Abstract
Background:
To date, the prognostic significance of acellular mucin pools in tumors from patients with locally advanced rectal cancer (LARC) undergoing preoperative chemoradiotherapy (CRT) and subsequently obtaining pathological complete response (pCR) has not been well determined. Our current study aimed to explore the prognostic impact on these patients of acellular mucin pools.
Methods:
We collected clinical data from 117 consecutive LARC patients who achieved pCR after preoperative CRT and then underwent radical resection. Two groups of patients were generated, according to the presence or absence of acellular mucin pools. The 5-year disease-free survival (DFS) and overall survival (OS) rates were compared between the two groups of patients.
Results:
A total of 27 (23.1%) patients presented with acellular mucin pools. At a median follow-up period of 64 months, patients with acellular mucin pool showed a 5-year DFS rate (96.3% versus 83.7%, p = 0.110) and 5-year OS rate (100% versus 87.5%, p = 0.054) statistically similar to those of patients without acellular mucin pools. In univariable and multivariable Cox regression analyses, the presence of acellular mucin pools was not determined as an independent risk factor for DFS [hazard ratio (HR): 0.222; 95% confidence interval (CI): 0.029–1.864; p = 0.145] or OS (HR: 0.033; 95% CI: 0.000–9.620; p = 0.238).
Conclusions:
Acellular mucin pools had no significant prognostic impact on LARC patients showing pCR after preoperative CRT.
Introduction
It has been widely recognized that preoperative chemoradiotherapy (CRT) combined with total mesorectal excision (TME) is an effective treatment pattern for locally advanced rectal cancer (LARC).1,2 As shown in our previous report, preoperative CRT has a well-established benefit of contributing to tumor response, and, thus, offers an opportunity to patients with low-lying rectal cancer undergoing sphincter-preserving resection.3,4 In addition, 20–30% of patients successfully achieve pathological complete response (pCR) after preoperative CRT, which indicates a favorable prognosis.5–7 Nevertheless, pCR does not absolutely represent disease-free survival (DFS) after radical treatment, in that 7.7–11% of patients ultimately develop distant metastasis postoperatively.8,9 Accurate pathological staging is important to determine the recurrence risk and thus provide guidance for postoperative treatment decisions. In this context, there is an urgent need to define pathological parameters related to the survival outcome of patients with pCR.
Some studies have reported that acellular mucin pools are present in 40% of resected rectal specimens after preoperative CRT.10,11 Specifically, for a specimen characterized by both pCR and acellular mucin pools, in order to confirm the true pCR, it is important to identify whether acellular mucin pools are part of the residual tumor or a type of treatment response. However, the prognostic implications of acellular mucin pools for patients undergoing preoperative CRT remain controversial. A previous study reported that acellular mucin pools were indicative of aggressive biological behavior of the malignant tumor, which leads to worse prognosis, 11 while other results suggested that acellular mucin pools in rectal cancer after preoperative CRT were not associated with poor prognostic outcomes. 12 To date, although several studies have focused specifically on investigating the prognostic significance of acellular mucin pools of pCR specimens, results have failed to reach a unified agreement due to limited sample numbers. 13 As it is vital to identify the prognostic implications of acellular mucin pools in LARC patients showing pCR after preoperative CRT, this was the aim of our current study.
Patients and methods
Patient selection
The current study collected clinical data from 577 consecutive LARC patients undergoing preoperative chemoradiotherapy and surgery from December 2003 to April 2014 at Sun Yat-sen University Cancer Center, China. The selection criteria for the patients included: (1) pretreatment stage II/III disease (2010 American Joint Committee on Cancer staging system), (2) radical resection for rectal tumor, and (3) postoperative specimen confirmed as pCR. Ultimately, we excluded a total of 460 patients, including 33 patients with distant metastases before CRT, 41 patients receiving palliative tumor resection, 385 patients with non-pCR tumors, and 1 patient having inadequate follow-up data. As a result, 117 patients were selected for this study (Figure 1). General patient characteristics, tumor traits, treatment details and follow-up data were recorded from our both electronic medical record system and follow-up tracking system. All procedures were conducted in accordance with the ethical standards of the 2013 World Medical Association Declaration of Helsinki. The requirement for informed consent was waived due to the retrospective nature of the current study; patient data were kept confidential. The current study was approved by the institutional review board of the Independent Ethics Committee at Sun Yat-sen University Cancer Center.

Flowchart representing the selection of eligible patients for the present study.
Treatment
Patients were given preoperative radiation therapy (RT) via three-dimensional conformal radiation therapy (3D-CRT) or intensity-modulated radiation therapy (IMRT), with a total dose of 46–50 Gy delivered in daily fractions of 2.0 Gy daily on 5 consecutive days every week. Concurrent chemotherapy included two regimens: (1) the XELOX regimen was performed as 130 mg/m2 oxaliplatin administered intravenously on day 1, and 1000 mg/m2 capecitabine administered orally twice daily on days 1–14 for a 3-week cycle; (2) the capecitabine alone regimen was performed as 1000 mg/m2 capecitabine administered orally twice daily on days 1–14 for a 3-week cycle. If a patient could not tolerate the full dose, or suffered from severe toxic effects, the RT or chemotherapy was stopped. At 6–8 weeks after completion of CRT, the patients were scheduled for TME. XELOX regimen adjuvant chemotherapy was recommended 3–6 weeks after surgery.
Pathological assessment
All resection embedded specimens were assessed by two pathologists. The definition of pCR was the absence of viable tumor cells, with or without fibrotic masses or acellular mucin pools on the proximity to primary tumor and lymph nodes. 14 Acellular mucin pools are defined as mucin pools composing over 10% of the entire tumor lesion area as revealed by hematoxylin-eosin (H&E) staining. 12 A pathologist, Shixun Lu, reviewed all samples initially diagnosed as ‘acellular mucin pools’. Patients were divided into acellular mucin pool and non-acellular mucin pool subgroups based on the presence or absence of acellular mucin pools in rectal primary tumor, as shown in Figure 2.

Representative pathologic micrographs of H&E staining of specimens from patients achieving pCR after preoperative CRT. (a) Presence of acellular mucin pools and (b) absence of acellular mucin pools.
Follow up
Patients were recommended to undergo a follow up every 3 months for 2 years after tumor resection, and then every 6 months for 3–5 years. Routine assessment included clinical check-up, carbohydrate antigen (CA19-9) and carcinoembryonic antigen (CEA) testing, abdominopelvic ultrasonography, and chest X-ray. The recommended Chest/abdominal/pelvic computed tomography (CT) and colonoscopy were conducted annually. DFS was defined as the duration from rectal tumor resection to the last follow-up date or tumor recurrence, and overall survival (OS) was recognized as the duration from rectal tumor resection to the last follow-up date or death from any cause. The last follow up was conducted in May 2019.
Statistical analysis
Analysis of total data was performed via IBM SPSS statistics Version.25 (IBM SPSS INC., Chicago, IL, USA). Chi-squared or Fisher’s exact test was applied to compare categorical parameters, which were presented as numbers of cases followed by percentages. Continuous variables were compared using Student’s t test or the Mann–Whitney U test, and were presented as means (standard deviation). Expected 5-year DFS and OS rates were estimated and compared using Kaplan–Meier log-rank test. Potential prognostic factors were selected using univariate Cox models, and statistically significant parameters were further determined by multivariate Cox models. Hazard ratios (HRs) and 95% confidence intervals (CIs) were both generated. p values less than 0.05 were considered statistically significant with two-sided statistical tests.
Results
Patient characteristics and treatment
Among the 117 patients [median age 56 years (range 19–85 years)], 80 (68.3%) were male. After treatment, 27 (23.1%) patients showed acellular mucin pools in rectal specimens, whereas 90 (76.9%) patients did not. Patient characteristics and treatments were compared between the acellular mucin pool group and non-acellular mucin pool group. As a result, age, sex, distance of inferior tumor margin from the anal verge (DAV), pretreatment tumor differentiation, pretreatment CEA, clinical stage, preoperative chemotherapy regimen, total radiation dose, surgical procedure, and patient acceptance of adjuvant chemotherapy were comparable between the two groups (Table 1).
Comparison of the clinicopathologic characteristics and treatment patterns of patients showing pCR with acellular mucin pools and without acellular mucin pools.

APR, abdominoperineal resection; CEA, carcinoembryonic antigen; DAV, distance of the inferior tumor margin from the anal verge; LAR, low anterior resection; pCR, pathological complete response; SD, standard deviation.
Oncological outcomes
During the median follow-up period of 64 months (range 4–125 months), 16 (13.6%) patients developed tumor recurrence, including 1 with locoregional recurrence, 14 with distant metastasis, and 1 with both locoregional recurrence and distant metastasis. As a result, 12 (10.3%) patients died of disease progression. For all patients, the 5-year DFS and OS rates were 90.3% and 86.4%, respectively. The 5-year DFS rate was 96.3% for patients with acellular mucin pools, and 83.7% for patients without acellular mucin pools; these values were comparable (p = 0.110) (Figure 3a). Similarly, the 5-year OS rate was 100% for patients with acellular mucin pools, and 87.5% for patients without acellular mucin pools; these values were not significantly different (p = 0.054) (Figure 3b).

Kaplan–Meier curves comparing 5-year (a) DFS and (b) OS rates based on the presence of acellular mucin pools in patients with pCR after preoperative CRT and radical surgery.
Prognostic analysis of clinical factors
In univariate analysis, clinical T4 stage (HR: 3.972, 95% CI: 1.438–10.972, p = 0.008), poor tumor differentiation (HR: 3.002, 95% CI: 1.089–8.273, p = 0.034), and abdominoperineal resection (HR: 2.844, 95% CI: 1.058–7.644, p = 0.038) were associated with shorter DFS. In multivariate analysis, clinical T4 stage (HR: 4.251, 95% CI: 1.494–12.097, p = 0.007) and abdominoperineal resection (HR: 4.263, 95% CI: 1.480–12.276, p = 0.007) were independent prognostic factors for DFS (Table 2). For OS, univariate analysis revealed that clinical T4 stage (HR: 4.706, 95% CI: 1.413–15.675, p = 0.012), poor tumor differentiation (HR: 7.034, 95% CI: 2.229–22.196, p = 0.001), and abdominoperineal resection (HR: 6.610, 95% CI: 1.788–24.433, p = 0.005) were associated with shorter OS, while a radiotherapy dosage of 50 Gy (HR: 0.253, 95% CI: 0.067–0.946, p = 0.041) and preoperative chemotherapy with the XELOX regimen (HR: 0.208, 95% CI: 0.054–0.808, p = 0.023) were associated with prolonged OS. In multivariate analysis, clinical T4 stage (HR: 5.460, 95% CI: 1.580–18.865, p = 0.007), poor tumor differentiation (HR: 6.687, 95% CI: 1.851–24.158, p = 0.004), and abdominoperineal resection (HR: 10.886, 95% CI: 2.568–46.138, p = 0.001) were negative prognostic factors for OS (Table 3).
Univariate and multivariate analyses of prognostic factors for the DSF of patients with pCR after preoperative chemotherapy.
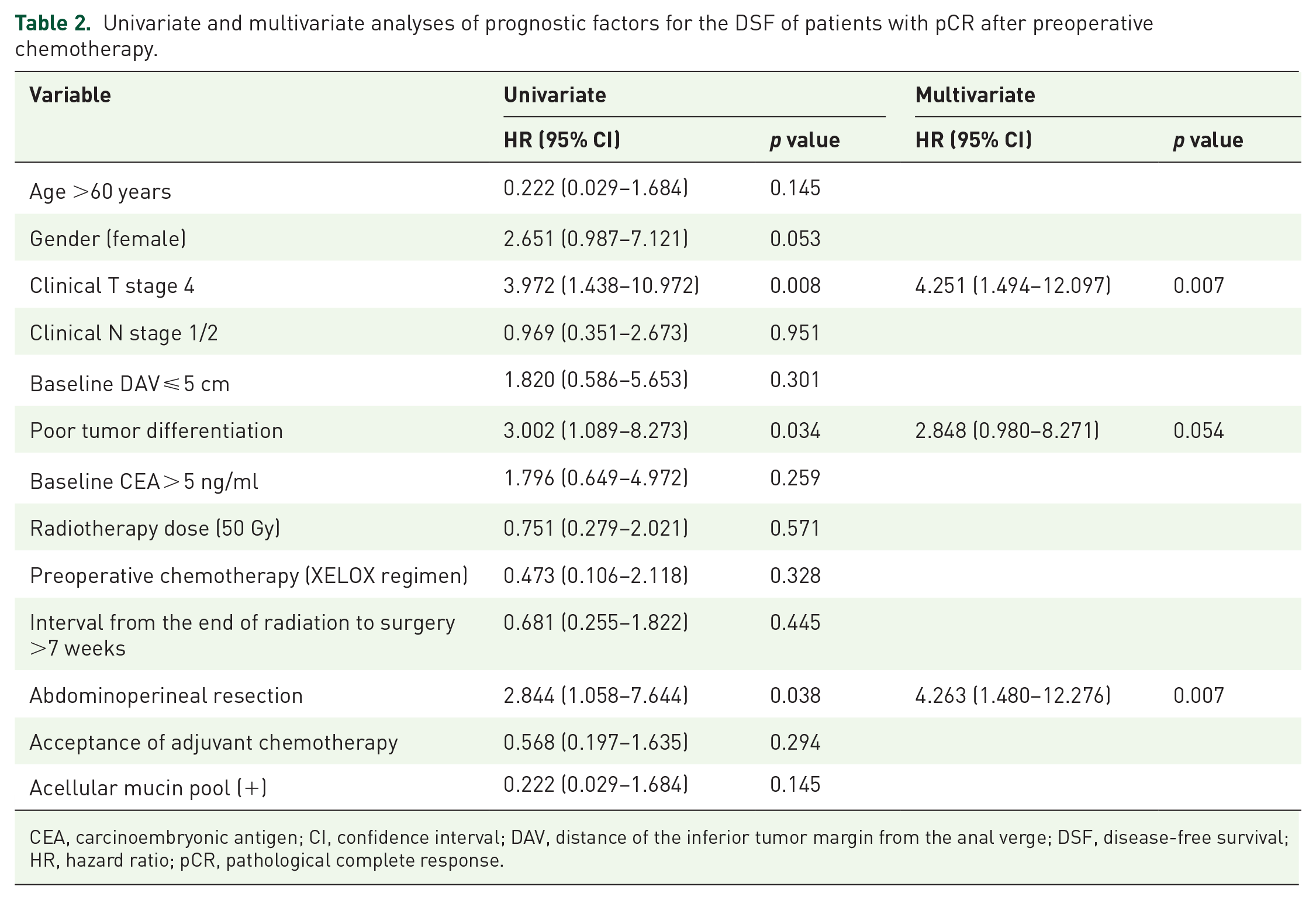
CEA, carcinoembryonic antigen; CI, confidence interval; DAV, distance of the inferior tumor margin from the anal verge; DSF, disease-free survival; HR, hazard ratio; pCR, pathological complete response.
Univariate and multivariate analyses of prognostic factors for the OS of patients with pCR after preoperative chemotherapy.

CEA, carcinoembryonic antigen; CI, confidence interval; DAV, distance of the inferior tumor margin from the anal verge; HR, hazard ratio; pCR, pathological complete response.
Discussion
Our current study reported a 27% prevalence of acellular mucin pools present in rectal specimens from patients with pCR following preoperative CRT. The presence of acellular mucin pools did not show an association of any patient characteristics, tumor trait, or treatment pattern. In addition, our results revealed comparable long-term survival among patients with pCR with and without acellular mucin pools. Thus, we suggest that acellular mucin pools after preoperative CRT do not have a significant prognostic impact on LARC patients who achieve pCR.
To date, five studies have revealed the prevalence rates of acellular mucin pools in rectal specimens from patients showing pCR (Table 4).9,13–17 Among these studies, the proportion of acellular mucin pools on rectal tumor specimens was identified in between 16.1% and 34.2% of cases. In the current study, the prevalence of acellular mucin pools was 27%, and thus falls within this range of 16.1–34.2%. Therefore, the existence of acellular mucin pools in rectal specimens from patients achieving pCR is not a rare phenomenon, which warrants careful pathological assessment. A study by Lim and colleagues revealed that the presence of acellular mucin pools was related to poor tumor differentiation, male gender, and a short duration between CRT and tumor resection. 15 In contrast, other studies, including ours, observed that the presence of acellular mucin pools was not related to any specific patient characteristics, tumor trait, or treatment.16,18 The association of various clinical factors with the presence of acellular mucin pools after treatment with preoperative CRT needs further validation in future studies.
Studies of patients with locally advanced rectal cancer showing pCR after preoperative chemoradiotherapy followed by total mesorectal excision.

DFS, disease-free survival; OS, overall survival; pCR, pathological complete response.
Reviewing the available series of studies, the prognostic implications of acellular mucin pools in resected rectal specimens from patients showing pCR are still not well understood and remain under debate. Recently, a study by Sun and colleagues demonstrated that pCR patients with acellular mucin pools have a poorer DFS rate (67.4% versus 96.5%, p = 0.003) compared with those without acellular mucin pools, which revealed acellular mucin pools as a possible indicator of aggressive biological behavior of the tumor. 9 Similarly, despite a lack of statistical evidence, Campos-Lobato and colleagues and Bhatti AB and colleagues both observed a tendency for pCR patients with acellular mucin pools to present increased distant metastasis and reduced DFS and OS rates.16,18 Inconsistent with the results of these latter two studies, Lim and colleagues demonstrated that the presence of acellular mucin pools had no significant association with poor outcome in DFS (97.0% versus 93.0%, p = 0.131) and OS (96.8% versus 95.9%, p = 0.314) rates, which were rather similar between the patients with and without acellular mucin pools, respectively. 15 In addition, a study by Smith and colleagues reported similar 7-year DFS (81% versus 87%, p = 0.764) and OS (85% versus 92%, p = 0.954) rates in patients with and without acellular mucin pools, respectively. 17 In our study, we even found a trend that patients with acellular mucin pools have a slightly superior 5-year DFS (96.3% versus 83.7%, p = 0.110) and OS (100% versus 87.5%, p = 0.054) rates than those without acellular mucin pools.
The inconsistency in these results can be attributed to the following: determination of the prognostic implication of acellular mucin pools is constrained by small sample size.16,18 As shown in Table 4, the number of patients showing pCR with acellular mucin pools was very small, ranging from 11 to 35 patients in each study, which impaired the statistical power to identify the survival difference between the two groups. Another important reason concerns the accuracy of the pathological diagnosis of acellular mucin pools. Distinct acellular mucin pools were well defined as constituting an area over 10% of the entire tumor lesion area. However, the assessment of acellular mucin pools involves a combination of the following factors: the amount of acellular mucin pools in the entire tumor lesion, tumor cell cellularity in mucin pools, and the location of acellular mucin pools in the bowel wall. 12 However, most of the studies cited did not note details of the assessment of acellular mucin pools in rectal tumor specimens; subtle differences in the pathological assessment of acellular mucin pools inevitably exist among studies, which might lead to different results. In addition, the distinct follow-up duration was not a negligible factor. In the studies by Lim and colleagues, Campos-Lobato and colleagues, and Bhatti AB and colleagues, the median follow-up duration was less than 5 years.15,16,18 The relatively short follow up may not accurately determine the effect of acellular mucin pools on long-term survival.
To date, the necessity for adjuvant chemotherapy for LARC patients who achieve pCR after preoperative CRT and radical surgery is still unclear.19–21 In the current study, 85.2% (23/27) of patients with acellular mucin pools received adjuvant chemotherapy. On account of the small sample size of the patients showing acellular mucin pools, we could not define the efficiency of adjuvant chemotherapy for acellular mucin pools. The therapeutic role of adjuvant chemotherapy in patients showing pCR and acellular mucin pools urgently needs to be demonstrated. Accordingly, postoperative management decisions should be judicious of particular patient characteristics and tumor traits. Our previous findings showed that oxaliplatin-containing adjuvant chemotherapy may offer survival benefits to patients showing pCR after preoperative CRT and radical surgery, especially to those aged <60 years. 22 Additionally, the current study found that the clinical T4 stage, poor tumor differentiation, and abdominoperineal resection were negative prognostic factors for patients achieving pCR after preoperative CRT. Thus, we considered that adjuvant chemotherapy should also be recommend to patients with these risk factors.
Several limitations of our study need to be pointed out. Firstly, the current study was conducted using a retrospective method with a limited volume size of patients from a single center. Patient selection and treatment bias were unavoidable. Secondly, the lack of validation for the results from centers was another limitation of the study. Thirdly, pretreatment molecular features of tumors that might be related to the occurrence of acellular mucin pools, such as expression of the mismatch repair protein (MMR) and the mutation of KRAS, BRAF, and PIK3CA oncogenes, were also not investigated. In the cohort in the study by Sun and colleagues, positive mucin 1 staining was significantly associated with the presence of acellular mucin pools, and the expression level was higher in patients with acellular mucin pools than in patients without acellular mucin pools. 9 Fourthly, we failed to record the number and location of acellular mucin pools in the bowel wall. The prognostic implication of different patterns of mucin pools warrants further investigation.
Conclusion
In summary, our current study provided more concrete evidence that acellular mucin pools are not associated tumor cells, and thus have no significant adverse impact on the long-term survival of patients achieving pCR after preoperative CRT. We look forward to studies with large sample numbers that will further investigate this topic and provide a better understanding of the prognostic significance of acellular mucin pools.
Footnotes
Acknowledgements
Authors Lin Zhang, Huajie Guan and Qiuyun Luo contributed equally to this work. We want to thank the pathologist Shixun Lu from the Department of Pathology at Sun Yat-sen University Cancer Center for assessment of acellular mucin pools.
Author contributions
LZ, HJG, and JHP performed data analysis and drafted the manuscript; JHP, QYL, LFY, and YLM collected the clinical data and follow-up results; ZZP, XJW, and JZL supervised the current study; JZL proofread the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Natural Science Foundation of Guangdong Province (grant number 2017A030310204, 2018A030310260), Medical Scientific Research Foundation of Guangdong Province (grant number A2017545), National Natural Science Foundation of China (grant number 81772595), and Sun Yat-sen University Clinical Research 5010 Program (grant number 2015024).
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The current study was performed in accordance with the ethical standards of the World Medical Association Declaration of Helsinki of 2013. The requirement for informed consent was waived because of the retrospective nature of the current study.
Consent for publication
Not applicable.
