and other cytokines like IFN γ, IL-17A, and IL-955,58 may increase. The expression of genes associated with T cell differentiation has also been postulated to vary; TNF-α and suppressor of cytokine signaling 1 expression are increased during early disease, while IL-4 receptor, growth factor independence 1, IL-1 receptor like 1, peroxisome proliferator-activated receptor gamma, and IL-5 expression are elevated later during UC course.55,57
Early intervention and impact on prognosis
Crohn’s disease
The acknowledgment of ‘early disease’ is the key to a prompt therapeutic action. The rationale for treating early disease, using a treat-to-target approach, is based on the evolving immunological milieu, and on the accumulating damage and disability associated with later CD stages. Throughout the years, several authors have evaluated the impact of targeting early CD on disease outcomes (Table 1). Most of the available evidence comes from the use of anti-TNF drugs, either alone (2212–21,23–25,29,31,32,34,35,36,37,38,41 out of 29 studies) or in combination with immunosuppressants (77,17,30,34,38,39,40); the single use of immunosuppressants was evaluated in five studies,17,21,28,33,38 while vedolizumab was studied once. 22 Although ‘early disease’ definition and the outcomes sought varied widely, data from observational studies and from post-hoc analysis of RCTs have shown that patients who received therapy in the early stage of disease have better outcomes than those who received treatment later. Short-term and medium-term outcomes were evaluated in nearly all studies regarding early CD management. In general, individual studies suggested that prompt medical intervention may improve the possibility of reaching clinical response and/or remission,7,13,14,19,20,22,24,29,30,32,35,36,38 reduce inflammatory biomarkers (C-reactive protein and fecal calprotectin), 20 and increase endoscopic response and/or remission.15,20,22,25 Also, in a recent meta-analysis, 26 which pooled individual patient data from 16 RCT evaluating biologic-treated CD patients, disease duration impacted the rate of clinical remission (odds ratio = 0.75; 95% confidence interval = 0.61–0.92) in patients with a diagnosis more than 18 months prior treatment start; yet, the placebo effect was also higher in that subset of patients. On the other hand, conflicting results were reported twice; Sandborn et al. 12 did not find acceptable clinical outcomes in patients who started anti-TNF in the first 2 years since diagnosis, while Nuij et al. 49 described that early anti-TNF treatment was not associated with higher mucosal healing. These discrepancies may be due to the low number of patients in the ‘early’ groups, as well as to inappropriate selection of patients for therapy, which may have led to suboptimal treatment and outcomes. The meta-analysis published by Ben-Horin et al. 26 did not find an association between early therapeutics with biologics and higher reduction in C-reactive therapy. In addition, these authors were not able to define a specific time point on CD history that could clearly impact the proportional biologic/placebo risk for remission (defined as the treatment effect), suggesting that disease progression may be influenced more strongly by other variables besides disease duration.
Concerning long-term outcomes, early introduction of immunosuppressants17,21,28, 33 ,39 or anti-TNF drugs17,18,21,39,44 was associated with a significant reduction in the probability of requiring CD-related surgeries. Also, patients receiving treatment early in disease course had fewer disease-related complications17,21,39 and need for hospitalization.14,21,41 Besides that, some authors have stated that intervention within 18 months after CD diagnosis may prevent and even reverse bowel damage. 40 On the other hand, the impact of early treatment on patients’ quality of life, work productivity, and disability remains largely unexplored.
Postoperative recurrence is a setting that mimics early CD. According to the literature, up to 90% of the patients experience recurrence within 1 year of surgical removal of all macroscopically identifiable disease. 59 Although no accurate biomarkers of recurrence exist, some studies have described the association with higher preoperative C-reactive protein levels and neutrophil-to-lymphocyte 60 ratio, while others highlight the role of postoperative fecal calprotectin. 61 Despite this, the relevance of postoperative prophylactic therapy in high-risk patients (e.g. several CD-related surgeries, smoking habits, penetrating disease) is well stated, as is the need for early screening colonoscopy after 1 year, in which evidence of macroscopic disease (Rutgeerts score of i2 or more) usually prompts therapeutic institution or escalation. 62 The drugs usually recommended as prophylactic therapy are thiopurines and/or anti-TNF. Although the body of evidence is relatively small, the prophylactic use of anti-TNF has been reported to prevent clinical and endoscopic recurrence in the first 2 years, yet the effect on long-term outcomes remains elusive. 63 The immunological mechanisms underlying recurrence after macroscopic resetting are still being investigated but may involve upregulation of several genes (TNF-α, IFNγ, IL23A, and IL17A) and pathways (mitochondrial dysfunction and JAK-STAT). 64
Ulcerative colitis
Little evidence is available regarding early intervention in UC. The existing data (Table 2) derive from retrospective observational studies, which evaluated different endpoints (rate of colectomy, secondary loss response, UC-related hospitalization, among others), at diverse time points. Overall, these studies18,22,44,45,47,50,51 failed to identify the differences in treatment response and prognosis, in patients receiving early therapeutic interventions, mostly biologics. The effect of aminosalicylates in early versus non-early disease has not been explored. Furthermore, a retrospective cohort study 43 showed that shorter disease duration is independently associated with increased risk of treatment failure in biologic-treated UC patients. On the other hand, biologic therapy early in the course of the disease was a negative prognostic marker. Likewise, another retrospective study observed that disease duration was not associated with response to vedolizumab in patients with UC. 22 In line with this, a recent meta-analysis, 26 which explored early biologic treatment (⩽18 months), concluded that early treatment of UC patients was not associated with higher rates of clinical remission after induction therapy. Some individual studies have analyzed long-term outcomes,2,18,47,48,50 such as hospitalization and surgery. However, once again, earlier treatment did not have a positive impact. Although most of these assumptions are not encouraging regarding the benefit of early interventions in UC, considering the retrospective nature of the studies, the conclusions to be drawn are limited. In this context, prospective studies comparing outcomes from patients treated at different time points, after UC diagnosis (early versus late disease) would provide relevant information for more generalized conclusions.
Tools to estimate disease progression
Crohn’s disease
Considering that preventing structural changes is the cornerstone of CD therapeutics, the introduction of objective tools to quantify that damage is of utmost importance. The Lémann index (Crohn’s Disease Digestive Damage Score), which incorporates clinical, endoscopic, imaging, and surgical features from all segments of the digestive tract, was the first and unique instrument to do so. 65 This index, whose score ranges from 0 to 140, considers damage location (upper tract, small bowel, colon/rectum, and anus), extent (each organ is divided into several segments, totalizing 30), severity (grade 0–3) and reversibility (concerning stricturing, penetrating and perianal lesions), and was recently validated. 66 This index is usually calculated using data from magnetic resonance imaging and colonoscopy, yet recent preliminary studies have shown that bowel ultrasound may be a reliable alternative for transmural evaluation. 67 Since its development, several reports have explored its ability to predict long-term disability. Gradual elevations in the Lémann index have been reported to occur in at least 60% of the patients in the first decade after diagnosis, 68 being associated with a significant increase in need for steroids and healthcare utilization, including surgeries and hospitalization. 69 Likewise, Lémann index at diagnosis has been reported to be a reliable predictor of the risk of abdominal surgery in the first years after diagnosis. 70 On the other hand, Lémann index’s responsiveness to therapeutic interventions was evaluated in less and smaller studies,71,72 requiring further confirmation. In a study of 30 patients treated with anti-TNF, a Lémann index score of 4.8 was the best cutoff to define the presence of bowel damage, while receiving this therapeutic agent was effective in stopping the progression of bowel damage in 83% of the subjects. 71 In another study enrolling 35 patients under anti-TNF, the early introduction of this biologic was associated with lower bowel damage scores. 73 Although this score has some limitations, particularly its complexity and need for trained gastroenterologists and radiologists, it has good predictive performance and should be incorporated in all disease-modification CD trials as a secondary endpoint, as pointed out by the SPIRIT initiative. 66 Conversely, its application into routine clinical practice will rely on further simplification or widespread training initiatives.
Ulcerative colitis
To date, as far as the authors know, there are no indexes to quantify colonic damage associated with UC. Some tools have been developed for cross-sectional imaging in UC (e.g. Tsuga’s colorectal ultrasound criteria, and a simplified magnetic resonance colonography index), but these tools lack validated cutoff values for extent of disease severity, being their use very restricted and not validated. 74
Future perspectives
The strengths and weaknesses of the current evidence on early IBD, the anticipated treats and opportunities are summarized in Figure 3. The definition of early disease based not only on time, but also on immunological characteristics is critical to guide therapeutic decisions, particularly in CD, in which the impact of prompt therapy on prognosis has been described. However, to avoid overtreatment, it is also essential to identify the patients with early disease that will present an indolent course. Indeed, considering that up to 20% of the CD patients will present a ‘benign’ and non-progressive disease course, it is important to select biomarkers to recognize the patients that would benefit from close monitoring and, if needed, from an accelerated ‘step-up’ approach, rather than early therapeutic ‘top-down’ intervention. In a recent study, Yanai et al. 75 demonstrated that routine measurements, such as body mass index, vitamin B12, white blood cells count, and alanine aminotransferase, at the time of diagnosis may predict the likelihood of an indolent course with a 90% accuracy, on the following 12 months. The validation of this model in longer follow-ups is pending. Also, it remains unknown whether drug de-escalation shall be considered in CD patients that started treatment in early disease stages; the results of ongoing trials, like the CURE trial (NCT03306446) may shed some light on this topic.

Strengths and weaknesses of current evidence on early management of IBDs, anticipated opportunities, and threats (SWOT analysis).
Concerning UC, the concept of early disease is less defined and the evidence of the benefits of early therapeutic interventions is very scarce and pertains, in general, to retrospective studies. The available evidence shows no relevant benefit of early treatment in UC patients. It is expected that upcoming studies, such as the SPRINT (ongoing randomized multicenter study, EudraCT number: 2020-003420-16), which will compare the efficacy of top-down and step-up approaches in UC patients diagnosed up to 2 years and followed-up for 3 years, will bring relevant data on this regard. Also, it remains to be clarified whether early treatment impacts more ambitious UC targets, such as ‘disease clearance’, which combines clinical, endoscopic, and histological remission.2,76
Conclusions
This scoping review summarizes the definition, pathophysiology, and data on early therapeutic intervention in patients with CD and UC, discussing its strengths and limitations. Since 2008, the interest in early CD increased, and continuous efforts to uniformize its definition resulted in the publication, in 2012, of the Paris consensus, which incorporates the concept of ‘time since diagnosis’ as well as medication data. However, the adherence to this definition has been modest. In fact, more than half of the studies published thereafter still define early CD studies only in a time-based fashion (disease diagnosed up to 2 years). Regarding UC, the definition of early disease is still not consensual. Although definitions are strictly related to time since diagnosis, it must be stressed that ‘true’ early disease comes from an early diagnosis free from diagnostic delay.
From a pathophysiological point of view, the expression profile of several cytokines is different in early phases of disease. Particular attention shall be devoted to IL-12, IFN γ, TNF-α (in the case of CD), and IL-23 (for UC). Therefore, further studies on cytokines profile may help to clarify definitions from an immunological perspective.
Overall, the available evidence supports that recognizing earlier stages of CD and adopting an early therapeutic intervention improves symptoms’ control, endoscopic activity (short- and medium-term outcomes), and reduces bowel disability, disease-related hospitalizations, and surgeries (long-term outcomes). However, these trends cannot be extended to UC, as, to date, no comparative studies have shown that prompt treatment impacts patients’ prognosis. Long-term high-quality RCTs are needed to further clarify these aspects. Also, moving the focus away from medical therapy, it is important to acknowledge that, for certain cases, early surgery may be the first therapeutic option, offering the best outcomes for a particular patient, either in short term or in long term.
This scoping review has some limitations; first, different ‘early disease’ definitions have been applied by the included studies; second, the therapeutic interventions and the outcomes sought varied widely; third, no formal assessments of literature quality were performed. Further studies are needed to explore the immunological signature of early disease, which will guide the identification of biomarkers to predict non-indolent progressive disease that may benefit from a ‘watch-and-wait’ strategy (particularly in CD). Immunological data may also be crossed with other patient-related characteristics with influence on disease progression, guiding decision-making and, gradually, stepping into tailored treatment schemes for each patient’s profile.
