Abstract
Background:
Approximately 15–30% of locally advanced rectal cancer (LARC) patients achieved pathological complete response (pCR) after neoadjuvant chemoradiotherapy (CRT) and total mesorectal excision, but the clinical significance of adjuvant chemotherapy (ACT) for pCR patients remains unclear.
Objectives:
To determine whether LARC pCR patients can benefit from the administration of ACT.
Design:
Single center retrospective study.
Methods:
This study retrospectively included 280 LARC patients who achieved pCR after CRT and surgery from 2011 to 2019. The information of patients was recorded. Main outcome measures included 5-year disease-free survival (DFS) and 5-year overall survival. Subgroup analysis was conducted on whether pCR patients with acellular mucin pools received ACT or not.
Results:
A total of 74/280 (26.4%) patients were identified with acellular mucin pools. Disease recurrence occurred in 38/280 (13.6%) patients, and in the subgroup of patients with acellular mucin pools, 15/74 (20.3%) patients developed distant metastases. The existence of acellular mucin pools was associated with worse DFS (79.7% versus 88.8%, P = 0.037). Among pCR patients with acellular mucin pools, 9/25 (36.0%) of non-ACT patients occurred recurrence, and ACT was beneficial for improving DFS (hazard ratio: 0.245; 95% confidence interval: 0.084–0.719; P = 0.010).
Conclusions:
The existence of acellular mucin pools may represent a sign of invasive tumor biology, which indicated a negative prognosis. ACT can improve the prognosis of patient with acellular mucin pools, so ACT should be considered for them.
Introduction
The incidence of colorectal cancer is increasing in the world. 1 15–30% of locally advanced rectal cancer (LARC) patients can achieve pathological complete response (pCR) after chemoradiotherapy (CRT) and total mesorectal excision, which was associated with better long-term survival outcome.2–4 However, 7.7–12.8% of pCR patients still occurred recurrence and metastasis after surgery.5,6 Therefore, it is meaningful to find ways to improve the prognosis of pCR patients.
The National Comprehensive Cancer Network recommended adjuvant chemotherapy (ACT) for patients who have received CRT, regardless of postoperative pathological diagnosis. 7 At the same time, European Society for Medical Oncology guidelines suggested that ACT can be used for high-risk patients. Nevertheless, there was still no consensus among experts on the definition of high-risk patients, which generally included poor differentiation, T4 tumors, the circumferential resection margin, inadequate lymph node dissection, and extramural vascular invasion. Although the results of imaging examinations provided information for judging tumor regression and clinical staging,8,9 there was still a lack of guiding indicators for further treatment after surgery.
Consequently, it remains unclear what subgroup of pCR patients should be treated with ACT. Several studies based on National Cancer Data Base showed that ACT can improve the overall survival (OS) of LARC pCR subgroup, such as patients with positive lymph nodes at baseline.10–13 The necessity of ACT in pCR patients remains controversial, 14 but the downstaging of pCR patients by CRT therapy15,16 showed that tumor biology responsive to therapy. Therefore, pCR patients may constitute a chemosensitive subset and benefit from ACT.
In previous studies, 19.0–42.4% of rectal cancer specimens resected after CRT treatment existed acellular mucin pools.17,18 Whether acellular mucin pools were residual tumors or therapeutic response has been controversial. Some researchers have observed that the existence of acellular mucin pools was linked to poorer outcomes, 19 The incidence of distant metastasis in rectal cancer pCR patients with acellular mucin pools was significantly higher.20,21 Their research demonstrated that the existence of acellular mucin pools may mean that tumors are more aggressive. If the presence of acellular mucin pools indicated a negative prognosis, ACT must be considered in pCR patients with acellular mucin pools. However, no previous studies have performed subgroup analysis of pCR patients with acellular mucin pools. So we conducted this research to analyze the prognostic role of the acellular mucin pools in pCR patients and identify whether ACT was associated with survival outcomes in pCR patients with acellular mucin pools.
Methods
Patients
Between November 2011 and November 2019, a total of 1760 patients diagnosed with LARC received neoadjuvant CRT, followed by radical surgery at the Sixth Affiliated Hospital of Sun Yat-sen University, China. In this research, the treatment information and prognosis of these patients were analyzed retrospectively. Selection criteria for patients included: (1) rectal cancer patients with preoperative staging of stage II/III did not have distant metastasis; (2) patients underwent radical resection, and postoperative pathology was confirmed as pCR; and (3) follow-up information was complete. The exclusion criteria for patients included the following: (1) multiple primary colorectal cancer, (2) the patient had a history of malignant tumors, and (3) local excision was performed. We eliminated 1480 patients, including 10 pCR patients with incomplete information, 1470 patients with non-pCR tumors. Finally, this retrospective study screened 280 eligible patients into our study (Figure 1).

Flow chart for screening suitable patients into this study.
The clinicopathological information and follow-up data were all obtained through the hospital database.
Treatment
The tumor and entire pelvis were treated with radiotherapy 23–25 doses, with a total dose of 46.0–50.4 Gy from Monday to Friday. Patients received preoperative therapy with 4–6 cycles of fluorouracil (folinate 400 mg/m2 intravenous infusion, fluorouracil 400 mg/m2 intravenous infusion, and fluorouracil 2.4 g/m2 intravenous continuous infusion for 48 h) and oxaliplatin 85 mg/m2 intravenously each chemotherapy treatment cycle and received radiotherapy during the 2–4 cycles. After surgery, patients received 6–8 cycles of postoperative mFOLFOX6 chemotherapy, with additional radiotherapy at the physician’s discretion.
Pathological assessment
Two pathologists specializing in colorectal cancer independently re-reviewed the hematoxylin-eosin (H&E) sections of all rectal cancer pathological specimens to determine whether there were acellular mucin pools in the sample. According to American Joint Committee on Cancer guidelines, complete pathological response was defined as no tumor cells were found in the resected specimen and lymph nodes after surgery. The integrity of the examined area was assured when submitted for histopathological analysis to ensure that there are no epithelial cells in the mucin pools. In addition, when routine histological analysis cannot determine whether there were no residual tumor cells, the specimen was further judged by immunohistochemistry. Pathologists individually reviewed specimens from 280 patients, and if the aggregated results differed, the specimens were reviewed and discussed to determine.
Follow-up
All patients were followed up for 5 months, and their health status and physical examination information were recorded. This study follow-up until the date of death or end of the study’s follow-up (March 2022). Routine examination items include clinical physical examination, carcinoembryonic antigen (CEA) testing, and imaging examination. In addition, patients were recommended to undergo computed tomography and colonoscopy every year.
Statistical analysis
All data analyses in this research were performed by IBM SPSS Statistics 26.0. The classification parameters were mainly carried out by chi-square test, and the number and percentage of cases were recorded. Mann–Whitney U-test or Student’s t-test was used to compare continuous variables and express the mean. The Kaplan–Meyer log-rank test was used to compare the effects of influencing factors on 5-year disease-free survival (DFS) and 5-year OS, and the comparison was made. For the potential prognostic factors, the univariate Cox model was used to analyze, and then the multivariate Cox model was used to further determine the statistically significant parameters. Hazard ratios (HRs) and 95% confidence interval (CIs) were output. When the two-sided P value < 0.05, we consider it to be statistically significant.
The reporting of this study conforms to the STROBE statement. 22
Results
In total, 1760 patients received radical surgery after CRT between 2011 and 2019, of whom 290 achieved pCR and 10 pCR patients with incomplete information. The average age of 280 patients was 53.1 years (range: 22–77 years), and 172 patients (61.4%) were male. After the surgery, 74 cases (26.4%) of the rectal specimens were identified as having acellular mucin pools. In the group of pCR patient with acellular mucin pools, 49 patients received ACT. The characteristics, treatment, and pathological characteristics of patients are summarized in Tables 1 and 2. There was no perineural invasion, lymphovascular invasion, and positive circumferential resection margin in all patients.
Comparison of the clinical features and treatment measures of LARC pCR patients with and without acellular mucin pools.

CEA, carcinoembryonic antigen; LARC, locally advanced rectal cancer; pCR, pathological complete response.
Comparison of the clinical features and treatment measures of LARC pCR patients with acellular mucin pools.

ACT, adjuvant chemotherapy; CEA, carcinoembryonic antigen; CRT, neoadjuvant chemoradiotherapy; LARC, locally advanced rectal cancer; pCR, pathological complete response.
Oncological outcomes
After a mean follow-up of 55 months (5–108 months), 38 of 280 patients (13.6%) occurred tumor recurrence, of which 38 had distant metastasis, 4 had local recurrence and distant metastasis. Finally, 17 patients (6.1%) died of aggravated disease during follow-up. There was a significantly difference in 5-year DFS rate between the pCR patients with or without acellular mucin pools (79.7% versus 88.8%, P = 0.037) (Figure 2(a)). ACT treatment had no significant effect on DFS and OS in pCR patients (Figure 3). In the subgroup of patients with acellular mucin pools, 15 of 74 patients (20.3%) occurred distant metastasis and 6 patients (8.1%) died of aggravated disease during follow-up. Comparably, patients with acellular mucin pools had improved 5-year DFS rates (87.8% versus 64.0%, P = 0.003) and 5-year OS rates (95.9% versus 84.0%, P = 0.027) after receiving ACT (Figure 4).

Kaplan–Meier estimates for oncological outcomes in patients with pCR: (a) DFS and (b) OS.
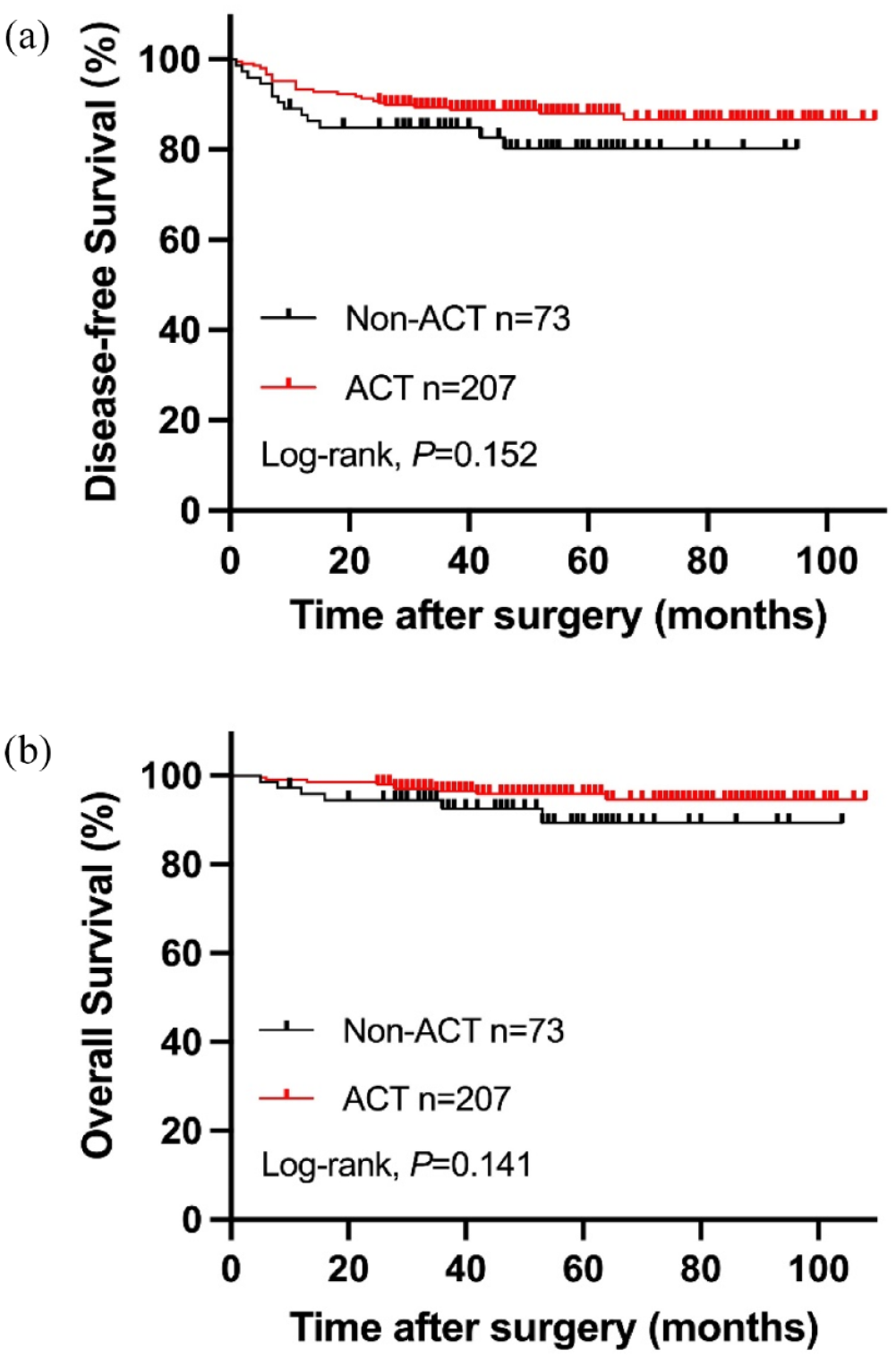
The Kaplan–Meier curve analysis of DFS (a) and OS (b) for LARC pCR patients treated with or without ACT.

The Kaplan–Meier curve analysis of DFS (a) and OS (b) for LARC pCR patients with acellular mucin pools, treated with or without ACT.
Prognostic analysis
In exploring the influencing factors of DFS in pCR patients with acellular mucin pools, univariate analysis showed that clinical N stage positive, pre-CRT CEA > 5 ng/mL will lead to worse DFS. ACT was beneficial to the improvement of DFS. Moreover, in the multivariate analysis, ACT was independent influencing factors of DFS (HR = 0.245, 95% CI = 0.084–0.719, P = 0.010) (Table 3). In addition, ACT and pre-CRT CEA > 5 ng/mL were associated with OS, but multivariate analysis showed that only pre-CRT CEA > 5 ng/mL was independent factors of OS (HR = 8.866, 95% CI = 1.014–77.533, P = 0.049) (Table 4).
Cox regression analysis of prognostic factors for the DFS of LARC pCR patients with acellular mucin pools.

ACT, adjuvant chemotherapy; CEA, carcinoembryonic antigen; CI, confidence interval; DFS, disease-free survival; HR, hazard ratio; LARC, locally advanced rectal cancer; pCR, pathological complete response.
Cox regression analysis of prognostic factors for the OS of LARC pCR patients with acellular mucin pools.

ACT, adjuvant chemotherapy; CEA, carcinoembryonic antigen; CI, confidence interval; HR, hazard ratio; LARC, locally advanced rectal cancer; OS, Overall survival; pCR, pathological complete response.
Discussion
To our knowledge, this study is the first subgroup analysis of pCR patients with acellular mucins pools and found that ACT treatment can improve DFS in pCR patients with acellular mucins pools.
The American College of Pathologists recommended that acellular mucin pools be seen as a therapeutic response rather than a residual tumor, but there was not enough evidence to confirm.20,23 In fact, previous studies have showed that acellular mucin pools formed after CRT were not the same as mucinous adenocarcinoma for their distinct composition and characteristics. Meanwhile, the intensity of radiotherapy affected the formation of acellular mucin pools. 24 Our results showed that acellular mucin pools were found preponderantly in preoperative chemotherapy cycles > 4. Thus, the presence of acellular mucin pools was linked to chemotherapy intensity.
Nevertheless, previous studies suggested that it was inappropriate to treat acellular mucin pools only as therapeutic responses, because there may be an intermediate natural history of the colloidal response with or without residual tumor cells. 24 Yanwu Sun et al. demonstrated that the acellular mucin pool was associated with significantly reduced DFS rate in patients received preoperative CRT and radical surgery. 21 Moreover, the study from Kaneko et al. revealed that rectal cancer patients with acellular mucin pools were more prone to distant metastases. 20 These studies showed that the presence of acellular mucin pools may mean that primary tumors were more aggressive. Our results showed that patients with acellular mucin pools have a higher rate of distant metastases and that acellular mucin pools were risk factors for 5-year DFS and 5-year OS. Theoretically, pre-existing tumor micrometastases cannot be completely treated in all patients. Furthermore, due to the genetic biology and molecular differences between primary tumors and metastatic cells, even in patients who acquired pCR, metastatic cells may still survive and lead to systemic tumor recurrence. 25 So we think that the presence of acellular mucin pools may be an indicator of aggressive tumor biology. In the presence of acellular mucin pools, we should be alert to the possibility of micrometastases.
If the presence of an acellular mucin pool is associated with worse prognosis, then pCR patients with acellular mucin pools should probably be considered for ACT. Although American guidelines recommend use of ACT in LARC patients, the necessity of ACT for pCR is unclear. The study from Fang He et al. revealed that ACT treatment in pCR patients did not affect 3-year survival outcomes. In addition, some studies have proved that pCR patients have a similar prognosis after received ACT.26–28 Nevertheless, distant metastasis of pCR patients can still reach more than 10%.29,30 The study from Jian-Hong Peng et al. proved that ACT is an independent protective factor of DFS, which indicated that ACT may be beneficial to patients who achieve pCR after CRT. 31 Furthermore, Polanco et al. showed that ACT was beneficial for the prognosis of pCR patients with T3/T4 clinical stage and node positive. 32 In addition, Dossa et al revealed that only patients with lymph node positive could benefit from ACT to improve OS. 12 The main problem at present is that there are no clear indicators of which pCR patients will benefit from the addition of ACT. In our research, pCR patients did not benefit from ACT, but the subgroup of pCR patients with acellular mucin pools received ACT was beneficial to the improvement of DFS. Actually, the potential advantage of ACT in improving DFS for pCR patients also could be detected in some small comparative studies.27,33 A plausible explanation for our findings was that tumor downstaging after CRT may indicate favorable tumor biology. This sensitivity to treatment allowed patients to further benefit from ACT. 34 The existence of acellular mucin pools may represent a sign of invasive tumor biology in rectal cancer specimens. So ACT for pCR patients with acellular mucin pools may be beneficial by potentially eradicating residual micrometastatic disease. To improve the prognosis of pCR patients, we suggested that pCR patients with acellular mucin pools receive ACT. In the meantime, we recommend closer follow-up of patients with acellular mucin pools.
In multiple studies, acellular mucin pools were found in 16.1–34.2% of rectal cancer specimens from pCR patients,35,36 which was not an uncommon event. Consequently, acellular mucin pools require careful consideration of their true clinical significance. It was undeniable that there were some limitations in our study such as retrospective, single center, and limited sample quantity. We hope that more rigorous prospective studies can demonstrate the importance of ACT in patients with acellular mucin pools.
Conclusion
In conclusion, our study provided more concrete evidence on the questions raised, showing that the acellular mucin pools may represent a sign of invasive tumor biology and lead to a decrease in DFS. ACT treatment was beneficial in improving prognosis of pCR patients with acellular mucin pools, so ACT should be considered for them.
Supplemental Material
sj-doc-1-tag-10.1177_17562848221117875 – Supplemental material for Clinical significance of adjuvant chemotherapy for pathological complete response rectal cancer patients with acellular mucin pools after neoadjuvant chemoradiotherapy
Supplemental material, sj-doc-1-tag-10.1177_17562848221117875 for Clinical significance of adjuvant chemotherapy for pathological complete response rectal cancer patients with acellular mucin pools after neoadjuvant chemoradiotherapy by Mian Chen, Jianwei Zhang, Yujie Hou, Huashan Liu, Xinjuan Fan, Shuangling Luo, Zhanzhen Liu, Huanxin Hu, Sicong Lai, Liang Kang and Liang Huang in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
