Abstract
Dysbiosis, a loss of balance between resident bacterial communities and their host, is associated with multiple diseases, including inflammatory bowel diseases (nonspecific chronic ulcerative colitis and Crohn’s disease), and digestive functional disorders. Probiotics, prebiotics, synbiotic organisms and, more recently, pharmabiotics, have been shown to modulate the human microbiota. In this review, we provide an overview of the key concepts relating to probiotics, prebiotics, synbiotic organisms, and pharmabiotics, with a focus on available clinical evidence regarding the specific use of a unique pharmabiotic, the strain
Introduction
The complex interaction between microbiota and humans is currently recognized as fundamental for balance (eubiosis) and life development. Medical research has shown that the balance lost (dysbiosis) between resident bacterial communities and their host can lead to multiple diseases.1,2 There are many conditions associated with dysbiosis, including metabolic diseases (e.g. obesity, fatty liver, cardiovascular conditions, etc.), infectious processes (acute diarrhea, antibiotic-associated diarrhea and
Since dysbiosis was recognized as a pathophysiological mechanism, it has been proposed that microbiota modulation through drugs and food (probiotics, prebiotics, synbiotic organisms and, recently, pharmabiotics) may aid in restoring the eubiotic condition. Moreover, the subject has drawn interest from the scientific community as well as among the general public. Recommendations are usually heard in the media promoting the use of ‘probiotics’ as a helpful measure to maintain a healthy condition.1–3
It is essential to acknowledge that each previously stated term is different from the other, and the evidence for their benefit is heterogeneous; meaning that not all of them work in the same way and it should not be assumed that the effects of a strain, a prebiotic or pharmabiotic, will be similar in all conditions. This article presents a detailed review of dysbiosis-related concepts frequently associated with gastrointestinal conditions, and the specific use of the strain
Generalities and definitions
The human microbiota consists of a wide variety of bacteria, viruses, fungi, and other unicellular microorganisms. Bacteria control the gut microbiota, and these are represented mainly by the phyla
nutrient degradation and absorption;
degradation of non-digestible carbohydrate (e.g. plant polysaccharides);
intestinal barrier maintenance;
protection against pathogens (inhibition of pathogen attachment to the intestinal epithelium);
modulation and correct maturation of the immune system;
participation in intestinal health;
production of a variety of metabolites, such as vitamins and short-chain fatty acids (SCFAs).
Each human houses around 10–100 quintillion microorganisms (~1000 different species). The gut microbiota is composed of indigenous members which colonize the intestinal mucosa, and by the transitory microbiota derived from ingested food.4,7,8 The human microbiome is the group of genes inside the microbial cells which influences four health areas: (a) nutrition, (b) immunity, (c) behavior, and (d) disease. 8
The human microbiota starts its development at birth. Cesarean section, a milk formula nutrition, a diet high in fat and sugar, the use of antibiotics, and excessive hygiene, adversely affect the health of the microbiota. Infant gut microbiota matures in the first 3 years of life. 9
Prebiotic
Under the auspices of the International Scientific Association for Probiotics and Prebiotics (ISAPP), an expert panel recently reviewed the definition and scope of prebiotics. 10 A prebiotic is defined as a non-viable substrate that serves as a microbiota nutrient and is selectively fermented by the microbiota, leading to specific changes in the host’s gastrointestinal microbiota composition and/or activity, resulting in a health benefit.10,11
The definition expands the concept of prebiotics in order to include potential non-carbohydrate substances, making it possible to apply the concept to body parts other than the digestive system (e.g. vagina, skin), and multiple food categories. Thus, other substances fall within the updated definition, such as polyphenols and polyunsaturated fatty acids converted to their respective conjugated fatty acids, assuming that convincing evidence is presented in favor of their beneficial health effect. 10
The difference between dietary fiber and prebiotics is that defined groups of microorganisms exclusively ferment the latter, while dietary fiber (pectins, cellulose, and xylan) is used by most colonic microorganisms. 12
Prebiotics have health benefits in the digestive tract (e.g. pathogen inhibition, stimulation of the immune system), the cardiovascular system (e.g. reduced blood lipid counts, impact on insulin resistance), mental health (e.g. metabolites that have an effect on brain function, energy and cognition), and the bone system (e.g. mineral bioavailability), among others. Therefore, prebiotics can improve human health and reduce the risk of diseases mediated by aberrations in the microbiota. 10
Probiotic
When administered in adequate amounts, live microorganisms (bacteria or fungus), provide health benefits to the host, for example, species of
Probiotic microorganisms used in human nutrition. 12 Reproduced with permission from MDPI.

Mostly used in pharmaceutical products.
Mostly used as food additives.
Qualified presumption of safety microorganisms.
Synbiotic
A product containing probiotics and prebiotics which facilitates the
increased count of
maintenance of the microbiota balance;
improved hepatic function in cirrhotic patients;
increased immunomodulating capacities;
bacterial translocation prevention and reduction of nosocomial infections in surgery. 14
Synbiotic microorganisms used in nutrition are:
12
Postbiotic
Non-viable bacterial products or metabolic bioproducts of probiotic microorganisms with biological effects on the host. They are considered an effective alternative method to increase the potential and functionality of each probiotic strain. As the understanding of the host–microbiota metabolic axis advances, the usage of postbiotic molecules has become a prominent strategy to treat many inflammatory diseases, since these molecules mimic the beneficial therapeutic effects of probiotics while avoiding the risk of administering live microorganisms into a host with a compromised immune system. Most SCFAs (⩾95%) are primarily generated in the colon; the microorganisms’ metabolic bioproducts, including acetate (two carbons, C2), propionate (three carbons, C3), and n-butyrate (four carbons, C4), have been shown to generate multiple modulatory effects within the host. The metabolic pathways that modulate such beneficial effects act by altering cytokine release, cell recruitment, and survival at the inflammatory site to induce pro-resolutive activities.9,10
Figure 1 shows the distinction between a prebiotic and a non-prebiotic based on the ISAPP consensus; namely, selective use distinguishes prebiotics from other substances. Prebiotics must be selectively utilized and have adequate evidence of a health benefit for the target host. In addition, they must not be degraded by target host enzymes. The main prebiotics are FOS and galactooligosaccharides (GOS). Certain soluble fermentable fibers are possible prebiotics and some other types of dietary fiber can be prebiotics.
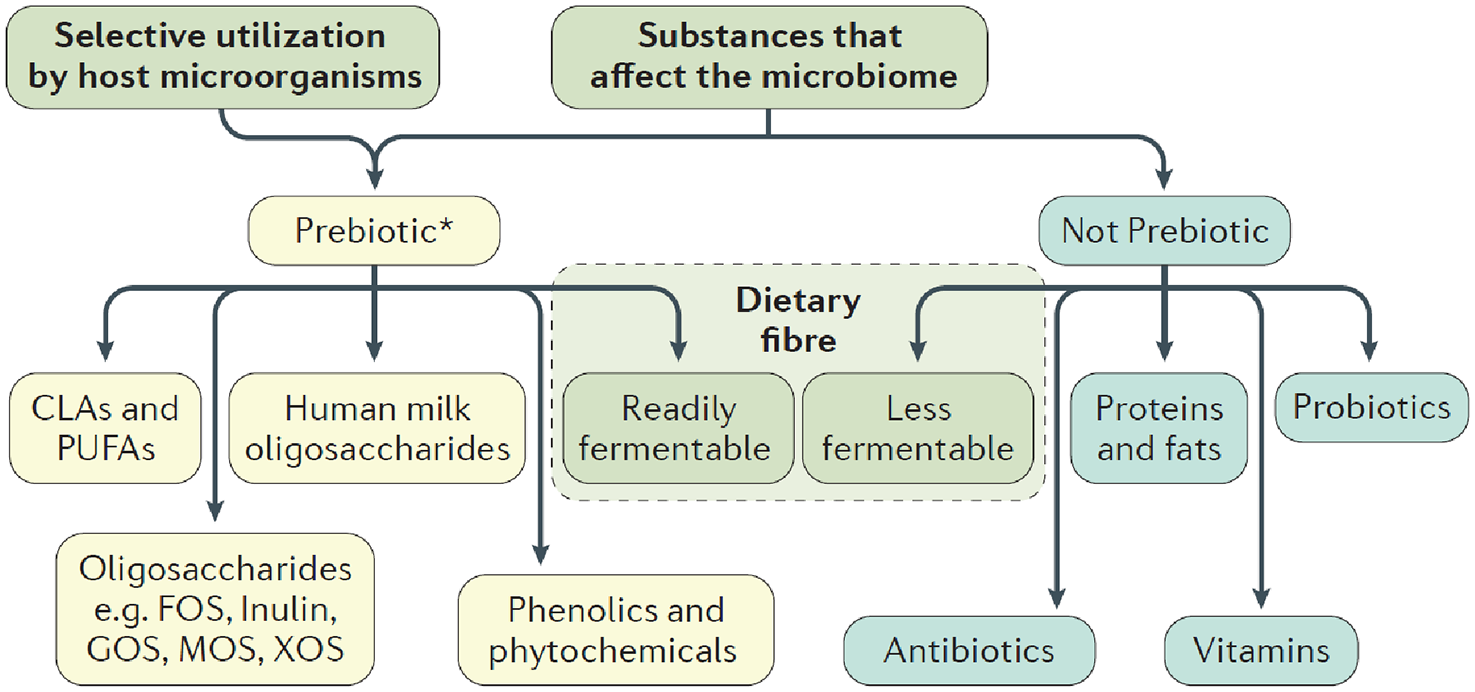
The distinction of a prebiotic based on the proposed definition. Reproduced with permission from Macmilllan/Springer Nature. 10
Pharmabiotic
This term was introduced to comprise any biological entity that is extracted from the microbiota and can exert influence in the microbiota to produce a therapeutic benefit. Therefore, this concept goes beyond live organisms and encompasses dead organisms, their components, and bioproducts. 15 Thus, some postbiotics can be pharmabiotics, but not all postbiotics are pharmabiotics. It has been suggested that pharmabiotics include pharmaceutical probiotics with specific evidence of generating health benefits and with proven positive physiological effects, or with the capacity to play a pharmacological role in diseases.15,16 Hence, pharmabiotics comply with two criteria: 16 (a) they provide physiological and pharmacological health benefits against diseases; and (b) they serve as prevention and treatment for medical conditions.
Dysbiosis and reinstatement of microbiota
Dysbiosis of the intestinal ecosystem (any compositional change in the intestinal resident commensal communities in relation to the community of healthy subjects) contributes to the development of certain diseases that can be reversed with favorable alterations caused by probiotics.1,13
As in other organs, the proper function of the gut microbiota depends on a stable cell composition; in this case, it consists primarily of the bacteria phyla
Environmental impacts, such as the use of antibiotics or diet itself, can result in structural changes of the microbial community. Such variations can lead to the loss of organisms that are beneficial to their host and to a subsequent overgrowth of pathobionts (organisms that, under normal circumstances, live as commensals or symbionts but whose overgrowth could harm). There are three types of dysbiosis in the intestinal ecosystem: 1
loss of beneficial microorganisms;
pathobionts expansion;
loss of the total diversity of microorganisms.
When dysbiosis occurs, the need to restore a healthy microbiota becomes evident. It can be carried out through a fecal microbiota transplant from a healthy donor, although the easiest way is through the administration of dietary supplements, which can be done through multiple mechanisms (fecal microbiota transplant, consumption of prebiotics, probiotics, and postbiotics), as shown in Figure 2.1,9
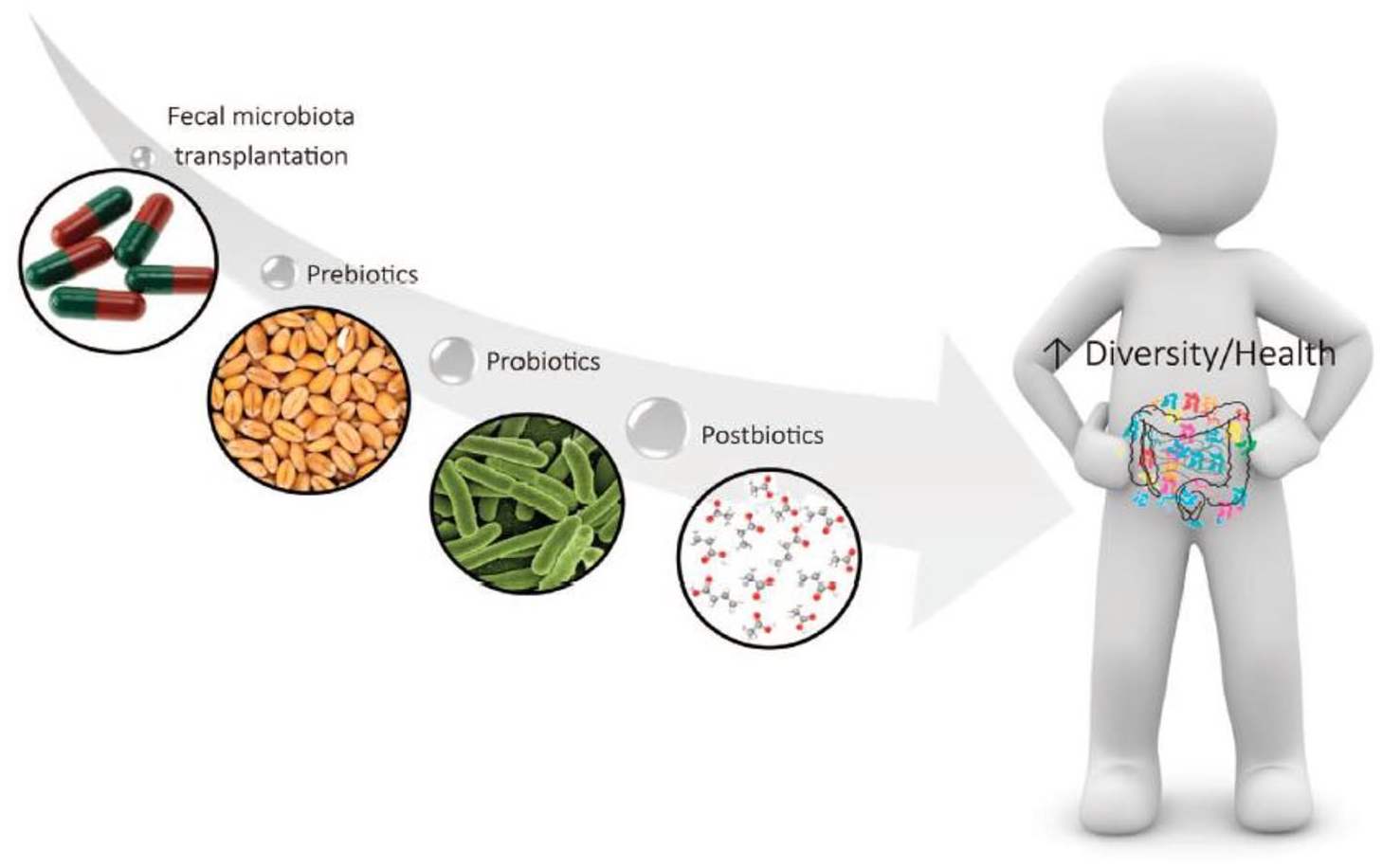
Mechanisms of gut microbiota modulation. 9 Reproduced with permission from Wiley–Blackwell.
The microbiota microorganisms can be grown; therefore, they can be used as probiotics in pills, or they can be included with food. Given the importance of microbial diversity, a single microorganism may not work effectively, but rather an entire group of microorganisms will provide maximum health benefit. Currently, yogurts and probiotics are beginning to introduce multiple strains of microorganisms.
1
The ability to modify the composition and the metabolic signatures of this microbial population is now possible through dietary and non-dietary interventions.10,18 Components which can beneficially modify the microbiota have been studied in the last 20 years. Currently, FOS, inulin and GOS are the most widely studied for their favorable effects on the growth of
All interindividual variability of the gut microbiota can be classified into three groups, called enterotypes, which can be defined as a network of microbial populations dominated by the presence of one of these three genuses:
Benefits of probiotics, nutribiotics, and pharmabiotics
There are six general mechanisms through which probiotics perform their beneficial effects, and there are essential differences between probiotic species and their strains.3,7,12,13
(1) Antimicrobial effects: probiotics can have the following antimicrobial effects: intestinal lumen alterations, production of antimicrobial molecules, inhibition of pathogen adhesion and cell invasion, competitive inhibition of pathogens, and antitoxin effects. These effects can result in intestinal pH reduction, production of bacteriocins, defensins and conjugated bile acids, competition for adhesion sites and resources (iron and nutrients), production of antitoxins, toxin expression prevention, and interference with the host’s response to toxins, thereby preventing
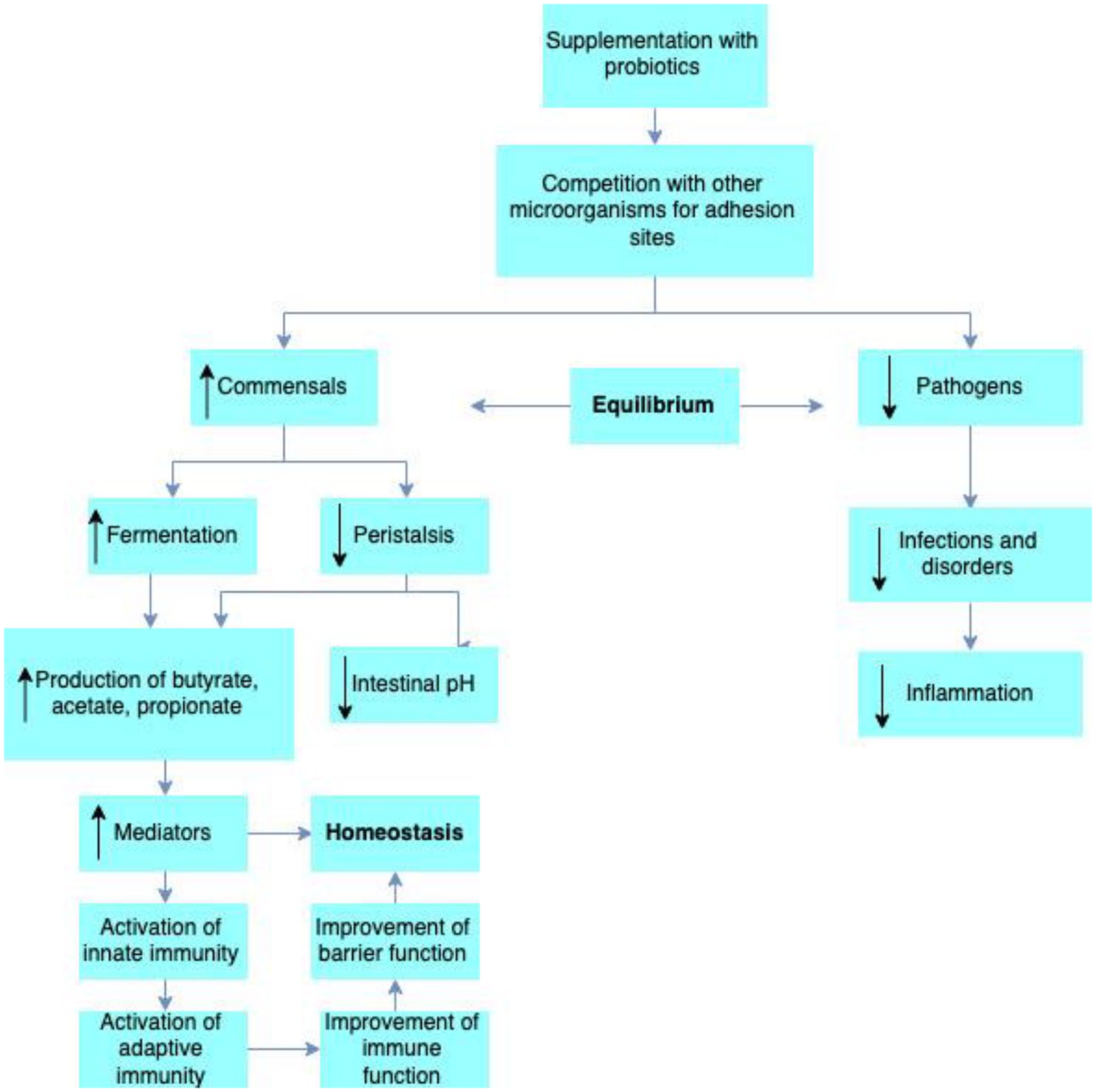
Probiotics’ action mechanisms in homeostasis maintenance. Modified, with permission from Wageningen Publishers. 3
(2) Inhibition of bacterial toxins production: probiotics can absorb and fix toxins to their cell wall, resulting in less intestinal absorption of toxins. Probiotics can also metabolize mycotoxins (e.g. aflatoxins).
(3) Competition with pathogens for adhesion to the epithelium and nutrients: coaggregation of probiotic strains can lead to the formation of a protective barrier over the intestinal epithelium which prevents colonization with pathogenic bacteria.
(4) Strengthening of the mucosal barrier integrity: increased mucus production can reinforce the epithelium barrier, disturbing the surface proteins, and leading to strengthening of the narrow intercellular joints and secretion of water and chloride, which, in turn, has an influence over the mucus interaction between cells and cell stability, and increases the function of the intestinal epithelium.
(5) Influence over other body organs through the immune system and production of neurotransmitters: gamma-amino-butyric acid (GABA), tryptophan, catecholamine, acetylcholine, and 5-hydroxytryptamine (5-HT or serotonin).
(6) Immunomodulation: adhesion of probiotics to the epithelium results in SCFA production with a consequent reduction in the production of pro-inflammatory cytokines, increased anti-inflammatory cytokines, priming of dendritic cells, induction of regulatory T lymphocytes, and an impact on B lymphocytes. These events can lead to a reduction of apoptosis mediated by tumor-necrosis-factor-alpha, increase production of interleukin-10 and antibodies, as well as an increase in secretory immunoglobulin A, resulting in the prevention of atopic dermatitis, CDAD, AAD, infectious diarrhea, and cancer.
The immunostimulant effect which is induced by probiotics is also displayed by increased immunoglobulin production, increased activity of macrophages and lymphocytes, as well as stimulation of interferon production. The immunomodulatory effects of the gut microbiota, including probiotic bacteria, are based on three apparently contradictory phenomena:
induction and maintenance of immunotolerance to environmental antigens (ingested and inhaled);
induction and control of immunological reactions to bacterial or viral pathogens;
inhibition of auto aggressive and allergic reactions.
The positive effects of probiotics can be used to restore the natural microbiota after antibiotic therapy. Another function is to counterattack pathogenic intestinal microbiota activity introduced by food and contaminated environmental elements. Therefore, probiotics can effectively inhibit the development of pathogenic bacteria such as
A positive effect of probiotics has also been confirmed in digestive processes, the management of food allergies, candidiasis, and tooth decay. Probiotic microorganisms such as
Thus, probiotics can present nutritional and/or therapeutic properties. For this reason, the concepts of nutribiotics and pharmabiotics emerged. A nutribiotic comprises probiotic microorganisms or its bioproducts, which are considered to have nutritional properties. Nutribiotics can be present in food, food products, or dietary supplements, and they are subject to sanitary regulations required to guarantee food safety and nutritional guidelines. Consequently, nutribiotics satisfy three criteria: (a) they provide benefits in the form of food or dietary supplements; (b) they work as a therapy for nutritional problems and, (c) they contribute to the maintenance of human health. 16
Nutraceuticals are defined as food or dietary components that play a beneficial role in modifying and maintaining physiological function in order to sustain human health; hence, nutribiotics can be classified as nutraceuticals.
What are lactobacilli?
Along with other aerobic and anaerobic bacteria,
Indications for and safety of lactobacilli
The Mexican Consensus on Probiotics notes that gastrointestinal disorders in which the benefit of lactobacilli has been demonstrated include the following: 25
AAD;
acute infectious diarrhea in children and adults;
prevention of nosocomial diarrhea in children;
prevention of recurrence of diarrhea in children and adults due to
avoiding adverse events from the eradication of
irritable bowel syndrome;
chronic constipation in adults;
lactose intolerance;
concomitant use with standard therapy for the induction or maintenance of remission in mild or moderate chronic non-specific ulcerative colitis in adults;
use after treatment with antibiotics, induction or maintenance of remission of pouchitis in adults;
prevention of necrotizing enterocolitis in preterm infants;
fatty liver;
hidden and manifest hepatic encephalopathy;
in lactating women or infants at high risk of developing an allergy.
Safety of lactobacilli
Probiotics are used widely and their safety has been proven in millions of individuals for many years. Nevertheless, it is important to consider that, since they are bacteria, there is always the possibility that probiotics can behave as infectious agents. Reports of severe infections are rare in the literature and are almost always associated with comorbidity (cancer, cirrhosis, cholecystolithiasis). In a study of 1176 patients, bacteremia associated with probiotics occurred in 0.2%. 26
Lactobacillus acidophilus
Even though
The strain
It is important to differentiate between probiotics (live organisms) and heat-treated strains, where the organisms are dead.27,29
The characterization of heat-treated strains can be divided into two broad categories. The first category includes probiotic physiology that can be demonstrated
As mentioned previously, when
It has similar activity to antibiotics. Secreted molecules present in the culture of
It has intravacuolar bactericidal activity. Demonstrated effect against
It has adhesive and cytoprotective properties. Creation of a biofilm that protects enterocytes against diffusely adherent
It has bacteriostatic action; its effect against

Summary of the mechanisms of action of
Safety of L. acidophilus LB, scientific support
A review of 57 clinical trials showed that the administration of probiotics and/or synbiotic organisms in immunocompromised adults (human immunodeficiency virus infection, critical, surgical, autoimmune disease patients) is safe.
14
In particular, the safety of heat-treated and lyophilized
In addition, the use of heat-treated and lyophilized
In another controlled clinical trial, conducted in Ecuador, 80 infants aged 1–24 months with acute diarrhea, probably infectious, lasting less than 72 h, received 10 billion
In Thailand, a controlled clinical trial was also conducted in 73 infants aged 3–24 months with acute diarrhea of less than 5 days’ duration, who received
On the other hand, Xiao
Benefits of L. acidophilus LB in different gastrointestinal pathologies
Acute diarrhea
Probiotics are used as a supplement to rehydration therapy in the treatment of infectious diarrhea. Results have been positive and remarkably consistent in terms of shortening the duration of the episode and reducing the frequency of evacuation.7,33
Randomized and non-randomized clinical trials have demonstrated the therapeutic efficacy of lyophilized and heat-treated cells and culture media together with oral rehydration solution therapy for the treatment of acute, well-established rotavirus-induced acute watery diarrhea in infants (Table 2).32,34–39 Thus, for example, in children with infectious diseases treated with
Overview of clinical therapeutic effects of

Drug: sachet or capsule pharmaceutical forms (Lacteol®) containing lyophilized and heat-treated combination of 10 billion
ORS, oral rehydration solution.
In a controlled clinical trial, oral rehydration plus placebo, and oral rehydration plus
A systematic review and meta-analysis of controlled clinical trials documented that the use of
Chronic diarrhea
A controlled, randomized clinical trial of 137 patients with chronic diarrhea compared the administration of two capsules per day of
Antibiotic-associated diarrhea
Antibiotic therapy alters the gut microbiota and results in diarrhea. In clinical studies that demonstrated their effectiveness, several types of probiotics were started 1–2 days after initiating antibiotic therapy with doses that ranged from 107 to 1010 per day and continued for 1–4 weeks after the discontinuation of the antibiotic. 42
In around one third of cases, diarrhea is related to the overgrowth of
Discussion
Even when there are studies showing the efficacy and safety of
In its recent guidelines, the American Gastroenterological Association (AGA) suggests avoiding the use of probiotics in children with acute infectious gastroenteritis (conditional recommendation). In adults and children with antibiotic treatment, the AGA suggests the use of
However, in a recent systematic review, the authors found a total of four randomized clinical trials using non-viable
Conclusion
Nowadays, it is recognized that it is important to maintain the gut microbiota through diet and, when, as a result of disease, antibiotic use, or other causes, dysbiosis develops through the use of supplements. These can be nutraceuticals or pharmabiotics. Currently, there is sufficient evidence to consider that the administration of
Footnotes
Acknowledgements
Under the direction of the authors, technical editorial assistance was provided by David P. Figgitt, PhD, ISMPP CMPP™, Content Ed Net, with funding from Laboratorios Carnot.
Conflict of interest statement
José María Remes Troche is a member of advisory boards for Asofarma, Takeda and Laboratorios Carnot and has given lectures for Asofarma, Takeda, Alfa Sigma, Sanfer, Menarini, and Laboratorios Carnot.
Enrique Coss Adame declares being a member of advisory boards for Takeda Mexico, Asofarma Mexico, and Grünenthal Mexico, and a speaker for Takeda, Asofarma, Grünenthal, and Alfa Sigma.
Miguel Ángel Valdovinos Díaz declares no conflicts of interest associated with this manuscript.
Octavio Gómez Escudero declares no conflicts of interest associated with this manuscript
María Eugenia Icaza Chávez declares no conflicts of interest associated with this manuscript
José Antonio Chávez-Barrera declares no conflicts of interest associated with this manuscript.
Flora Zárate Mondragón declares no conflicts of interest associated with this manuscript.
José Antonio Ruíz Velarde Velasco declares no conflicts of interest associated with this manuscript
Guillermo Rafael Aceves Tavares declares being a speaker for Laboratorios Carnot.
Marco Antonio Lira Pedrín declares no conflicts of interest associated with this manuscript.
Eduardo Cerda Contreras declares no conflicts of interest associated with this manuscript.
Ramón Isaías declares no conflicts of interest associated with this manuscript.
Héctor Guerra López and Rodolfo Solana Ortiz are employees of Laboratorios Carnot.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
