Abstract
Background and Aims:
Crohn’s disease (CD) can be complicated by intestinal fibrosis. Pharmacological therapies against intestinal fibrosis are not available. The aim of this study was to determine whether pathways involved in collagen metabolism are upregulated in intestinal fibrosis, and to discuss which drugs might be suitable to inhibit excessive extracellular matrix formation targeting these pathways.
Methods:
Human fibrotic and non-fibrotic terminal ileum was obtained from patients with CD undergoing ileocecal resection due to stenosis. Genes involved in collagen metabolism were analyzed using a microfluidic low-density TaqMan array. A literature search was performed to find potential anti-fibrotic drugs that target proteins/enzymes involved in collagen synthesis, its degradation and its recognition.
Results:
mRNA expression of collagen type I (COL1A1, 0.76 ± 0.28 versus 37.82 ± 49.85, p = 0.02) and III (COL3A1, 2.01 ± 2.61 versus 68.65 ± 84.07, p = 0.02) was increased in fibrotic CD compared with non-fibrotic CD. mRNA expression of proteins involved in both intra- and extracellular post-translational modification of collagens (prolyl- and lysyl hydroxylases, lysyl oxidases, chaperones), collagen-degrading enzymes (MMPs and cathepsin-K), and collagen receptors were upregulated in the fibrosis-affected part. A literature search on the upregulated genes revealed several potential anti-fibrotic drugs.
Conclusion:
Expression of genes involved in collagen metabolism in intestinal fibrosis affected terminal ileum of patients with CD reveals a plethora of drug targets. Inhibition of post-translational modification and altering collagen metabolism might attenuate fibrosis formation in the intestine in CD. Which compound has the highest potential depends on a combination anti-fibrotic efficacy and safety, especially since some of the enzymes play key roles in the physiology of collagen.
Introduction
Fibrosis in any organ is the result of chronic injury, leading to a disturbed balance in the formation and degradation of an extracellular matrix (ECM) rich in collagen. 1 Crohn’s disease (CD) is an inflammatory bowel disease showing a heterogeneous phenotype. The phenotype, which can change over time, can be classified according to the Montreal classification in non-stricturing/non-penetrating (B1), stricturing (B2), and penetrating disease (B3).2–6 A stricturing phenotype, which is characterized by intestinal fibrosis, occurs mainly in CD (70%). However, it can also occur in ulcerative colitis (UC; 1.5–11.2%), upon radiation injury, or upon chronic allograft dysfunction after intestinal transplantation.7–11 In CD, thickening of the intestinal wall causes symptomatic fibrotic stenosis due to narrowing of the lumen, which requires surgery. In UC, it will lead to thickening and shortening of the colon. 12 The mechanisms involved in transmural intestinal fibrosis may be comparable with those of pathological collagen accumulation in other organs, and drugs tested for fibrosis in other organs might be applicable to intestinal fibrosis as well. So far, despite the tremendous amount of research performed on renal and hepatic fibrosis, pharmacological treatments have become available only for fibrotic conditions such as idiopathic pulmonary fibrosis.13,14 No pharmacological therapies against intestinal fibrosis are available so far.
The interstitial matrix of the intestine consists of the fibrillary collagens type I, III and V, and in addition collagen type VI. 15 The non-fibrillar collagen type IV is the main component of the basement membrane, which creates the barrier between the epithelium on the gut luminal side and the lamina propria of the intestine. 16 In addition, intestinal ECM (as ECM of any other organ) consists of fibronectin (FN1) and presumably of elastin, as well as the proteoglycans decorin, biglycan (BGN), and fibromodulin.17,18 An increase, especially in the amount of interstitial collagens, is accompanied with thickening and stiffening of the intestinal wall, thereby causing stenosis due to the luminal stricture of the intestine. 19 Net deposited ECM is the result of a complex balance between factors involved in collagen synthesis (including post-transcriptional modification) versus collagen degradation. Even though fibrogenesis in the intestine is presumably similar to fibrosis in other organs, the expression of genes involved in collagen homeostasis has not been evaluated before.
Generally, formation of collagen starts with transcription of procollagen mRNA in the nucleus, leading to synthesis on ribosomes of three polypeptide α-chains that are released into the endoplasmic reticulum for post-transcriptional modification.20,21 Within the endoplasmic reticulum, certain lysine and proline residues from the α-chains are hydroxylated by lysyl hydroxylases (LHs) and prolyl 3- and 4- hydroxylases (P3H, P4H), respectively. Some of the hydroxylysine residues are subsequently glycosylated by collagen glycosyltransferases. During hydroxylation and glycosylation, the three procollagen-chains are assembled and further stabilized by formation of intra- and inter-molecular disulfide bonds. 20 Triple helix formation requires the aid of chaperones like heat shock protein 47 (HSP47) and FK506 binding protein 10.22,23 Procollagens are then transported out of the cell via the Golgi apparatus. In the extracellular space, the N- and C- terminal propeptides are enzymatically cleaved off [by enzymes from the “a Disintegrin and Metalloproteinase with Thrombospondin motifs” (ADAMTS, N-terminal) family and by Bone Morphogenetic Protein 1 (BMP1, C-terminal)]. Subsequently, collagens are assembled into fibrils and cross-linking is induced by lysyl oxidases/lysyl oxidase-like (LOX, LOXL) enzymes. Degradation of collagens occurs mostly via matrix metalloproteinases (MMPs). MMPs are Zn2+-dependent endopeptidases that can degrade a plethora of ECM proteins, including collagen. 24 Degradation products of ECM can have chemotactic properties and MMPs are able to activate or degrade several non-ECM substrates like cytokines/chemokines or growth factors. The MMPs thereby play a central role in ECM remodeling as well as in intestinal inflammation. MMPs 1, 2, 3, and 9 activity is elevated in the active mucosal inflammation of both patients with CD and UC, and the balance between MMPs and tissue inhibitors of MMPs (TIMPs) is altered in inflammatory bowel disease (IBD).25,26
Candidate drugs against intestinal fibrosis mostly target pathways of (intestinal) fibrosis such as the mitogen-activated protein kinase (MAPK), Rho-associated protein kinase (ROCK), and transforming growth factor β (TGF-β) pathways.27,28 However, genes involved in the assembly of the ECM also comprise targets that can inhibit ECM formation or alter its molecular structure in such a way that ECM will be degraded faster. 29 Here, we show an upregulation of mRNA expression of genes involved in collagen metabolism in intestinal fibrosis affected terminal ileum of patients with CD. These results reveal possible drug targets, which are reviewed in this paper.
Methods
Fibrosis affects terminal ileum from patients with CD and from non-fibrotic control patients
Fibrotic and non-fibrotic terminal ileum from patients with CD undergoing ileocecal (re-)resection because of stenosis was obtained. All included patients with CD had purely stricturing phenotype (Montreal B2, Table 1). The fibrotic/stenotic and the non-fibrosis affected (resection margin) regions were identified macroscopically during surgery. Non-fibrotic non-CD affected tissue was obtained from patients undergoing right-sided hemicolectomy because of an adenocarcinoma (non-cancer affected ileal resection margin, Table 1). All tissue obtained was freshly collected and stored by a laboratory technician in the operating room immediately after resection. Samples were fixed in Tissue-Tek® (O.C.T. Compound, Sakura® Finetek) in the operation room, and frozen in isopentane on dry ice before being stored at −80°C until further use.
Characteristics of the patients with CD and controls of which terminal ileum was obtained.

CD, Crohn’s disease; HBI, Harvey Bradshaw Index.
Isolation of RNA
To isolate RNA, 10 Tissue-Tek sections (10 μm thick) containing the full thickness (verified by hematoxylin and eosin staining) of the intestinal wall, were cut using a cryostat. Sections were dissolved in TRIzol (Invitrogen, Life Technologies, Carlsbad, CA, USA), after which total RNA was isolated according to the manufacturer’s protocol. To avoid genomic DNA contamination, samples were treated with DNase I, Amp Grade (Invitrogen) according to the manufacturer’s protocol.
Reverse transcription and TaqMan® gene expression assays
Equal amounts of RNA were reverse transcribed using the Reverse Transcription System (Promega, Madison, WI, USA). Subsequently, complementary DNA was used for quantitative real-time polymerase chain reaction (RT-qPCR) in a microfluidic card-based low-density TaqMan array (Applied Biosystems, Foster City, CA, USA), which enables simultaneous measurement of mRNA expression of 44 genes we selected (Supplemental Table S2). RT-qPCR was performed by loading 100 ng of cDNA per sample using the ViiA™ 7 Real-Time PCR System (Applied Biosystems). The following settings were used: 50°C for 2 min, 95°C for 10 min, and the next two steps were repeated for 50 cycles: 95°C for 12 s and 60°C for 1 min. Threshold cycle numbers >40 were excluded from the analysis. Patients/pairs were removed from the analysis if there was no detectable expression (Ct > 40) in either one of the pairs. Delta-Ct values were calculated using GAPDH as a reference gene. Expression in the figures is presented as 2–∆Ct on a logarithmic scale.
Statistical analysis
Data were statistically analyzed and visualized with graphs using GraphPad Prism software (v6.0). All data was considered to be non-parametric. A Wilcoxon paired signed rank test was used to compare analysis of fibrotic versus non-fibrotic ileum from the one patient with CD for gene expression as well as quantification of immunohistochemistry (IHC). A Mann-Whitney U test was used to compare with CD-affected non-fibrotic ileum to non-CD-affected non-fibrotic ileum, and to compare age at surgery. Differences were considered significant at a p value of < 0.05. Median interquartile range (IQR) values are presented in the text and figures.
Literature search
A literature search was performed to find (potential) drugs that target the proteins/enzymes transcribed from genes involved in collagen fibril synthesis and degradation, detected in CD patients with stricturing phenotype by a microfluidic card-based low-density TaqMan array. A comprehensive literature search was conducted to identify relevant drugs. The electronic exploration involved keyword searches in Pubmed. The following search criteria were used (all fields) (“gene name” or “protein name”) and (“inhibitor”, “antagonist” or “agonist”). Targeting drugs tested in vivo/in silico, in vivo in animals or in vivo in humans are listed separately in Table 2.
Possible drug targets in collagen metabolism in intestinal fibrosis.
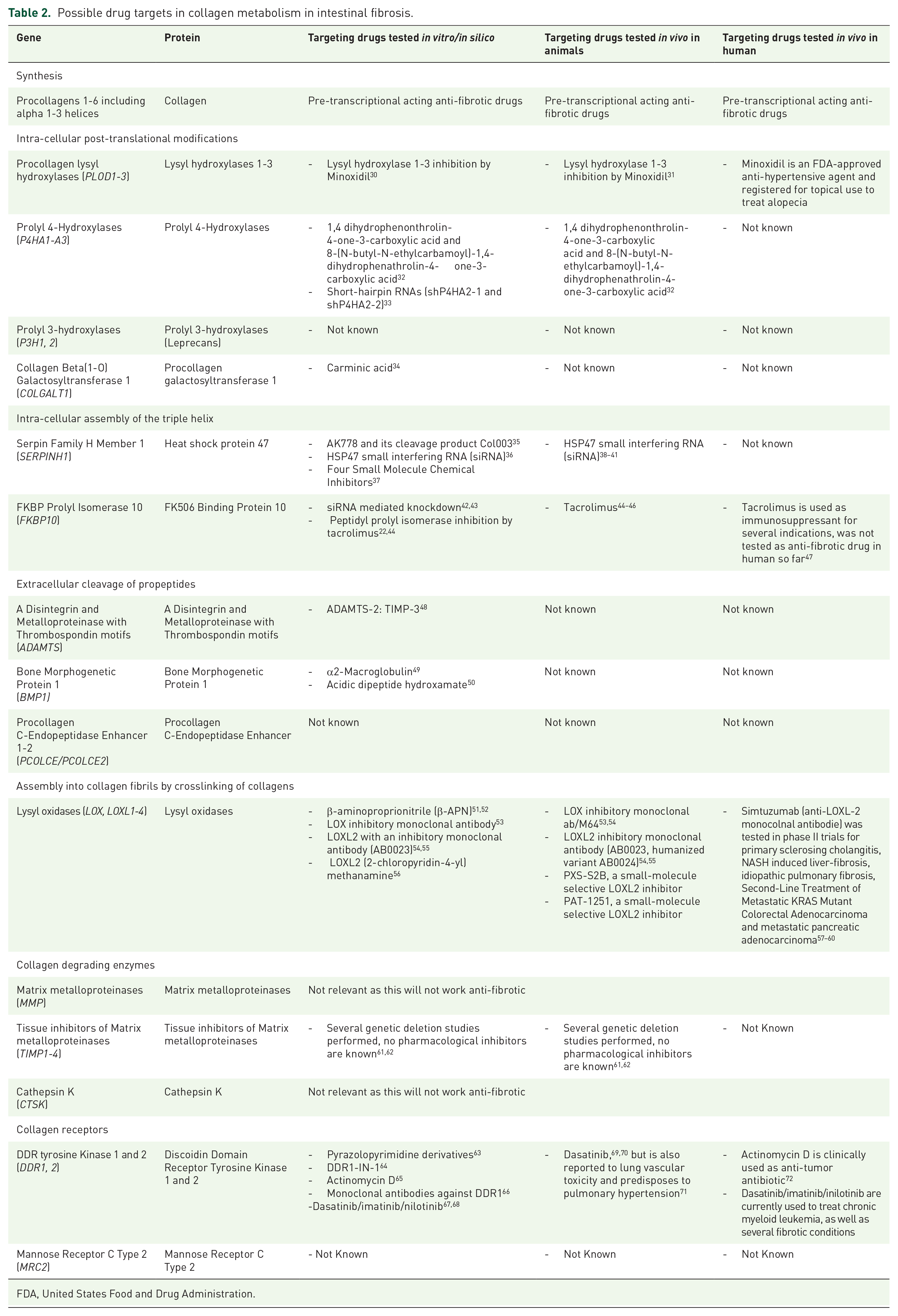
FDA, United States Food and Drug Administration.
Ethical considerations
Patients gave written informed consent for anonymous use of patient data and resected parts of human intestine according to the code of conduct for responsible use of surgical left-over material (See: “Code goed gebruik voor gecodeerd lichaamsmateriaal”, Research Code University Medical Center Groningen, http://www.rug.nl/umcg/research/documents/research-code-info-umcg-nl.pdf).
Results
Cohort characteristics
This descriptive cohort study included seven patients with fibro-stenotic CD who underwent ileocecal resection and four patients with adenocarcinoma who underwent right-sided hemicolectomy. All patients with CD had a stricturing disease phenotype and had either ileal [n = 4 (57.1%)] or ileocolonic [n = 3 (42.9%)] disease. On average, they were 33.6 years old (range 21.1–54.5) and had suffered from CD for 6.4 years (range 1.8–16.0). All patients had clinically active disease before they underwent ileocecal resection [moderate disease n = 3 (42.9%), severe disease n = 4 (57.1%)], and they used several different anti-inflammatory drugs before surgery (Table 1). As controls, patients who right-sided hemicolectomy due to adenocarcinoma were included at a mean age of 73.1 years (range 69.1–78.2). These patients were significantly older than the patients with CD (p = 0.008). All included patients with CD were female.
Expression of fibrosis markers is increased in macroscopically fibrosis-affected terminal ileum
Using a microfluidic card-based low-density TaqMan array, mRNA expression of a variety of ECM proteins were investigated. mRNA expression of procollagens type I, III, IV, V and IV was increased in fibrotic CD compared with non-fibrotic CD. Especially expression of collagen type I (COL1A1, 0.76 ± 0.28 versus 37.82 ± 49.85, p = 0.02) and III (COL3A1, 2.01 ± 2.61 versus 68.65 ± 84.07, p = 0.02) was increased in the fibrosis and the CD affected part compared with non-fibrotic CD (Figure 1A). Upregulation of these procollagens was considered as a positive control for the correct selection of the fibrosis-affected region. mRNA expression of other ECM proteins such as elastin (ELN, 0.01 ± 0.02 versus 0.07 ± 0.10, p = 0.03), FN1 (0.53 ± 0.43 versus 2.50 ± 10.52, p = 0.02), and BGN (0.1 ± 0.12 versus 2.83 ± 4.46, p = 0.03) was also increased in fibrotic CD compared with non-fibrotic CD (Figure 1B). mRNA expression of alpha-smooth muscle actin (generally considered as a marker for myofibroblasts), was also elevated in the fibrosis and CD-affected region compared with non-fibrotic CD (ACTA2, 1.03 ± 3.98 versus 16.57 ± 47.31, p = 0.02, Figure 1C).
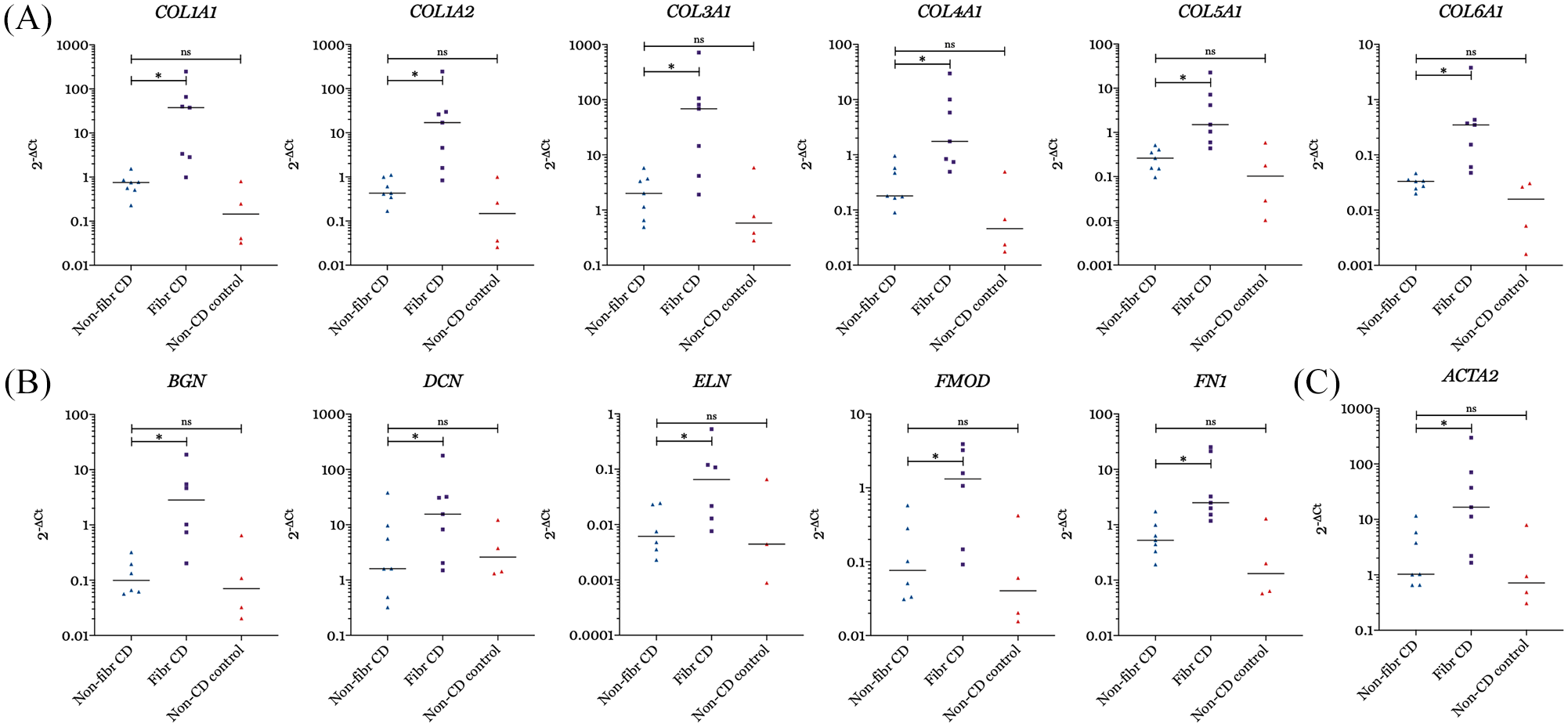
mRNA expression of procollagens 1–6 (A), of extracellular matrix molecules BGN, DCN, ELN, FMOD, FN1 (B), and of ACTA2 in fibrotic versus non-fibrotic terminal ileum of patients with CD, and versus non-CD control terminal ileum.
Expression of intra- and extra-cellular modification of collagen fibrils is increased in macroscopically fibrosis-affected terminal ileum
Enzymes involved in intracellular post-translational modification of the collagen fibril, were also upregulated in fibrotic CD compared with non-fibrotic CD. Expression of lysyl hydroxylases 1–3 (PLOD1, 0.05 ± 0.06 versus 0.40 ± 0.38, p = 0.02; PLOD2, 0.23 ± 0.13 versus 0.58 ± 0.69, p = 0.05; PLOD3, 0.06 ± 0.01 versus 0.29 ± 0.40, p = 0.02), prolyl 4-hydroxylases (P4HA1, 0.37 ± 0.43 versus 1.04 ± 2.10, p = 0.02); P4HB, 2.37 ± 3.72 versus 9.48 ± 14.54, p = 0.02) and prolyl-3-hydroxylases 1-3 (P3H1, 0.022 ± 0.037 versus 0.39 ± 0.54, p = 0.03; P3H2, 0.05 ± 0.27 versus 0.33 ± 1.77, p = 0.03; P3H3, 0.03 ± 0.16 versus 0.39 ± 0.91, p = 0.02) was increased in the fibrosis- and CD-affected region compared with non-fibrotic CD (Figure 2A–C). Expression of P4HA2 and P4HA3 was not detectable. Expression of chaperones HSP47 and FK506 binding protein 10 (SERPINH1, 0.06 ± 0.02 versus 0.68 ± 0.62, p = 0.03; FKBP10, 0.04 ± 0.07 versus 0.51 ± 0.35, p = 0.031) was also increased in fibrotic CD compared with non-fibrotic CD (Figure 2D). mRNA expression of the enzymes related to cleavage of the N- and C- terminal propeptides (ADAMTS2, 0.06 ± 0.08 versus 0.27 ± 0.38, p = 0.03; ADAMTS14, 0.01 ± 0.01 versus 0.01 ± 0.06, p = 0.06) and bone morphogenetic protein 1 (BMP1, 0.07 ± 0.05 versus 0.30 ± 1.26, p = 0.031) was increased in fibrotic CD compared with non-fibrotic CD. Also, the expression of collagen receptors discoidin domain receptor tyrosine kinase 2 (DDR2, 0.17 ± 0.09 versus 0.49 ± 1.72, p = 0.016) and of mannose receptor C type 2 (MRC2, 0.07 ± 0.06 versus 0.36 ± 0.80, p = 0.016) was increased in the fibrosis- and CD-affected tissue compared with non-fibrotic CD.
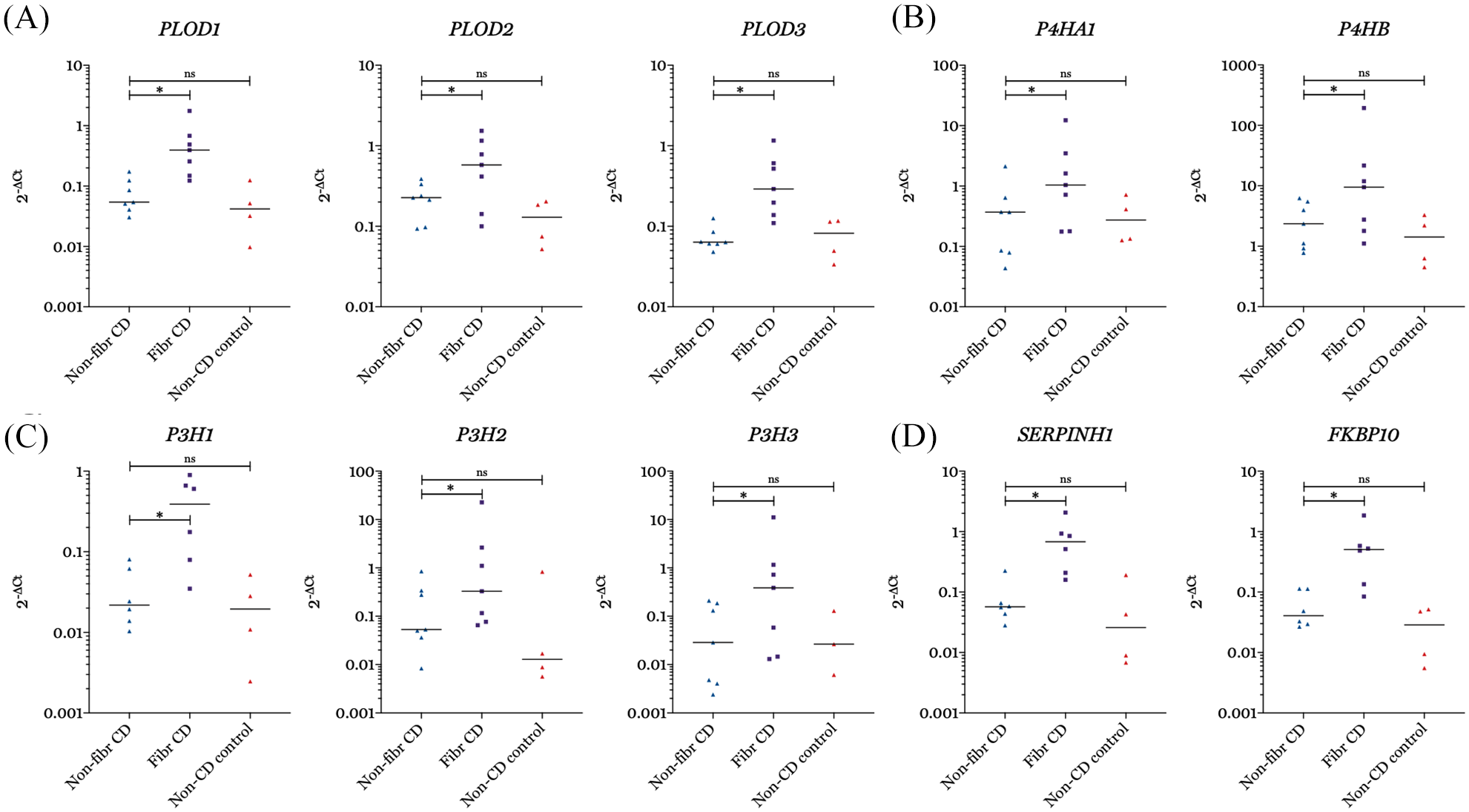
mRNA expression of lysyl hydroxylases 1–3 (PLOD1–3) (A), of prolyl 4-hydroxylases (P4HA1 and P4HB) (B), of prolyl 3-hydroxylases (P3H1, P3H2, P3H3) (C), and of heat shock protein 47 (SERPINH1) and of FK506 binding protein 10 (FKBP10) (D) in fibrotic versus non-fibrotic terminal ileum of patients with CD, and versus non-CD control terminal ileum.
Expression of MMPs and their tissue inhibitors is increased in macroscopically fibrosis-affected terminal ileum
Net deposition of collagen depends on the balance between formation and degradation. Collagens are degraded by matrix-metalloproteinases, which are inhibited by tissue inhibitors of MMPs. MMP1 (0.02 ± 0.24 versus 1.22 ± 0.88, p = 0.031) and MMP14 (0.16 ± 0.21 versus 1.51 ± 2.95, p = 0.031) was upregulated in fibrotic CD compared with non-fibrotic CD, as well as TIMP1 (0.85 ± 3.17 versus 22.78 ± 47.61, p = 0.016).
No differences in expression of the genes described in this study involved in collagen metabolism was observed between non-CD non-fibrotic control tissue and non-fibrotic CD (Figures 1–4 and Supplemental Table S1).

mRNA expression of ADAMTS2, ADAMTS14 and BMP1 (A), PCOLCE2 (B), and LOXs LOXL1–4 (C) in fibrotic versus non-fibrotic terminal ileum of patients with CD, and versus non-CD control terminal ileum.

mRNA expression of collagen degrading enzymes MMPs MMP1, MMP14, and CTSK (A), and of TIMP1 (B) in fibrotic versus non-fibrotic terminal ileum of patients with CD, and versus non-CD control terminal ileum.
Discussion
To our knowledge, this is the first study examining the expression of genes coding for enzymes involved in the metabolism (such as post-translational modifications) of collagens in intestinal fibrosis in CD. We aimed to reveal a gene signature with potential targets for drugs against intestinal fibrosis based on gene expression of CD-affected fibrotic versus non-fibrotic terminal ileum from the same patient using a microfluidic card-based low-density TaqMan array. A literature search revealed several drugs that interfere with the metabolism of collagens that could be candidates for drug treatment against intestinal fibrosis (Table 2). Some of these drugs are already used clinically for other (fibrotic) conditions, whereas some have been tested only either in vivo in animals or in vitro in cell culture models. Others have been tested only in biochemical models such as binding assays to determine biochemical half-maximal binding concentrations or were identified using screening technologies for small molecule discovery. Outside of the scope of this study, comprehensive reviews and studies are available on pharmacological inhibition of (pre-transcriptional/translational) pathways leading to a lowered production of ECM.73,74
The expression of enzymes involved in the intracellular and extracellular post-translational modifications of collagens (see Table 2) has never been described for intestinal fibrosis in CD. However, expression of several collagens and post-translational modulators of collagens was assessed in colorectal-cancer-associated fibrosis using comparative liquid chromatography with mass spectrometry. 75 In a study by Afik et al., PLOD1-3 and P4HA1 protein expression was upregulated in colorectal cancer-associated fibrosis compared with more distal non-fibrosis-affected colon tissue, which is in line with our results. This study also reports on increased protein expression of BGN and FN1 in colorectal cancer-associated fibrosis, which is in line with our results. 75 Upregulation of PLOD1–3 mRNA was also observed in fibrotic conditions such as idiopathic pulmonary fibrosis. 31 Furthermore, upregulation of LOXL-2 has been reported in renal fibrosis and inhibition of LOXL-2 by several inhibitors in mice in vivo successfully reduced the expression of fibrosis markers in several studies.76,77 A relative increase in protein expression of collagen type III over type I in fibrostenotic CD compared with inflamed or non-disease affected intestinal tissue reported previously was not observed in this cohort at the mRNA level.78,79
Intracellular post-translational modifications
Pharmacological inhibition of the intracellular post-translational modifications (lysyl hydroxylase, prolyl-3 and -4 hydroxylase and glycosyltransferase, Table 2) could inhibit collagen formation and thereby fibrosis, but these enzymes are pivotal to human physiology. Therefore, the inhibition should be very enzyme specific and ideally be targeted to the fibrosis-affected area in order to be effective without causing severe side effects. The importance of these genes is confirmed by the fact that genetic mutations in the genes coding for these enzymes (resulting in aberrant synthesis, degradation and/or modification) can lead to syndromic disorders in musculoskeletal or connective tissues such as osteogenesis imperfecta type VIII (mutation in P3H1), Bruck syndrome type 2 (mutation in PLOD2 resulting in a form of osteogenesis imperfecta), Ehlers-Danlos syndrome type VIA (mutation in PLOD1), and severe myopia in the absence of musculoskeletal abnormalities (P3H2). 29 No mutations are found in genes coding for the P4HA a-subunits, perhaps indicating that loss of PH4 enzyme function leads to premature death of the embryo. 29 Pharmacological inhibition of lysyl hydroxylases 1-3 by minoxidil upon stimulation with transforming growth factor beta (TGF-β) was tested in vitro in primary human fibroblasts by Zuurmond et al. Even though a concentration-and time-dependent reduction in LH1-3 mRNA was observed, no effect on the total number of pyridinoline cross-links in the collagen matrix was observed, and the authors therefore conclude that minoxidil is unlikely to be anti-fibrotic in these concentrations. 30 Shao et al. did observe an anti-fibrotic effect of minoxidil in mice in bleomycin-induced pulmonary fibrosis without reporting side effects in these animals. 31 The side effects of minodoxil when used systemically in humans are, however, severe, which makes further testing unattractive. However, specific inhibition of lysyl hydroxylase-2 (PLOD-2) could be attractive as reduction of the pyridinoline cross-links between collagen molecules facilitates easier degradation by endogenous proteinases (i.e., MMPs and cathepsin K). This because cross-linked collagen can be degraded effectively only by MMP-13 and cathepsin K, in contrast to non-crosslinked collagen, which can be degraded by several MMPs. 29 Inhibition of another hydroxylase, P4H, was tested by administering two small-molecular inhibitors (both Phenanthrolinones, see Table 2) to rats. 32 These compounds were well tolerated in the rat at doses producing sustained inhibition of collagen hydroxylation. Procollagen molecules which are less hydroxylated will accumulate in the endoplasmatic reticulum (ER), thereby causing ER-stress, which triggers ER-stress-mediated apoptosis of myofibroblasts.80,81 However, P4H inhibition would be effective as an anti-fibrotic target only if the inhibition is constantly maintained, as collagen will be rapidly hydroxylated after P4H activity is restored. 32
Intracellular assembly of the triple helix
Inhibition of the collagen chaperone HSP47 and the FK506 binding protein 10 has promising anti-fibrotic potential. HSP47 expression is upregulated in intestinal fibrosis.23,82–84 The anti-fibrotic potential of inhibition of HSP47 is shown by the deletion of Hsp47 in hepatic stellate cells isolated from Cre-LoxP system Hsp47 floxed mice, which led to ER stress-mediated apoptosis of the collagen-producing cells. 81 Another study shows that local (submesothelial) delivery of Hsp47 siRNA conjugated with cationized gelatin microspheres could suppresses peritoneal fibrosis in mice. 38 The use of microRNA and small siRNA for several indications in vivo in humans has progressed to several phase II and III clinical trials. 85 The activity of the other collagen chaperone FK506 binding protein 10 (peptidyl prolyl isomerase) can be inhibited by the widely used immunosuppressive tacrolimus. Next to its immunosuppressive properties, tacrolimus can inhibit the chaperone activity of FK506 binding protein 10, and is thereby proposed to have anti-fibrotic properties as well. Results from human embryonic kidney 293-cells and normal human dermal fibroblasts indicate that FK506 binding protein 10 peptidyl prolyl isomerase inhibition activity (which can be inhibited by tacrolimus) is linked to pyridinoline cross-linking by specifically mediating the dimerization of LH2. 22 Thereby, tacrolimus not only inhibits the collagen chaperone activity of FK506 binding protein 10, but also decreases the number of pyridinoline cross-links, making the produced collagen more easily degradable by MMPs. Animal studies showed that tacrolimus can prevent alcohol- or carbon tetrachloride (CCl4)-induced liver fibrosis in rats by inhibiting synthesis of type I collagen polypeptides, without affecting expression of collagen mRNAs. 45 Results are, however, conflicting since both Patsenker et al. (liver fibrosis induced by CCl4 and bile duct ligation) and Frizell et al. (liver fibrosis induced by CCl4) showed that tacrolimus was not able to inhibit fibrosis formation, but even enhanced fibrogenesis in the liver.46,86 The clinical experience with tacrolimus for CD is limited, but remission rates of 44% (range, 7–69%) and response rates of 37% (range, 14–57%) are reported for luminal CD. 87 No studies were found investigating the incidence of stricturing Crohn’s disease in patients receiving tacrolimus versus other immunosuppressives. Cohort studies from kidney transplant recipients show that tacrolimus as the current standard calcineurin inhibitor therapy post-renal transplantation, is not superior in preventing progression to interstitial fibrosis compared with cyclosporin or sirolimus. 88
Extracellular cleavage of propeptides
Inhibition of the cleavage of C- and N- terminal propeptides could have anti-fibrotic potential as well since it could inhibit the formation of an irreversibly stable collagenous ECM, thereby making it more easily degradable by collagenases that are able to degrade cross-linked collagen. 48 In vitro studies have shown that TIMP-3 can inhibit the procollagen N-proteinase activity of ADAMTS-2, thereby inhibiting procollagen processing in mouse embryonic fibroblasts. Upon stimulation with TIMP-3, reduced amounts of mature α1(I) chains were observed. 48 Administration of TIMP-3 in vivo in a model for fibrosis has not been performed so far. Inhibition of bone morphogenic protein-1 proteinase activity (BMP-1) was tested only in vitro using (modified forms of) α2-macroglobulin and acidic dipeptide hydroxamate. Also, these compounds have not been tested in in vivo models of organ fibrosis. Therapeutic inhibition of procollagen C-endopeptidase enhancer (PCOLCE; an enhancer of BMP-1 activity in vitro and in vivo), might have the same effect as therapeutic inhibition of BMP-1. In chronic-pressure-overload-induced cardiac fibrosis, PCOLCE2-null hearts demonstrated a decreased collagen content and a lower muscle stiffness compared with wildtype chronic pressure overloaded hearts. 89
Assembly into collagen fibrils by crosslinking of collagens
The general hypothesis is that targeting extracellular cross-linking by lysyl oxidases [including lysyl oxidase-like (LOXL)] might cause an increase in net degradation of collagen and other ECM molecules, thereby resulting in less fibrosis. Furthermore, it is generally hypothesized and proven in animal studies that inhibition of LOX or LOXL (e.g. anti-LOX or -lysyl oxidase-like 2 antibodies) decreases tumor stiffness and suppresses metastasis.53,90 In vivo studies on liver fibrosis showed that LOXL2 mediates collagen crosslinking and fibrotic matrix stabilization during liver fibrosis, and independently promotes fibrogenic differentiation of hepatic progenitor cells. By blocking these two convergent profibrotic pathways, therapeutic LOXL2 inhibition attenuates both parenchymal and biliary fibrosis and promotes fibrosis reversal. 91 However, even though inhibition of LOX(L) was very promising in pre-clinical in vitro and in vivo studies, several phase II studies testing the effect of the LOXL2 monoclonal antibody simtuzumab did not show an anti-fibrotic or antimetastatic effect.57–60 This study is the first to show upregulation of LOX and LOXL1-4 in CD-associated fibrosis in the terminal ileum. In vivo studies using animal models for intestinal fibrosis with LOX(L) knock-out animals or LOX(L) inhibitors, have not yet been performed.
Collagen-degrading enzymes
Based on their function as collagenases, inhibition of MMPs or CTSK is not expected to have anti-fibrotic efficacy. However, mRNA and protein levels of MMPs and CTSK are known to be upregulated in IBD, and it is known that these enzymes have other, for example chemotactic, properties as well.24,26 Therefore, inhibition of MMPs or CTSK might have an (indirect) anti-fibrotic effect. Especially MMP-9 is suggested to play a role in intestinal fibrosis and fistulae formation. Inhibition of MMP-9 in a heterotopic transplant model for intestinal fibrosis did reduce collagen content of the intestinal graft after induction of fibrosis by transplantation. 92 Furthermore, expression of MMP-9 is a marker of mucosal healing, and increased local or serologic expression of MMP-9 is related to penetrating CD.92–94 Because inhibition of MMP-9 could simultaneously inhibit degradation of ECM and reduce fistulae associated fibrosis, this therapy may be superior for stricturing or penetrating CD compared with current immunosuppressive agents.
The importance of Mmp-9 in intestinal inflammation in mice was recently questioned. 95 Very well-controlled studies comparing intestinal (colonic) inflammation induced by dextran sodium sulfate (DSS, both acute and chronic) and 2,4,6-trinitrobenzenesulfonic acid (TNBS) between Mmp-9 knockout and wildtype mice, did not show a difference in the degree of intestinal inflammation or fibrosis induced. Inhibition of MMP-9 with bio-active peptides did not improve DSS-induced colitis, but the effect of inhibition of MMP-9 on intestinal fibrosis was not tested. De Bruyn et al. suggest that upregulation of MMP-9 is a consequence rather than a cause of inflammation of the colon, and question whether MMP-9 represents a disease target in IBD. 95 Differences in the pathophysiology of fibrosis between colon and (terminal) ileum, and pathophysiological differences between the models used by de Bruyn et al. and Goffin et al. allow further testing of MMP-9 inhibitors for stricturing and penetrating CD.92,95
Several MMP inhibitors are being tested in clinical trials as it was hypothesized that metastasis of cancer could be reduced by inhibition of MMP-mediated degradation of tumor-associated fibrosis, thereby reducing cancer progression. 96 Due to a lack of inhibitory specificity, and insufficient knowledge about the pleiotropic substrates and opposing effects of MMPs (on tumor growth/angiogenesis/modulation of immune response), these trials have all failed. 96 Especially since ECM remodeling is believed to be part of the pathophysiology of IBD, further testing of inhibitors of (other) MMPs still holds promise. 97
Whether inhibition of the TIMP-1 would have anti-fibrotic properties by reducing TIMP-mediated inhibition of MMP-activity remains to be studied. In a model of obstructive nephropathy-induced interstitial fibrosis, no amelioration of renal fibrosis was observed in Timp-1 knockout mice. 62
Collagen receptors
Similar to other tyrosine kinase receptors, DDR kinase receptors (DDR-1 and -2) regulate fundamental cellular processes such as adhesion, migration, proliferation, and differentiation. Furthermore, they influence ECM remodeling via activation of MMPs. 98 DDR1 is found mostly in epithelial cells, whereas DDR2 is confined to cells of mesenchymal origin. 98 Inhibition of DDRs by tyrosine kinase inhibitors (dasatinib/imatinib/inilotinib, which are currently clinically used to treat chronic myeloid leukemia) has anti-fibrotic potential. 67 A significant decrease in collagen deposition of injured arteries of DDR1-null mice was observed. 99 Furthermore, type I collagen-dependent upregulation of DDR2 expression in hepatic stellate cells (HSC) establishes a positive feedback loop in activated stellate cells, leading to further proliferation and enhanced invasive activity of HSC. 100
A limitation of this study is that number of patients included is small; therefore, this study was not powered to reveal differences in, for example, the expression of fibrosis genes depending on the therapy patients were using. However, the validity of the data increases due to the fact that the samples from non-fibrotic CD versus fibrotic CD are from the same patient and are therefore paired. Ideally, expression of the detected fibrosis genes should be validated in a replication cohort. Another limitation of this study is that mechanisms by which inhibition of post-translational modification and processing of collagens might attenuate the development of fibrosis in CD patients are only proposed. Mechanistic in vitro/in vivo data proving that inhibition of post-translational modification of collagens can inhibit fibrosis formation using one of the proposed mechanisms is not provided and was not the aim of this study. Efficacy for intestinal fibrosis and the underlying mechanisms will be explored in future research using, for example, cell culture of intestinal fibroblasts, precision-cut intestinal slices, or a heterotopic transplant animal model of intestinal fibrosis.101–103
Conclusion
Therapeutic inhibition of fibrosis for patients with CD is not yet possible, but several drugs acting on factors involved in both pre- and post-transcriptional regulation of deposition of collagens and other extracellular matrix molecules are available. Which compound has the highest potential will depend on a combination of safety and anti-fibrotic efficacy. A drug against intestinal fibrosis would ideally be targeted to an enzyme/receptor that is uniquely expressed in a fibro-stenotic intestine in order to minimize systemic side effects. Since fibrosis formation occurs over a long period of time, clinical trials of long duration, a large number of patients, and selecting intestinal fibrosis-relevant endpoints, are warranted.73,104 Furthermore, the best route of application of the drug should be determined. This might be intravenous, but could also be oral for example, making use of sustained release by the ColoPulse technology (film coated tablets of targeted delivery in the lower intestinal tract) or topical administration for more distal (radiation-induced) fibrosis.105,106 Moreover, sustained release from the staple of a stapled anastomosis might be optional. However, meta-analysis of CD patients post-strictureplasty showed that (with an overall 5-year surgical recurrence rate of 28%), recurrence occurs mainly at non-strictureplasty sites in 90% of patients (whereas the site-specific recurrence rate was 3%). 107 Furthermore, caution should be taken with anti-fibrotic therapy, because disturbance of the balance between collagen formation and degradation may induce a shift towards degradation to such an extent that fistulae or abscesses can occur. The anti-fibrotic capacity of some of the reviewed drugs for fibro-stenotic CD could be further unraveled, and their efficacy can be tested in in vitro and in vivo models for (CD-associated) intestinal fibrosis. In conclusion, inhibition of post-translational modification of collagens might be suitable to inhibit fibrosis formation in the intestine in CD.
Supplemental Material
Supplementary_data_WTvH_collagens_intestinal_fibrosis_TAIGdocx – Supplemental material for Intestinal stenosis in Crohn’s disease shows a generalized upregulation of genes involved in collagen metabolism and recognition that could serve as novel anti-fibrotic drug targets
Supplemental material, Supplementary_data_WTvH_collagens_intestinal_fibrosis_TAIGdocx for Intestinal stenosis in Crohn’s disease shows a generalized upregulation of genes involved in collagen metabolism and recognition that could serve as novel anti-fibrotic drug targets by Wouter Tobias van Haaften, Tjasso Blokzijl, Hendrik Sijbrand Hofker, Peter Olinga, Gerard Dijkstra, Ruud A. Bank and Miriam Boersema in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to thank the students of the Prometheus Kidney team of the Department of Surgery of the University Medical Center Groningen for collecting the surgical resection material from patients with CD and healthy controls.
Author contributions
WTvH: sample and data collection, analysis of the data, drafting of the manuscript. TB: sample and data collection, HS: sample collection. PO: critically reviewing the manuscript, GD: sample collection, critically reviewing the draft, critically reviewing the manuscript. RAB: data collection, critically reviewing the manuscript. MB: sample and data collection, analysis of the data, critically reviewing the manuscript.
Conflict of interest statement
WTvH has received funding to print his thesis from Ferring b.v., Teva b.v., Tramedico b.v. and Mylan b.v. GD reports outside the submitted work grants from Takeda and Abbvie, Fees for advisory boards from Cosmopharma and Mundipharma, speakers fees from Pfizer, Janssen pharmaceutical and Takeda.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
