Abstract
The potential of endoscopic evaluation in the management of inflammatory bowel diseases (IBD) has undoubtedly grown over the last few years. When dealing with IBD patients, histological remission (HR) is now considered a desirable target along with symptomatic and endoscopic remission, due to its association with better long-term outcomes. Consequently, the ability of endoscopic techniques to reflect microscopic findings
Keywords
Introduction
Endoscopy plays a crucial role in the management of patients with inflammatory bowel diseases (IBD), as it represents the mainstay of both initial diagnostic assessment and evaluation of disease activity.1,2 To date, endoscopic remission is the main treatment target as mucosal healing (MH) is associated with better long-term efficacy outcomes.3–5 However, there is growing evidence that, in addition to MH, the objective of attaining histological remission (HR) might result in further improved outcomes.6,7 In fact, histological disease activity can be detected in up to 40% of patients with endoscopic remission and is associated with a greater risk of clinical relapse. 8
It is currently recommended that endoscopic procedures should be performed with high definition white-light endoscopy (HD-WLE) in order to ensure optimal evaluation of the mucosa. 2 Importantly, residual inflammatory activity persists even if HD-WLE identifies a normal-appearing mucosa. 9 Besides the certain discordance between HD-WLE and histopathology, there is a multitude of histological scoring systems, which makes disease activity estimation more complex. 10 Of note, in recent years, several methods have been introduced to ameliorate the evaluation of the mucosa by endoscopists. New diagnostic devices, such as confocal laser endomicroscopy (CLE), have been developed to better predict histopathology, but their real incorporation in daily practice is not yet well defined. 11 The use of artificial intelligence (AI) systems to standardize operator-subjectivity in IBD and molecular imaging have also come to light as captivating areas of reasearch. 12 In addition, endoscopic surveillance is fundamental in the prevention of colorectal cancer in IBD and the use of modalities such as chromoendoscopy, which involves the spraying of topical dyes to enhance visualization of the mucosa has become the gold standard for dysplasia detection. 13 The Surveillance for Colorectal Endoscopic Neoplasia Detection and Management in Inflammatory Bowel Disease Patients (SCENIC) recommendations highlighted the superiority of HD-WLE over standard definition (SD)-WLE and, most importantly, put emphasis on the preferential use of dye-chromoendoscopy (DCE) over WLE, in guiding targeted biopsies. 14 Despite the authoritative position of the SCENIC consensus, debate is still ongoing, as further research has produced contradictory results, with a recent meta-analysis failing to show a significant superiority of DCE over HD-WLE in dysplasia identification [relative risk (RR) = 1.36; 95% confidence interval (CI): 0.84–2.18]. 15 Moreover, long examination time, need for adequate training, and additional costs for dye spraying might limit the wider application of this technique.16,17 In this context, dye-less chromoendoscopy or virtual chromoendoscopy (VCE) is emerging for its advantageous role in IBD surveillance. 18 In this review, we aim to summarize the current evidence on the most recent endoscopic techniques and devices in two cornerstones of IBD management: evaluation of disease activity and colorectal cancer surveillance. The review also focused on the strengths and limitations of the new advances to provide physicians with an updated insight on what is feasible and effective in a real-life setting.
Methods
We searched the PubMed, Ovid MEDLINE and EMBASE databases using the following keywords ‘endoscopy’, ‘dye-chromoendoscopy’, ‘virtual chromoendoscopy’, ‘endocytoscopy’, ‘confocal laser endomicroscopy’, ‘capsule enteroscopy’, ‘artificial intelligence’, ‘molecular imaging’ individually or in combination with ‘IBD’, ‘Inflammatory Bowel Disease(s)’, ‘Crohn’s disease’, ‘CD’, ‘Ulcerative Colitis’, or ‘UC’. The search focused on full text papers published in English up to December 2020. Abstracts were selected when relevant. No publication date restrictions were imposed. Finally, articles were included in this review if they were relevant, while additional publications were identified through their reference lists. Ethics approval and informed consent were not required for this review.
Endoscopic technologies for the assessment of mucosal inflammation
Endoscopy plays a key role in both Crohn’s disease (CD) and ulcerative colitis (UC). However, in patients with CD inflammation is extended to deeper layers and additional cross-sectional imaging techniques are required to assess transmural healing. 19 Rapid advancements in endoscopic technologies including DCE, VCE, endocytoscopy (EC), and CLE have enabled a more precise assessment of mucosal inflammation (Figure 1). 18

Rectum flat elevated lesion (IIa + IIb according to the Paris classification), pit Patter IIIL–IV according to the Kudo classification. (a) WLE. (b) LCI. (c) BLI. Histologic examination: high grade dysplasia.
Dye-chromoendoscopy
DCE was introduced more than a decade ago. In this procedure, dye agents (e.g., methylene blue and indigo carmine) are applied to improve the detection of the mucosal surface and any potential abnormalities.
20
The interest in DCE derives from the clear benefit in the adenoma detection rate (ADR) in the context of IBD surveillance (Figure 2).
21
Several studies have demonstrated the role of this technology in accurately determining mucosal inflammation.22–24 A paradigmatic trial randomized 263 clinically inactive, UC patients at a 1:1 ratio to undergo conventional colonoscopy or colonoscopy with DCE.
24
A significantly improved correlation between endoscopic assessment of disease extent and histologic data was observed in the group of patients undergoing DCE (89%

Descending colon flat lesion (IIb according to the Paris classification), pit Patter IIIL according to the Kudo classification at the same specific points. (a) WLE. (b) DCE. (c) Detail with DCE. Histologic examination: biopsies on the white area compatible with chronic inflammation; biopsies on the lesion compatible with low grade dysplasia.
Dye-less chromoendoscopy
Unlike DCE, which utilizes staining agents and has a long procedure time, dye-less chromoendoscopy or VCE [narrow band imaging (NBI), Fuji intelligent color enhancement (FICE), i-scan, blue light imaging (BLI), and linked color imaging (LCI)] can be performed easily without complex equipment (Table 1). 25
Dye-less chromoendoscopy techniques.

BLI, blue light imaging; FICE, flexible imaging color enhancement; LCI, linked color imaging; NBI, narrow band imaging; OE, optical enhancement; VCE, virtual chromoendoscopy.
NBI uses an optic filter that reduces the light spectrum emitted from the endoscope, thus being absorbed by hemoglobin and providing an enhanced image of the mucosal vascularity.
26
FICE and i-scan belong to DCE. A digital post-processing system is used to enhance the real-time vascular and surface images.
25
Among most advanced developments in light emitting technologies, BLI is based on the not-filtered emission of short wavelength blue light, which is selectively absorbed by hemoglobin.
27
By increasing shades of reddish and whitish tones through the BLI light, an image-enhanced endoscope called LCI, increases slight differences in the red region of the mucosa that are related to inflammation and neoplasia (Figure 3).
28
A prospective study compared structural and vascular alterations of Peyer’s paths (PPs) between 60 IBD patients and 23 healthy controls (HC) through the use of NBI.
29
IBD patients had significant abnormalities in the vessels of PPs (e.g., branch-like structures) compared with HC (

Flat elevated lesion in the ascending colon (IIa according to the Paris classification), pit pattern serrated (IIO according to the Kudo classification). (a) WLE. (b) LCI. (c) BLI. Histologic examination: serrated lesion without dysplasia.
Endocytoscopy
Endocytoscopy (EC) is an endoscopic imaging technique that allows histological analysis of
Confocal laser endomicroscopy
Confocal laser endomicroscopy (CLE) is an additional endomicroscopic technique that allows for tissue analysis with a maximum depth of imaging of 250 μm.
42
In UC patients in remission, CLE revealed a significant rate of mucosal pathologic abnormalities such as impaired and distorted crypt regeneration, sustained inflammation, and abnormal vascular patterns (all
Most relevant studies on available techniques for the assessment of mucosal inflammation.
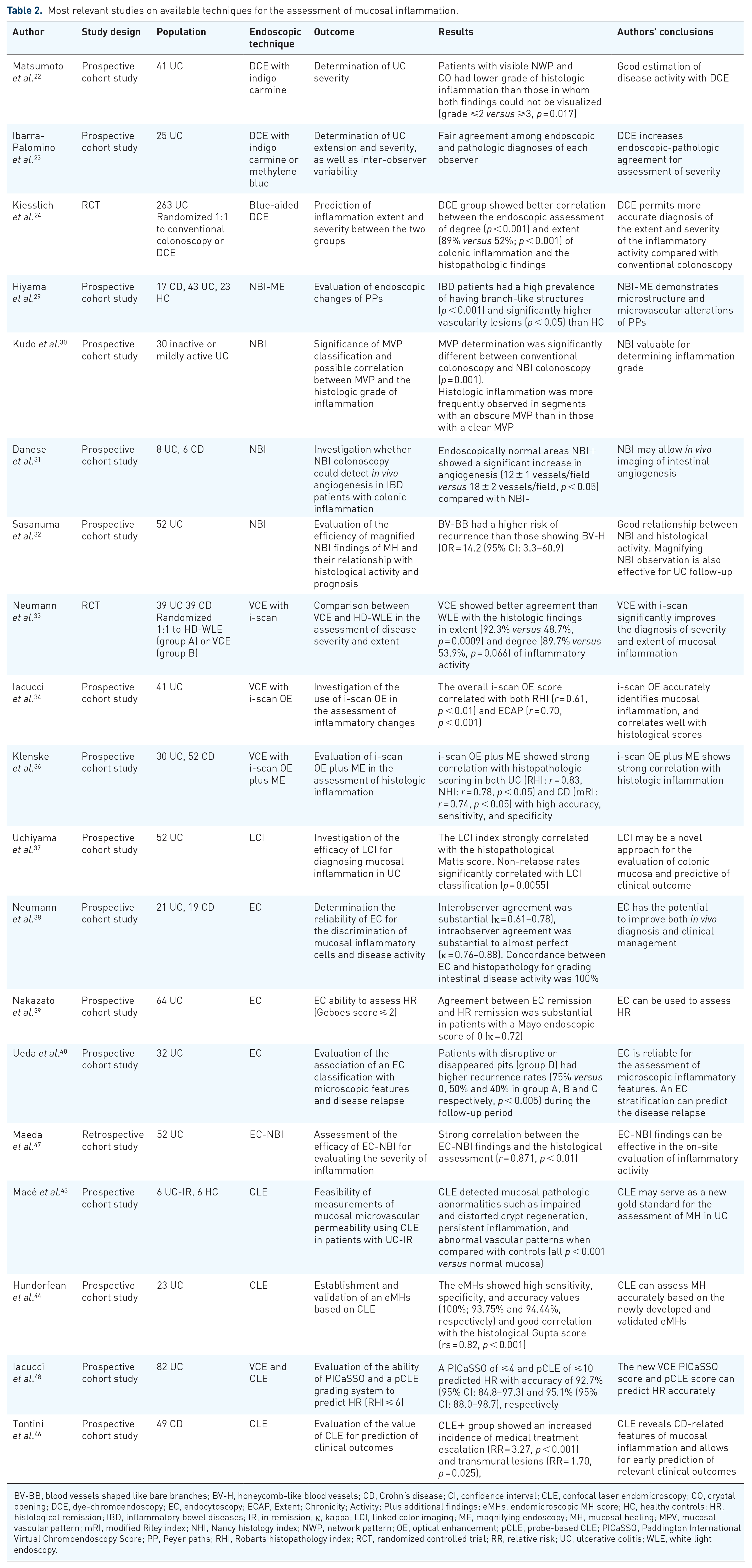
BV-BB, blood vessels shaped like bare branches; BV-H, honeycomb-like blood vessels; CD, Crohn’s disease; CI, confidence interval; CLE, confocal laser endomicroscopy; CO, cryptal opening; DCE, dye-chromoendoscopy; EC, endocytoscopy; ECAP, Extent; Chronicity; Activity; Plus additional findings; eMHs, endomicroscopic MH score; HC, healthy controls; HR, histological remission; IBD, inflammatory bowel diseases; IR, in remission; κ, kappa; LCI, linked color imaging; ME, magnifying endoscopy; MH, mucosal healing; MPV, mucosal vascular pattern; mRI, modified Riley index; NHI, Nancy histology index; NWP, network pattern; OE, optical enhancement; pCLE, probe-based CLE; PICaSSO, Paddington International Virtual Chromoendoscopy Score; PP, Peyer paths; RHI, Robarts histopathology index; RCT, randomized controlled trial; RR, relative risk; UC, ulcerative colitis; WLE, white light endoscopy.
Endoscopic technologies for the detection of dysplasia
Patients with longstanding colonic IBD have an increased risk of developing colorectal cancer (estimated prevalence is approximately 3.5% in both longstanding UC and CD).49,50 Subsequently, 8–10 years after disease onset, IBD patients are enrolled in surveillance colonoscopy programmes. 2
At the moment DCE is recognized as the gold standard method for dysplasia surveillance in IBD, as it was demonstrated to be superior to WLE for dysplasia detection. 14
Dye-chromoendoscopy
A prospective study by Picco
Dye-less chromoendoscopy
The first results from studies evaluating the effectiveness of VCE in revealing dysplastic lesions in IBD are now becoming available.
15
To compare the performance of DCE with NBI for the detection of neoplastic lesions in patients with long-standing UC, a multicenter prospective study randomized 1:1 131 patients to DCE (
Two studies by Hoffman
In addition, an updated meta-analysis conducted by El-Dallal
A new classification of lesions discovered during IBD surveillance, FACILE, (Frankfurt Advanced Chromoendoscopic IBD LEsions classification) was recently proposed.
60
It integrates four endoscopic findings (e.g., morphology, surface and vessel pattern, and inflammation), focusing on predictors of dysplasia (e.g., flat shape, irregular vessel and surface pattern, and signs of inflammation).
60
Finally, data about the use of either BLI or LCI to identify neoplastic lesions in patients with IBD are not available yet. However, these newer-generation endoscopic devices proved to be promising in gastrointestinal tumor surveillance in the general population.61–63 A prospective randomized study including 245 patients proved that BLI was more accurate in predicting the histopathology of colon polyps when compared with HD-WLE (92%
Endocytoscopy and CLE
At the moment, consistent evidence from using EC and CLE for dysplasia detection is lacking, probably due to the intrinsic difficulties of obtaining clear images during breathing and cardiac motion.
64
A prospective pilot study in 2011 aimed to evaluate feasibility and diagnostic accuracy of pCLE in UC surveillance.
65
Even though accuracy, specificity, and sensitivity of pCLE were 81%, 82%, and 65%, respectively, these performance measures were inferior to those provided by NBI/HD-WLE in real time (92%, 89% and 100%, respectively).
65
In line with these findings, a study assessing the diagnostic value of CE combined with integrated CLE (iCLE) reported a relatively good accuracy (86.7%, 95% CI: 78.1–95.3
Most relevant studies on available techniques for dysplasia detection in IBD.
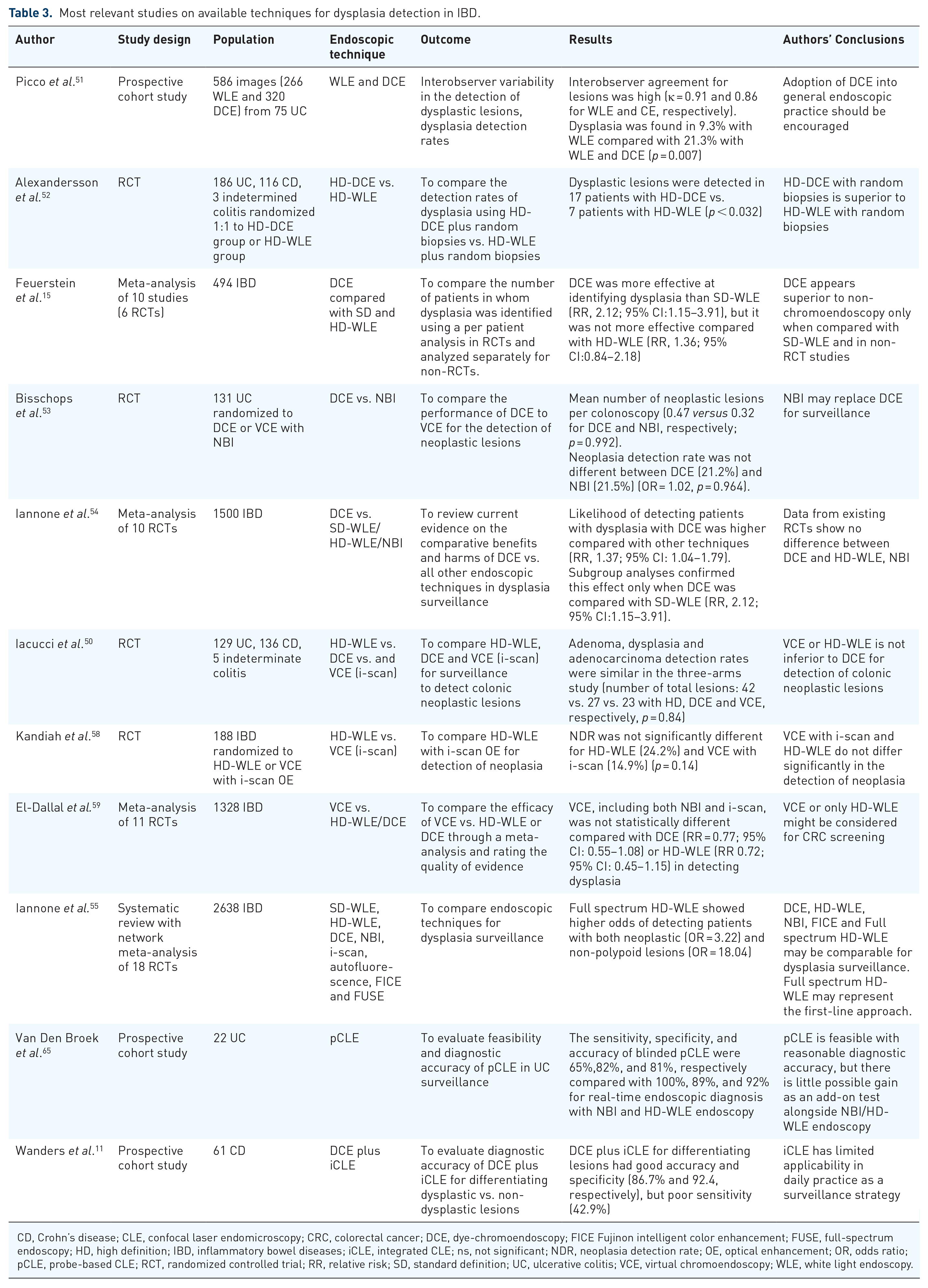
CD, Crohn’s disease; CLE, confocal laser endomicroscopy; CRC, colorectal cancer; DCE, dye-chromoendoscopy; FICE Fujinon intelligent color enhancement; FUSE, full-spectrum endoscopy; HD, high definition; IBD, inflammatory bowel diseases; iCLE, integrated CLE; ns, not significant; NDR, neoplasia detection rate; OE, optical enhancement; OR, odds ratio; pCLE, probe-based CLE; RCT, randomized controlled trial; RR, relative risk; SD, standard definition; UC, ulcerative colitis; VCE, virtual chromoendoscopy; WLE, white light endoscopy.
What is next: capsule endoscopy, artificial intelligence, and molecular imaging
Capsule endoscopy
Small bowel capsule endoscopy (SBCE) is a particularly sensitive instrument used to detect mucosal alterations in the small bowel.
2
It should be considered for patients with clinical suspicion of CD who have already undergone an endoscopy, as stated in more recent guidelines.
2
Since the introduction of the first wireless capsule endoscopy (CE) in the 2000s, several advances, such as improved resolution systems and new types of capsules, have been made to further increase diagnostic yield.
66
Several clinical trials have investigated the potential role of SBCE in MH assessment.67,68 A prospective, multicenter, case-series included 40 patients who underwent SBCE before therapy initiation and after achievement of clinical response.
67
The number of large ulcers detected through SBCE was significantly reduced after treatment (8.3 ± 1.4
Artificial intelligence
The broad term AI generally refers to the ability of a computer to perform functions and reasoning that are typical of the human mind.
73
AI tools are used in the field of endoscopy, and studies evaluating AI systems for endoscopic support have shown good results.74,75 This can be highly valued also in IBD, where the endoscopic assessment can be influenced by operator subjectivity.
76
A computer-aided diagnosis (CAD) system in use with EC was developed to predict persistent histologic inflammation in UC patients.
47
CAD provided good performance measures, showing sensitivity, specificity, and accuracy of 74% (95% CI: 65–81%), 97% (95% CI: 95–99%), and 91% (95% CI: 83–95%), respectively.
47
A CAD system based on GoogLeNet architecture was applied to identify normal mucosa (Mayo endoscopic sub-score 0) and MH (Mayo endoscopic sub-score 0–1) in an independent test set of 3981 images from 114 UC patients.
77
This new system performed well, with AUCs of 0.86 and 0.98 in the two groups, respectively.
77
Moreover, a computer algorithm based on the integration of pixel color data from the redness map and vascular pattern recognition, called red density (RD), was tested in a validation cohort of 29 UC patients.
78
In this latter study, Bossuyt
Taking advantage of convolutional neural networks, a fully automated video analysis system was developed recently, aimed at generating Mayo endoscopic scores automatically. 81 The analysis of 264 videos from a multicenter clinical trial proved that this system was able to distinguish remission from endoscopic activity in more than 80% of videos reviewed, with a positive level of agreement with gastroenterologist scoring (kappa = 0.84). 81
Molecular imaging
Molecular imaging is based on the topical or intravenous administration of specific and label structures and the subsequent visualization of targeted structures through simultaneous confocal imaging.
82
Molecular imaging with fluorescent labeled antibody for membrane-bound TNF (mTNF) in conjunction with CLE can be used for
Multiphoton imaging is a molecular imaging technique that provides both high cellular resolution and great depth of penetration into the tissue without the need for labels.
85
An endoscopic application of this system, named multiphoton endomicroscopy (MPEM), has recently been proven to yield label-free
Discussion
Progress in endoscopy has paved the way for a more accurate approximation of histopathologic results. Histological modifications, inflammation, and healing are well reflected by endoscopic images provided by cutting-edge techniques. Furthermore, with the introduction of new advanced endoscopic modalities, such as VCE, most dysplastic lesions are visible and a smaller number of biopsy specimens is required, making surveillance less cumbersome for both patients and clinicians.53,87 Among image enhancement techniques, the available evidence suggests that dye-less chromoendoscopy is not inferior to the gold standard DCE in terms of neoplastic lesions detection.48,53,54,59 Moreover, dye-less chromoendoscopy modalities offer the advantage of requiring less procedure time without additional spray-agent costs (median withdrawal time: 15
EC and CLE are also available, and they provide the clear advantage of allowing
In addition, SBCE might be seen not only as a diagnostic tool in patients with suspected small bowel CD and normal endoscopy and magnetic resonance enterography (MRE), but also as an accurate investigation method to evaluate MH in small-bowel CD patients. Capsule retention leading to an urgent endoscopic or surgical intervention remains one of the main concerns of this device; as a result, it is not considered in patients with known or suspected stenotic disease or with a history of bowel obstruction. Of note, the new Pillcam Crohn’s capsule with a pan-enteric video system is a promising and safe tool for the assessment of MH in selected populations such as pediatric patients in order to avoid endoscopy deep sedation and biopsy-related complications. The use of pan-enteric CE for activity monitoring should be encouraged in centers where video capsule is a routine examination, and the staff are adequately trained to read images. Due to its likely cost-effectiveness (mean total 20-year cost per patient: £38,043
AI represents a revolutionary technique in the endoscopy field, providing an objective and accurate evaluation of disease severity. AI offers enormous potential to improve the quality of endoscopic procedures and ultimately to reduce the number of biopsy specimens.
91
The use of AI in endoscopy is also expected to increase the ADR and help non-expert endoscopists in the challenging differentiation between inflammatory and neoplastic lesions, although this will need to be confirmed in studies involving IBD patients. Finally, implementing molecular imaging in endoscopy could provide physicians with a real-time
Conclusion
The goal of HR in IBD management has certainly influenced new advances in the endoscopic field. Endoscopy is now asked to accurately describe inflammatory disease activity and promptly identify suspected lesions during the ongoing procedure, thus getting as close as possible to microscopic findings. Dye-less chromoendoscopy has already demonstrated to be feasible and cost-effective in a real-life setting, whereas endomicroscopy systems still appear as complex and costly techniques. New pan-enteric video capsules might enrich the endoscopic armamentarium for monitoring disease in specific populations. Further clinical studies are expected to confirm the great expectations placed on AI tools and molecular imaging.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848211005692 – Supplemental material for Rediscovering histology: what is new in endoscopy for inflammatory bowel disease?
Supplemental material, sj-pdf-1-tag-10.1177_17562848211005692 for Rediscovering histology: what is new in endoscopy for inflammatory bowel disease? by Virginia Solitano, Ferdinando D’Amico, Mariangela Allocca, Gionata Fiorino, Alessandra Zilli, Laura Loy, Daniela Gilardi, Simona Radice, Carmen Correale, Silvio Danese, Laurent Peyrin-Biroulet and Federica Furfaro in Therapeutic Advances in Gastroenterology
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
V. Solitano, F. D’Amico, A. Zilli, L. Loy, D. Gilardi, S. Radice, and C. Correale declare no conflict of interest. M. Allocca received consulting fees from Nikkiso Europe and lecture fees from Janssen and Pfizer. G. Fiorino received consultancy fees from Ferring, MSD, AbbVie, Takeda, Janssen, Amgen, Sandoz, Samsung Bioepis, and Celltrion. S. Danese has served as a speaker, consultant, and advisory board member for Schering-Plough, AbbVie, Actelion, Alphawasserman, AstraZeneca, Cellerix, Cosmo Pharmaceuticals, Ferring, Genentech, Grunenthal, Johnson and Johnson, Millenium Takeda, MSD, Nikkiso Europe GmbH, Novo Nordisk, Nycomed, Pfizer, Pharmacosmos, UCB Pharma, and Vifor. L. Peyrin-Biroulet has served as a speaker consultant and advisory board member for Merck, Abbvie, Janssen, Genentech, Mitsubishi, Ferring, Norgine, Tillots, Vifor, Hospira/Pfizer, Celltrion, Takeda, Biogaran, Boerhinger-Ingelheim, Lilly, HAC- Pharma, Index Pharmaceuticals, Amgen, Sandoz, Forward Pharma GmbH, Celgene, Biogen, Lycera, Samsung Bioepis, and Theravance. F. Furfaro received consulting fees form MSD and Abbvie and lecture fees from Janssen and Pfizer.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
