Abstract
Background:
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus enters the cells via angiotensin-converting enzyme 2 receptor; therefore, tissues expressing this receptor are potential targets for infection. Although many studies have observed gastrointestinal (GI) symptoms in coronavirus disease 2019 (COVID-19) patients, prevalence and clinical impact are still uncertain due to the heterogeneity of reports and obstacles to generalization.
Methods:
In this cross-sectional study, we included symptomatic patients requiring hospital admission, with a confirmed diagnosis of COVID-19 by nasopharyngeal polymerase chain reaction test, between 18 March and 30 May 2020. Demographic data, symptoms at onset, vital signs, and laboratory tests at admission were recorded.
Results:
In all, 300 patients were included (57%M, 43%F). GI symptoms were mainly diarrhea (13%), anorexia (4.3%), vomiting (3%), and abdominal pain (2.3%). Overall, males were younger (68 years versus 76 years; p = 0.01); patients with GI manifestations at disease onset required significantly faster hospital admission and showed larger GI complication rates. GI symptoms were associated with abnormal high aspartate aminotransferase and alanine aminotransferase serum titers, especially in male patients.
Conclusion:
Our study on an Italian population during the outbreak of the COVID-19 pandemic shows that GI symptoms are part of the spectrum of the SARS-CoV-2 infection and could be the only manifestations at disease onset. Although patients with GI symptoms were associated with faster hospital admission and liver involvement, prognosis was not affected.
Keywords
Introduction
Coronaviruses are single, plus-stranded RNA viruses that infect different animal species, including humans, and cause respiratory, neurological, and hepatic diseases. 1 Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus has been shown to enter the cells via angiotensin converting enzyme 2 (ACE2) receptor; therefore, any tissue that expresses this receptor is a potential target for infection.
Gastrointestinal (GI) epithelial cells2,3 and alveolar type II cells of the lung 4 highly express ACE2 receptors and studies have, in fact, shown that SARS-CoV-2 infects the GI tract via its viral receptor ACE2. Moreover, viral RNA has also been isolated from stool specimens of patients with coronavirus disease 2019 (COVID-19), a fact that could imply fecal–oral transmission in addition to droplet transmission.5,6 An increasing number of studies have reported a range of GI symptoms, including diarrhea, nausea, vomiting, and abdominal discomfort, 7 in patients with COVID-19 disease.
Unfortunately, many of these studies have been performed on Asian populations. In particular, as noted in a recent publication, 8 81% of the reported studies are from China, 29 of which (39%) from the Hubei Province. Symptom prevalence varies from 15.7% to 21.6%, often without any distinction regarding presentation along with or before respiratory symptoms. Considering the relationship between GI symptoms and their clinical impact, isolated studies have reported significantly higher rates of severe acute respiratory distress syndrome (ARDS) and liver injury.9,10
The aim of this study was to evaluate the prevalence of GI symptoms in confirmed hospitalized cases of SARS-CoV-2 infection in a Caucasian population. Moreover, we evaluated the percentage of GI manifestations presenting as primary symptoms of disease, as well as their correlation with disease severity, outcome, and laboratory findings.
Methods
Study design and data source
This cross-sectional study was conducted on a cohort of COVID-19 patients who were admitted in three Regional Hospitals (SS. Annunziata, Chieti; Giuseppe Mazzini, Teramo; and San Liberatore, Atri) of the Abruzzo Region in Central Italy. The study was approved by the local ethics committee (Comitato Etico delle Province di Chieti e Pescara, approval number 07/18.03.2021). We included consecutive symptomatic patients requiring hospital admission, with a confirmed diagnosis of COVID-19 infection on nasopharyngeal (NP) polymerase chain reaction testing for SARS-CoV-2 from 18 March to 30 May 2020; all subjects were de-identified appropriately. Data on patients not requiring hospitalization or managed on an ambulatory basis were unavailable and thus not considered for the purposes of this study. We excluded non-adults (<18 years of age), pregnant patients, and/or clinically suspect cases where SARS-CoV-2 NP swab results were inconclusive/negative.
We recorded demographic data, clinical symptoms at the onset, both typical (fever, myalgia, cough, fatigue, sore throat, headache) and GI (ex-novo occurrence of diarrhea, abdominal pain, nausea, vomiting, and anorexia), comorbidities (coronary heart disease, hypertension, diabetes, neoplasia, chronic renal failure, cerebrovascular disease, chronic lung disease), vitals sign at presentation, and laboratory tests at admission [blood count, C-reactive protein levels, aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyl transferase, alkaline phosphatase, Na+, K+, albumin, total serum protein, creatinine, myocardial enzymes, blood glucose, pancreatic enzymes, lactate dehydrogenase]. Diarrhea was defined as the passing of loose stools >3 times per day. The date of disease onset (defined as the day when first symptom of any type was noticed) and the date of GI symptom onset were recorded. Clinicians evaluated symptoms on admission by interview according to requirements of the study protocol. Therapeutic decisions were independently taken by the local medical staff after achieving individual consent to treatment as per current local practice.
GI complications (bleeding, acute pancreatitis, acute liver disease) and outcomes [exitus, intensive care unit (ICU), remission] were also recorded. Grade of ARDS were evaluated by PaO2/FIO2 ratio, according to the Berlin classification 11 : mild (200 mm Hg < PaO2/FIO2 ⩽ 300 mm Hg), moderate (100 mm Hg < PaO2/FIO2 ⩽ 200 mm Hg), and severe (PaO2/FIO2 ⩽ 100 mm Hg). Criteria for ICU admission were respiratory distress with peripheral SpO2 <92% despite the use of high-flow nasal cannula or ventilatory support with non-invasive ventilation (NIV) or continuous positive airway pressure. Hospital discharge of non-ICU patients required a negative SARS-CoV-2 polymerase chain reaction NP swab with no need of supportive O2 therapy. All three centers adopted the same criteria for disease severity, ICU admission, and hospital discharge.
The reporting of this study conforms to the STROBE statement. 12
Statistical analysis
Statistical analysis was performed using the IBM SPSS software version 20 (SPSS Inc, Armonk, NY, USA). Results for continuous variables are presented as mean ± confidence interval (CI) or median with CI (95% CI), as appropriate. Categorical variables were analyzed using the chi-square test; for continuous variables, Student’s t test or Mann–Whitney U test was used as appropriate. A p value of <0.05% was considered as statistically significant.
Results
A total of 344 patients were recruited; we excluded 2 patients under 18 years of age, 27 registry duplicates, and 15 negative subjects on NP swab despite the radiological imaging suggestive of SARS-CoV-2-related disease. In total, 300 patients [M = 172 (57%), F = 128 (43%), M/F = 4/3] were enrolled with mean age of 70.3 years (95% CI: 68.51–72.20 years). Male patients were statistically younger than females (respectively 68 years, 95% CI: 6.33–70.96 versus 76 years, 95% CI: 69.66–75.64; p = 0.01). Patient characteristics and symptomatic onset are shown in Table 1 and Figure 1, respectively; comorbidities of male and female patients with GI manifestations of SARS-CoV-2 infection are shown in Table 2. Patients who developed GI symptoms did not have predisposing conditions such as inflammatory bowel disease or chronic liver disease at admission. No use of immunosuppressants was noted.
Characteristics of patients with and without GI symptoms.
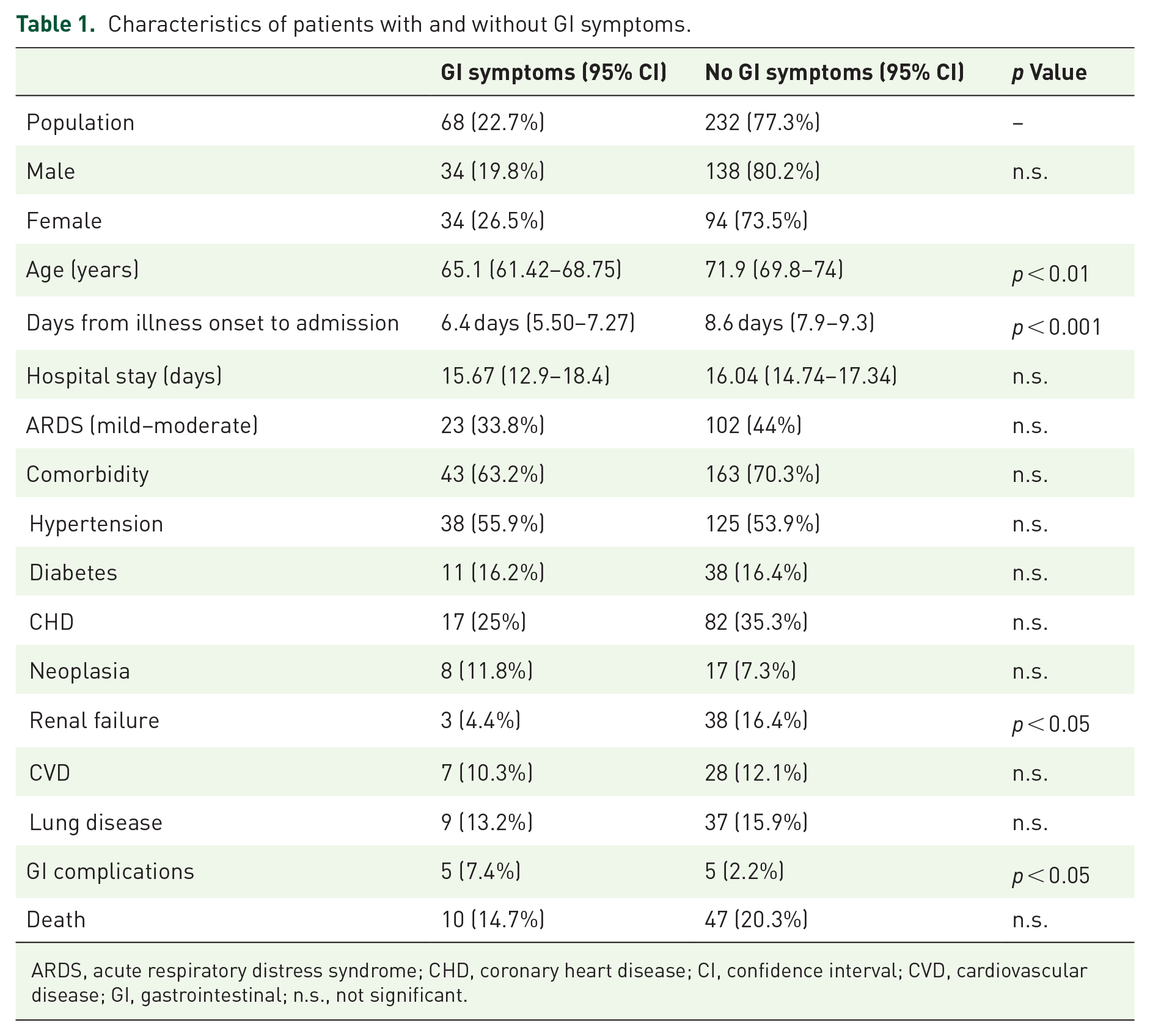
ARDS, acute respiratory distress syndrome; CHD, coronary heart disease; CI, confidence interval; CVD, cardiovascular disease; GI, gastrointestinal; n.s., not significant.

Distribution of symptoms in the subject population.
Distribution of comorbidities in male and female patients with GI manifestations of SARS-CoV-2 infection.
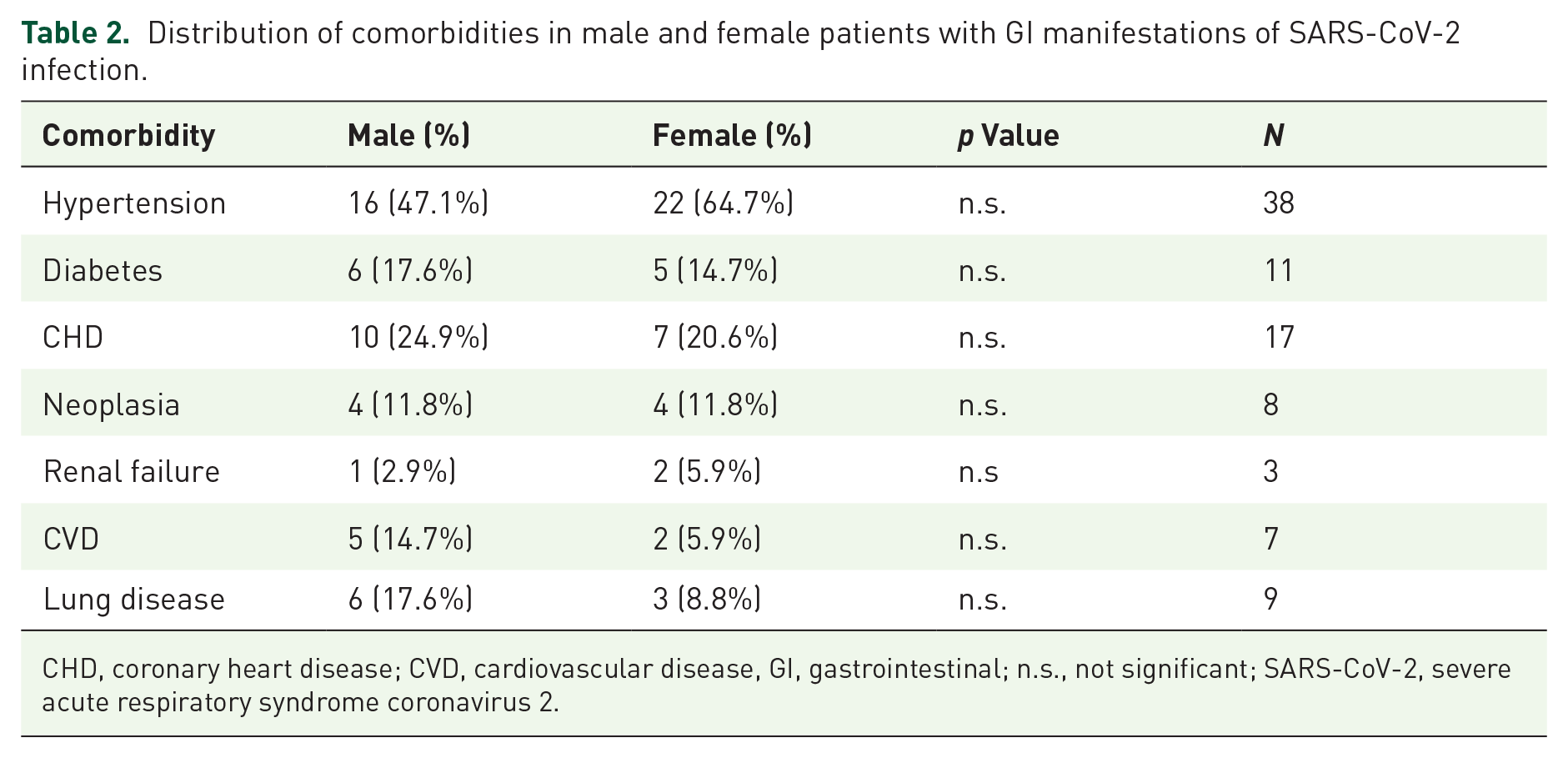
CHD, coronary heart disease; CVD, cardiovascular disease, GI, gastrointestinal; n.s., not significant; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
The overall prevalence of GI symptoms occurring before hospitalization (with or without typical symptoms) was 22.7% (68/300); relative prevalence was diarrhea (n = 39; 13%) with a mean bowel movement of 4.3/day (95% CI: 3.96–4.71), anorexia (n = 13; 4.3%), vomiting (n = 9; 3%), and abdominal pain (n = 7; 2.3%). Out of 68 patients presenting with GI symptoms, 60 patients experienced also typical symptoms while 8 (11.8%) presented GI symptoms alone (Figure 1).
GI involvement in 10.4% (31/300) of cases, alone or accompanying other typical symptoms, was present at disease onset. Patients presenting with GI symptoms were significantly younger (respectively: 65.1 years, 95% CI: 61.42–68.75 versus 71.9 years, 95% CI: 69.8–74; p < 0.01). Analysis by sex demonstrated that this trend was statistically significant in females (p < 0.002) but not in males (Table 3). The average time from symptom onset to hospital admission was 8.1 ± 6.4 days. Patients presenting GI symptoms required a significantly faster hospital admission (Table 1). No statistical differences were observed regarding duration of hospital stay or prevalence of respiratory distress, comorbidity, and death. GI complications (bleeding, acute pancreatitis, acute liver disease) were significantly more frequent in patients with GI symptoms at presentation [odds ratio (OR) 3.6, 95% CI: 1.01–12.8, p < 0.05; Table 1]. After stratification by sex, this effect was only present in male patients (OR 9.07, 95% CI: 1.59–51.8, p < 0.05; Table 3).
GI involvement by sex.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; GI, gastrointestinal; n.s., not significant.
Considering laboratory findings, no significant differences between mean values of the tested parameters were observed, with the sole exception of a lower mean potassium concentration in the sera of COVID-positive patients (p < 0.05), albeit within the normal range (Table 4). The presence of GI symptoms was associated with abnormal high AST and ALT serum titers; however, this reached statistical significance for ALT titers only (Figure 2). Stratification by sex showed that abnormal titers of both AST and/or ALT were associated with GI symptoms in males (p < 0.05) but not in females (Figure 2 and Table 3). We did not find any association between GI symptoms and serum abnormalities including hypokalemia, hypophosphatemia, hypoalbuminemia, etc., as defined by respective cutoffs.
Mean values of blood and respiratory parameters.

ALP, alkaline phosphatase; ALT, alanine aminotransferase; ARDS, acute respiratory distress syndrome; AST, aspartate aminotransferase; CI, confidence interval; CRP, C-reactive protein; γGT, gamma-glutamyl transferase; LDH, lactate dehydrogenase; WBC, white blood cell.

Prevalence of hypertransaminasemia overall and by sex.
Discussion
In this study, we aimed to evaluate the prevalence of GI symptoms in hospitalized patients with COVID-19. To this end, we assessed the GI involvement in one of the largest Caucasian populations of COVID-19 patients recruited for this purpose. Overall, nearly a quarter (22.7%) of patients experienced GI symptoms prior to hospital admission, with more than 10% of cases showing GI symptoms at disease onset. Our data suggest that GI symptoms are part of the SARS-CoV-2 infection symptom spectrum, in addition to respiratory and neurological symptoms. This is further suggested by the observation that in 3% of population enrolled in our study GI symptoms were the only manifestations at disease onset.
Although this association is currently accepted, for diarrhea in particular, similar studies evaluating the prevalence of GI symptoms in COVID-19 patients are unfortunately highly heterogeneous 13 so that the exact burden of GI involvement remains uncertain.14,15 Data from the literature are mainly from Asian populations, and seldom from the western world. In the metanalysis of 60 studies on a total of 4243 patients, 13 88.3% (n = 53) of the included publications were from China, while only one patient was from Europe 16 and one patient from the United States, 5 the latter including only three patients (the first three cases reported by the United States). In this metanalysis, the pooled prevalence of GI symptoms was 17.6%; however, this was heavily dependent on patient origin as in Chinese patients, prevalence of GI symptoms was less than half that of the non-Chinese (16.1%, 95% CI: 10.9–23 versus 33.4%, 95% CI: 15.2–58.3, respectively). In a recent publication on 318 confirmed COVID-19 patients from the Massachusetts area hospitals (USA), 17 the overall prevalence of any GI symptom was 61.3%, with loss of appetite and diarrhea accounting for 34.8% and 33.7% of all GI symptoms, respectively. Other authors from the United States reported a 20.6% of prevalence of GI symptoms, with diarrhea as the main symptom in 14.7% of patients. 18 In an Italian cohort from Milan, 19 the overall prevalence of diarrhea was 27.1%, while vomiting was reported in 4% of cases, while in another cohort from Rome, 20 48.7% of patients referred diarrhea at admission.
Most studies have not been specifically designed to evaluate GI involvement in SARS-CoV-2 infection; in fact, in many of those studies, only diarrhea was actually recorded, often without the use any objective measure (e.g. bowel movement per day).21–25 Moreover, the populations studied to date were substantially different with regard to demographics and medical setting (e.g. medical division, ICU), inclusion methodology, data collection, disease severity criteria used, and patient stratification.
The presence of GI symptoms is not surprising considering that the virus can directly infect and colonize the mucosa of the GI tract. Some studies performed during the previous SARS-CoV epidemic had already shown a tropism for the GI tract and the presence of viral RNA in the feces. 26 SARS-CoV-2 RNA has been isolated in fecal samples in the first case of COVID-19 in the United States 5 ; this was subsequently confirmed in other series where viral RNA in feces was detected in 34.6–48.1% of cases.13,27,28 Importantly, viral RNA in feces showed remarkably slower clearance rates compared to NP samples (mean difference 7.1 days, 95% CI: 1.2–13.0), remaining detectable even after negative NP swab in 27% of patients (pooled prevalence, 95% CI: 15–44%). 28 Moreover, Xiao et al. 29 reported having isolated infectious viral particles on fecal samples from COVID-19 patients, suggesting a possible direct fecal–oral or fecal–respiratory transmission through aerosolized feces. This observation appears extremely important considering that among our patients with GI symptoms, 11.8% reported GI symptoms exclusively. In two other studies, involving 204 and 1141 patients, similar observations were made in 3% and 16% of cases, respectively, therefore indicating a direct attack of the virus on the mucosa of the GI tract rather than a mere translocation. Those data, although referring only to a small number of patients, if projected to all infected patients globally,10,30 become an important health problem in terms of viral diffusion: indeed, if at least some GI symptoms were to be considered as probable signs of SARS-CoV-2 infection in otherwise asymptomatic patients, a significant number of possibly infected subjects would automatically be considered symptomatic and therefore rapidly intercepted based on GI symptoms alone. Interestingly, in our population, patients with GI symptoms were significantly younger on average than in those without. Although data on younger adults and/or patients not requiring hospitalization are lacking to confirm this association, it has been observed on pediatric patients where studies show that GI involvement is very frequent.31,32
Analysis of laboratory data showed no particularly distinctive features in patients with and without GI symptoms: the only significant differences were increased liver enzymes (AST/ALT) and reduced (but within normal range) mean potassium levels. Although hypokalemia could be related to diarrhea or vomiting, our analysis did not show an association with GI symptoms; therefore, it is necessary to consider other etiologies such as some degree of renal impairment, dehydration, and/or alteration of the renin–angiotensin–aldosterone system.
Regarding liver damage, although there are consistent data showing increased transaminases in COVID-19 patients,33,34 the clinical significance of hypertransaminasemia in this setting is uncertain. Many authors suggest that altered liver function tests, although very common in patients already at admission, are still moderate overall and do not seem to represent a major factor in determining a worse outcome in COVID-19 disease. 35 However, this observation could possibly be linked to the high heterogeneity of recent publications, owing to a lack of standardized terms and definitions (population, liver function test cutoffs, definition of disease severity, control for other known severity factors or comorbidities, etc.). For example, in a recent study by Leal et al., during hospitalization, the authors observed elevated serum AST and ALT in 65.2% and 62.7% of patient, respectively, mostly slightly elevated. Further analysis showed that when considering transaminase values two times the upper limit of normal, percentages were halved. In accordance to our results, they also found more frequent transaminase abnormalities in males (AST, p = 0.004; ALT, p = 0.001). 36 A recent meta-analysis published by Sharma et al. 37 suggests that acute liver injury and elevated liver enzymes could be significantly associated with COVID-19 poorer outcomes (e.g. oxygen saturation <90%, invasive mechanical ventilation, ICU admission, severe disease, and in-hospital mortality). More studies are needed to better identify the role of concomitant liver disease and drug use.
In our study, patients with GI symptoms showed a greater probability of experiencing GI complications. Regarding outcomes, no differences were observed in terms of need for NIV, length of hospital stay, or mortality between groups, in line with other Italian and European studies.19,20,36,38 However, bearing in mind that only hospitalized patients were included in this study, our data showed significantly lower times from disease onset to hospital admission in patients with GI symptoms. Somewhat similarly, a study on 207 patients with confirmed COVID-19 infection reported an increased risk of hospitalization among subjects with digestive symptoms (adjusted OR 4.84, 95% CI: 1.68–13.94). In contrast to our findings, Pan et al. 10 showed that patients with digestive symptoms had a longer hospital length of stay (9 days versus 7.3 days, p = 0.013). Furthermore, the authors noted that, as the severity and duration of COVID-19 disease increases, GI symptoms increase as well, in line with current evidence that severe COVID-19 patients are more likely to show GI symptoms and liver function abnormalities, 15 possibly due to the inflammatory burden, pharmacological therapies or other comorbidities, and not vice versa. Finally, a metanalysis of 35 studies, on a total of 6686 patients with COVID-19, showed that while patients with GI involvement had an increased risk of ARDS (OR 2.96, 95% CI: 1.17-7.48) and liver injury (OR 2.71, 95% CI: 1.52-4.83), the pooled rates of discharge, length of hospital stay, and mortality did not show significant differences. 39
This study has three main limitations: first, its retrospective design, which reduces its level of evidence. Second, although among unfavorable outcomes transfer to ICU was considered, further follow-up data and final outcome of the transferred patients were unavailable; thus, data on overall mortality could not be shown. Third, consistent follow-up on discharged patients was unavailable in the immediate aftermath of the first wave of COVID-19. Thus, data collection on long-term GI complications or novel manifestations was not feasible. Some studies have reported that GI sequelae after COVID-19 infection (mostly diarrhea, loss of appetite, nausea, abdominal discomfort, and acid reflux) are observed with high variability, ranging from 16% to 44% at 3 months from discharge, particularly in patients initially presenting with severe disease.40,41 However, such estimations are hindered by heterogeneous definition of symptoms and manifestations, as well as variable methods and duration of follow-up. Given the known high prevalence of post-infectious generic GI symptoms, 42 prospective controlled protocols may be necessary to elucidate the severity and duration of such sequelae and their relevance to patients.
In conclusion, our study on an Italian population recruited during the outbreak of the COVID-19 pandemic shows that GI symptoms are part of the spectrum of the SARS-CoV-2 infection and could be the only manifestations at disease onset. This may be of significant importance as post-lockdown protocols are put in place and restrictions are relaxed based on the fluctuating epidemiological burden, especially if this were to be confirmed in the pediatric or unvaccinated population. Considering laboratory findings, only hypertransaminasemia was clearly associated with GI manifestations, in line with current knowledge regarding the GI tropism of this virus. Although patients showing GI involvement required faster hospital admission, clinical outcomes did not differ significantly between the two groups. In light of other reports, our observations could have been affected by the specific type of subjects recruited. Indeed, access to patient data for this study was limited to non-ICU patients. While need for ICU admittance was recorded as an outcome in our study, patients put on intensive care were not further followed. It is possible that GI involvement could differentially affect further outcomes during ICU stay. Given the current state of the pandemic, less-than-optimal vaccinations rates, the possible need of repeat vaccinations to maintain immunity, and the likelihood of COVID-19 becoming an endemic disease, further studies are needed regarding the impact of GI involvement in the SARS-CoV-2 infection.
Footnotes
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
