Abstract
Background:
We aimed to study the safety of cold snare polypectomy (CSP) for colorectal polyps in patients administered periprocedural antithrombotic agents.
Methods:
We searched the PubMed, Embase, and Cochrane Library databases through June 2021. The primary outcomes were the rates of delayed and immediate bleeding (requiring endoscopic hemostasis). Secondary outcomes included thromboembolic events. Meta-analysis using odds ratios (ORs) and corresponding 95% confidence intervals (CIs) was performed to compare the outcomes.
Results:
Seventeen studies, including five randomized trials, were included. Over 96% of polyps were ⩽1 cm. The pooled rates of delayed and immediate bleeding for patients receiving CSP and periprocedural antithrombotic agents were 1.6% and 10.5%, respectively. Both the delayed (OR = 4.02, 95% CI = 1.98–8.17) and immediate bleeding (OR = 5.85, 95% CI = 3.84–8.89) rates were significantly higher in patients using periprocedural antithrombotic agents than in non-users. Although both antiplatelet agents and anticoagulants increased the risk of delayed bleeding, the risks associated with the use of direct oral anticoagulants (DOACs; 2.5%) or multiple agents (3.9%) were particularly high. Compared to their counterparts, diminutive polyps and uncomplicated lesions not requiring hemoclipping were associated with lower risks of delayed bleeding (pooled estimates of 0.4% and 0.18%, respectively). Thromboembolic risk was similar among patients using and not using periprocedural antithrombotic agents.
Conclusions:
CSP with periprocedural antiplatelet agents and warfarin may be feasible, especially for diminutive polyps. However, drug discontinuation should be considered with the use of DOACs or multiple agents which entail higher bleeding risk even with hemoclipping.
Introduction
Because of its excellent efficacy and safety, cold snare polypectomy (CSP) is the standard treatment for colorectal polyps of ⩽10 mm under clinical guidelines.1,2 In contrast to hot snare polypectomy (HSP), CSP entails more superficial cutting and less damage to submucosal arteries than HSP does, 3 thus minimizing adverse events like perforation and delayed bleeding.
Although CSP’s safety is generally established, it has not been thoroughly demonstrated in patients on periprocedural antithrombotic agents, including antiplatelet and anticoagulant agents. Traditionally, polypectomy has been considered to have a high risk of bleeding (>1%); therefore, current guidelines recommend periprocedural discontinuation of antithrombotic agents except for aspirin.4,5 However, whether such recommendations apply in CSP for small colorectal polyp removal is unclear. Millions of patients worldwide are taking antithrombotic agents,6,7 and such patients exhibit an increased risk of colorectal neoplasms, 8 more than 80% of which are ⩽10 mm. 9 Therefore, determining the optimal approach in the present era of CSP prominence is invaluable.
Until recently, studies had been evaluating the safety of CSP for colorectal polyp removal in conjunction with periprocedural antithrombotic agents.10–20 However, these studies tested different antithrombotic agents and whether the use of periprocedural antithrombotic agents influences the risk of thromboembolism remains unclear. Therefore, we conducted this systematic review and meta-analysis to explore the bleeding and thromboembolism risks of CSP with and without antithrombotic agents.
Methods
Search strategy
We independently performed comprehensive literature searches of three databases, namely, the PubMed, Embase, and Cochrane Library online databases, for articles published before June 2021. All searches were performed using the same search strategy, which is summarized in Supplementary Appendix 1. After excluding duplicate records and irrelevant studies, two of us (J-HY and C-WL) independently performed manual reviews of the references from the selected articles to identify other potentially relevant studies and determine their eligibility. In case of discrepancies, consensus was sought through one-on-one discussion. When disagreement remained unresolved, J-YW made a final judgment.
Selection criteria and data extraction
Studies were included if they investigated the clinical outcomes of CSP for colorectal polyps among patients who used periprocedural antithrombotic agents as well as least one of the outcomes (immediate or delayed bleeding rate). In this study, antithrombotic agents referred to antiplatelet agents (typically aspirin or thienopyridines including clopidogrel, prasugrel and ticagrelor, and cilostazol) and anticoagulants (typically warfarin and direct oral anticoagulants (DOACs)).
Studies were excluded if (1) patients underwent only HSP, (2) outcome data were incomplete for patients who underwent CSP and received antithrombotic agents, (3) antithrombotic agents were discontinued for all patients in the intervention group, or (4) a patient group overlapped with one of another included study. The following data were independently extracted from eligible studies: the name of the first author, year of publication, country of origin, number and characteristics of patients, study design and treatment modality, type of antithrombotic agents used, and primary and secondary outcomes. All data were extracted as originally stated or after appropriate calculations. If the necessary data were unavailable, attempts were made to contact the corresponding author for additional information.
Outcome assessment
The primary outcomes in this study were the delayed and immediate bleeding rates associated with CSP with periprocedural antithrombotic agents. Delayed bleeding referred to severe hematochezia that developed within 30 days after polypectomy and required endoscopic evaluation and treatment. Immediate bleeding was defined, in accordance with relevant studies, as active spurting or persistent oozing (for 30–300 seconds) that required endoscopic hemostasis. We performed patient-wise and polyp-wise meta-analysis for both outcomes and compared patients or polyp cases treated with antithrombotic agents with those not treated with antithrombotic agents or for which such treatment was discontinued. The secondary outcomes were periprocedural thromboembolic events and minor hematochezia occurring within 30 days of the procedure’s completion. Thromboembolic events included acute cerebrovascular or cardiovascular events. Minor hematochezia referred to uncomplicated, self-limited rectal bleeding within 30 days after polypectomy.
Statistical analysis
All meta-analyses were performed using Comprehensive Meta-Analysis Version 3.3.070 (Biostat, Englewood, NJ, USA) with random-effects models. Odds ratios (ORs) were used for the analysis of discrete variables. Corresponding 95% confidence intervals (CIs) were used to compare the outcomes of the intervention and control groups. In this study, we used the Mantel–Haenszel method to calculate ORs because zero primary outcome events were often observed; continuity correction was used in the case of double-zero events to facilitate the meta-analysis. 21 The pooled effect size was considered statistically significant if the ranges of 95% CIs of the ORs excluded 1. The I2 statistic was used to evaluate statistical heterogeneity, which was considered statistically significant if I2 was >50% or if a chi-square test result had a P value of <0.1.
Sensitivity analysis and risk-of-bias assessment
For all meta-analyses, we evaluated the robustness of pooled effect estimates by excluding one study at a time. In addition, we used variables including study design (prospective or retrospective), status of antithrombotic agent use in the control group (interruption or non-use), concomitant use of cold forceps polypectomy, and inclusion of polyps >10 mm for sensitivity analysis of delayed bleeding rate. We also sought clinical predictors of the bleeding outcomes after CSP by meta-analyzing the available data.
Risk of bias was assessed using the Cochrane risk-of-bias assessment tool for randomized control trials and Newcastle–Ottawa scale for retrospective studies. Publication bias was evaluated using a funnel plot along with Egger’s test, for which P < 0.1 was considered a positive result. The delayed bleeding rate in patients receiving and not receiving periprocedural antithrombotic treatment was selected for publication bias testing.
Results
Search results and baseline characteristics of included studies
The review process is illustrated using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart (Supplementary Figure 1) and summarized in the PRISMA checklist (Supplementary Appendix 2). The search and review process identified 17 eligible studies from 763 records for the final analysis.3,7,10–20,22–25 The baseline characteristics of the included studies are summarized in Table 1 and Supplementary Table 1. Eight prospective studies (including five randomized controlled trials) and nine retrospective studies were analyzed. Most studies were conducted in East Asia,3,7,11–17,19,20,23–25 and the remaining studies were conducted in Italy, 10 the United States, 22 and Australia/New Zealand. 18
Baseline characteristics of included studies.

SD, standard deviation.
Including patients with hot snare polypectomy.
Including lesions undergoing mucosal biopsy. Subsequent meta-analyses were performed on patients or lesions that underwent cold snare polypectomy.
Patient and lesion characteristics and antithrombotic agents
This systematic review included 12,894 patients (70.4% male) with 26,197 (53.4% right- and 46.6% left-sided) colorectal lesions. The median age ranged from 65 to 73 years. The polyps were mostly flat (36.6%) or sessile (59.3%), and more than 96% of them were ⩽1 cm in size. Larger polyps, which accounted for up to 18% of polyps, were generally subjected to HSP and thus were excluded from outcome analysis.7,22,25 In addition, although a few studies used cold forceps polypectomy concomitantly,7,10,11,22 at least 91.7% of the lesions were removed through CSP alone.
In studies included in our meta-analysis, 76.1% of patients who underwent CSP used antiplatelet agents, 44.7% used anticoagulants, and 13.4% used multiple agents, namely both antiplatelet agents and anticoagulants (Supplementary Table 2).3,7,10,12–19,22–25 Aspirin was the most used antiplatelet agent (53.1%), followed by clopidogrel. Among patients taking oral anticoagulants, the proportions on warfarin (52.8%) and DOACs (47.2%) were similar. Heparin bridge therapy was employed for 6–37% of patients in some studies.11,13,19,25 The most common indications for antithrombotic agents were coronary artery disease, atrial fibrillation, and cerebrovascular disease. Although all studies evaluated the outcomes of patients undergoing CSP with continued antithrombotic agent use, the control groups ceased antithrombotic agent use in several studies7,11,16,18,19 and some allowed continuation of aspirin use in the control group.7,16,18,22 Moreover, three real-world studies11,13,19 reported antithrombotic agent discontinuation in subgroups of patients (44–69%) within the periprocedural antithrombotic agent group.
Primary outcomes
Delayed bleeding and immediate bleeding rates
The bleeding rates and thromboembolic outcomes reported by the included studies are summarized in Table 2. For CSP with continued antithrombotic agent use, the pooled delayed bleeding rate across all studies was 1.6% (95% CI = 0.9–2.6%; Figure 1(a)), indicating a significantly higher risk than non-use or interrupted use of antithrombotic agents in both the patient-wise (OR = 4.02, 95% CI = 1.98–8.17, P < 0.001; Figure 1(b)) and polyp-wise (OR = 4.17, 95% CI = 1.78–9.79, P = 0.001, Figure 1(c)) analyses. The pooled estimate of the immediate bleeding rate was 10.5% (95% CI = 6.0–17.9%; Figure 2(a)). A significantly greater risk was also associated with ongoing antithrombotic treatment in the patient-wise (OR = 5.85, 95% CI = 3.84–8.89, P < 0.001; Figure 2(b)) and polyp-wise (OR = 2.41, 95% CI = 1.92–3.01, P = 0.001; Figure 2(c)) analyses.
Bleeding and thromboembolic events in included Studies.

DeB, delayed bleeding; N/A, not available; TEE, thromboembolic events
Including patients and polyps eligible for outcome assessment.
Rate per polyp.

(a) Pooled delayed bleeding rate after CSP in patients receiving periprocedural antithrombotic therapy. Heterogeneity: I2 = 35.5%, τ2 = 0.293, P = 0.091. (b) Delayed bleeding rate after CSP in patients receiving periprocedural antithrombotic therapy (ATT) compared with those discontinuing or not receiving antithrombotic therapy (NAT). Heterogeneity: I2 = 0, τ2 = 0, P = 0.908. (c) Delayed bleeding rate following CSP among polyps in patients receiving periprocedural antithrombotic therapy (ATT) and those discontinuing or not receiving antithrombotic therapy (NAT). Heterogeneity: I2 = 0, τ2 = 0, P = 0.993.

(a) Pooled bleeding rate after CSP in patients receiving periprocedural antithrombotic therapy. Heterogeneity: I2 = 68.2%, τ2 = 0.302, P = 0.013. (b) Immediate bleeding rate after CSP in patients receiving periprocedural antithrombotic therapy (ATT) and those discontinuing or not receiving antithrombotic therapy (NAT). Heterogeneity: I2 = 0, τ2 = 0, P = 0.785. (c) Immediate bleeding rate after CSP among polyps in patients receiving periprocedural antithrombotic therapy (ATT) and those discontinuing or not receiving antithrombotic therapy (NAT). Heterogeneity: I2 = 37.1%, τ2 = 0.04, P = 0.145.
Clinical predictors of bleeding risk with various antithrombotic agents
Several factors, including age,10,19 use of hemoclipping,13,19,22 and characteristics of polyps such as size,10,13,14,16,17,19,24 polypoid morphology,14,17,24 and rectal location,14,17 were associated with immediate or delayed bleeding rates after CSP in the included studies. However, meta-analyses could be performed for only prophylactic clipping and polyp size. Hemoclipping was associated with an increased odds of delayed bleeding (OR = 9.25, 95% CI = 2.26–37.78, P = 0.002). Furthermore, compared with diminutive polyps (⩽5 mm), polyps sized 6–10 mm were associated with significantly higher immediate (OR = 2.91, 95% CI = 1.92–4.40, P < 0.001; Table 3) and delayed bleeding rates (OR = 5.77, 95% CI = 1.96–16.98, P = 0.001). In addition, among three studies that compared CSP and HSP with periprocedural antithrombotic agents, HSP demonstrated a significantly higher risk of delayed bleeding (OR = 2.80, 95% CI = 1.41–5.55, P = 0.003; Supplementary Figure 2).
Meta-analyses of bleeding risks after cold snare polypectomy with regard to size, prophylactic clipping, and type of antithrombotic agent.
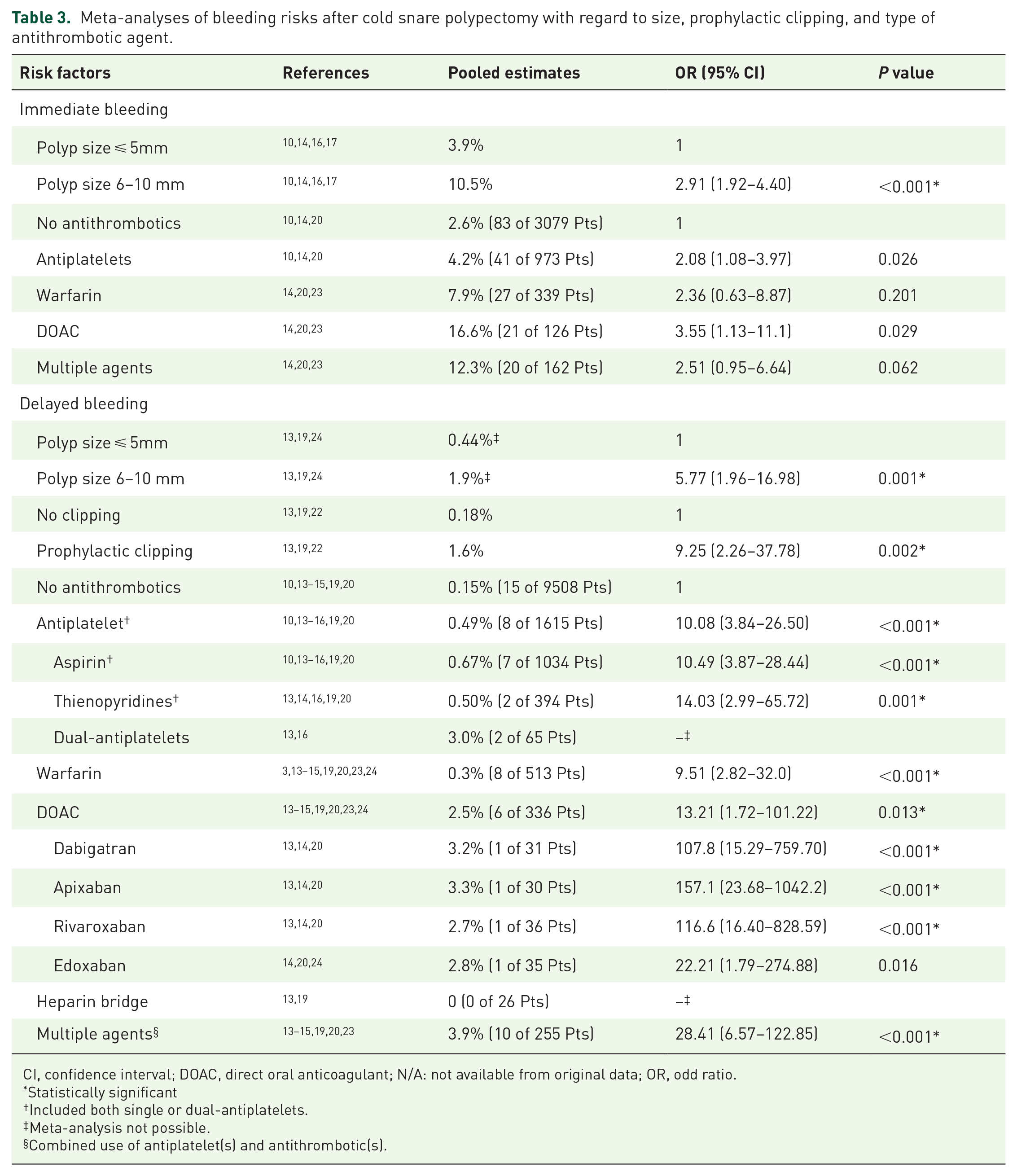
CI, confidence interval; DOAC, direct oral anticoagulant; N/A: not available from original data; OR, odd ratio.
Statistically significant
Included both single or dual-antiplatelets.
Meta-analysis not possible.
Combined use of antiplatelet(s) and antithrombotic(s).
We explored the effects of individual antithrombotic agents on CSP-related bleeding. Four articles reported immediate bleeding rates for CSP with specific types of antithrombotic agents.10,14,20,23 Antiplatelet agent use (OR = 2.08, 95% CI = 1.08–3.97, P = 0.026) and DOAC use (OR = 3.55, 95% CI = 1.13–11.1, P = 0.029) were associated with significantly greater risks of immediate bleeding than was non-use of antithrombotic agents. Moreover, five studies investigated delayed bleeding outcomes in patients who underwent CSP without antithrombotic agents,13–15,19,20 finding that patients who used antiplatelet agents (0.49%, OR = 10.08, 95% CI = 3.84–26.50, P < 0.001), warfarin (0.3%, OR = 9.51, 95% CI = 2.82–32.0, P < 0.001), DOACs (2.5%, OR = 13.21, 95% CI = 1.72–101.22, P = 0.001), or multiple agents (3.9%, OR = 28.41, 95% CI = 6.57–122.85, P < 0.001) had a significantly higher bleeding rate then patients who did not use any of them. Notably, CSP involving aspirin alone was associated more with delayed bleeding than was antithrombotic agent non-use, with the pooled bleeding rate being 0.67% (OR = 10.49, 95% CI = 3.87–28.44, P < 0.001). All types of DOAC (dabigatran, apixaban, and rivaroxaban) exhibited similar pooled bleeding rates, whereas the data on heparin bridge therapy were insufficient for meta-analysis.
Secondary outcomes
30-day risk of thromboembolism and minor hematochezia
Six of the included studies reported data on thromboembolic events within 30 days of the procedure. No significant difference in event rates between patients using and not using periprocedural antithrombotic agents was observed (OR = 1.24, 95% CI = 0.42–3.68, P = 0.702; Figure 3). The thromboembolic events were acute coronary syndrome in six patients and stroke in four patients. Notably, one patient developed ischemic stroke on postpolypectomy day 6 despite having undergone heparin bridge therapy. 25 Furthermore, minor hematochezia was reported in only two studies with conflicting results.16,18
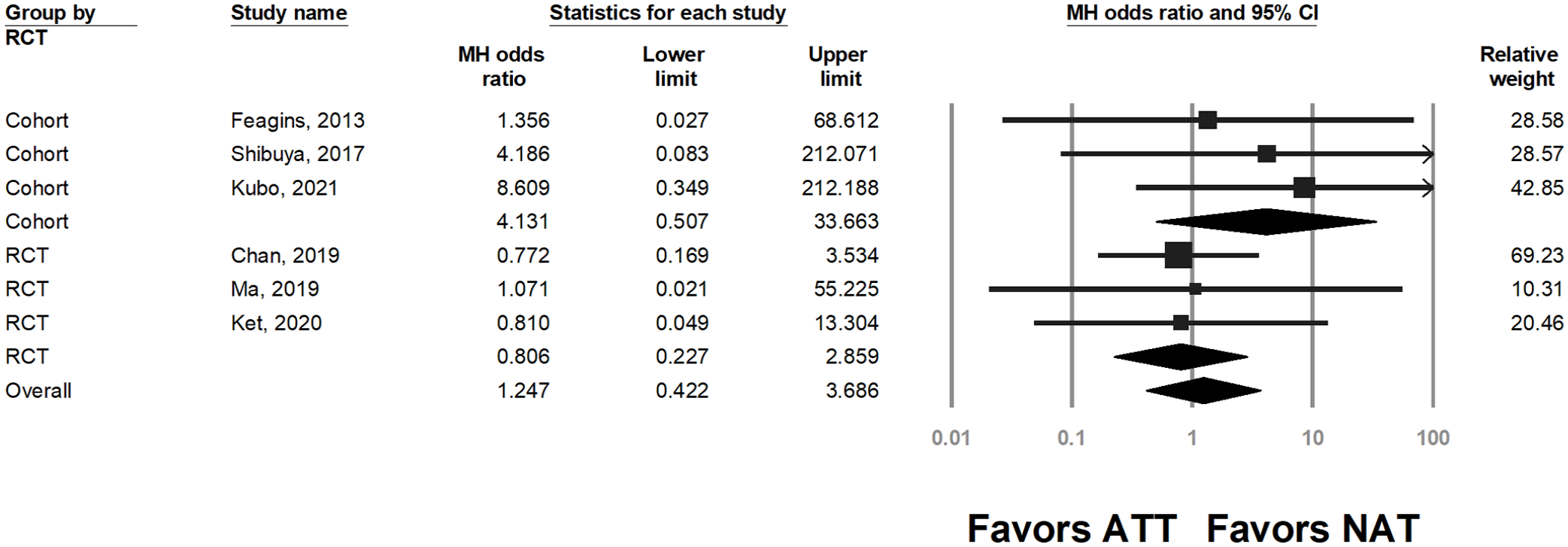
Thromboembolic event rate in patients continuing periprocedural antithrombotic therapy (ATT) and those discontinuing or not receiving antithrombotic therapy (NAT). Heterogeneity: I2 = 0, τ2 = 0, P = 0.831.
Sensitivity analysis and risk-of-bias assessment
In this review, the meta-analyses in the rates of delayed and immediate bleeding and thromboembolic events between patients who used periprocedural antithrombotic agents and those who did not were statistically robust. Similarly, the subgroup analysis of bleeding risk with respective antithrombotic agents and prophylactic clipping was also robust. Further sensitivity analysis of delayed bleeding rate by predefined subgroups is shown in Supplementary Table 3. Notably, studies that were conference abstracts or studies comparing continued and ceased antithrombotic agent use failed to demonstrate difference of delayed bleeding rates between the two groups. The rates of delayed bleeding were also similar between patients in prospective and retrospective studies (1.3% vs. 2.2%, P = 0.297).
Finally, the risk-of-bias assessment did not reveal any obvious risk of bias among the studies (Supplementary Table 4 and Supplementary Table 5). Publication bias was deemed to be absent, whether based on a funnel plot (Supplementary Figure 3) and Egger’s test (P = 0.449).
Discussion
In this systematic review and meta-analysis, we explored the pooled rates of delayed bleeding, immediate bleeding, and thromboembolic events in patients receiving CSP and periprocedural antithrombotic agents and compared them with those of patients who had discontinued use of or not used these drugs for CSP. The risk of delayed bleeding and immediate bleeding among patients receiving periprocedural antithrombotic treatment is higher than those who have no periprocedural antithrombotic agents. However, the risk of delayed bleeding varies with different types of drugs. Moreover, polyp size and the initial requirement of hemoclipping are key risk factors for delayed bleeding.
An association between delayed bleeding and CSP has been deemed rarely among genera population in the literature.26–28 Although antithrombotic agents increase the risk of bleeding, the risk of bleeding remains lower than that after traditional HSP.3,7,22,24,25 Given the low pooled rate (<1%) of delayed bleeding after CSP and treatment with antiplatelet agents or warfarin, these might be considered low-risk treatments. 4 However, the immediate bleeding rate was more variable, ranging from 0% to nearly 20%.26,29,30 In this study, we calculated an overall pooled estimate at 10.5% among patients taking antithrombotic agents, which did not exceed the rate after HSP in previous studies.16,24 Notably, compared with the rate of delayed bleeding, the immediate bleeding rate might be more susceptible to bias when b endoscopists are not blinded. For instance, two randomized trials with blinded endoscopists16,18 in this review did not demonstrate significant differences between the immediate bleeding rates of patients receiving and not receiving antithrombotic agents. Therefore, although immediate bleeding is more likely with antithrombotic agents, the exact magnitude of this difference may be lower than the estimation in this study.
Another noteworthy question regards the influence of hemoclipping on delayed bleeding involved in CSP with antithrombotic treatment. Among the included studies, endoscopists employed hemoclipping when requiring hemostasis or when lesions presenting a high risk of delayed bleeding were present. However, such practice does not prevent delayed bleeding based on our findings. It is not clear whether the observation truly reflects the ineffectiveness of hemoclipping after CSP with antithrombotics, or there is confounding due to intrinsic high risk of bleeding among the lesions with hemoclipping. Since hemoclipping remains the most practical strategy for bleeding prevention, and we would not suggest limiting its use on the basis of the present finding alone. Nevertheless, further randomized controlled trials or large observation cohorts may further elucidate this issue.
In this systematic review, we investigated the relative bleeding risk for individual antithrombotic agents, which may be statistically infeasible with a single cohort. Remarkably, the use of aspirin was associated with a significantly higher risk of delayed bleeding than non-use, albeit at a modest magnitude. Cessation of aspirin use before HSP has been considered unnecessary because an increased risk of bleeding has not been observed.4,31 These findings may be explained by several means. First, we did not exclude patients using dual antiplatelet agents from the subgroup analysis of aspirin. Although the patients with dual antiplatelets were too few (65 cases with two delayed bleeding)13,16 to perform meta-analysis, this might partly influence the result. In addition, the meta-analysis for specific agents included all retrospective studies, which encompassed far more control group patients than intervention group patients; thus, the effect estimates tended to be overestimated because of the discrepancy in case numbers between the two groups. In addition, although did not compare warfarin use directly with that of DOACs, the pooled risk estimate of delayed bleeding for warfarin use was much lower than expected (0.3%). This result differs from two recent large cohort studies suggesting equivalent or higher risk of bleeding associated with warfarin use after polypectomy procedures.32,33 HSP, however, was the main treatment modality investigated in those two studies. Two possible reasons explain why warfarin was associated with a lower risk than was expected. First, warfarin has a more variable blood concentration than DOACs. In fact, the included retrospective cohort studies did not report periprocedural international normalized ratios, and the other two randomized trials did not report any delayed bleeding with warfarin use, despite reporting a mean international normalized ratio of 2:2.5.3,24 Second, the fundamental mechanism of bleeding after CSP may be different from that after HSP. 3 Therefore, we suggest that further large trials evaluate the risk of bleeding after CSP with periprocedural antithrombotic agents, especially aspirin and warfarin.
The decision to continue or cease antithrombotic agent use relies not only on the risk of bleeding but also on that of thromboembolism. Medication interruption may be strongly considered for patients taking DOAC and multiple agents, given the obviously higher risk of periprocedural bleeding from CSP compared to thromboembolic events. Notably, patients using multiple agents, despite representing a minority, can be particularly challenging to treat because their risks of both bleeding and thromboembolic events are high. Hence, a case-by-case basis and consultation with cardiovascular specialist is appropriate. Although this review could not draw conclusions regarding heparin bridge therapy which has been suggested in patients with high risk of periprocedural thromboembolism, previous studies have suggested that continuing antithrombotic agents (rather than using heparin bridge therapy) with colonoscopy and CSP on subcentimeter polyps may be equally safe 24 or even safer11,25,34 than using heparin bridge therapy in terms of delayed bleeding among patients with a high risk of thromboembolism. In addition, patients having small polyps and taking antiplatelet agents or warfarin may not require drug interruption for CSP given the slightly the risk increase.
The strength of this meta-analysis is its review of numerous recent studies and its meta-analysis of the risks of bleeding and thromboembolic events in patients who underwent CSP with and without antithrombotic agents, as well as its comparison of the effects of individual types of antithrombotic agent. Our findings may provide insight for clinical practice and the management of periprocedural antithrombotic treatment for colonoscopy and polypectomy. However, this study has several limitations. First, the meta-analysis included both randomized trials and retrospective studies, and the control group contained both patients who did not use or who discontinue antithrombotic agents. Nevertheless, the sensitivity analysis may have assisted in reducing bias. Second, we were unable to meta-analyze the risk of bleeding with heparin bridge therapy, but several studies have suggested a high bleeding rate associated with this approach.11,25,34 As previously mentioned, in patients for whom continual anticoagulant use is deemed necessary, proceeding with CSP with DOAC treatment may be an acceptable alternative. Third, we could not analyze patients with clopidogrel or dual-antiplatelets use which may be of high clinical interest because of a paucity of data. Finally, the included studies had heterogenous patient populations and diverse distributions of antithrombotic agents, and most of the studies were conducted in Asian countries. However, the statistical heterogeneity was generally low, and we performed random-effects model for meta-analyses in order to obtain more robust results. A prospective, multicenter database would be most valuable for answering the remaining questions.
In conclusion, this systematic review demonstrated that the use of antithrombotic agents, such as that of DOACs or multiple agents, can substantially increase the risk of bleeding, and immediate bleeding requiring hemostasis may be predictive of delayed bleeding. However, antiplatelet agents and warfarin present only a modestly bleeding risk, and their uninterrupted usage, particularly for diminutive polyps, may be acceptable. Further prospective trials are required to validate these findings.
Supplemental Material
sj-doc-1-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-doc-1-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-docx-4-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-5-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-docx-5-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-6-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-docx-6-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-rtf-7-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-rtf-7-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-10-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-tif-10-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-11-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-tif-11-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-8-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-tif-8-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-9-tag-10.1177_17562848211070717 – Supplemental material for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis
Supplemental material, sj-tif-9-tag-10.1177_17562848211070717 for Safety of cold snare polypectomy with periprocedural antithrombotic agents for colorectal polyps: a systematic review and meta-analysis by Jen-Hao Yeh, Wen-Lun Wang, Chih-Wen Lin, Ching-Tai Lee, Cheng-Hao Tseng, Po-Jen Hsiao, Yu-Peng Liu and Jaw-Yuan Wang in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
J-HY performed the database search, article review, statistical analysis, and manuscript writing. W-LW was responsible for the statistics and assessed the risks of bias, as well as the manuscript writing. C-WL also performed the database search and study design. C-TL was responsible for article review and assessment of bias risks. P-JH helped data retrieval, reviewed the analyzed data, and provided suggestions for manuscript writing. C-HT assisted with the refinement of methods and discussion. Y-PL was responsible to review the manuscript and help revision. J-YW designed the study and revised the manuscript critically.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the EDAHP110009 project of E-Da Hospital granted to Dr. Jen-Hao Yeh. This work was also supported by grants through funding from the Ministry of Science and Technology (MOST 109-2314-B-037-035, MOST 109-2314-B-037-040, MOST 109-2314-B-037-046-MY3, MOST110-2314-B-037-097) and the Ministry of Health and Welfare (MOHW109-TDU-B-212-134026, MOHW109-TDU-B-212-114006, MOHW110-TDU-B-212-1140026) and funded by the health and welfare surcharge of on tobacco products, and the Kaohsiung Medical University Hospital (KMUH110-0R37, KMUH110-0R38, KMUH110-0M34, KMUH110-0M35, KMUH110-0M36, KMUHSA11013, KMUH-DK(C)110010, KMUH-DK(B)110004-3) and KMU Center for Cancer Research (KMU-TC109A04-1) and KMU Center for Liquid Biopsy and Cohort Research Center Grant (KMU-TC109B05) and KMU Office for Industry-Academic Collaboration (S109036),Kaohsiung Medical University. In addition, this study was supported by the Grant of Taiwan Precision Medicine Initiative, Academia Sinica, Taiwan, R.O.C.
Writing assistance
This article has English editing by Wallace Academic Editing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
