Abstract
Advances in minimally invasive tissue resection techniques now allow for the majority of early colorectal neoplasia to be managed endoscopically. To optimize their respective risk–benefit profiles, and, therefore, appropriately select between endoscopic mucosal resection, endoscopic submucosal dissection, and surgery, the endoscopist must accurately predict the risk of submucosal invasive cancer and estimate depth of invasion. Herein, we discuss the evidence and our approach for optical evaluation of large (⩾ 20 mm) colorectal laterally spreading lesions.
Introduction
Informed by irrefutable evidence over the last decade, minimally invasive endoscopic resection techniques have emerged as the primary treatment strategy for large (⩾20 mm) colorectal laterally spreading lesions (LSLs).1,2 Endoscopic mucosal resection (EMR) is safer and more cost-effective than surgery.3–5 As high-quality EMR continues to evolve,6–12 the risks of clinically significant post-EMR bleeding, 13 deep mural injury, 14 and recurrence have largely been mitigated.15,16 Its primary limitation is the need for piecemeal resection when lesion size exceeds 20 mm. 14 If submucosal invasive cancer (SMIC) is identified on histopathology, margin assessment is limited, and, therefore, cure cannot be ascertained. Moreover, coagulation artifact could potentially obscure other high-risk histopathological features.
Herein lies the benefit of endoscopic submucosal dissection (ESD), as it allows for organ-sparing curative resection for superficial SMIC (⩽1000 μm; S-SMIC) in the absence of other high-risk features (poor differentiation, lymphovascular invasion, high-grade tumor budding, positive margins).2,17,18 However, ESD is a technically challenging and time-consuming technique. 19 Its universal application has been questioned given the low frequency of S-SMIC in colorectal neoplasia alongside its modest R0 resection frequency. 20 Recent cost-effective analyses demonstrate that a selective resection algorithm using both EMR and ESD is the optimal approach. 21 It is therefore incumbent on the endoscopist to accurately identify potential SMIC and estimate SMIC depth of invasion so as to select the appropriate resection technique.
With the dissemination of image enhanced endoscopy (IEE), optical evaluation is now the primary method for performing SMIC risk-stratification prior to endoscopic resection. In this review, we appraise the evidence on the performance of optical evaluation to diagnose SMIC and quantify depth of invasion for large LSLs.
Gross morphological features
Lesion size
Lesion size is arguably the critical gross morphological feature for SMIC risk stratification. Inherent to the complexity of large LSL management is the heightened frequency of SMIC compared with smaller lesions. Among 12 North American and European cohorts, the frequency of SMIC in 51,510 diminutive (⩽ 5 mm) and 16,177 small (6–9 mm) polyps were 0.04% and 0.3%, respectively. 22 In a single North American cohort of 3068 medium (10–19 mm) polyps, the frequency of SMIC was 0.9%. 23
Conversely, in a prospective cohort of 2277 LSLs ⩾ 20 mm by Burgess and colleagues, 24 the frequency of SMIC was 7.6%. When stratified by lesion size, significant differences in the frequency of SMIC were identified (20–29.9 mm 5.7%, 30–39.9 mm 6.8%, 40–49.9 mm 6.7%, ⩾50 mm 11.6%; p = 0.002). On multiple logistic regression analysis, size was independently associated with SMIC [odds ratio (OR) 1.1, 95% confidence interval (CI) 1.0–1.2]. These findings are supported by a recent systematic review and meta-analysis by Bogie and colleagues, 25 of 48 studies assessing the frequency of SMIC in LSLs ⩾ 10 mm. Among 10 studies evaluating LSLs ⩾ 20 mm, the pooled frequency of SMIC was 11.3%.
Lesion location
Location is another important variable in therapeutic decision making for large LSLs, specifically the rectum, given the heightened morbidity and risk of permanent ostomy formation of distal colorectal surgery. 26
In the above study by Burgess and colleagues, 24 the frequency of SMIC was significantly higher in LSLs located in the rectum or sigmoid colon (13.1%) compared with more proximal lesions (5.2%; p < 0.001). On multiple logistic regression analysis, rectosigmoid location was independently associated with SMIC (OR 1.9, 95% CI 1.3–2.8). This is reinforced by the systematic review and meta-analysis by Bogie and colleagues, 25 whereby LSLs with SMIC were more often located in the distal colon compared with the proximal colon (OR 2.5, 95% CI 1.2–5.0).
Lesion morphology and topography
Lesion morphology and topography classification not only facilitates SMIC risk stratification but allows for a universal language among endoscopists (Figures 1 and 2). The most widely used morphology classification is the Paris classification.27,28 Lesion topography is most commonly classified by the presence of a granular, non-granular, or mixed appearance.

Predominant Paris classification morphologies: (a) 0-Ip, (b) 0-Is, (c) 0-IIa+Is, (d) 0-IIa, (e) 0-IIb, (f) 0-IIa+c.

Topography classification: (a) granular, (b) non-granular, (c) mixed.
In the systematic review and meta-analysis by Bogie and colleagues, 25 the pooled frequencies of granular homogenous type, granular nodular mixed type, non-granular flat elevated type, and non-granular pseudo-depressed type were 35.4%, 26.1%, 33.0%, and 5.5%, respectively. The corresponding pooled SMIC frequencies were 0.5%, 10.5%, 4.9%, and 31.6%, respectively.
In the study by Burgess and colleagues, 24 the frequency of SMIC significantly differed by lesion morphology (Paris 0-IIa 4.9%, Paris 0-Is 10.5%, Paris 0-IIa+Is 10.4%; p < 0.001; any Paris 0-IIc component 29.3%; p < 0.001) and by lesion topography (granular 4.7%, any non-granular component 13.8%; p < 0.001). On multiple logistic regression, Paris 0-Is morphology (OR 2.7, 95% CI 1.6–4.6), Paris 0-IIa+Is morphology (OR 2.5, 95% CI 1.5–4.1), and any non-granular component (OR 2.8, 95% CI 1.9–4.2) were independently associated with SMIC.
Unique morphological features
Unique morphological features have been evaluated in an attempt to identify a pathognomonic optical characteristic for SMIC. In a prospective multicenter study of 2123 lesions >10 mm by Puig and colleagues, 29 non-lifting, chicken skin sign, edge retraction, depressed areas, fold convergence, induration, ulceration, and polyp over polyp were all significantly associated with deep SMIC (>1000 μm; D-SMIC; all p < 0.04). Of note, statistical analyses to assess for independent association with D-SMIC were not performed. Moreover, a universal definition for many of these features has not been established, which impairs both their widespread application and diagnostic performance. This has been demonstrated in a recent systematic review and meta-analysis of 33 studies and 31,568 lesions by Backes and colleagues, 30 which evaluated the diagnostic performance of gross morphological features. For diagnosing SMIC, gross morphological feature (non-granularity, any kind of depression, presence of large nodule, sclerous wall change, surface redness), sensitivity and specificity ranged from 21% to 46% and 81% to 100%, respectively. For diagnosing D-SMIC, gross morphological feature (non-granularity, any kind of depression, spontaneous bleeding, irregular surface, ulcer or erosion, white spots, exudate) sensitivity and specificity ranged from 18 to 68% and 80 to 98%, respectively.
Image enhanced endoscopy
Whereas gross morphological features play an important role in SMIC risk stratification, unreliable diagnostic performance limits their ability to act as the foundation of a selective resection algorithm.
Image-enhanced endoscopy, either by virtual- or dye-chromoendoscopy, with or without traditional magnification, allows for examination and identification of pit pattern and microvascular pattern changes consistent with SMIC (Figure 3). 31 Multiple classifications have been created. For pit pattern evaluation, the most commonly used is the Kudo pit pattern classification. 32 For microvascular pattern evaluation, the narrow-band imaging (NBI) international colorectal endoscopic (NICE) classification 33 and the Japanese NBI expert team (JNET) classification have emerged as the predominant classifications. 34 Although challenging even among experts, the basis of all optical evaluation classification schemes is identifying a demarcated area with either a disordered or a disrupted pit pattern or microvascular pattern consistent with invasive disease.
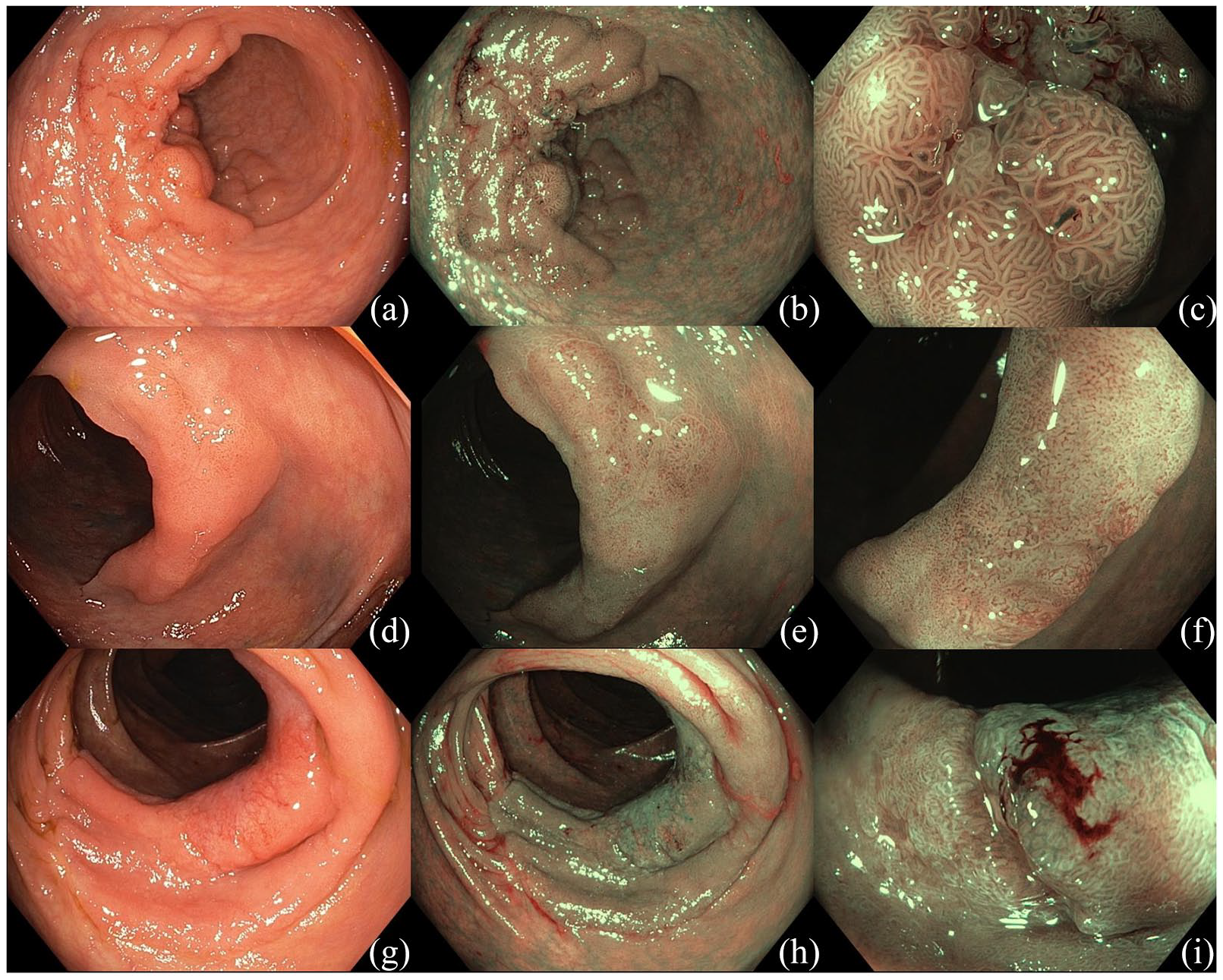
Optical evaluation with image enhanced endoscopy. (a–c) A 50-mm 0-IIa+Is mixed laterally spreading lesion in the recto-sigmoid junction. On narrow-band imaging with near-focus a homogenous surface pattern (NICE II, JNET IIa, Kudo IV) is identified consistent with a benign adenoma. (d–f) A 30-mm 0-IIa non-granular laterally spreading lesion in the transverse colon. On narrow-band imaging with near-focus a demarcated area with surface pattern disruption (JNET IIb, Kudo Vi) is identified consistent with superficial submucosal invasive cancer. (g–i) A 40-mm 0-IIa non-granular laterally spreading lesion in the ascending colon. On narrow-band imaging with near focus a demarcated area with surface pattern disruption (NICE III, JNET III, Kudo Vn) is identified consistent with deep submucosal invasive cancer.
Virtual chromoendoscopy without traditional magnification
In the above study by Puig and colleagues, 29 the diagnostic performance of the NICE classification was assessed. All lesions were assessed without near-focus or traditional magnification. A total of 89 (4.2%) lesions were diagnosed with D-SMIC. The NICE criteria for diagnosing D-SMIC had a sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of 58.4%, 96.4%, 41.6%, and 98.1%, respectively. Using a conditional inference tree, the NICE classification was found to most accurately identify lesions with D-SMIC with pedunculated morphology, ulceration, depressed areas, and nodular-mixed type affecting diagnostic accuracy.
Among studies solely evaluating LSLs ⩾ 20 mm, Backes and colleagues prospectively evaluated 343 large LSLs using the Hiroshima classification to diagnose SMIC and D-SMIC. 35 For SMIC, sensitivity, specificity, PPV, and NPV were 78.7%, 94.2%, 68.5%, and 96.5%, respectively. For D-SMIC, sensitivity, specificity, PPV, and NPV were 63.3%, 99.0%, 86.4%, and 96.5%, respectively.
In the above study by Burgess and colleagues, 24 the frequency of SMIC was significantly higher among lesions with Kudo V pit pattern compared with Kudo I–IV pit patterns (56.6% versus 4.7%; p < 0.001). On multiple logistic regression analysis, Kudo V pit pattern was the strongest predictor of SMIC (OR 14.2, 95% CI 8.6–23.4). The sensitivity, specificity, PPV, and NPV of Kudo V pit pattern for SMIC was 40.4%, 97.5%, 56.6%, and 95.3%, respectively.
Virtual chromoendoscopy with traditional magnification
Virtual chromoendoscopy can be coupled with traditional optical magnification to facilitate microvascular pattern characterization. However, it is important to note that traditional magnification is neither widely available nor used by western endoscopy centers.
In the above systematic review and meta-analysis by Backes and colleagues, 30 the pooled sensitivity and pooled specificity of NBI with traditional magnification to diagnose SMIC were 85% and 94%, respectively. For D-SMIC, the pooled sensitivity and specificity were 77% and 98%, respectively.
Using the JNET classification, Sumimoto and colleagues retrospectively evaluated and applied the JNET classification to 2933 colorectal lesions. 36 The sensitivity, specificity, PPV, NPV and accuracy for type IIa lesions (adenomatous pathology with low-grade dysplasia) were 74.3%, 92.7%, 98.3%, 38.7%, and 77.1%, respectively. For type IIb lesions (adenomatous pathology with high-grade dysplasia or S-SMIC), the sensitivity, specificity, PPV, NPV, and accuracy were 61.9%, 82.8%, 50.9%, 88.2%, and 78.1%, respectively. For type III lesions (adenomatous pathology with D-SMIC), the sensitivity, specificity, PPV, NPV, and accuracy were 55.4%, 99.8%, 95.2%, 96.6%, and 96.6%, respectively.
Dye-chromoendoscopy with magnification
Dyes, most commonly indigo carmine or crystal violet, sprayed on the lesion’s surface can be used to accentuate the surface pit pattern during optical evaluation with magnification. In the above systematic review and meta-analysis by Backes and colleagues, 30 the sensitivity and specificity of magnified dye chromoendoscopy for diagnosing SMIC were 90% and 96%, respectively. For D-SMIC, the sensitivity and specificity were 81% and 95%, respectively. Of note, no differences in performance between magnified NBI and magnified dye-chromoendoscopy were identified.
Covert submucosal invasive cancer
Large LSLs are the critical subgroup that require optical evaluation for the purpose of SMIC risk stratification. However, there is limited data on its application for large LSLs, with most studies having a mixed cohort of diminutive, small, medium and large polyps. In studies that assess solely large LSLs, optical evaluation has shown modest performance characteristics, specifically low sensitivity, for the diagnosis of both SMIC and D-SMIC. Therefore, a risk of invisible or covert SMIC exists.
In the study above by Burgess and colleagues, 24 after removing lesions with visible or overt SMIC based on optical evaluation, lesion size, rectosigmoid location, Paris 0-Is morphology and Paris 0-IIa+Is morphology were independently associated with a risk of covert SMIC. Moreover, by utilizing lesion location, morphology, and topography, a lesion subgroup at high risk (>10%) of covert SMIC was identified (Paris 0-Is non-granular, Paris 0-IIa+Is non-granular, distal Paris 0-IIa+Is granular). These lesions are ideal candidates for ESD.
Approach to optical evaluation and treatment selection
To perform high-quality optical evaluation, begin by manipulating the patient’s position (transitioning to supine or right lateral decubitus), to utilize the effect of gravity and shift the fluid pool to the opposing colorectal wall. This will maximize exposure of the lesion’s epithelial surface. The lesion should be generously irrigated to remove any residual mucous or debris. However, care should be taken not to traumatize the epithelial surface.
Beginning with high-definition white-light, estimate the lesion’s size, location, morphology, and topography. Unique morphological features may be identified, but are unlikely to be present in the absence of pit pattern or microvascular pattern changes consistent with SMIC. Subsequently, proceed with IEE (virtual chromoendoscopy or dye chromoendoscopy, dependent on the availability of appropriate technology and local expertise). Although not our practice, if features of SMIC are identified with virtual chromoendoscopy, there may be a role for supplementary dye chromoendoscopy as is currently advocated by some European and Japanese experts.
Regardless of the IEE modality used, rigorous photo-documentation is critical and each phase of optical evaluation should be captured. Optical evaluation should be performed both in anterograde and retroflexed positions, where feasible.
Given the morphological- and topographical-specific frequencies of SMIC alongside the performance of optical evaluation in large LSLs, in flat (Paris 0-IIa, Paris 0-IIb) lesions without optical features consistent with SMIC, EMR should be the primary resection modality. In lesions with optical features consistent with S-SMIC, ESD should be considered dependent on local availability and expertise. In lesions with normal optical evaluation but a heightened risk of covert SMIC, ESD should be considered as well. If features of D-SMIC are detected, these lesions should be referred to surgery or multi-disciplinary team discussion.
Artificial intelligence
Artificial intelligence (AI) carries the potential to mitigate the operator-dependent limitations of optical evaluation for SMIC diagnosis and quantifying depth of invasion.37–40 However, the predominant focus has been the management of diminutive polyps so as to implement the American Society for Gastrointestinal Endoscopy (ASGE) preservation and incorporation of valuable endoscopic innovations (PIVI) “resect and discard” and “diagnose and leave” strategies. 41
Using a consecutive image dataset of 5843 endocytoscopy images (375 lesions), Takeda and colleagues developed a machine learning AI platform to differentiate benign and invasive colorectal neoplasia. 42 Endocytoscopy is a form of IEE which allows for x380 magnification and allows for in vivo assessment of both nuclei and gland lumens. In a test set of 200 images, 94% were assessable by the endocytoscopy AI-platform. Sensitivity, specificity, accuracy, PPV, and NPV to distinguish between invasive and benign lesions were 89.4%, 98.9%, 94.1%, 98.8%, and 90.1%, respectively. Similar to traditional magnification, endocytoscopy is neither widely available nor used by western endoscopy centers.
Conclusion
Minimally invasive resection techniques have revolutionized the management of early colorectal neoplasia. However, appropriate technique selection is predicated on the ability of optical evaluation to diagnose invasive disease and stratify by clinically relevant depths of invasion. An appreciation of the risk of SMIC, based on lesion size, location, morphology, and topography is critical to the effective application of optical evaluation. For large LSLs, optical evaluation has shown modest performance characteristics. Therefore, further refinement is needed to optimize its clinical application. Moreover, this emphasizes the importance of understanding and quantifying the risk of covert SMIC alongside the potential for AI to circumvent endoscopist-imposed limitations in the future. The ultimate goal being the creation of a robust selective resection algorithm for the colorectum.
Footnotes
Author contributions
Drafting of the article: Neal Shahidi; Critical revision of the article for important intellectual content: Sergei Vosko, W. Arnout van Hattem, Mayenaaz Sidhu, Michael J. Bourke; Final approval of the article: Michael J. Bourke
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Neal Shahidi is supported by the University of British Columbia Clinician Investigator Program.
Conflict of interest statement
Michael J. Bourke: Research Support: Olympus Medical, Cook Medical, Boston Scientific.
The remaining authors have no conflicts of interest to disclose.
