Abstract
It is frequently challenging to make the correct diagnosis in patients with biliary strictures. This is particularly important as errors may have disastrous consequences. Benign-appearing strictures treated with stents may later be revealed to be malignant and unnecessary surgery for benign strictures carries a high morbidity rate.
In the first part of the review, the essential information that clinicians need to know about diseases responsible for biliary strictures is presented, with a focus on the most recent data. Then, the characteristics and pitfalls of the methods used to make the diagnosis are summarized. These include serum biomarkers, imaging studies, and endoscopic modalities. As tissue diagnosis is the only 100% specific tool, it is described in detail, including techniques for tissue acquisition and their yields, how to prepare samples, and what to expect from the pathologist. Tricks to increase diagnostic yields are described. Clues are then presented for the differential diagnosis between primary and secondary sclerosing cholangitis, IgG4-related sclerosing cholangitis, cholangiocarcinoma, pancreatic cancer, autoimmune pancreatitis, and less frequent diseases. Finally, algorithms that will help to achieve the correct diagnosis are proposed.
Keywords
Introduction
In patients presenting with a cholestatic clinical profile, intrahepatic and/or extrahepatic biliary strictures frequently present a diagnostic challenge to determine their benign or malignant nature. Appropriate diagnosis is essential to avoid missing malignancy in benign-appearing strictures, or unnecessary surgical exploration for benign disease mimicking malignancy. Benign causes of biliary strictures include surgery [most often cholecystectomy and liver transplantation (LT)], pancreatitis [acute, chronic (CP), or autoimmune (AIP)] and primary sclerosing cholangitis (PSC). 1 The latter should be distinguished from secondary sclerosing cholangitis diagnoses, such as IgG4-related sclerosing cholangitis (IgG4-SC), recurrent pyogenic cholangitis, ischaemic cholangiopathy, or AIDS-associated cholangiopathy, using key diagnostic elements that are available for differential diagnosis of these cases. PSC poses a significant problem due to the increased risk of developing a cholangiocarcinoma (CCA), mainly in cases of dominant biliary stricture. On the other hand, malignant biliary strictures are most frequently due to CCA and pancreatic cancer. 2
Investigations used to distinguish benign from malignant aetiologies include blood tests [carbohydrate antigen 19-9 (CA19-9), carcinoembryonic antigen (CEA)], imaging studies [computed tomography (CT) scan, magnetic resonance cholangiopancreatography (MRCP)], endoscopic modalities [endoscopic retrograde cholangiopancreatography (ERCP), endoscopic ultrasonography (EUS), cholangioscopy], and tissue sampling (brush cytology/biopsy). Among the standard circulating markers of pancreaticobiliary cancers, CA19-9 is the gold standard against which other markers are evaluated.3,4 Despite its relatively high false-negative and false-positive rates, serum CA19-9 can be useful for some differential diagnoses, for assessment of unresectability, and as a prognostic factor following tumor resection. Imaging plays an important role in the diagnostic process for differentiating benign from malignant biliary strictures. Duct hyperenhancement, ductal wall thickening, long irregular and asymmetric shape of the biliary stricture, regional lymph node enlargement, or a mass lesion are some of the imaging features suggestive of malignancy. 5 Tissue sampling is of paramount importance for achievement of adequate results. Despite this multimodality approach, these methods of evaluation suffer from low sensitivity and low negative predictive value for diagnosis of malignant biliary strictures. Therefore, the discovery of novel clinically relevant biomarkers might lead to better diagnostic accuracy. This review provides an overview of the different benign and malignant diseases associated with biliary strictures.
Diseases
Algorithms for the evaluation of patients with a cholestatic pattern caused by biliary stricture(s) are presented in Figures 1 and 2.

Algorithm for the evaluation of patients with a cholestatic clinical pattern caused by biliary stricture(s).

Algorithm for the evaluation of indeterminate biliary strictures. Note that surgery may be indicated in some patients before completing all the steps.
Benign biliary strictures
In Western countries, iatrogenic injury (cholecystectomy, LT) is the most common cause of benign biliary stricture; other causes include PSC, IgG4-SC, CP, Mirizzi syndrome, recurrent pyogenic cholangitis, acquired immune deficiency syndrome (AIDS)-associated cholangiopathy, and chemotherapy-induced sclerosing cholangitis. 1
Primary sclerosing cholangitis
PSC is a chronic liver disease of unknown aetiology, characterized by inflammation and concentric periductal fibrosis that may involve the intra- and extra-hepatic bile ducts, leading to multifocal biliary strictures.7–9 Its incidence ranges from 0.5 to 1.3 per 100,000 person-years, with a prevalence of 1–16 per 100,000 persons. It is predominantly seen in men (male/female ratio: 2/1) with a peak of incidence in the third and fourth decades. Secondary sclerosing cholangitis is characterized by a similar multifocal biliary stricturing process due to identifiable causes such as long-term biliary obstruction by choledocholithiasis. 8
Clinical features
Approximately half of patients are asymptomatic at the time of diagnosis. Non-specific symptoms such as fatigue and pruritus are common at presentation. 10 Fever, jaundice, and right upper quadrant pain may also be present due to transient bacterial cholangitis. Continued destruction of bile ducts may lead to end-stage liver disease and portal hypertension.
Strictures may be present in both the intra- and extra-hepatic bile ducts (70%), or the intra-hepatic (25%) or extra-hepatic (<5%) bile ducts alone. The gallbladder and cystic duct may also be involved. 11 A dominant stricture develops during follow up in 36–50% of patients12–14; this is defined at ERCP as a stricture with a diameter of ⩽1.5 mm in the common bile duct (CBD) and/or ⩽1.0 mm in an hepatic duct within 2 cm of the main hepatic confluence. 15
Cholangiography is normal in <5% of patients who have “small-duct PSC” but 20% of them will develop large-duct PSC over a 7- to 10-year period. 16 Small-duct PSC is associated with a lower risk of LT and death without LT than large-duct PSC. 17 Inflammatory bowel disease (IBD) is associated with 60–90% of PSC in European and North American populations, with ulcerative colitis (UC) five times more frequent than Crohn’s disease. PSC patients with UC present a higher risk of LT or death without LT compared with patients with Crohn’s disease [hazard ratio (HR), 1.56; p < 0.001] or without IBD (HR, 1.15, p = 0.002). 17
PSC-related complications include CCA, gallbladder stones (25% of patients) and carcinoma, colon carcinoma as well as cholestasis-associated problems.7,8 The annual incidence of CCA is 0.5–2% in PSC, with a lifetime risk of 10–20%. 18 The diagnosis of CCA is difficult, particularly in the presence of a dominant biliary stricture. These should be carefully evaluated, particularly if they are discovered soon after the diagnosis of PSC (approximately half of CCAs are diagnosed within the first year after PSC diagnosis). 9 Risk factors for CCA include older age at the time of PSC diagnosis (HR, 1.02 per 1-year increase in age),17,18 smoking, alcohol consumption, a long history of IBD, and coexisting UC with colorectal neoplasia. No screening strategy is recommended but a sensible attitude is, if the patient opts for surveillance after information about risks, to propose annual screening using magnetic resonance imaging (MRI) and CA19-9; if a dominant biliary stricture and/or rise in CA19-9 is observed, biliary brushings for conventional and fluorescence in situ hybridization (FISH) cytological examination (see below) should be obtained, 19 or cholangioscopy performed. 20 Rapid clinical deterioration associated with a progressive biliary dilatation in the setting of a dominant biliary stricture should raise a strong suspicion of CCA.21,22 PSC increases the risk of colorectal cancer and dysplasia with an odds ratio (OR) of 3.2 when compared with patients with IBD without PSC.23,24 Gallbladder mass lesions develop in 3–14% of patients.11,25 Due to the high risk of malignancy associated with gallbladder polyps in PSC and the potential for increased cholecystectomy-related morbidity in these patients, guidelines recommend cholecystectomy either for gallbladder polyps of any size,26,27 or with a lower size threshold than in non-PSC patients. 28 Cholestasis-related complications include metabolic bone disease, fat-soluble vitamin deficiencies, and choledocholithiasis. 29
The estimated median survival until LT or PSC-related death is 20.6 years with the main causes of mortality being CCA (32%) followed by liver failure (18%), LT complications (9%), and colorectal carcinoma (8%). 18 The Amsterdam cholangiographic classification of PSC correlates with patient prognosis. 30
Diagnostic work up
At diagnosis, serum aminotransferases are typically <300 IU/L and serum bilirubin is within normal values. Increased serum IgG4 values have been reported in about 10% of patients; IgG4-SC should be differentiated from PSC. 31
The major diagnostic criteria for PSC are multifocal, short, annular strictures of the intrahepatic and/or extrahepatic bile ducts, alternating with areas of normal or slightly dilated segments, giving a “beaded” appearance (Figure 3); secondary sclerosing cholangitis should be excluded (Figure 1). With the progression of fibrosis, the peripheral ducts become poorly visible, giving a “pruned tree” appearance. MRCP has gained priority over ERCP for diagnosing PSC due to its non-invasive character 15 ; the overall diagnostic accuracy of MRCP is 90% (97% for ERCP).32,33 Liver biopsy is not required to diagnose large-duct PSC; it is required only to diagnose small-duct PSC or a coexisting disease such as overlapping autoimmune hepatitis. 9 The most typical, but infrequent, histopathological finding is fibrous obliteration of small bile ducts with periductal concentric replacement by connective tissue in an “onion skin” pattern.

A 31-year-old man with severe ulcerative pancolitis and primary sclerosing cholangitis.
Diagnostic evaluation of a dominant stricture suspicious for CCA should include CEA, CA19-9 serum analysis, contrast-enhanced cross-sectional imaging, plus ERCP, possibly with single operator cholangioscopy and/or EUS-guided sampling.15,20 (Figure 1). Sampling of hilar masses should be avoided in locations where LT is offered for perihilar CCA due to the risk of tumor seeding.2,34–36
Differential diagnosis
Secondary sclerosing cholangitis may be caused by many diseases, including malignancy, and some of these are detailed below (Figure 1) 8 :

A 30-year-old Vietnamese woman with acute cholangitis, septicaemia, and shock.

Types of biliary strictures in IgG4-SC. Type 1: isolated stricture of the distal common bile duct. Type 2: diffuse strictures of the intra- and extra-hepatic bile ducts with (Type 2a) or without (Type 2b) prestenotic dilatation. Type 3: hilar stricture and distal common bile duct stricture. Type 4: isolated hilar stricture. Reproduced from Kamisawa et al. 31

A 36-year-old man with asthenia, abdominal pain, and pruritus.

A 36-year-old man with recurrent abdominal pain and fever. AIDS had been diagnosed 12 years previously and treated episodically with antiretroviral drugs.
Acute/chronic pancreatitis
In the setting of pancreatitis, a CBD stricture may develop because of acute pancreatitis, compression by a pseudocyst, or, in the case of CP, periductal fibrosis. Biliary strictures complicate the course of CP in 3–23% of patients, 48 usually in its advanced stages, and may be transient.
Clinical features
The clinical presentation of biliary stricture secondary to CP is variable. Some patients remain asymptomatic, whereas jaundice develops in 30–50% of patients. Secondary biliary cirrhosis occurs in 7% of patients. 48
Diagnostic work up
CT-scan or MRCP shows biliary obstruction and possibly compression by a pseudocyst. Other features of CP (e.g. parenchymal atrophy, calcifications, dilated main pancreatic duct) may be visible. 49
Differential diagnosis

A 76-year-old man with painless obstructive jaundice, increased bilirubin (6 mg/dl, normal values <1.2 mg/dl) and IgG4 (4.96 g/l, normal values <1.26 g/l) in the serum. CT-scan in the portal venous phase (A, coronal plan) shows dilatation of the common bile duct (arrow) and main pancreatic duct (arrowhead) upstream from a pancreatic head mass (star). At 2D-MRCP (B), a stricture of the distal common bile duct (arrow) and main pancreatic duct (arrowhead) associated with mild irregularities of the secondary pancreatic ducts are demonstrated. Diagnosis was autoimmune pancreatitis.
Post-surgical biliary strictures
In Western countries, iatrogenic strictures account for up to 80% of all benign strictures. 1
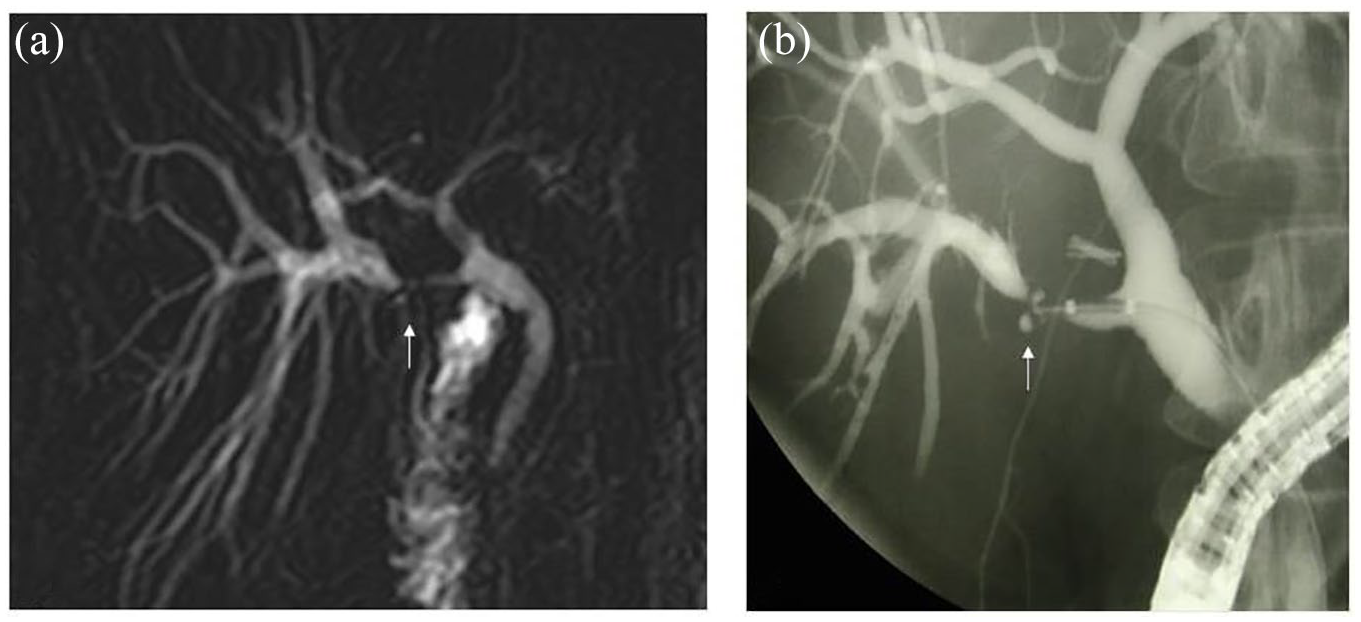
Post-cholecystectomy stricture of an aberrant right posterior biliary duct (low insertion on the common bile duct) shown on 2D-MRCP (A, arrow) and endoscopic cholangiogram (B, arrow).
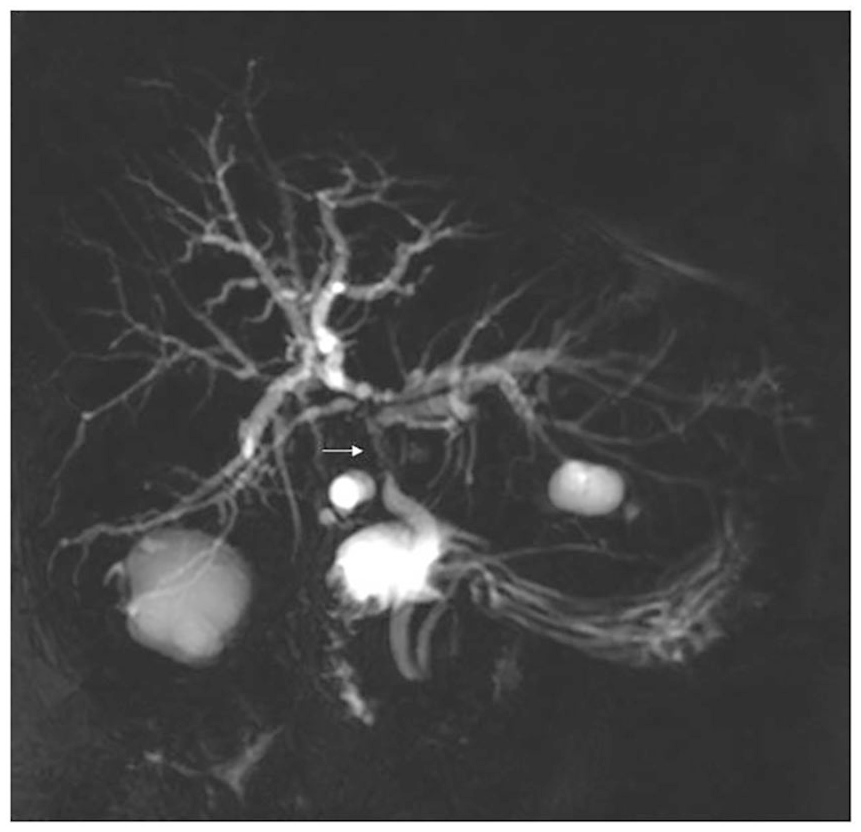
A 62-year-old woman with cholestasis 8 months after liver transplantation for hepatocellular carcinoma developed cirrhosis related to hepatitis C virus.
Mirizzi syndrome
Mirizzi syndrome, the obstruction of the common hepatic duct by a gallstone impacted at the gallbladder neck or cystic duct, occurs in 0.1% of gallstone disease.64,65 Common presenting symptoms include fever, right upper quadrant pain, and obstructive jaundice. MRCP or even ultrasonography may be diagnostic, showing stone(s) within the cystic duct or neck of the gallbladder as well as extrinsic narrowing of the common hepatic duct with upstream dilatation. 66 It has been classified into four types, depending on the presence and importance of a defect in the common hepatic duct. 67 Differential diagnosis with an underlying CCA or gallbladder carcinoma should be made.
Others
Other extremely rare causes of benign biliary strictures include sarcoidosis, 68 adenomyomatosis, 69 inflammatory cholangiopathy related to mastocytosis, 70 eosinophilic cholangitis, and follicular cholangitis. 71
Some features of benign biliary strictures may suggest a diagnosis (Table 1).
Benign biliary strictures.
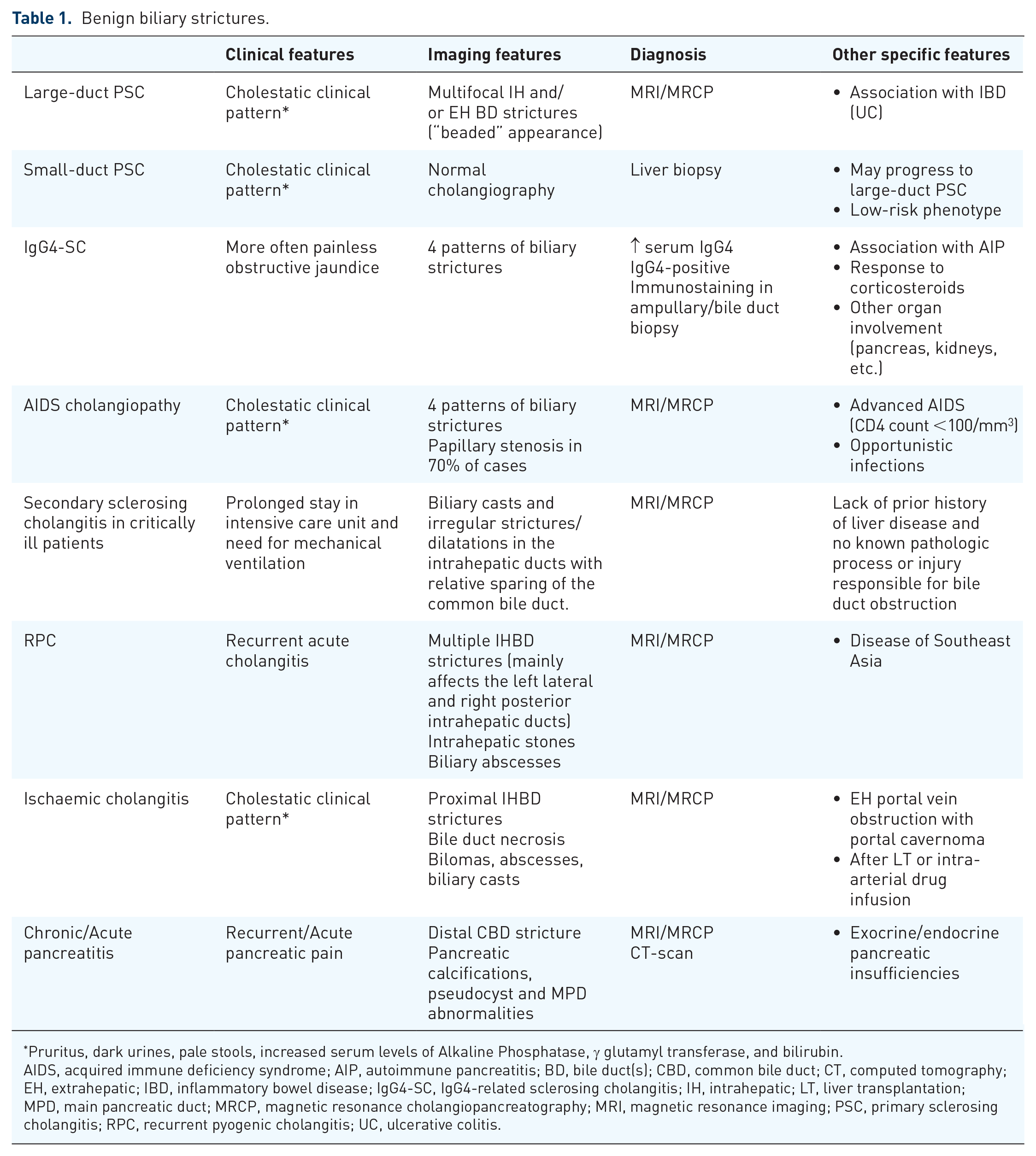
Pruritus, dark urines, pale stools, increased serum levels of Alkaline Phosphatase, γ glutamyl transferase, and bilirubin.
AIDS, acquired immune deficiency syndrome; AIP, autoimmune pancreatitis; BD, bile duct(s); CBD, common bile duct; CT, computed tomography; EH, extrahepatic; IBD, inflammatory bowel disease; IgG4-SC, IgG4-related sclerosing cholangitis; IH, intrahepatic; LT, liver transplantation; MPD, main pancreatic duct; MRCP, magnetic resonance cholangiopancreatography; MRI, magnetic resonance imaging; PSC, primary sclerosing cholangitis; RPC, recurrent pyogenic cholangitis; UC, ulcerative colitis.
Malignant biliary strictures
CCA and pancreatic cancer are the two most common causes of malignant biliary strictures. 2
Pancreatic cancer
The incidence of pancreatic cancer varies between 7.7 (Europe and North America) and 2.2 (Africa) per 100,000 people, with little difference between genders and a peak incidence in the seventh and eighth decades. 72 Due to its low survival rate (9% at 5 years), it is the seventh leading cause of cancer death in industrialized countries, and the third leading cause of cancer death in the United States (US). Risk factors include cigarette smoking, increased body mass index, heredity, CP, and diabetes. 73
Clinical features
Approximately 60–70% of tumors occur in the cephalic area and typically cause jaundice, weight loss, and steatorrhea, whereas tumors in the body and tail usually cause pain and weight loss. 74 Pancreatic cancer should be considered in patients presenting with acute pancreatitis and no causative factor, a recent onset of diabetes, or unexplained thrombophlebitis. 75 Physical findings may include a dilated gallbladder (Courvoisier’s sign), and, rarely, an abdominal mass, ascites, and left supraclavicular adenopathy.
Diagnostic work up
A meta-analysis (52 studies, 5399 patients) found that CT-scan, MRI, transabdominal ultrasonography, and EUS present similarly high accuracies for the diagnosis of pancreatic cancer (90%). 76 Guidelines recommend CT-scan for the detection, staging, and assessment of resectability of suspected pancreatic cancer.77,78 A hypoattenuating pancreatic mass associated with secondary signs such as biliopancreatic ductal dilatation, parenchymal atrophy, or contour abnormalities is highly suggestive of pancreatic cancer (Figure 11). 18 Fluorodeoxyglucose positron emission tomography ( 18 F-FDG PET)/CT may detect masses not seen at CT-scan and differentiate pancreatic malignancy from CP and AIP.79,80

A 48-year-old man with painless obstructive jaundice, loss of weight, and increased CA19-9 serum level (14,000 U/l, normal values <37 U/l).
Differential diagnosis
Focal CP and AIP are the two benign processes most commonly mistaken for pancreatic malignancy. A history of prolonged alcohol abuse and smoking, young age, and diffuse pancreatic ductal changes favor CP, whereas AIP is suspected in the presence of increased levels of serum IgG4. In a meta-analysis (11 studies, 1294 patients), the sensitivity/specificity of IgG4>130–140 mg/dl for distinguishing AIP from pancreatic cancer was 72%/93%. 81 Some authors have suggested increasing the IgG4 cut-off value to avoid missing a cancer (280 mg/dl: specificity of 99%). 6 Other features that may help to diagnose AIP from pancreatic cancer include biopsy (infiltration by IgG4-positive plasma cells), other organ involvement, and a corticosteroid trial if diagnostic criteria are not fulfilled.31,42
Differential diagnosis also includes pancreatic neuroendocrine tumors, lymphoma, and a variety of other rare conditions.
Cholangiocarcinoma
CCA is rare (incidence in the US, 1.3 per 100,000/year) but it is becoming more frequent worldwide. 82 CCA occurs slightly more often in men, (male/female ratio, 1.3:1) during the sixth to eighth decade. CCAs are classified according to their location as intrahepatic, perihilar (also called Klatskin tumours, two-thirds of CCAs), and distal,83,84 or according to their macroscopic appearance as mass-forming, periductal-infiltrating, or intraductal growth. 85 Klatskin tumours are further classified according to the level of biliary obstruction from type I to IV using the Bismuth-Corlette classification. 86 Risk factors include hepatitis C virus infection, chronic biliary inflammation (e.g. PSC, parasitic biliary infestation), and some types of choledochal cysts and anomalous pancreaticobiliary junctions.87,88
Clinical features
Tumour location dictates the symptoms and signs of CCA. Intrahepatic CCAs may cause a dull constant pain in the right upper quadrant, whereas hilar and extrahepatic CCAs cause jaundice, pain, and weight loss. Cholangitis is rare at presentation. At clinical examination, hepatomegaly, tumor mass, or Courvoisier’s sign may be detected. 89
Diagnostic work up
All guidelines recommend MRI or CT-scan for the diagnosis of CCA. 90 At imaging, upstream biliary tract dilation is detected, except in the case of peripheral intrahepatic CCAs, but the tumour itself may not be detected. MRI/MRCP is highly accurate (98%) for identifying biliary obstruction (Figure 12), 91 but is less able to differentiate benign from malignant causes. 92 CT-scan and MRI protocols continuously improve, with the latest techniques currently involving T1- and T2-weighted sequences as well as diffusion-weighted imaging and late-phase sequences with hepatocyte-specific contrast agents. This has allowed for marked improvements but some aspects still remain problematic with both MRI and CT-scans (e.g. evaluation of the longitudinal extent of the lesion). The performance characteristics of CT-scan and MRI for diagnosis and staging of CCA have recently been reviewed. 93 Another technique, 18 F-FDG PET/CT, has demonstrated sensitivity and specificity of 82% and 75% respectively, for diagnosing CCA, with better performance for intra- than extra-hepatic CCA. 94

A 74-year-old man with cholestasis, loss of weight, and increased CA19-9 serum level (190 U/l, normal values <37 U/l).
Differential diagnosis
Depending on tumour location, differential diagnosis may include choledocholithiasis, ampullary tumours, and all other causes of biliary stricture. It is particularly difficult if no mass is detected on cross-sectional imaging (Figure 12). In difficult cases, intraductal imaging, such as intraductal ultrasound (IDUS), cholangioscopy, and confocal laser endomicroscopy, may be used. The interested reader is referred to technical reviews for in-depth information.95,96 IDUS is ideally performed before biliary stenting (3D-IDUS may be recorded for interpretation by a specialist) and helps to distinguish benign from malignant strictures better than EUS, particularly for tumours at the hilum or mid-bile duct. Its accuracy for malignancy is >80% in patients with negative ERCP cytology and histology, and it may serve to guide biopsy sampling, thus improving the accuracy of histological diagnosis. 97 At cholangioscopy, dilated and tortuous vessels are the strongest predictor of malignancy and a recent meta-analysis reported that the sensitivity/specificity of cholangioscopy images for the diagnosis of malignant strictures was 85%/83%, respectively, whereas cholangioscopy-guided biopsy sampling yielded a sensitivity/specificity of 60%/98%, respectively. 98 Confocal laser endomicroscopy uses a 0.96-mm in-diameter probe that can be passed through most endoscopy channels and some catheters or needles. However, this technique is losing momentum due to its high cost, need for training, and low specificity. 99 In a meta-analysis, the sensitivity/specificity of confocal laser endomicroscopy for discriminating benign and malignant pancreaticobiliary strictures was 87%/76%, respectively. 100
Gallbladder carcinoma
The incidence of gallbladder carcinoma widely varies in the world, with the highest incidences in India and South America (it is the primary cause of cancer death among women in Chile). 101 Gallbladder carcinoma affects two to six times more women than men, its incidence steadily increases with age with a peak in the sixth and seventh decades. Gallstones are present in 65–90% of patients with gallbladder carcinoma 102 ; other risk factors include porcelain gallbladder, gallbladder polyps, and anomalous pancreaticobiliary junction.103,104
Clinical features
Early diagnosis is difficult because most patients present with non-specific findings of right upper quadrant pain, jaundice, and weight loss. Jaundice has been associated with unresectable disease. Patients with advanced disease may also present with a palpable gallbladder mass, hard nodular liver, and malignant ascites.
Diagnostic work up
Three major patterns of presentation have been described on ultrasonography, CT-scan, or MRI: a focal or diffuse mural thickening (20–30%), an intraluminal polypoid mass originating from the gallbladder wall (15–25%), and a subhepatic mass replacing or obscuring the gallbladder (45–60%). 105
Standard circulating markers of pancreaticobiliary cancers
Carbohydrate antigen 19-9
CA19-9 is the only biomarker for pancreatic cancer currently approved by the FDA. Although it is suboptimal, it is the gold standard against which other markers are evaluated. In particular, it is superior to CEA in terms of sensitivity for pancreatic cancer with a similar specificity. 106
Screening (asymptomatic subjects)
Subjects at average risk for biliopancreatic cancer
The US Preventive Services Task Force recently reaffirmed its recommendation against screening for pancreatic cancer in asymptomatic adults at average risk for pancreatic cancer. 107 In a large study involving 58,498 asymptomatic subjects, serum CA19-9 was elevated in 581 (1%), and cancers were found during follow up in 25, including 6 (0.01%) pancreatic cancers. 108
Subjects at increased risk of pancreaticobiliary cancer
Screening for pancreatic adenocarcinoma presents a theoretical interest as the interval between tumor initiation and cancer is approximately 10 years. 109 High-risk patients include those with familial pancreatic cancer, Peutz-Jeghers syndrome, hereditary CP, hereditary colon cancer (Lynch syndrome or mismatch repair mutations), and seven specific gene mutations. 110 Current protocols use imaging and, very rarely, CA19-9 measurement, to screen such patients.111,112 New-onset diabetes is another high-risk situation for pancreatic cancer in which CA19-9 seems inadequate.113,114
Screening for CCA in PSC patients using biochemical markers is not recommended in the guidelines, 9 although this screening is performed widely (see above). Some authors have proposed using different cut-off values of CA19-9 depending on the patient’s CA19-9 biosynthesis activity and to exclude patients with a low biosynthesis activity (this was assessed by determining allelic variants of fucosyltransferases 2 and 3); with these modifications, the sensitivity/specificity of CA19-9 for CCA diagnosis in 392 PSC patients increased to 78%/90%. 115
Diagnosis
For the diagnosis of pancreatic cancer and CCA, the sensitivity/specificity of CA19-9 was approximately 75%/75% in two large meta-analyses, with a slightly higher specificity for CCA at 84%.3,4 For pancreatic cancer, a cut-off value of 37 U/ml was used, and controls had benign pancreatic disease. The parallel combination of CA19-9 and CEA (test positive if any of the two markers was higher than the cut-off value) yielded a sensitivity/specificity of 85%/71%. 4 For CCA, the cut-off values varied from 20 to 200 U/ml and controls had benign hepatobiliary disease except in 5 of 31 studies (healthy controls). 3
Limitations of CA19-9 for the diagnosis of biliopancreatic malignancy include:
False-negative cases
Patients of the Lewis (a–b–) blood group who cannot synthesize CA19-9 represent 7–10% of the Caucasian population (Lewis blood typing may be performed using hemagglutination assays). In these patients, the dosage of CEA and of cancer antigen 125 (CA125) could be an alternative (sensitivity of the parallel combination, 74%). 116 For pancreatic cancers <2 cm, the sensitivity of CA19-9 is approximately 50%; it increases with higher stages of cancer.117,118
False-positive cases
CA19-9 may be increased in patients with hepatocellular, ovarian, bronchial, colon, and gastric cancers. 118 Some benign conditions also consistently increase CA19-9 values:
Cholestasis and cholangitis of any cause, but the increase is generally limited. 40 In the presence of cholangitis or cholestasis, interpretation of CA19-9 values should be delayed until the acute condition has resolved, or a higher cut-off value should be used. In the case of cholestasis, in particular PSC, a cut-off of 129 U/ml, 119 or the ratio of the fold-increase of CA19-9 to that of total bilirubin, has been proposed to increase specificity. 120 Of note, CEA is not affected by cholestasis.
Diabetes mellitus is associated with increased CA19-9 values, particularly if metabolic control is poor. 121 Higher cut-off values have also been proposed.
Despite its limitations, CA19-9 may be useful, including in difficult clinical situations such as for differentiating pancreatic carcinoma from CP and AIP. In the first scenario, the sensitivity/specificity of CA19-9 was 81%/81% in a meta-analysis (34 studies, 5186 patients); 122 in the second scenario, the combination of CA19-9 (<74 U/ml) and IgG4 (>1.0 g/l) has been proposed as a better diagnostic test (sensitivity/specificity 94%/100%) than IgG4 alone. In both scenarios, imaging techniques are crucial, including EUS supplemented with sampling if indicated. 123
Prognosis and follow up during treatment
Unresectability of pancreaticobiliary tumours is suspected if CA19-9 values before treatment are particularly high but optimal cut-off values remain elusive.124–126
Following the resection of pancreatic adenocarcinoma, patient survival has been associated with (i) a decrease in CA19-9 values at the first postoperative measurement and (ii) a first post-operative CA19-9 value <200 U/ml (analysis was restricted to patients with preoperative total bilirubin <2 mg/dl). 127 These two factors predicted patient survival independently of T and N stages. In patients who receive neoadjuvant therapy for pancreatic cancer, CA 19-9 decrease is associated with survival. 128 Conversely, an increase in CA19-9 values during treatment may indicate tumour progression before it is detected by imaging studies. 129
Carcinoembryonic antigen
CEA is used routinely in clinical practice; it is overexpressed in patients with cancers (e.g. colorectum, pancreas, breast, and lung) and in a series of benign conditions, including pancreatitis, IBD, and cigarette smoking. The sensitivity/specificity of CEA for the diagnosis of suspected pancreatic cancer was 36%/87% in a meta-analysis of 11 studies that used benign diseases as controls; 5 µg/l was the cut-off value in most studies. 4 CEA also provides prognostic value, in particular for biliary cancer: in 190 patients with a CCA in various locations, preoperative CEA predicted overall survival independently of tumour stage, 130 and this was confirmed in a series of 805 patients with intrahepatic CCA. 131
Tissue sampling
EUS-guided sampling can be performed if a mass is detected at the level of the biliary stricture, or not, albeit with a lower accuracy, 132 and it simultaneously allows pathological staging of loco-regional and distant lymph nodes, of the left liver lobe, and of ascites undetected by other imaging techniques. 133
ERCP also allows sampling. This may be performed at the level of a biliary stricture or the papilla if an ampullary neoplasm or IgG4-SC is suspected. In the biliary tree, sampling is recommended every time that a stricture with no established or evident diagnosis (e.g. anastomotic stricture following LT) is demonstrated. 134 Once depreciated, the technique is simple and it allows patients to be spared invasive procedures. 135
Finally, bile may be collected through a nasobiliary or percutaneous drain and then prepared for cytopathological examination by centrifugation. This technique is cumbersome but it can provide acceptable sensitivity if properly applied on serial samples. 136
Materials and methods
Sample acquisition
At EUS, fine needle aspiration (FNA) and fine needle biopsy (FNB) needles can be used; the latter have been introduced more recently and present either a special geometry or a side-slot at the needle tip. The method is described thoroughly in a guideline. 137 Briefly, the needle, with or without a stylet, is introduced into the target mass or at the level of the biliary stricture under continuous EUS control, suction is applied, back and forth movements are performed with the needle fanning throughout the lesion, residual negative pressure is suppressed, and the needle is withdrawn.
Cytopathological sampling at ERCP is not as well standardized. Different brush models are available without differences in yield; they all provide material for cytopathological examination. The brush, located inside a catheter, is introduced into the bile duct, moved back and forth within the stricture at least 10 times under pulsed mode continuous fluoroscopy to ensure correct brush positioning, pulled into the sheath still located immediately below the structure, and the unit brush/sheath is finally removed. In a pilot study, injecting 5–15 ml of saline into the bile duct after brushing and collecting this fluid increased the sensitivity for cancer diagnosis by 24% (absolute difference). 138 The grasping basket is another device that provides more material and allows for a higher diagnostic accuracy than brushing139,140; it is available in some countries only. Soehendra’s stent retriever is sometimes used to pass very tight strictures, and may provide material adequate for cytopathological examination. 141 Pathological examination of retrieved biliary stents has been abandoned due to its unacceptably low sensitivity and the availability of alternatives. 142
For biopsy sampling at ERCP, standard or specifically designed biopsy forceps may be used. In contrast with brushing, biopsy sampling usually requires a biliary sphincterotomy, with its associated risks. The biopsy forceps are introduced transpapillary and positioned against the biliary stricture under fluoroscopic guidance or direct visual control during cholangioscopy. This latest technique is usually reserved for patients with unsatisfactory diagnosis after a standard work up because it is not widely available and, compared with the standard technique, it is associated with higher morbidity, 143 longer procedure duration, and higher costs. 144 Despite all these drawbacks, it has been suggested to be the most cost-effective method in PSC patients. 20 Biliary biopsy samples are processed and examined as any other gastrointestinal biopsy sample except if rapid on-site pathological evaluation (ROSE) is performed.
Sample preparation
All material required for sample preservation is prepared before sampling to limit preservation artefacts. Direct smears are prepared by collecting the specimen onto a glass slide and spreading it in an evenly thin layer. Training is required to avoid common pitfalls such as air-drying and thick smears that make observation of cellular details difficult. 145 Smears may be allowed to dry or be fixed immediately, according to the cytopathologist’s preference. If a brush is used, it is placed into liquid after smearing and the brush sheath is rinsed into the brush vial for liquid-based cytology. 146 Smears are prepared in the laboratory using an automated process, overcoming the pitfalls associated with manual preparation. Cell-blocks are prepared by centrifuging the liquid medium, collecting the pellet and embedding it into paraffin. The sample is then processed as a biopsy specimen, allowing specific tests such as immunocytochemistry and genetic analysis. A part of the sample is stored in case additional preparation proves useful after initial cytopathological examination. 145
Sample interpretation
Rapid on-site cytopathological evaluation
ROSE is available only to a minority of endoscopists, 147 and this has led to various work-arounds, such as microscopic examination by the endoscopist rather than a cytopathologist or telecytopathology using WhatsApp.148,149 During EUS-guided sampling, ROSE allowed clinicians to perform fewer needle passes compared with delayed sample interpretation in two randomized controlled trials (RCTs). Diagnostic accuracy, adverse events, and costs were similar.150,151 During ERCP, in a pilot study, 152 biliary brushing was repeated if ROSE showed an inadequate sample and this yielded an unusually high sensitivity for cancer diagnosis (83%). Similarly, biliary biopsy samples have been subjected to ROSE in pilot studies, yielding high sensitivities for cancer diagnosis (76–97%) but false-positives are a concern.153,154 Obtaining a firm diagnosis of malignancy during ERCP would also allow immediate decision-making, for example to select a biliary stent model. 134
Delayed pathological examination
It is recommended that standardized diagnostic categories be used in the cytopathological report [i.e. non-diagnostic, negative, atypical, neoplastic (benign or other), suspicious and positive]. 155 Vague diagnoses such as “dysplasia” are not clinically useful and may be reclassified into one of the diagnostic categories cited above, allowing for increased sensitivity for cancer diagnosis.156,157
Many authors recommend considering the former category “highly atypical suspicious for cancer” or the Papanicolaou classes “atypical” and “suspicious malignancy” as equivalent to “positive” for malignancy in order to increase sensitivity for cancer diagnosis. 158 As some authors have reported a low specificity (83%) with this strategy, 159 this option should be discussed in a multidisciplinary team. In particular, interpretation of atypical diagnoses should be made with care in patients with PSC or those with a stent in place before brushing. 160
The inter-observer variability of cytopathological diagnoses is a concern, and, if no cytopathologist dedicated to biliopancreatic diseases is locally available, samples can be sent to a reference centre. For example, in a retrospective study of 129 biliary brushings with a sensitivity for cancer diagnosis of 36% at initial smear reading, re-reading by two dedicated pathologists increased sensitivity to 61% while specificity remained at 100%. 161
Ancillary techniques such as FISH are increasingly being applied. These may use a commercial kit developed for the urinary tract (UroVysion), 162 or probes specific to pancreaticobiliary cancers, 163 and are primarily used to increase sensitivity, 162 but may also be used in new applications, such as for prognosis in CCA. 164
Diagnostic accuracy
Single approach
A meta-analysis (three comparative studies, 294 patients) reported higher sensitivity (79% versus 45%) and similar specificity (100% versus 98%) for EUS-guided sampling versus ERCP sampling for the diagnosis of suspected malignant biliary strictures. The negative predictive values were low with both approaches, at 47% and 34%, respectively. 165 A more recent study confirmed, in 50 patients, the higher sensitivity of EUS-guided sampling versus ERCP brushing plus biopsy sampling (94% versus 60%), particularly for extraductal lesions and those >1.5 cm. 166 Non-comparative studies support these results:
For EUS-guided sampling, two meta-analyses (6 and 20 studies, 196 and 957 patients, respectively) reported sensitivities of 66–80% and specificities of 100–97%, respectively, with higher sensitivity reported in patients with a mass detected at EUS.132,167 In the community, the diagnostic accuracy of EUS-guided sampling may be lower than that reported in studies from large centers. 147
For ERCP, a meta-analysis (18 studies, 814 patients) found that biliary brushing had a sensitivity of 48% (59% versus 41% for strictures related to biliary versus pancreatic cancer, respectively; p < 0.0001), with wide variations between studies (from 9% to 93%). 168 Specificity was close to 100%. For biliary biopsy sampling, the mean sensitivity was 55% (63% versus 46% for strictures related to biliary versus pancreatic cancer, respectively; p = 0.02), also with large variations between studies (from 32% to 82%). Combining brushing and biopsy sampling increases the sensitivity by 8–34% in absolute value (Table 2).
Sensitivity of brushing, biopsy sampling, and a combination of these techniques for diagnosis of malignant biliary strictures.
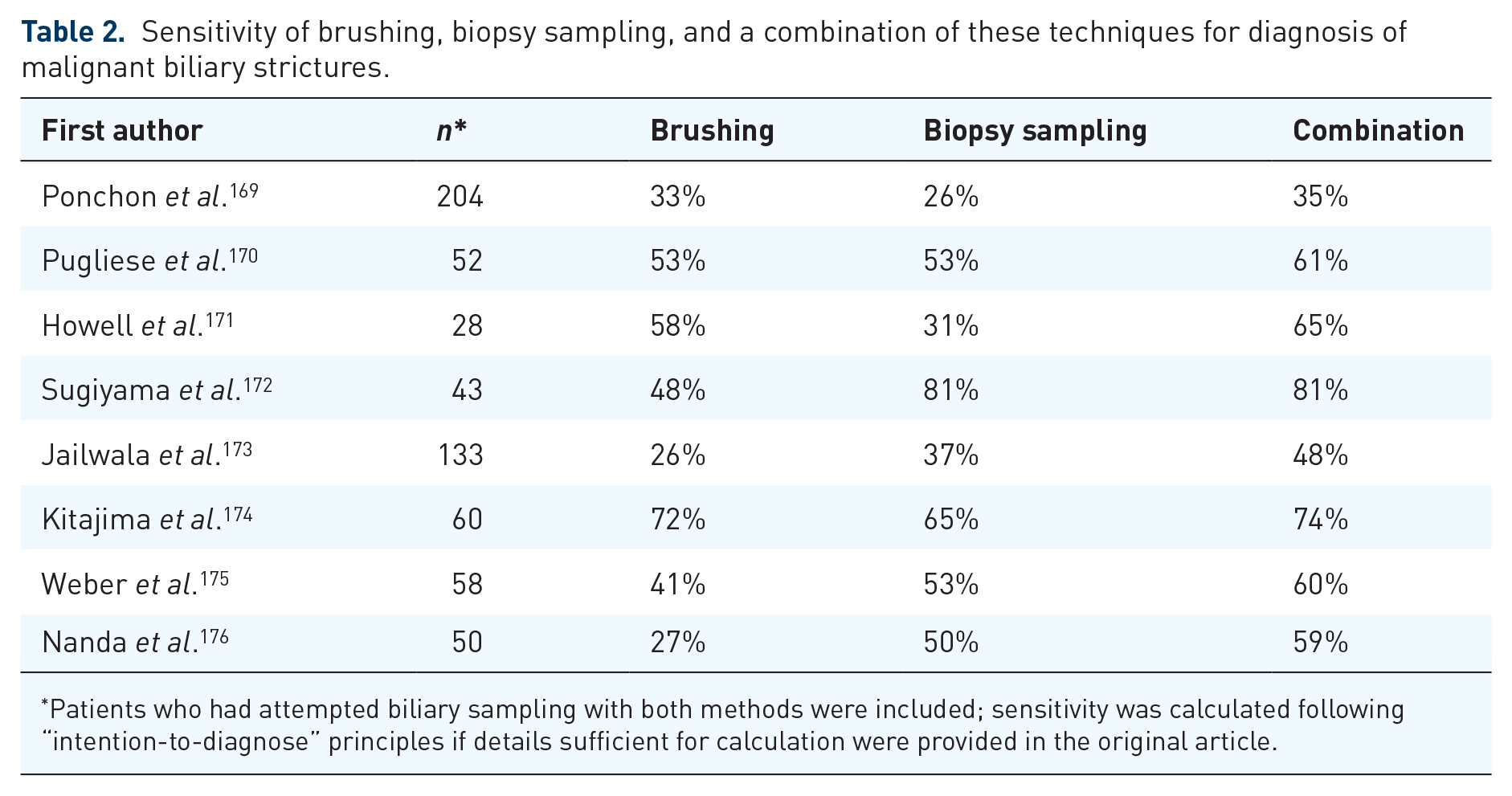
Patients who had attempted biliary sampling with both methods were included; sensitivity was calculated following “intention-to-diagnose” principles if details sufficient for calculation were provided in the original article.
Combined approaches
A meta-analysis (10 studies) found that the incremental benefit of EUS for the diagnosis of malignant extrahepatic biliary strictures compared with brushing at ERCP was 14% (95% CI, 7–20%), 177 and was greater for distal strictures or strictures related to extrinsic compression.
EUS-guided sampling and ERCP sampling/treatment may be performed during a single session. Using this so-called “tandem” approach, a recent study (239 patients) reported diagnostic accuracies of 61% (ERCP), 76% (EUS-guided sampling), and 87% for both studies combined. The accuracy of EUS-guided sampling was higher than that of ERCP for pancreatic masses but not for biliary lesions. 178
How to reach a high sensitivity for the pathological diagnosis of malignant biliary strictures?
With biliary brushings, a learning curve exists for the endoscopist–cytopathologist team (in a series the sensitivity for cancer increased with experience, from 44% to 71%), 179 whereas it seems less problematic with biliary biopsy samples. Endoscopists with suboptimal results at ERCP sampling are likely to obtain rapid improvements by following these recommendations:
Finally, adding EUS-guided sampling to ERCP permits practitioners to obtain one positive diagnosis in seven patients with negative ERCP samples. 177
The clinical approach to biliary strictures
The diagnostic approach begins with a careful patient history and physical examination: a history of surgery (cholecystectomy or LT), autoimmune disease (IBD or AIP), pancreatitis, gallstones, HIV status, or chemotherapy is vital knowledge needed to narrow the differential diagnosis.
Figure 1 summarizes the initial work up proposed for patients with a cholestatic clinical pattern. Abdominal ultrasonography reveals the level of obstruction with an accuracy of >90%. It may also show the cause of obstruction but it is highly operator dependent. 76 CT-scan or MRI is the next step to identify the cause of obstruction. Some authors suggest to prefer MRI and CT-scan for intrahepatic and distal bile duct strictures, respectively 188 ; however, the evidence supporting this diagnostic approach is limited. Duct hyperenhancement and thickness ⩾3, 4, or 5 mm are independent predictors of malignancy, 5 while other criteria may also be helpful (longer, irregular, and asymmetric biliary stricture, regional lymph node enlargement >1 cm, abrupt cut-off on cholangiography, or a mass lesion). 189 In patients with CP, MRI with diffusion-weighted imaging may be particularly useful. At this stage, standard biochemical tests should be complemented with complete blood count, CA19-9, CEA, IgG, IgG4, and HIV serology.
Biliary strictures that remain indeterminate will likely require invasive procedures (Figure 12) that allow sampling and, in the case of EUS, detection of masses missed by other techniques and locoregional tumor staging. After a complete work up, some patients still have no diagnosis and exploratory surgery may be indicated. This should be discussed in a multidisciplinary team and with the patient as about 15% of patients with suspected perihilar CCA are found to have a benign diagnosis after resection and post-operative mortality remains about 10% in many Western referral centers. 190
Footnotes
Acknowledgements
The authors wish to thank a medical writer, Sandy Field, for critical revision of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
