Abstract
Background/Aims:
Fecal incontinence (FI) is a common, debilitating condition that causes major impact on quality of life for those affected. Non-surgical treatment options include anorectal biofeedback therapy (BF) and percutaneous tibial nerve stimulation (PTNS), usually performed separately. The aims of the current study were to determine the feasibility, tolerability, safety, and efficacy of performing a combined BF and PTNS treatment protocol.
Methods:
Female patients with urge FI were offered a novel pilot program combining BF with PTNS. The treatment protocol consisted of 13 weekly sessions: an educational session, followed by 5 combined BF and PTNS sessions, 6 PTNS and a final combined session. Anorectal physiology and clinical outcomes were assessed throughout the program. For efficacy, patients were compared with BF only historical FI patients matched for age, parity, and severity of symptoms.
Results:
A total of 12/13 (93%) patients completed the full program. Overall attendance rate was 93% (157/169 sessions). Patient comfort score with treatment was rated high at 9.8/10 (SD 0.7) for PTNS and 8.6/10 (SD 1.7) for the BF component. No major side effects were reported. A reduction of at least 50% in FI episodes/week was achieved by 58% of patients by visit 6, and 92% by visit 13. No physiology changes were evident immediately following PTNS compared with before, but pressure during sustained anal squeeze improved by the end of the treatment course. Comparing outcomes with historical matched controls, reductions in weekly FI episodes were more pronounced in the BF only group at visit 6, but not week 13.
Conclusions:
In this pilot study, concurrent PTNS and anorectal biofeedback therapy has been shown to be feasible, comfortable, and low risk. The combined protocol is likely to be an effective treatment for FI, but future research could focus on optimizing patient selection.
Introduction
Fecal incontinence (FI) is a chronic, debilitating condition with enormous consequences for both patients and the community. 1 Since FI is believed to be a heterogeneous disorder, initial management is symptom based. 2 Correction of any underlying abnormal stool form and/or frequency, if present, is the first step. Additional conservative therapies include dietary, medical, and psychological modifications, which are estimated to benefit a quarter of patients. 3 When a conservative approach fails, treatment with anorectal biofeedback (BF) is often recommended. A multitude of prospective and randomized trials have consistently shown short and long-term improvement in around 60% to 70% of patients treated with BF. 4
Neuromodulation is an effective therapeutic modality in the treatment of patients suffering from pelvic floor disorders. 5 For patients with FI who fail BF, the only well-established option at present is neuromodulation by an implantable sacral nerve stimulator. 4 This treatment is invasive, with potential for significant complications, and is associated with substantial costs. 6 Therefore, there is a need for a less invasive and more economical method of performing neuromodulation. Percutaneous tibial nerve stimulation (PTNS) has the potential to address these disadvantages. With this method, neuromodulation is delivered through a disposable needle electrode inserted adjacent to the posterior tibial nerve at the CEPHALAD to the medial mallelous of the ankle. Although PTNS is a well-proven and recognized treatment for urge urinary incontinence, 7 in the last decade its role in treating patients with FI is slowly being elucidated. A systemic review, including six prospective studies of PTNS for treating FI, suggested PTNS results in significant improvements in 50–70% of patients. 8 Subsequently, two randomized controlled trials comparing PTNS with sham treatment yielded conflicting results,9,10 although subgroup analysis in the negative trial has suggested benefit in patients with urge FI. In all of these studies, patients were required to fail conservative management, but not specifically anorectal biofeedback, before undergoing neuromodulation, and in none of the studies was PTNS evaluated in combination with BF.
We thus hypothesized that the combination of PTNS and BF for treating patients with FI, especially patients with urge incontinence, would be a valuable addition to the current standard of care of BF treatment only. Our aims, therefore, were (a) to determine the feasibility and safety of performing combined BF therapy and PTNS, (b) to assess for immediate and short-term changes in anorectal physiology following neuromodulation, and (c) to compare outcomes with patient controls undergoing BF alone.
Methods
Patients
A prospective, single-arm, non-randomized, pilot study was performed in the Neurogastroenterology Unit, Royal North Shore Hospital. Consecutive patients with FI referred for BF completed the Rome III Integrative Questionnaire, 11 the Hospital Anxiety and Depression (HAD) scale, 12 the Fecal Incontinence Severity Index (FISI) 13 and a symptom-based questionnaire. After digital rectal examination, patients underwent anorectal manometry (Dentsleeve International, Canada) and balloon expulsion testing (4 mm intravenous tube with a party balloon tied at the end) as previously described in detail. 14 If clinically indicated, further testing including anorectal ultrasound, defecating proctogram and/or colonoscopy were performed to rule out a structural/organic cause for the patients’ symptoms.
Inclusion criteria for the study included (a) female gender, (b) age > 18 and <80 (c) fecal incontinence, defined as an uncontrolled passage of fecal material occurring at least 2–4 times a month, in the last 6 months, (d) urge symptoms: defined as having an urge sensation before the uncontrolled passage, on most occasions, (e) failure of a trial of diet and bulking or laxative therapy in an effort to restore normal bowel movements, (f) a minimum score of 8 on the FISI score, and (g) willingness to give written informed consent and willingness to comply with the study protocol.
Exclusion criteria included (a) presence of an overt organic anorectal disorder, (b) a medical condition or medication that may compromise PTNS/BF treatment, including hypercoagulability state or anti-coagulation medication, latex allergy, implanted stimulator, pacemaker or defibrillator, sciatica symptoms, peripheral neuropathy or peripheral vascular disease, (c) a history of psychological illness or condition such as to interfere with the patient’s ability to understand the requirements of the study, (d) women lactating, pregnant, or of childbearing potential who were not willing to avoid pregnancy during the study, and (e) previous anorectal biofeedback training or neuromodulation.
Patients who satisfied these criteria were given an oral and written explanation detailing the BF and PTNS procedures and trial, and offered participation in the study. All patients included in the study gave written informed consent. The study was approved by the Local Area Human Research Health Committee (reference number HREC/15/HAWKE/428) and was registered on the Australian New Zealand Clinical Trials Registry (reference number ACTRN12616000056493).
Anorectal biofeedback treatment
All patients received anorectal BF training according to a pre-defined protocol based on the American Neurogastroenterology and Motility Society and the European Society of Neurogastroenterology and Motility (ANMS-ESNM) recent consensus statement. 15 This consisted of six 30–60 min weekly sessions, four instrumented, with a gastroenterologist-supervised nurse specialist. The protocol consisted of (a) education regarding the anatomy of normal defecation, (b) advice on correct toilet positioning, (c) diaphragmatic breathing with manometric feedback aiming to achieve normalization of rectal pressure on strain, and use of diaphragmatic breathing for urge resistance training, (d) manometric and surface electromyography (Neomedix, Sydney, Australia) based biofeedback for quick, sustained and half maximum anal squeezes aimed to improve amplitude and duration of anal squeeze pressure, (e) manometric-based biofeedback aiming to improve rectoanal co-ordination with synchronous rise in rectal pressure with anal sphincter relaxation, (f) rectal sensory training, and (g) balloon expulsion training where appropriate. A reinforcement BF training session was performed at week 13 of the study.
PTNS (Endotherapeutics Pty Ltd, Sydney, Australia) was performed in 12 consecutive weekly sessions. Following the insertion of a needle electrode adjacent to the posterior tibial nerve cephalad to the medial malleolus of the ankle, the maximum treatment intensity was determined by increasing the stimulus intensity slowly until the patient’s great toe began to curl or until a tingling sensation was perceived in the medial aspect of the foot. Each PTNS treatment session lasted 30 min at a sub-maximal stimulation level, with the patient in the semi-supine position.
Following the screening visit, week 1 visit of the study was dedicated to educational aspects of the BF and PTNS treatments, informed consent, and advice regarding medication use and toileting habits (supplementary Table 1). On weeks 2 to 6 and week 13 visits, BF training was performed followed by PTNS. On weeks 7 to 12 visits, patients received PTNS treatment alone. On week 2,5,9 and 13 visits, anorectal manometry (ARM) studies were performed immediately before and after the 30 min of PTNS treatment. At the end of treatment, all patients were offered off-study, optional, reinforcement PTNS sessions at 3 months from end of study and thereafter at 6 month intervals. A follow up combination PTNS and BF visit at 12 months was also offered.
Outcome measures
At week 1 (baseline), 6 and 13 (end of study) visits, all patients filled out the FISI, a quality of life questionnaire (SF36) 16 and a 10 cm visual analogue scale (VAS) for (a) global bowel satisfaction (score anchors: 0 = completely unsatisfied; 10 = completely satisfied), (b) impact of bowel dysfunction on quality of life (score anchors: 0 = no impact; 10 = most impact) and (c) control of bowel functions (score anchors: 0 = no control; 10 = most control). Before treatment, VAS for willingness to participate in the therapeutic course (score anchors: 0 = not willing; 10 = very willing) and following treatment VAS for comfort with treatment (score anchors: 0 = most uncomfortable; 10 = most comfortable) were also completed. Anorectal manometry with sensory testing was performed on the screening visit and repeated on visits 2,5,9 and 13 immediately before and after PTNS treatment.
Our primary outcome measures for feasibility were completion rate, percentage of visit attendance, and VAS score for ‘patient comfort with treatment’ for BF and PTNS at visits 6 and 13. For our secondary objectives, physiological ARM measurements before and after PTNS treatment at week 2,5,9 and 13 visits were compared, as well as changes during the treatment compared with baseline. Lastly, for assessing efficacy, outcome measures of change in frequency of FI episodes, FISI and VAS scores were compared with a group of age and gender-matched historical controls undergoing standard BF without PTNS in the same Unit.
Sample size requirements
Although designed as a pilot study, sample size requirement was determined a priori, for the primary contrast, to be
Statistical analysis
With the exception of the analysis of changes in physiology within visits, comparisons within groups over time were carried out using Wilcoxon signed-ranks tests. For the analysis of physiology within visits, a series of repeated-measures analysis of variance (ANOVAs) were conducted. The predictor variable in each ANOVA was visit number (2
Results
Patients
A total of 29 consecutive female patients with urge FI presenting for BF treatment were screened. In total, 12 patients were excluded (1 recurrent rectal prolapse surgery, 1 peripheral vascular disease, 1 peripheral neuropathy, 1 sciatica, 1 myasthenia gravis, 1 low anterior resection and radiotherapy, 5 with frequency of FI episodes not meeting minimal requirements for the study, and 1 with no consistent urge symptoms on specific questioning). Of the remaining 17 patients, 13 agreed to participate in the study (Figure 1).
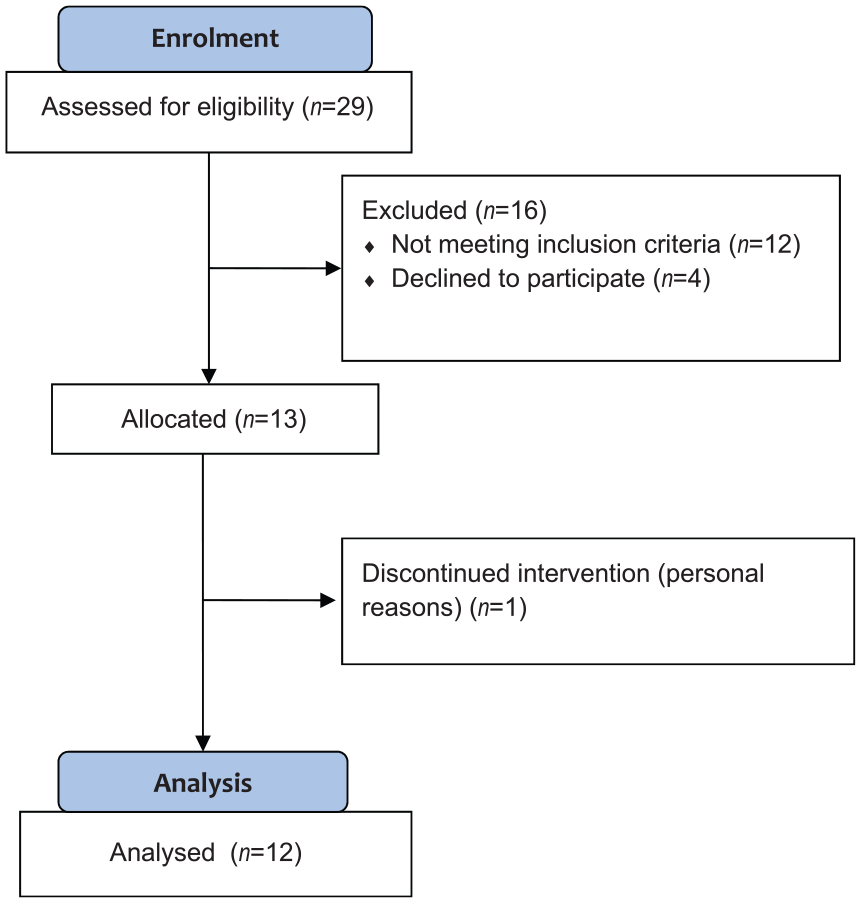
Participant flow diagram.
Thus, 13 patients (mean age 54, SD 14) were enrolled, all female patients with urge FI. Average FI episodes/week was 2.8 (SD 2.2), with 11/13 patients reporting 2 or more FI episodes/week. Mean FISI score was 30 (SD 10) and duration of FI symptoms was 11 years (SD 10). In 54% of patients, previous obstetric trauma was noted (3rd degree tear or instrumented delivery), and a single patient had concomitant constipation according to Rome criteria. In total, seven patients had elevated (>7) HAD anxiety scores, but none had elevated HAD depression (all <7).
Feasibility, safety, and tolerance of combined treatment
A total of 12 patients (93%) completed the full program. A single patient withdrew from the study after visit 3 due to personal reasons, and follow up data was not available for this patient. Overall attendance rate, including the withdrawn patient, was 93% (157/169 sessions). Mean treatment amplitude used for PTNS was 5 mA (SD 3). At week 6 visit, patients rated VAS comfort with combined treatment as 8.8/10 (SD 1.8) and on week 13 final visit, mean comfort score was rated as 9.8/10 (SD 0.7) for PTNS and 8.6/10 (1.7) for the BF component. No major side effects were reported.
Changes in physiology: immediately before and after PTNS and between visits
Within visits, anorectal physiology measured immediately after PTNS did not differ from values immediately before at all time-points measured (week 2,5,9 and 13 visits). Comparing physiology measures at week 5,9 or 13 with baseline, no changes in anal sphincter resting, squeeze, or cough pressures were seen. Some improvement was seen in the anal sustained pressure at end of treatment visit 13 compared with baseline (Table 1).
Symptom severity, quality of life measures and anorectal physiology parameters during combined treatment with anorectal biofeedback (BF) and percutaneous tibial nerve stimulation (PTNS) (

Visual analogue scale ranging from 0 to 10, with lower scores indicating decreased severity.
Visual analogue scales ranging from 0 to 10, with higher scores indicating decreased severity.
FI, fecal incontinence; NA, not available.
Efficacy of treatment
Changes in symptom severity, quality of life measures, and physiology during treatment course are shown in Table 1. There was significant improvement in FISI, VAS quality of life, control, and satisfaction at week 6 visit (
12 months follow-up
In total, 8 of the 12 patients completing combination treatment were available for 12-month follow up. A total of five of these eight patients performed reinforcement PTNS sessions as offered following completion of the 13-week treatment phase. One patient underwent sacral nerve stimulator (SNS) implantation due to incomplete resolution of symptoms. Two patients who were improved at the end of treatment were more symptomatic again at 12 months, although one of these patients received reinforcement PTNS sessions. The remaining five patients (63%) displayed sustained benefit at 12 months, with all but one of these patients arriving for all PTNS reinforcement sessions.
Improvements in FISI and VAS scores for impact on quality of life, control, and satisfaction with bowel movements were also maintained at 12 months, but although the average number of weekly FI episodes was numerically lower, this was non-significant compared with baseline.
Comparison with historical controls
The 12 patients completing treatment were compared with 24 female patients with urge FI who recently completed BF as a stand-alone treatment for their symptoms. Similar to patients in this study, these patients underwent 6 weekly BF sessions and a follow up reinforcement BF session at week 13, according to an identical standardized BF protocol. Study patients were matched in 1:2 design with age, parity, and severity matched patient controls.
Baseline characteristics of the two groups are compared in Table 2 and outcomes of the two groups are compared in Table 3. At baseline, duration of symptoms was shorter in the matched controls (5
Baseline demographics, clinical and psychological features of patients undergoing percutaneous tibial nerve stimulation with anorectal biofeedback (PTNS +BF)
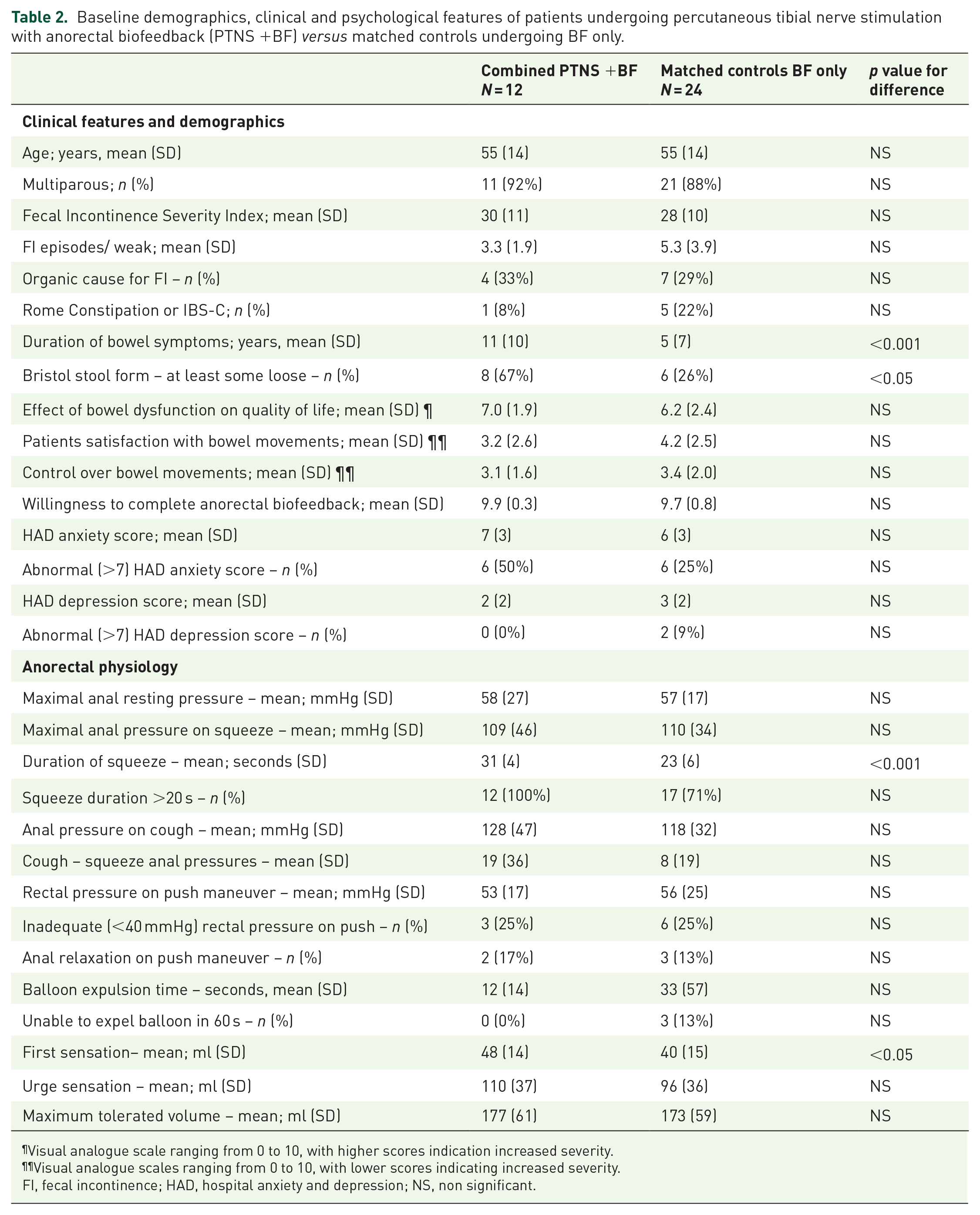
Visual analogue scale ranging from 0 to 10, with higher scores indication increased severity.
Visual analogue scales ranging from 0 to 10, with lower scores indicating increased severity.
FI, fecal incontinence; HAD, hospital anxiety and depression; NS, non significant.
Changes during and after biofeedback (BF): percutaneous tibial nerve stimulation with anorectal biofeedback (PTNS +BF)
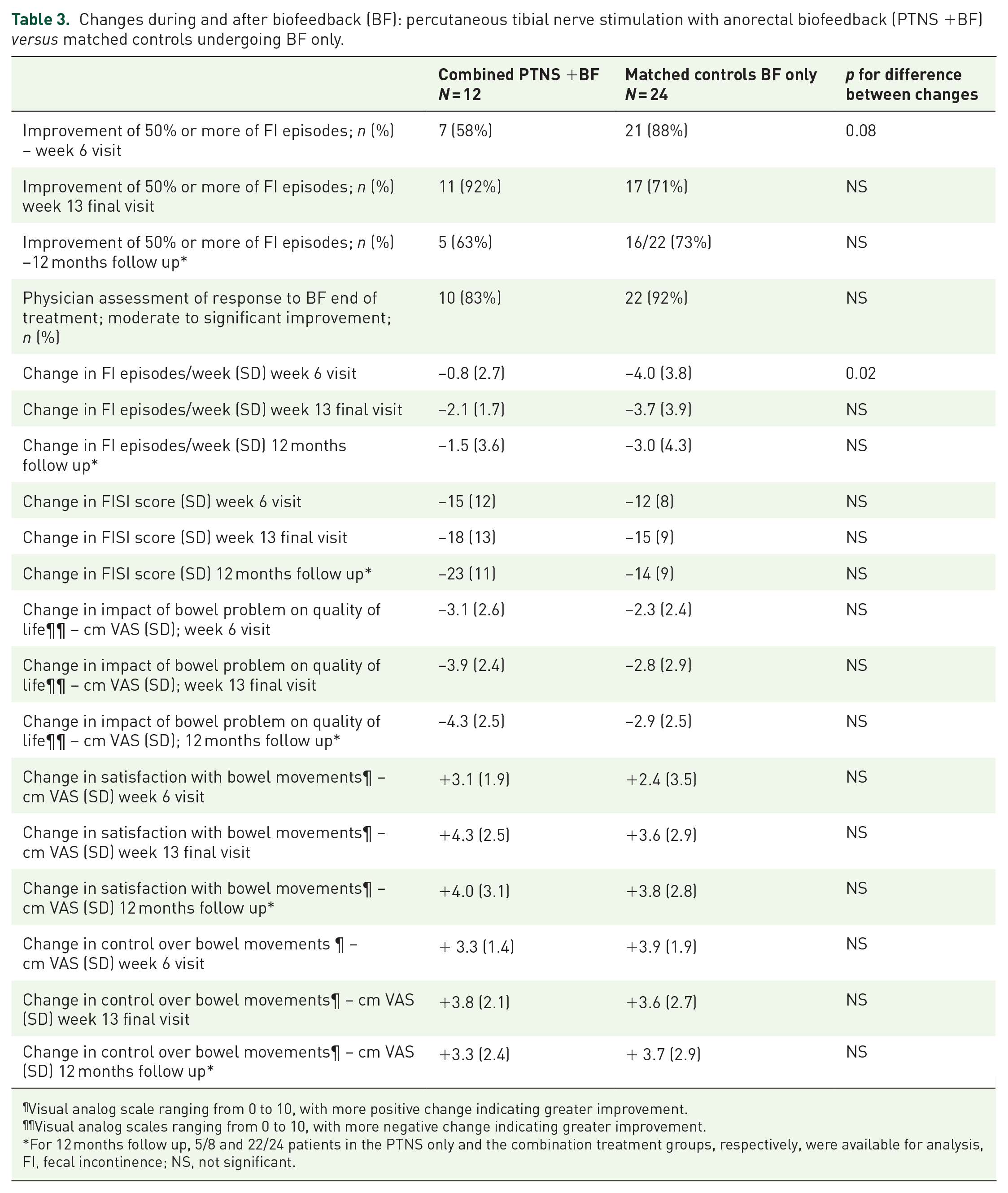
Visual analog scale ranging from 0 to 10, with more positive change indicating greater improvement.
Visual analog scales ranging from 0 to 10, with more negative change indicating greater improvement.
For 12 months follow up, 5/8 and 22/24 patients in the PTNS only and the combination treatment groups, respectively, were available for analysis,
FI, fecal incontinence; NS, not significant.
Discussion
In this pilot, non-randomized, single-arm study, the feasibility and safety of an augmented biofeedback protocol combining PTNS and BF for treating female patients with urge fecal incontinence is demonstrated. These primary outcomes were met with a 93% completion rate, 93% session attendance rate, no major adverse events, and a high level of patient reported comfort with PTNS (9.8/10), BF (8.6/10), and the combination of the two modalities (8.8/10). This is despite the relatively cumbersome visits required in the study protocol and the recurrent needle placement and anorectal studies performed during the visits.
In an effort to understand the mechanism of neuromodulation by PTNS, anorectal manometry and sensory testing was repeated on four of the treatment sessions, both immediately before and after PTNS. No immediate changes in any of the measurable variables was detected. This is in keeping with studies performed in patients with sacral nerve stimulators, where a lack of physiology changes have been seen in the ‘on’ and ‘off’ mode of stimulation. 17 In addition in our study, no significant changes were seen in post-treatment physiology as compared with baseline, with the exception of a higher sustained squeeze pressure. This, however, is in contrast to some previous reports in both BF and PTNS. 18 As the variables most likely to improve following BF, for example anal squeeze pressure, 19 and following PTNS, for example anal resting pressure and rectal sensory volumes, 18 were numerically improved in our study but did not reach statistical significance the likely explanation for this finding is the small patient numbers. As physiological changes were defined as a secondary endpoint in the current study, power to detect change was limited, and it is possible that including more patients would replicate previous reports of some physiological improvements. It is interesting to speculate that with more sensitive physiology testing, differences may emerge, and future studies could include sensory testing with a barostat,20,21 motor evoked potentials 22 or measures of CNS function such as positron emission tomography.23,24
It is of interest that compared with the standard BF protocol, patients in the augmented protocol continued to improve after week 6, with the maximal benefits attained at week 13. This is similar to previous reports of the time line of improvement in PTNS. 18 Nevertheless, data regarding efficacy in this trial should be interpreted with caution, as this was a small cohort, with no sham treatment arm, in a selected group of patients. This probably explains the unusually high rates of success at the end of treatment (92%), even as assessed by the objective measure of 50% or more improvement in FI episodes. A better reflection of the true efficacy would perhaps be the follow up data, where 63% of patients available for follow up still had a successful outcome at 12 months, which is more in line with other publications.19,25 Comparison with matched historical controls did not a find a difference in response between groups. Although matched for age, parity, and severity, baseline differences between patients and controls, such as numerically higher frequency of weekly FI episodes, higher symptom duration, and higher frequency of loose stools, might account for some disparities in outcome measures. Furthermore, this comparison is underpowered to detect small but perhaps meaningful advantages of the augmented protocol. Still, a power calculation accounting for a 70% success rate of BF as a stand-alone therapy for FI and assuming a minimal clinically significant benefit of additional 10% by adding on PTNS, results in large (>250 in each group) patient numbers needed to prove this benefit. Such a future clinical trial is probably unrealistic.
The question stands as to which patients could or should be offered this augmented combination treatment protocol. In the current study, women with urge FI were included, making it reasonable to offer the protocol to these patients in the future. In addition, the recent
In conclusion, PTNS and anorectal biofeedback therapy can be performed concurrently and is feasible, comfortable, and low risk and the combined course is likely to be an effective treatment for FI. Potential benefits and appropriate patient selection for this combined treatment remain to be established. As with sacral nerve stimulation, the physiological mechanisms of action of PTNS remain unclear and deserve further study.
Supplemental Material
PTNS_Supplementary_Table_1 – Supplemental material for A novel combined anorectal biofeedback and percutaneous tibial nerve stimulation protocol for treating fecal incontinence
Supplemental material, PTNS_Supplementary_Table_1 for A novel combined anorectal biofeedback and percutaneous tibial nerve stimulation protocol for treating fecal incontinence by Yoav Mazor, Gillian M. Prott, Carol Sequeira, Michael Jones, Anastasia Ejova, John E. Kellow, Margaret Schnitzler and Allison Malcolm in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
YM, AM and MS planned the study. GMP and CS performed all studies. YM, GMP and CS collected all data, MJ, AE and YM conducted statistical analysis and YM, MS, MJ, AM and JEK interpreted the data and jointly drafted the manuscript. All authors reviewed and approved the final manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Colorectal Society of Australia and New Zealand (CCANZ) and the Kolling Institute (Douglas Piper Award)
YM received an IPRS/APA Australian government scholarship for his studies at the University of Sydney.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
