Abstract
Introduction:
Linaclotide, a guanylate cyclase C agonist that improves the symptoms of irritable bowel syndrome with predominant constipation (IBS-C), has been recently approved for IBS-C treatment. This study aimed to report real-world data on linaclotide treatment in China.
Methods:
This was a prospective multicentre study of the effectiveness of linaclotide treatment in patients with IBS-C from 10 primary medical institutions. Changes in defecation, abdominal symptoms, the IBS symptom severity scale (IBS-SSS), IBS quality of life questionnaire (IBS-QOL), Zung Self-Rating Anxiety Scale and Self-Rating Depression Scale in patients were evaluated to determine the drug’s clinical efficacy and safety.
Results:
We enrolled 97 patients (mean age: 52.39 ± 13.99 years), 55 of whom were women (56.7%). In terms of efficacy, the number of the patients’ defecation per week and Bristol stool form scale scores significantly increased at week 4 and week 12 compared with the values at the baseline. The baseline average IBS-SSS score was 211.01 ± 81.23. Of the patients, 24 had severe IBS-C, and their IBS-SSS scores at week 4 (51.81 ± 54.42) and week 12 (9.3 ± 30.39) significantly decreased and showed a pronounced improvement. The IBS-QOL total scores at week 4 and week 12 gradually decreased compared with that at the baseline and the QOL significantly improved. Treatment satisfaction rate was 79.3% in week 4 and 100% in week 12, showing a gradually increased satisfaction and significant differences. However, 11 cases (11.3%) had diarrhoea.
Conclusion:
Linaclotide has proved to be a safe and effective drug to improve IBS-C symptoms and severity.
Introduction
Irritable bowel syndrome (IBS) is a common gastrointestinal disorder that is mainly characterised by abdominal pain, abdominal distension and altered bowel habits. The Rome criteria classify IBS into the following four subtypes: IBS with predominant constipation (IBS-C), IBS with predominant diarrhoea (IBS-D), IBS with mixed bowel habits (IBS-M) and IBS unclassified (IBS-U).1,2 A literature review from Rome committees has revealed that the prevalence of IBS was 9.6% in Asia and 8.1% in North America, Europe, Australia and New Zealand. 3 The prevalence of IBS in China was 1–18% of Rome I/II and 5–16% of Rome III. 4
According to Rome IV, IBS-C is a subtype of IBS with > 1/4 of bowel movements with the Bristol stool form scale (BSFS) types 1 or 2 and < 1/4 of bowel movements with the Bristol stool types 6 or 7, mainly characterised by abdominal symptoms (e.g. abdominal pain and abdominal distension), reduced bowel movements, hard stools, laborious defecation and poor defecation. Approximately one-third of cases of IBS are IBS-C. 1 Reports have commonly recommended lifestyle changes or long-term medication to control symptoms in patients with IBS-C. 5 However, these treatments are mainly aimed at alleviating a single symptom, often with multiple switches of treatments and drugs without satisfactory efficacy. 6 A study of the burden of IBS disease conducted in China reported that the total annual expenditure per patient was estimated at RMB 18,262. 7 Thus, IBS imposes a huge financial burden on patients and healthcare systems. 8
Linaclotide is a 14-amino acid guanylate cyclase C agonist that is rarely absorbed into the blood. It was approved by the Food and Drug Administration (FDA) in 2012 for the treatment of adult chronic idiopathic constipation and IBS-C, and China also approved linaclotide for the treatment of adult IBS-C. Based on the results of multiple clinical trials in 2019, linaclotide treatment resulted in significantly improved abdominal pain and bowel movements compared with a placebo. 9 However, data from clinical trials may not accurately reflect the clinical practice setting.10,11 Real-world studies on linaclotide for IBS-C are few, and no real-world studies on linaclotide use in China or even in Asia have been conducted.12,13 Therefore, in this multicentre patient cohort, we aimed to determine the efficacy and safety of linaclotide in clinical practice by assessing changes in defecation, abdominal symptoms, the IBS symptom severity scale (IBS-SSS), the IBS quality of life questionnaire (IBS-QOL), the Zung Self-Rating Anxiety Scale (SAS) and the Self-Rating Depression Scale (SDS) in patients with IBS-C who underwent a 12-week treatment with linaclotide.
Methods
This study was a multicentre, prospective, single-arm, observational real-world study. Patients with IBS-C were recruited from 10 primary medical institutions in Shandong Province from 1 September 2020 to 31 March 2021.
The study was approved by the research ethics committee of the Second Hospital of Shandong University (LCLL-2021-004) and was registered at the Chinese Clinical Trial Registry (ChiCTR2100052551). Each patient signed an informed consent form, and all patient details were simultaneously de-identified. The inclusion criteria were as follows: voluntary participation in this study and ability to abide by the study regulations, understand and adhere to the follow-up plan, and correctly record medication, symptom assessment and adverse events; Chinese patients aged ⩾ 18 years; with IBS-C meeting the Rome IV classification; underwent colonoscopy in the previous 3 months and suspension of other medications for constipation for 1 week. The exclusion criteria were as follows: with contraindications to linaclotide, such as allergy to linaclotide or any excipients and known or suspected mechanical gastrointestinal obstruction; not expected to adhere to linaclotide alone; pregnant and lactating patients; participating in other clinical trials and those who were considered unsuitable for other reasons. This study was reported according to the Strengthening The Reporting of OBservational Studies in Epidemiology (STROBE) guidelines 14 (see checklist in Supplementary Material).
The study was conducted in the context of real clinical practice, and the enrolled patients took 290 µg linaclotide orally once a day, half an hour before breakfast and were followed-up for 12 weeks after the enrolment. Patient self-evaluation records were completed daily after group entry and were collected at each follow-up. The self-assessment records contained basic information on the patients, daily medication, defecation and abdominal symptoms (Table 1). Medications for diseases, except IBS-C, were permitted during the study and documented in Table 1; however, administration of enema or other laxatives was prohibited. The patients were followed-up once a week during 0–4 weeks and once at 8 and 12 weeks; both face-to-face and the information collection and symptom assessment scale were completed (Table 2). A final follow-up by telephone was conducted within 1 week from study withdrawal. A visit record was completed for each follow-up to assess changes in bowel movements, abdominal symptoms, treatment satisfaction, scores on the IBS-SSS, IBS-QOL, SAS and SDS 12 weeks after linaclotide treatment, and adverse events and withdrawal (Table 3).
Patient self-evaluation record form.

BSFS, Bristol stool form scale.
Scores of abdominal pain, abdominal distension, abdominal discomfort within 24 h. The range is 0–10 with higher scores indicating more severe symptoms.
Follow-up plan.
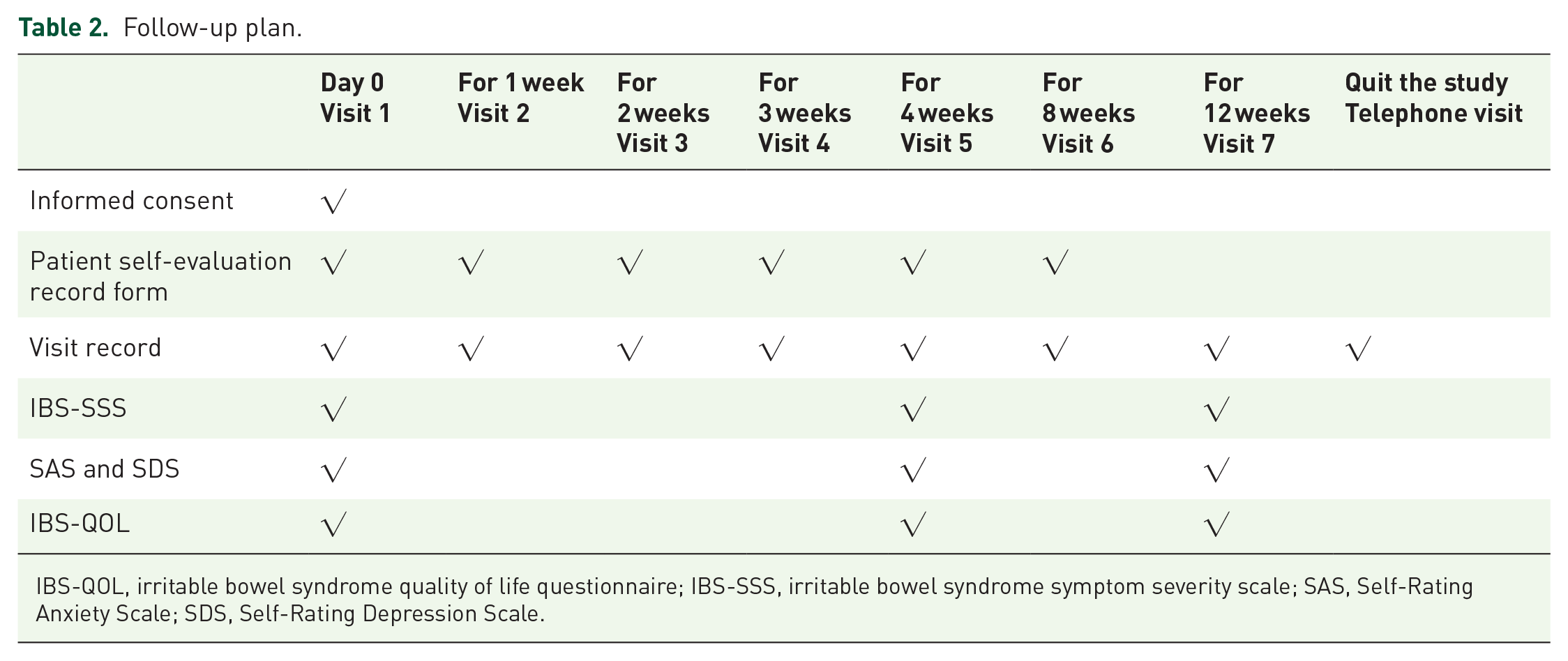
IBS-QOL, irritable bowel syndrome quality of life questionnaire; IBS-SSS, irritable bowel syndrome symptom severity scale; SAS, Self-Rating Anxiety Scale; SDS, Self-Rating Depression Scale.
Visit record.

BSFS, Bristol stool form scale; IBS-QOL, irritable bowel syndrome quality of life questionnaire; IBS-SSS, irritable bowel syndrome symptom severity scale; SAS, Self-Rating Anxiety Scale; SDS, Self-Rating Depression Scale.
This visit record was filled out by the assigned doctor for each visit.
Defecation frequency: number of defecations per week.
Scores of abdominal pain, abdominal distension and abdominal discomfort within 24 h. The range is 0–10 with higher scores indicating more severe symptoms.
The IBS-SSS has five domains with scores ranging from 0 to 100 and a total score of 0–500. High scores indicate more severe symptoms, and a reduction of 50 points is considered a marked improvement in symptoms. Disease severity was stratified using the IBS-SSS score as follows: normal: < 75, mild: 75 to < 175, moderate: 175 to < 300, and severe: ⩾ 300. The IBS-QOL is a disease-specific scale with an overall score of 34–170 (eight dimensions), with a high score indicating a lower QOL. The SAS and SDS comprehensively evaluate the status of anxiety and depression, respectively, based on the psycho-mental condition of the respondents for approximately a week. The standard cut-off scores for the SAS are < 50 for normal, 50–59 for mild anxiety, 60–69 for moderate anxiety and > 70 for severe anxiety. The standard cut-off scores for the SDS are < 53 for normal, 53–62 for mild depression, 63–72 for moderate depression and > 72 for severe depression.
Adverse events, tolerances and study withdrawal were recorded at each follow-up. The severity of adverse events, the causal relationship with linaclotide, and the tolerance and safety of the treatment were assessed according to the occurrence of adverse events.
Statistical analysis of data was performed using standard statistical methods and the SPSS 20.0 software (IBM Corporation, Armonk, NY, USA). Descriptive data are expressed as mean ± standard deviation (SD); a paired t-test was used to compare pre- and post-treatment follow-up results, and multiple group results comparisons were analysed using one-way analysis of variance (ANOVA) or Kruskal–Wallis methods. In addition, data on grade variables, such as patient satisfaction scores, were analysed using the Mann–Whitney U test. Significance was set at p < 0.05.
Results
Patient demographic and baseline characteristics
Overall, 120 patients with IBS-C, who all signed informed consent forms and met the Rome IV classification for IBS-C, were recruited from 10 centres. Some patients who did not provide complete visit records (n = 11) or were lost to follow-up (n = 12) were excluded from the study. The remaining 97 patients provided complete follow-up records, of whom 43 (44.3%) continued linaclotide administration at 12 weeks, 39 (40.2%) stopped linaclotide treatment before 4 weeks and 15 (15.5%) stopped linaclotide treatment at 4–12 weeks (Figure 1).

The participant selection process. Overall, 120 patients with IBS-C were recruited from 10 centres. Some patients who did not provide complete visit records (n = 11) or were lost to follow-up (n = 12) were excluded from the study. The remaining 97 patients provided complete follow-up records, of whom 43 (44.3%) continued linaclotide at 12 weeks, 39 (40.2%) stopped linaclotide treatment before 4 weeks and 15 (15.5%) stopped linaclotide treatment at 4–12 weeks.
Of the 54 patients (55.7%) without 12 weeks of linaclotide treatment, 30 quit the study due to significantly improved symptoms, 5 due to lack of efficacy, 11 due to excessive diarrhoea and 8 other patients withdrew for other reasons, such as economic issues and timing (Figure 1).
Finally, the 97 patients who met the linaclotide administration criteria were assessed for drug efficacy. The main baseline demographic and IBS-C related features were evaluated for patients who completed the study (Table 4). The mean age of the participants was 52.39 ± 13.99 years; 55 were female (56.7%), and 42 were male (43.3%). At the initial enrolment, the average number of defecations per week was 2.03 ± 1.25 and the BSFS was 2.18 ± 0.72. The average score of bloating was 3.18 ± 1.70; the average score of abdominal pain was 2.67 ± 1.91 and the baseline average IBS-SSS score was 211.01 ± 81.23; in this study, 24 patients (24.7%) had severe IBS-C. The number of participants who had anxiety was 12 (12.4%), with an average SAS score of 41.29 ± 11.46, while 15 patients had depression (15.5%) with an average SDS score of 42.37 ± 11.08. The IBS-QOL score was 57 ± 13.72.
Patient demographic and baseline characteristics.

BSFS, Bristol stool form scale; IBS-QOL, irritable bowel syndrome quality of life questionnaire; IBS-SSS, irritable bowel syndrome symptom severity scale; SAS, Self-Rating Anxiety Scale; SDS, Self-Rating Depression Scale.
Data are expressed as mean ± SD.
Effect of linaclotide treatment on bowel movements and abdominal symptoms
After linaclotide treatment, the number of defecations was 8.59 ± 3.61 at week 4 and 7.72 ± 2.03 at week 12 compared with the baseline (2.03 ± 1.25). The number of defecations significantly increased (p < 0.05) (Figure 2(a)). BSFS scores were 3.71 ± 0.84 at week 4 and 3.67 ± 0.68 at week 12, with a significant improvement in faecal traits (p < 0.05). Abdominal pain, abdominal distension symptoms and abdominal discomfort significantly improved at 4 and 12 weeks (p < 0.05) (Figure 2).

Effects of linaclotide treatment on bowel movements and abdominal symptoms. (a) After linaclotide treatment, the number of defecations significantly increased at weeks 4 and 12 (p < 0.05). (b) BSFS scores were significantly improved in faecal traits at weeks 4 and 12. (c)–(e) Abdominal pain, abdominal distension symptoms and abdominal discomfort significantly improved at 4 and 12 weeks (p < 0.05).
Effect of linaclotide treatment on IBS-SSS scores
The patients’ IBS-SSS score after linaclotide treatment was 51.81 ± 54.42 at week 4, which was significantly lower than that at the baseline (Figure 3(a)). The IBS-SSS score decreased to 166.72 ± 103.78 at week 4 in 58 patients, 38 of whom had mild and moderate IBS-C with an IBS-SSS score of 62.63 ± 57.21, which decreased from 105.1 ± 70.5. In 18 patients with severe IBS-C, the IBS-SSS score was 30.56 ± 45.01, which decreased from 287.5 ± 47.2. The improvement was more significant in patients with severe IBS-C (Figure 3(b) and (c)).

Effect of linaclotide treatment on the IBS-SSS scores. (a) The patients’ IBS-SSS scores after linaclotide treatment were significantly lower at weeks 4 and 12 than that at the baseline. (b) and (c) For patients with mild and moderate IBS-C and severe IBS-C, IBS-SSS scores decreased significantly on weeks 4 and 12, and the improvement was more significant in patients with severe IBS-C.
Similar changes were observed in the data analysis at week 12, with an average IBS-SSS score of 9.3 ± 30.39; the score was significantly reduced compared with that at the baseline (Figure 3(a)). The IBS-SSS score decreased from 225.1 ± 93.2 to 167.31 ± 72.72 at week 12 after treatment in 43 patients, 26 of whom had mild and moderate IBS-C with an IBS-SSS score of 14.42 ± 38.19. In 17 cases with severe IBS-C, the IBS-SSS score decreased from 313.24 ± 29.47 to 1.47 ± 6.06. The improvement was more significant in patients with severe IBS-C (Figure 3(b) and (c)).
At week 4, 58 patients’ data were analysed. The IBS-SSS score decreased by more than 50 points in 51 patients; IBS-SSS decreased by less than 50 points in 4 patients; the score remained the same in 3 patients; no increase in IBS-SSS score was observed. At week 12, the data of 43 patients were analysed. The IBS-SSS score decreased by more than 50 points in 42 patients, but the score was slightly (25 points) increased in 1 patient.
Effect of linaclotide treatment on IBS-QOL scores
The total IBS-QOL scores after linaclotide treatment were 45.6 ± 11.1 at week 4 and 39.77 ± 11.05 at week 12, showing a gradual decrease with significant improvements compared with the score at the baseline (Table 5).
Effects of linaclotide treatment on IBS-QOL scores.

Data are expressed as mean ± SD, the data were analysed using ANOVA and Significance was set at p < 0.05.
and # represent statistical differences between the tagged data.
After analysis, each dimension of the IBS-QOL score showed a gradual reduction and improvement (Table 5), and the dimension scale of emotional disorders, behavioural disorders, health concerns and social function showed a significant reduction (p < 0.05), whereas the dimensionality measures of somatic thoughts, dietary restriction, sexual behaviour and interpersonal relationship did not improve at week 4 but subsequently improved at week 12.
Effect of linaclotide treatment on SAS and SDS scores
The SAS scores after linaclotide treatment were 34.74 ± 9.54 at week 4 and 31.91 ± 14.49 at week 12, showing a gradual decrease and significant improvement of the patients’ anxiety levels (p < 0.05). Similarly, the SDS scores were 34.98 ± 10.39 at week 4 and 30.65 ± 13.74 at week 12, showing a gradual decrease and significant improvement in depressive symptoms (p < 0.05) (Figure 4).

Effects of linaclotide treatment on the SAS and the SDS scores. (a) The SAS scores after linaclotide treatment showed a gradual decrease and significant improvement of the patients’ anxiety levels at weeks 4 and 12 (p < 0.05). (b) The SDS scores also showed a gradual decrease and significant improvement in depressive symptoms at weeks 4 and 12 (p < 0.05).
Treatment satisfaction and adverse events
A satisfaction survey performed during linaclotide treatment revealed satisfaction rates of 29.79% at week 1, 48.19% at week 2, 79.31% at week 4 and 100% at week 12, which indicated progressive patient satisfaction and significant differences (p < 0.05). Partial or complete dissatisfaction with the treatment mainly occurred at weeks 1 and 2 (23 patients in week 1 and 15 patients in week 2) (Figure 5).
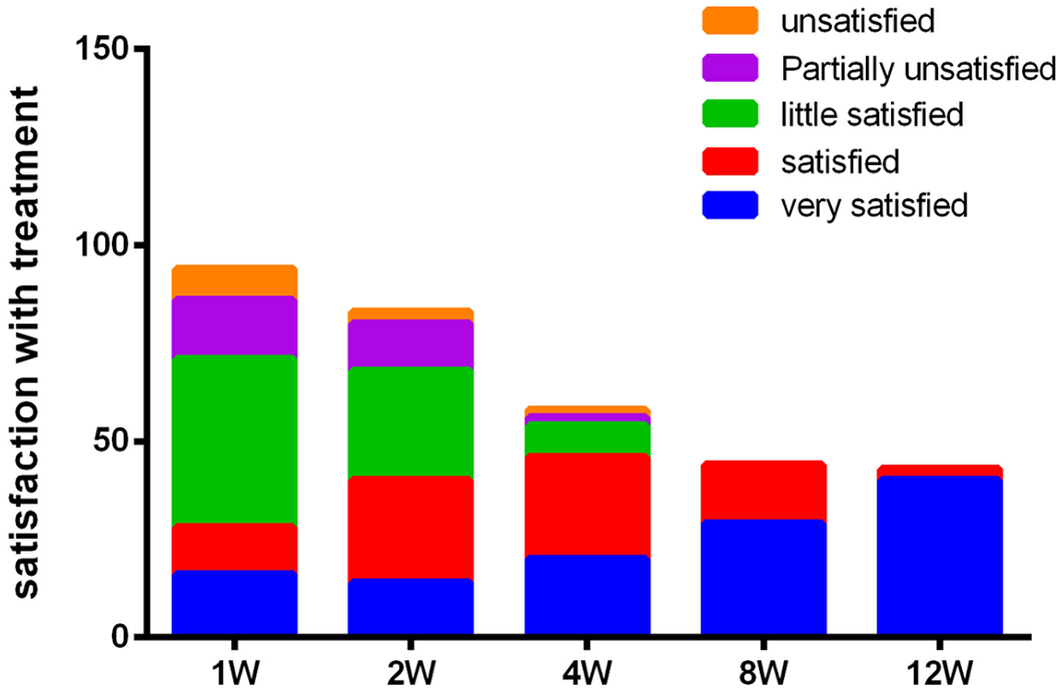
The satisfaction survey performed during the linaclotide treatment. The survey indicated progressive patient satisfaction and significant differences. Partial or complete dissatisfaction with the treatment mainly occurred at weeks 1 and 2.
In the 12-week study period, 11 patients (11.3%) reported experiencing diarrhoea. By week 4, nine patients developed diarrhoea, four of whom experienced diarrhoea in the first week, two in the second week and three in the third week. By week 12, the number of patients suffering from diarrhoea increased to 11. Five patients withdrew from the study because of nonsignificant efficacy and three patients because of the cost.
Follow-up results of 30 patients who stopped treatment early because of improved symptoms
In total, 30 patients withdrew from the study prior to its completion because of significant efficacy. Five patients took linaclotide again after a period of drug withdrawal. Among these 30 patients, the number of defecations at week 12 was 6.93 ± 2.41, which was significantly higher than that at baseline (2.23 ± 1.45) (p = 0.039). BSFS score at week 12 (3.53 ± 0.90) was significantly improved compared with that at baseline (2.30 ± 0.65) (p = 0.047). Similarly, the abdominal pain score at week 12 (0.60 ± 0.77) was significantly improved compared with that at baseline (2.17 ± 2.11) (p = 0.00). Notably, the treatment satisfaction rate was 100%.
Discussion
In this study, the symptom severity was significantly reduced based on the IBS-SSS score compared with that at the baseline, and linaclotide effectively and significantly improved abdominal distension and abdominal pain symptoms, with defecation frequency and BSFS at 4 and 12 weeks of treatment. This is consistent with the results of a linaclotide observational study in a clinical practice in Germany, which reported a significant reduction in abdominal pain and abdominal distension intensity and improvement in bowel frequency compared with those at the baseline. 15 The threshold for assessing the improvement in the total IBS-SSS score in this study was reduced by approximately 50 points, consistent with numerous studies using IBS-SSS for evaluation.16–18
Linaclotide treatment also significantly improved the QOL. A previous phase 3 clinical trial demonstrated that linaclotide treatment significantly improved QOLcompared with a placebo, with more than half of the patients demonstrating improved IBS-QOL scores after 12 weeks of linaclotide treatment. 19 This study’s results showed significant improvements in total IBS-QOL scores in all subscales, supporting the findings of previous studies. Individual analysis of the dimensions of the IBS-QOL score revealed that certain dimensions (mood disorders, behavioural disorders, health concerns and social function) showed a gradual reduction. Meanwhile, somatic thoughts, dietary restriction, sexual behaviour and relationships did not improve at week 4 but improved at week 12, indicating that their cognition required a long time to establish and change.
The most common adverse event associated with linaclotide treatment in this study was diarrhoea. The safety of linaclotide observed in this 12-week study was similar to that observed in phase 3 trials, and there was no indication of additional adverse events following long-term use. Diarrhoea was the most common adverse reaction associated with treatment,20,21 with 11.3% of patients reporting mild diarrhoea after linaclotide treatment and no patients developing severe diarrhoea. In clinical practice, diarrhoea is considered a marker of drug activity rather than an adverse event in many patients. 9
In this study, approximately 79.3% and 100% of patients were very satisfied or satisfied with the ability of linaclotide to relieve IBS-C symptoms at weeks 4 and 12, respectively, similar to a previous phase 3 IBS-C trial that showed that patients were relatively satisfied or very satisfied with the treatment effect at week 12. 22 The patients’ IBS-SSS scores after linaclotide treatment were 51.81 ± 54.42 at week 4 and 9.3 ± 30.39 at week 12. The satisfaction rate was basically consistent with the IBS-SSS score. There are two reasons for the gradual increase in the satisfaction rate to 100%: (1) the effect of linaclotide fully developed through the period of drug use and (2) patients with poor treatment response stopped taking the medication at an early stage.
At 12 weeks, linaclotide treatment was discontinued in 55.7% of the patients, while most patients stopped linaclotide in the first 4 weeks. The most common cause of withdrawal was symptom remission in approximately 30.9% of the patients, followed by adverse events in 11.3%. However, in two phase 3 clinical studies, linaclotide was stopped in 7.9% and 10.2% of the patients. This high withdrawal rate may be due to real-world studies reflecting the true wishes of patients, which may be higher than that in phase 3 clinical studies.20,21 Therefore, these results may represent a more actual result. Meanwhile, 30 patients stopped the treatment early because of improved symptoms. In the follow-up at week 12, the number of defecations, BSFS scores and abdominal pain scores were significantly improved. This indicates that linaclotide treatment perhaps elicits a long-lasting effect. Patients with IBS-C do not necessarily need to adhere to long-term medications; rather, the dosing frequency and interval of their medications can be adjusted according to their symptoms.
To reduce experimental bias, participants were required to stop taking laxatives for 1 week before enrolment and not take other laxatives during linaclotide treatment to accurately observe the therapeutic effect of linaclotide. Of course, we observed that some patients may have been lost to follow-up or dropped out of the study due to poor efficacy (five cases).
This study has some limitations. First, this study was observational in nature, with no control group but is reflective of the real-world data on linaclotide treatment. Second, the findings may have been influenced by confounding factors, such as diet and concomitant medications. Third, the sample size needs to be further increased.
Overall, the results of this multicentre, prospective, single-arm, observational study represent the first real-world study of linaclotide in China and demonstrate that linaclotide treatment effectively improves the severity of IBS-C and has good safety.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221092596 – Supplemental material for Linaclotide for treating patients with irritable bowel syndrome with predominant constipation: a multicentre study of real-world data in China
Supplemental material, sj-docx-1-tag-10.1177_17562848221092596 for Linaclotide for treating patients with irritable bowel syndrome with predominant constipation: a multicentre study of real-world data in China by Lan Liu, Weihao Zhang, Wei Zhao, Shuang Guo, Yaojun Wang, Xiaojun Lv, Bing Li, Haiping Wang, Enbin Xu, Quan Li, Qin Zhu, Xiao bo Gou, Weidong Zhao and Jianqiang Guo in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The author (Lan Liu) had received the Jinan Clinical Medical Science and Technology Innovation Program (202019101). The author thanks Dr Edward C Mignot, Shandong University, for linguistic advice.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Jinan Clinical Medical Science and Technology Innovation Programme (202019101).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data sets used and analysed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
