Abstract
Background:
The only effective treatment for celiac disease (CeD) is gluten free diet (GFD). However, GFD is restrictive and efforts are being made to explore alternative therapies including safer wheat varieties. Wheat variety C273 has been previously identified to have reduced load of intact T-cell stimulatory epitopes via in silico and in vitro analysis.
Methods:
Adult patients diagnosed with CeD and recovered on GFD were included in the study. Patients were randomised into two groups in a 2:1 ratio. Patients in group I had graded introduction of C273 wheat in diet, maintained for 24 weeks; in Group II, wheat was restricted with continuation of GFD. Clinical symptoms, serology [anti-tissue transglutaminase (anti-tTG), anti-endomysial antibody (anti-EMA)], circulating inflammatory biomarkers [intestinal fatty-acid binding protein (I-FABP), plasma citrulline, interferon-γ (IFN-γ)] and histology were evaluated periodically. Final evaluation was performed at week 28.
Results:
A total of 15 patients were enrolled (Group I: n = 10, Group II: n = 5). All patients except two in Group I remained compliant. None of the remaining eight patients in group I developed symptoms. No significant changes in serology (anti-tTG, anti-EMA) and histology were observed between the two groups at 28 weeks (p > 0.05). Significant changes in plasma citrulline(29.87 ± 8.98 versus 36.58 ± 3.09, p = 0.049) and IFN-γ (44.56 ± 9.74 versus 33.50 ± 3.68; p = 0.031) were observed in Group I.
Conclusion:
Consumption of C273 wheat did not result in development of symptoms or evident changes in serology and histology at 28 weeks. However, variations in circulating inflammatory markers were noted. Larger randomised trials are needed to corroborate these findings.
Clinical Trials Registry-India:
CTRI/2018/06/014521.
Introduction
Celiac disease (CeD) is an autoimmune gluten sensitive enteropathy caused by an inflammatory response to gluten (storage proteins found in the starchy endosperm of many cereals including wheat, barley and rye) in genetically predisposed individuals. 1 Gluten refers to a family of proteins called prolamins and is composed of a complex group of alcohol-soluble gliadins and glutenins that are resistant to proteolytic enzymes. 2 Upon deamidation by tissue transglutaminase (tTG), high affinity complexes are formed between deamidated gluten peptides and human leukocyte antigen (HLA) DQ2/8 present on antigen presenting cells (APCs). 3 The resultant T-cell activation produces diverse inflammatory cytokines [including interferon gamma (IFN-γ) and tumour necrosis factor alpha (TNF-α)] culminating in damage to intestinal mucosal integrity and functionality.4–6
The only definite treatment for CeD is strict exclusion of gluten from diet. However, cost and palatability of gluten free diet (GFD, defined as foods containing less than 20 parts per million of gluten or 20 mg/kg), 7 the presence of traces of gluten in many unlabelled food products and the psychological effects of eliminating gluten from diet, especially in wheat-consuming regions, makes practical application of GFD an onerous task. With an increasing understanding of molecular pathogenesis of CeD, several new therapeutic approaches targeting the pathophysiological mechanisms of CeD; including glutenases, gluten sequestrators, gut barrier modulators and tTG inhibitors, etc., have been evaluated in clinical trials.8–21 One of the novel approaches to treat CeD is to target the genetic diversity of α-gliadin genes encoding the T-cell stimulatory epitopes (Glia-α9, Glia-α20, Glia-α2 and Glia-α). We had, in our previous work, screened 43 Indian wheat genotypes by cloning their α-gliadin genes in an in silico analysis, and studied their CeD eliciting epitopes. After phylogenetic analysis, three varieties (C273, C591 and K78) were found to have a reduced load of intact T-cell stimulatory epitopes. 22 We then tested these three varieties in vitro for their antigenic peptides using T-cells (derived from duodenal mucosal biopsies) and peripheral blood monocytes (PBMCs) isolated from patients with CeD in remission (on GFD) and reported C273 variety as the least immunogenic. 23 The present randomized pilot trial appraises the innocuous nature of the wheat variety C273 through an in vivo exposure in patients with CeD in remission.
Methods
This was an open-label, randomised pilot study conducted in the Department of Gastroenterology, Dayanand Medical College and Hospital, Ludhiana, India between August 2017 and July 2018. The study was approved by Institutional Ethics Committee (ECR/715/Inst/PB/2015) and was registered with Clinical Trials Registry-India (CTRI/2018/06/014521). Written informed consent was obtained from all the patients.
Study population
HLA DQ2/8 positive adult (aged ⩾18 years) patients diagnosed with CeD [based on elevated serology (anti-tTG) and evidence of Marsh-Oberhuber 3 histology on duodenal biopsy] and on GFD for ⩾2 years, showing complete recovery [defined as absence of CeD symptoms; normal anti-tTG (IgA), negative anti-EMA (qualitative) and normal duodenal histology (Marsh-Oberhuber zero)] were enrolled.
Patients on GFD for <2 years, incomplete recovery [persistence of symptoms, elevated anti-tTG(IgA)/positive anti-EMA (qualitative) and/or evidence of histological activity], poor compliance to GFD and co-morbid conditions (inflammatory bowel disease, chronic liver disease, end stage renal disease, type 1 and type 2 diabetes mellitus, coronary artery disease, chronic obstructive pulmonary disease, psychiatric illness), immunocompromised state (human immunodeficiency virus infection, malignancy, history of intake of immunosuppressants) and pregnancy were excluded.
Randomisation and intervention
This was an open-label randomised pilot study. Consenting patients were divided into two groups according to computer based 2:1 randomisation:
Group I: Wheat allowed (only from C273 variety)
Group II: Wheat restricted (only GFD).
Wheat flour from preselected variety C273 was gradually introduced in the diet of the patients in Group I in an incremental fashion. Wheat variety C273 was grown in Punjab Agricultural University, Ludhiana, India, following standard cultivation practices in a controlled environment to prevent cross-contamination. Wheat grains were milled in a cabinet flour mill and sieved through a mesh (0.5 mm). Each patient was provided with a weekly pre-weighed measure of wheat flour to ensure compliance to the wheat variety C273 as well as to control the size of flatbread. The flatbread was prepared on a domiciliary basis by the patient/family member. In the first week, a single 20 g piece of flatbread (containing approximately 2 g of gluten) per day was introduced. Gradual increments in consumption of wheat were done over the next 3 weeks, after which full gluten exposure consisting of 80 g of flatbread (four pieces, gluten content 8 g/day) was continued for 24 weeks. (Figure 1) Patients in group II were continued on the same GFD as previously and no change was allowed after screening and enrolment.

Schematic representation of intervention in group I.
Weekly evaluations of dietary adherence to wheat variety C273 (including identification of ‘missed’ doses) in group I and GFD in group II were done using a patient-filled diet diary, and any deviations from the protocol were recorded.
Baseline evaluation
Clinical symptoms
The patients were asked about the presence of (gastrointestinal or non-gastro-intestinal) symptoms such as diarrhoea, abdominal distension, bloating, loss of appetite, abdominal pain, bony pains, anaemia, infertility, neurological symptoms, dermatological manifestations and weight loss. Any patient who had symptoms suggestive of active CeD was excluded.
Laboratory investigations
Haemoglobin; UniCelDxH 900 Coulter Cellular Analysis System (Beckman Coulter Ltd., High Wycombe, UK)
Anti-TTG (IgA); ELISA (Cat No. 3508, Aesku Diagnostics, Wendelsheim, Germany; Normal: <12 U/ml, Equivocal: 12–18 U/ml, Positive: >18 U/ml).
Anti-EMA (Qualitative); Indirect immunofluorescence (Cat No. 512.050, Aesku Diagnostics) with a primary dilution of 1:5.
Circulatory immune-biomarkers: We measured intestinal type fatty acid binding protein (I-FABP, detected in circulation on loss of enterocyte mass), 24 plasma citrulline (a non-protein amino acid synthesized by the enterocytes that predicts the severity of villous atrophy) 25 and interferon γ (IFN-γ, a pro-inflammatory cytokine produced by the interaction of gliadin fragments with T-cell receptors and has increased expression in patients with CeD with mucosal injury) to identify mucosal injury associated with consumption of gluten. 26 I-FABP (RayBiotech, Norcross, GA, USA), plasma citrulline (DRG, Marburg, Germany) and IFN-γ (RayBiotech), were measured by ELISA in the plasma. All the samples were tested in duplicate and read at 450 nm with a correction wavelength of 620 nm. For each protein, standard curves were established to quantify the levels in culture supernatants (R2 = 0.9937 for IFN-γ, R2 = 0.9933 for I-FABP and R2 = 0.9953 for citrulline). The assay sensitivity for IFN-γ and I-FABP were 15 pg/ml and 25 pg/ml, respectively. Plasma citrulline level ⩽10 μmol/l was taken as threshold for total villous atrophy. 25
Duodenal biopsy: All the enrolled patients underwent esophago-gastro-duodenoscopy (EGD), performed by a single blinded expert gastroenterologist (RM) using Olympus gastroduodenoscope (GIF-H170, Tokyo, Japan). A video was recorded maintaining the anonymity of the patient and a blinded second expert (AS) validated the findings after reviewing the video (percentage of agreement between the two experts: 97.02%, Cohen’s k: 0.87). Six biopsies (four from each quadrant in the second part of duodenum and two from the duodenal bulb) were collected in 10% formalin. The biopsy specimens were stained with haematoxylin and eosin and examined under 100× magnification by a single blinded expert pathologist (VN) and the observations were recorded. These findings were then reviewed by a blinded second expert (NS) and confirmed (percentage of agreement: 93.75%, Cohen’s k: 0.81). The morphometric analysis of the sections was performed on >10 well-oriented villi, and crypts, by a computerised image analyser and villous height to crypt depth (vh/cd) ratio and intraepithelial lymphocyte (IEL) count were calculated. Immune-staining using anti-human CD3+ antibody was done to aid the counting of IELs. Marsh-Oberhuber classification was used to report the histological findings. 27
Quantitative real-time reverse transcription polymerase chain reaction (RT-PCR): Total RNA was extracted from paraffin-embedded duodenal biopsy tissue specimens using Purelink® FFPE RNA Isolation Kit (Life Technologies, New York, NY, USA) and reverse-transcribed using SuperScript VILO cDNA Synthesis Master Mix (Invitrogen, New York, NY, USA). A real-time quantitative PCR, using human primer sequences for genes encoding IFN-γ (forward read – TGGGTCTGTCTCATCGTCAA, reverse read – TCCTCTGGCTGCTGGTATTT) and I-FABP-2 (forward read – CTGGAGCCTTGAGGGAAA, reverse read – GGACTGTGCGCCAAGAATAA), was used to quantify gene expressions (GoTaq® qPCR Master Mix, Promega, Madison, WI, USA) using the Bio-Rad 96 Real-Time System. Differences in the threshold cycle (ΔCt) number between the target genes and TATA-box binding protein (TBP) (forward read – CAGCGTGACTGTGAGTTGCT, reverse read – AACTGTGGTTCATGGGGAAA) were used to normalise the expression.
Follow up
All the patients in group I were followed up weekly for disbursement of the milled wheat flour, assessment of clinical symptoms and checking of diet diary for any potential deviations or non-compliance. Serology [anti-TTG(IgA), anti-EMA(IgA)], haemoglobin and circulatory inflammatory markers (IFN-γ, I-FABP and plasma citrulline) were performed at weeks 2, 6, 14, 22 and 28 in both groups I and II. Endoscopic duodenal biopsies were taken at week 28.
Statistical analysis
The data were described in terms of frequencies, relative frequencies, range, mean ± standard deviation (SD) and median, as appropriate. A comparison of quantitative variables between the groups was done using student t-test and paired t-tests for parametric data whereas Mann-Whitney U test and Wilcoxon test were used for non-parametric data. For comparing categorical data, Chi-square test was performed and exact test was used when the expected frequency was <5. Probability value (p-value) <0.05 was considered statistically significant. All statistical calculations for were done using Stata version 12 (Stata Corp LLC, College Station, TX, USA).
Results
A total of 52 patients with CeD on strict GFD for ⩾2 years were screened between August 2017 and October 2017. Of the 18 eligible patients meeting the inclusion criteria, 3 did not consent to participate and 15 patients were enrolled and randomised into two groups in a 2:1 ratio. Beginning November 2017, patients in Group I (n = 10) were subjected to a graded gluten exposure with wheat variety C273, and patients in Group II (n = 5) continued GFD (Figure 2). Baseline characteristics of the patients in two groups at the time of enrolment are shown in Table 1.
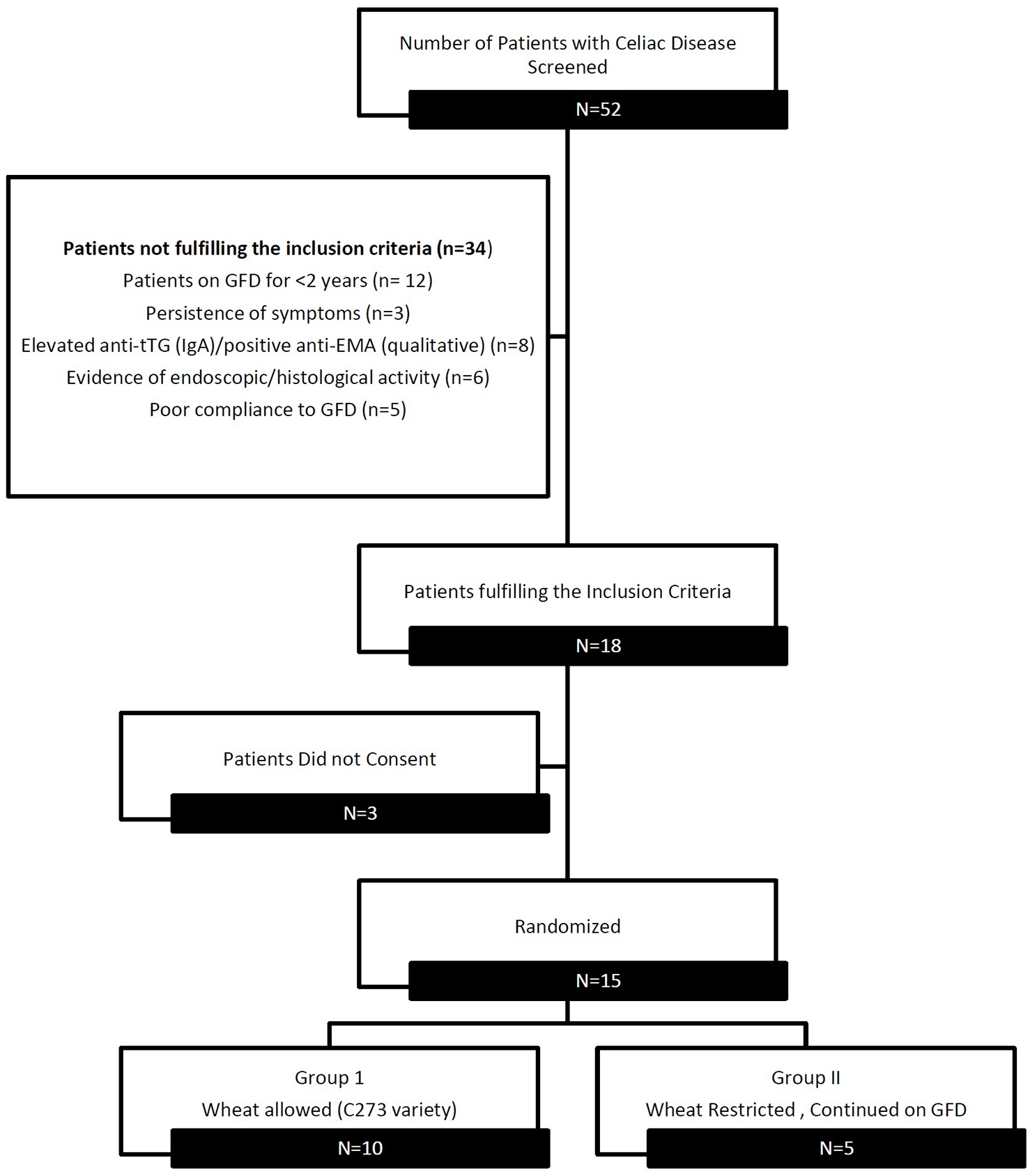
CONSORT diagram showing flow of patients.
Baseline characteristics of enrolled patients.

anti-EMA, anti-endomysial antibody; anti-tTG, anti-tissue transglutaminase; BMI, body mass index; GFD, gluten-free diet; HLA, human leukocyte antigen; I-FABP, intestinal fatty-acid binding protein; IFN-γ, interferon gamma; SD, standard deviation.
Intention-to-treat analysis
In the intention-to-treat (ITT analysis, out of 10 patients in group I, 2 defaulted (non-compliant) and consumed other varieties of wheat available in the market. These two patients developed diarrhoea and arthralgia, respectively. None of the remaining eight patients in group I (compliant) on wheat variety C273 experienced worsening of clinical symptoms. No significant differences were noted in mean anti-tTG values between groups I and II at the end of study period (31.45 ± 66.40 U/ml in group I versus 7.20 ± 4.09 U/ml in group II, p = 0.17). There were significant differences in IFN-γ (44.56 ± 9.7.4 pg/ml versus 33.50 ± 3.68 pg/ml, p = 0.031) and plasma citrulline (29.87 ± 8.98 μmol/l versus 36.58 ± 3.09 μmol/l, p = 0.049) between the two groups at week 28. I-FABP was also higher in group I (484.45 ± 173.60 pg/ml versus 448.62 ± 53.63 pg/ml in group II) though it failed to achieve significance (p = 0.103). No differences were observed between histology in the two groups at week 28 (Figure 3, Table 2).

Concentration of anti-tTG, IFN-γ, I-FABP and plasma citrulline in group I and group II at baseline and during follow-up visits (ITT analysis). (Units : Anti-tTG in U/ml, I-FABP and IFN-γ in pg/ml and Citrulline in μmol/l)
Between group outcome analysis.

anti-EMA, anti-endomysial antibody; anti-tTG, anti-tissue transglutaminase; ITT, intention-to-treat; I-FABP, intestinal fatty-acid binding protein; IFN-γ, interferon gamma; PP, per protocol; SD, standard deviation.
The changes in mean anti-tTG, IFN-γ and I-FABP levels were higher than baseline within group I as compared to group II, though these were statistically insignificant. Plasma citrulline levels, however, declined significantly within group I (36.49 ± 1.43 at week 0 versus 29.87 ± 8.98 at week 29, p = 0.047), whereas the levels increased within group II (Table 3). Two patients who defaulted and consumed other varieties of wheat available in the market (‘as an adventure’) demonstrated an increase in anti-tTG (220 U/ml and 16 U/ml versus baseline values of 10 U/ml and 8 U/ml, respectively), qualitative anti EMA titres (4+ and 3+ versus baseline negative titres in both the patients), I-FABP (803.19 pg/ml and 802.22 pg/ml versus baseline values of 469.39 pg/ml and 381.30 pg/ml, respectively) and IFN γ (528.91 pg/ml and 663.27 pg/ml versus baseline values of 433.78 pg/ml and 476.23 pg/ml, respectively). The plasma citrulline levels decreased in both the patients (10.99 µmol/l and 20.60 µmol/l versus baseline values of 36.43 µmol/l and 36.52 µmol/l, respectively). Duodenal biopsies of these patients revealed Marsh-Oberhuber 3b and Marsh-Oberhuber 2 disease, respectively.
Within group outcome analysis.

anti-EMA, anti-endomysial antibody; anti-tTG, anti-tissue transglutaminase; ITT, intention-to-treat; I-FABP, intestinal fatty-acid binding protein; IFN-γ, interferon gamma; PP, per protocol; SD, standard deviation.
Expression of the I-FABP and IFN-γ genes were compared in both groups at baseline and at the end of the study period. No significant differences were observed in the expression of IFN-γ and I-FABP genes in patients of group I (I-FABP 1.11 ± 0.42 versus 1.09 ± 0.45; p = 0.922 and IFN-γ 1.66 ± 1.58 versus 1.74 ± 1.63, p = 0.912; week 0 and week 28, respectively) and group II (I-FABP 1.53 ± 0.85 versus 1.27 ± 0.05, p = 0.514 and IFN-γ 1.26 ± 0.83 versus 1.27 ± 0.96, p = 0.979; week 0 and week 28, respectively).
The palatability and cooking quality of wheat variety C273 were compared with standard commercial wheat on a visual analogue scale (VAS) from 0 to 10 (0 means very poor, 10 means excellent). No differences were noted between wheat variety C273 (median VAS 8) and commercially available wheat (median VAS 9).
Per protocol analysis
In the per protocol (PP) analysis, eight patients in group I who adhered to the diet with wheat variety C273 [group I (compliant)] remained asymptomatic upto week 28. Two patients in group I (non-compliant) were excluded in per protocol analysis. No significant difference was observed in the mean titres of both the serological parameters [anti-tTG (9.81 ± 4.63 U/ml versus 7.20 ± 4.09 U/ml, p = 0.915) and anti EMA positivity (p = 1.000)] between groups I (compliant) and II. A significantly high IFN-γ concentration was seen at week 28 in group I (compliant) as compared with group II (40.80 ± 5.31 pg/ml versus 33.50 ± 3.68 pg/ml, p = 0.034); whereas the differences in I-FABP (404.89 ± 50.76 pg/ml versus 448.62 ± 53.63 pg/ml, p = 0.148) and plasma citrulline (33.39 ± 5.12 µmol/l versus 36.58 ± 3.09 µmol/l, p = 0.259) were insignificant. (Figure 4, Table 2) Seven patients (87.5%) in group I (compliant) had a normal EGD and were in histological remission (Marsh-Oberhuber 0), while one patient had scalloping of duodenal mucosal folds on EGD and Marsh-Oberhuber 1 histology.

Concentration of anti-tTG, IFN-γ, I-FABP and plasma citrulline in group I (compliant) and group II at baseline and during follow-up visits (per protocol analysis). (Units : Anti-tTG in U/ml, I-FABP and IFN-γ in pg/ml and Citrulline in μmol/l)
On assessing changes within groups at weeks 0 and 28, mean anti-tTG (IgA) and IFN-γ levels increased and plasma citrulline levels decreased in group I (compliant), but the change was not found to be statistically significant (7.05 ± 3.10 U/ml versus 9.81 ± 4.63 U/ml, p = 0.28; 38.91 ± 7.07 pg/ml versus 40.80 ± 5.31 pg/ml, p = 0.41 and 36.49 ± 1.62 μmol/l versus 33.39 ± 5.12 μmol/l, p = 0.14, respectively). No significant differences were observed in patients on GFD in group II (Table 3).
The gene expressions of I-FABP and IFN-γ did not change significantly from baseline in any of the groups (group I: I-FABP 1.20 ± 0.31 versus 1.04 ± 0.48, p = 0.445; and IFN-γ 1.62 ± 1.55 versus 1.44 ± 1.53, p = 0.796 and group II: I-FABP1.53 ± 0.85 versus 1.27 ± 0.51, p = 0.489 and IFN-γ 1.26 ± 0.83 versus 1.27 ± 0.96; p = 0.979 at week 0 and week 28, respectively).
Discussion
The present randomised pilot study is an in vivo extension of previous in silico (identification of specific allelic variants in α-gliadin region of wheat variety C273 especially CeD eliciting epitopes) 22 and in vitro (reduced potential of wheat variety C273 to stimulate CeD specific T-cells) studies. 23 Patients with CeD in remission on strict GFD for ⩾2 years were subjected to a 28-week graded gluten exposure with wheat variety C273. Patients on GFD acted as controls. On ITT analysis, no significant changes in anti-tTG, anti EMA, I-FABP and histology were observed in the two groups, indicating the lesser immunogenic potential of gluten from wheat variety C273. But a significant increase in circulating immune-biomarker IFN-γ (p = 0.031) and a significant decline in plasma citrulline levels (p = 0.049) in patients exposed to gluten was observed. None of the patients on PP analysis relapsed and/or presented with overt CeD. The only difference observed on PP analysis was an increase in circulating IFN-γ levels (p = 0.034) between group I (compliant) and group II at week 28.
A higher degree of polymorphism exists in gluten genes in individual wheat accessions, which may be related to distinct toxicity profiles of different wheat genotypes. C273 is a pre-green revolution pre-dwarfing era (when the estimated global prevalence of CeD was 0.03% and CeD was a rarity in India – the first case being reported in 1966)28–30 variety with a dry gluten content of 10.03% (similar to the gluten content in other varieties of wheat). 31 C273 wheat was known for quality traits such as grain hardness, keeping quality, colour, texture and flavour of flat bread. 32 However, being medium-tall and low yielding, it failed to popularise among cultivars and is no longer available commercially. 33 Findings from our in silico and in vitro studies led us to select and cultivate a special crop of this wheat variety (C273) for in vivo testing of immunogenicity.22,23
The use of circulatory inflammatory markers as predictors of intestinal damage and for monitoring disease activity in CeD is not validated but has been proposed to pre-date the changes in established serological markers (anti-tTG and anti-EMA) and histology. A recent study evaluating the efficacy of non-invasive CeD biomarkers in an Indian population concluded that lower levels of plasma citrulline (<30 µmol/l; normal > 30µmol/l) and higher levels of I-FABP [>1100 pg/ml; normal 535.3 (range 321.9–826.9) pg/ml] were consistent with active CeD. 34 No significant changes in circulating I-FABP levels, or gene expression in biopsy, were noted pre and post gluten exposure in the current study. However, a certain degree of mucosal injury was indicated by changes in levels of circulating IFN-γ and plasma citrulline. Interestingly, the gene expression for IFN-γ in duodenal biopsy did not change significantly after gluten exposure. Though IFN-γ is not specific to CeD and changes in its circulating levels could be altered due to a wide range of non-gastrointestinal acute and chronic pathologies, decline in plasma citrulline is specific for active CeD, which cannot be ignored and its clinical implications need to be studied prospectively in larger randomised trials.
In the ITT analysis, two patients defaulted at week 22. This was reflected by a rapid change in circulating inflammatory biomarkers (IFN-γ, I-FABP and plasma citrulline), whereas the change in anti-tTG serology was evident later (at week 28) (Figure 3). This observation vindicates the sensitivity of novel circulating immune biomarkers in predicting mucosal injury with gluten.
Leffler et al. had previously demonstrated that a 14-day gluten exposure with ⩾3 g of gluten/day induces histological and serological changes in the majority of adults with CeD. 35 However, no significant changes were noted in histology in our patients despite consumption of 8 g of gluten from C273 variety for more than 6 months. Absence of evident gluten-related symptoms and changes in serology or histology, even after a long follow up of 28 weeks (sufficient to result in histological evidence of villous atrophy) suggest potential immune-safety of C273 wheat variety.
Patient compliance to gluten exposure was crucial to ensure the validity of results. Dietary assessment was done using a patient-filled daily-diet diary. Frequent follow ups and weekly distribution of pre-weighed milled wheat flour from wheat variety C273 enabled us to follow patients in the treatment arm and ensure continued consumption of appropriate number of flatbreads (in their entirety) each day according to the protocol. Frequent follow ups and a strict compliance assessment methodology led to identification of two defaulters, which was substantiated by both serological and histological markers.
The strengths of our study include a sequential and scientifically logical in silico, in vitro and in vivo (albeit a pilot) approach to evaluation of wheat variety C273 in CeD. In the current in vivo analysis, we followed a holistic approach with serial evaluations of clinical symptoms, serology [anti-tTG (IgA), anti-EMA] and histology to assess response to gluten exposure. Serology is, however, not approved for use as a primary clinical trial outcome by the United States Food and Drug Administration (FDA) as substantial mucosal changes may occur without a significant change in serology. Therefore we also measured circulating I-FABP, plasma citrulline and IFN-γ in addition to analysing the mucosal immune landscape in duodenal biopsy (gene expression of I-FABP and IFN-γ) to identify mucosal injury with consumption of gluten.
We acknowledge a few limitations of our study. Apart from the small group sizes in both in vitro and in vivo studies, we lacked specific blood, urine and faecal biomarkers of exposure to gluten to assess compliance to the protocol in intervention arm. The T-cell and PBMC assays used in in vitro analysis have inherent limitations, and the current findings need to be interpreted with caution. Also, CeD is a heterogenous disease and the DQ2 and DQ8-restricted T-cells recognize deamidated peptides of different regions. The differential effect of wheat variety C273 on HLA subtypes could not be evaluated as only one patient in our study was HLA-DQ8+. Therefore extrapolating the findings of current study and application across the HLA subtypes is not reasonable at the moment.
To conclude, gluten exposure with gluten from C273 wheat variety did not induce significant changes in serology and histology of patients with CeD in remission. This pilot project serves as a roadmap to identify and test older landraces of wheat genotypes containing minimally harmful potentially safe gluten sequences in future. There is still a very long way to go before use of specific wheat varieties is considered a safe intervention in patients with CeD. Until then, the search for safe wheat is on.
Footnotes
Author contributions
All authors contributed to and agreed on the content of the manuscript, and the respective roles of each author are listed below:
Jasmine Grover: Conception and design; analysis and interpretation of the data; drafting of the article; critical revision of the article for important intellectual content; final approval of the article
Parveen Chhuneja: Conception and design; drafting of the article; critical revision of the article for important intellectual content; final approval of the article
Vandana Midha: Conception and design; drafting of the article, critical revision of the article for important intellectual content; final approval of the article
Arshdeep Singh: Collection, analysis and interpretation of the data, drafting of the article, critical revision of the article for important intellectual content; final approval of the article
Ramit Mahajan: Drafting of the article; critical revision of the article for important intellectual content; final approval of the article
Varun Mehta: Critical revision of the article for important intellectual content; final approval of the article
Ramneek Verma: Collection, critical revision of the article for important intellectual content; final approval of the article
Ekta Bansal: Collection, critical revision of the article for important intellectual content; final approval of the article
Dipak Deka: Critical revision of the article for important intellectual content; final approval of the article
Namita Bansal: Analysis; critical revision of the article for important intellectual content; final approval of the article
Neena Sood: Analysis; critical revision of the article for important intellectual content; final approval of the article
Vikram Narang: Analysis; critical revision of the article for important intellectual content; final approval of the article
Ajit Sood: Conception and design; collection, drafting of the article, critical revision of the article for important intellectual content; final approval of the article
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
