Abstract
Portal hypertension represents one of the major clinical consequences of chronic liver disease, having a deep impact on patients’ prognosis and survival. Its pathophysiology defines a pathological increase in the intrahepatic vascular resistance as the primary factor in its development, being subsequently aggravated by a paradoxical increase in portal blood inflow. Although extensive preclinical and clinical research in the field has been developed in recent decades, no effective treatment targeting its primary mechanism has been defined. The present review critically summarizes the current knowledge in portal hypertension therapeutics, focusing on those strategies driven by the disease pathophysiology and underlying cellular mechanisms.
Introduction
Advanced chronic liver disease (CLD) represents the fourth cause of death due to noncommunicable diseases worldwide. Its high mortality is mainly due to long-time asymptomatic stages during disease progression and, thus, is diagnosed after appearance of portal hypertension (PH) and its clinical complications such as hepatic encephalopathy, variceal hemorrhage or ascites. In fact, PH is the major driver of the complications of CLD, the measurement of hepatic venous pressure gradient (HVPG) being a predictor of the risk of clinical decompensation and survival in compensated cirrhotic patients.1,2
Clinically significant PH is defined as an HVPG of 10 mmHg or greater and depends on changes in portal blood flow or in intrahepatic vascular resistance (IHVR). During cirrhosis development, elevation in IHVR due to deregulation of hepatic cells and liver fibrosis is the primary factor involved in increasing portal pressure (PP) leading to secondary splanchnic vasodilation and extrahepatic shunt formation, which further aggravate PH and liver dysfunction. Notably, current treatments do not target the main underlying mechanism, but consist in extrahepatic vasoconstrictors [i.e. nonselective beta blockers (NSBB) and somatostatin] aimed at ameliorating PH by reducing the hyperdynamic circulation, or in strategies focused on the prevention of PH-derived complications such as infections or encephalopathy. In the specific case of NSBB, it should be noted that carvedilol may also exert intrahepatic vasodilatation due to its anti-α1-adrenergic properties. 3 Despite the extended use of these drugs as the most effective treatment for PH, a recent analysis based on over 100,000 cases showed that 30-day mortality following a decompensation in cirrhosis remains equal, or even higher, than that observed 10 years ago, 4 strongly suggesting there is a need for new therapeutic approaches for PH and CLD.
In this review we summarize the potential therapeutic strategies for the treatment of PH, which importantly derive from preclinical knowledge and mainly target the increased IHVR. These approaches consist of pharmacological, cell-derived or lifestyle interventions, as shown below.
Vasoprotective strategies
CLD is characterized by persistent injury of the liver due to different etiologies, including alcoholic and nonalcoholic steatohepatitis (NASH) or viral infections. This in turn leads to alterations of the hepatic microvasculature, which in combination with fibrosis constitute the main components of increased IHVR. This scenario of vascular alterations has led to the assessment of different vasoprotective strategies for the treatment of intrahepatic-derived PH, mainly vasomodulators, anticoagulants and antiangiogenic approaches.
Vasomodulators
One of the causes of the abovementioned microvascular alterations during CLD is a shift/alteration in the synthesis of vasoactive molecules in the liver, where vasoconstrictors prevail over vasodilators. Thus, several vasomodulator strategies aimed at enhancing intrahepatic vasodilation and normalizing excessive vasoconstriction have been recently proposed (Figure 1).
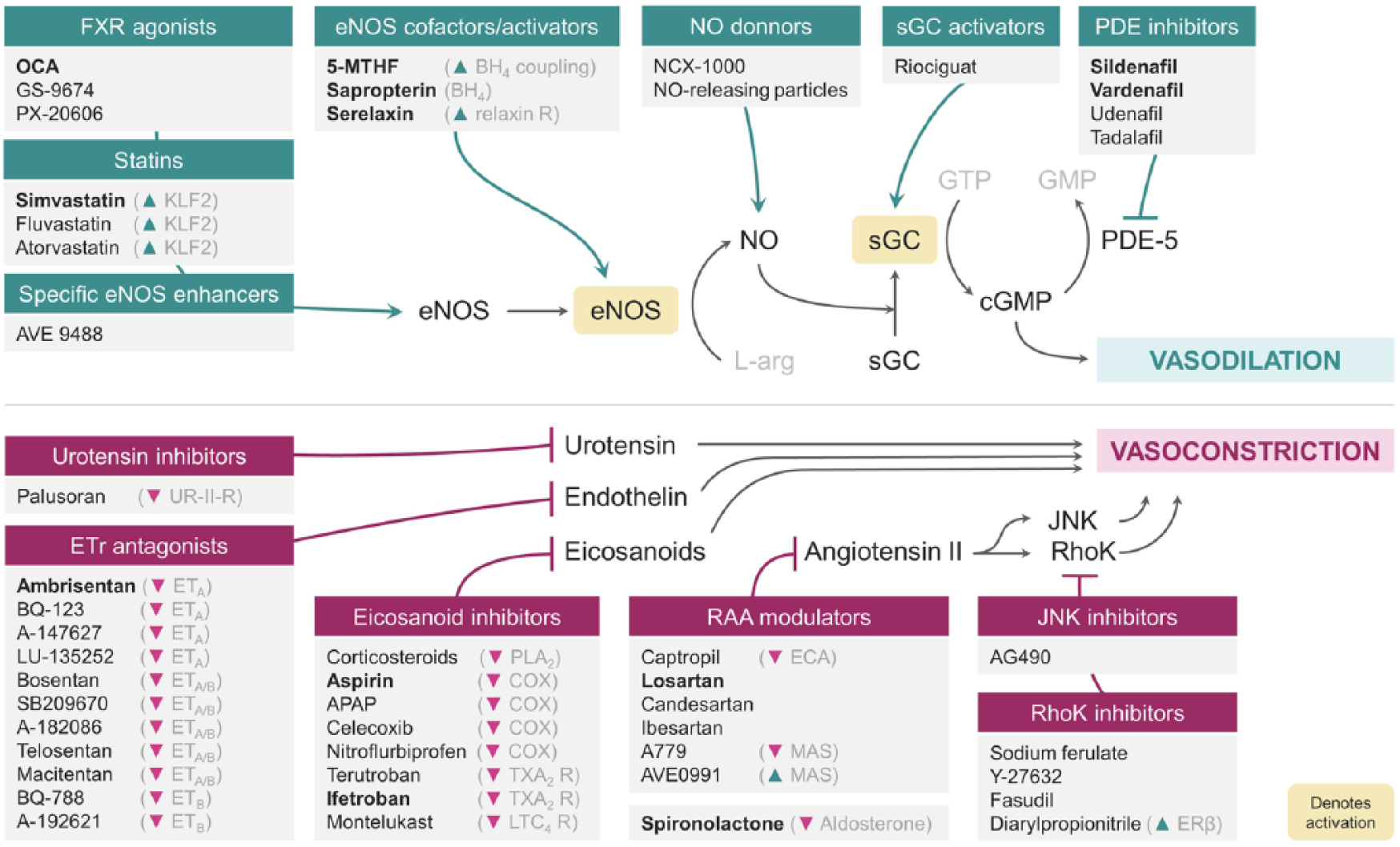
Schematic representation of different therapeutic approaches for portal hypertension focused on their liver vasomodulation effect.
Inhibition of vasoconstriction
One of the most potent vasoconstrictors that contributes to microvascular dysfunction in CLD is endothelin. This molecule is synthesized by the activated endothelium and acts as a ligand for the endothelin receptors (ET) on hepatic stellate cells (HSCs), which are the final effectors of vasoconstriction. Indeed, the use of antagonists targeting ET (either ETA, ETB or both) showed improvements in the hepatic sinusoid and in liver fibrosis in preclinical models5,6 and decreased PP in a small clinical study. 7 However, its possible beneficial effects were not further validated in human randomized-controlled trials (RCTs).8,9 Currently ongoing human RCTs evaluating the unspecific antagonist macitentan 10 and ambrisentan 11 will provide essential information to support or discard this therapeutic strategy in cirrhosis and PH.
Urotensin is another vasoconstrictor playing a role in PH, known to correlate with HVPG levels. 12 Similar to ET, blockage of the urotensin II receptor with the antagonist palosuran decreases PP in experimental models. 13 Nevertheless, there are no current studies on cirrhotic patients in order to validate the translatability of these results to the bedside.
Eicosanoids represent another important family of vasoconstrictors derived from the conversion of arachidonic acid. Indeed, cyclooxygenase (COX)-derived prostanoids (a subclass of eicosanoids) are overexpressed in the cirrhotic liver.14,15 In this regard, several drugs have been used to chronically inhibit either their synthesis (i.e. by COX inhibition with nitroflurbiprofen 16 or celecoxib17,18) or to block their receptor (e.g. thromboxane A2 receptor with terutroban 19 ). Although these strategies decreased PP in preclinical models of cirrhosis, there is only one ongoing RCT with the thromboxane receptor antagonist ifetroban. 20 Similar strategies aimed at reducing arachidonic acid metabolites focused on inhibiting epoxygenases using MS-PPOH 21 and cysteinyl leukotrienes with montelukast. 22
In addition to the increased synthesis of vasoconstrictors, the cirrhotic liver is also hypersensitive to these molecules. Thus, research aimed to inhibit the effect of not only hepatic vasoconstrictors, but also systemic ones. In this regard, the renin–angiotensin–aldosterone (RAA) system is a hormone system that controls blood pressure and fluid balance, playing also an important role regulating hepatic hemodynamics. Importantly, angiotensin II is a potent vasoconstrictor peptide with hepatic effects that favors the development of PH. 23 Several strategies have been developed to reduce its effects on the liver: angiotensin-converting-enzyme inhibitors (captopril) and angiotensin receptor blockers (losartan, candesartan, irbesartan) have been tested in preclinical and clinical settings and showed beneficial effects on reducing PH but mainly in compensated patients with Child A cirrhosis. 24 Given the potential of these drugs, some strategies aimed at focusing on the RAA system downstream pathway. Indeed, the Janus kinase inhibitor AG490 showed a PP-lowering effect additive to propranolol in preclinical models,25–27 while the Rho-kinase inhibitors Y-27632 28 and fasudil 29 showed comparable beneficial effects. Indeed, RhoA modulation using sodium ferulate 30 or diarylpropionitrile 31 showed beneficial effects in preclinical PH, and interestingly, pointed out the relevance of the latter (which is an estrogen-receptor β agonist) in postmenopausal women. On the other hand, the receptor Mas also represents an alternative downstream target of the RAA pathway. Its activation showed promising effects in experimental models of cirrhosis, also reducing PP and liver fibrosis. 32 Nevertheless, another study blocking the Mas receptor with A779 led to the same decrease in PP, 33 leaving the Mas receptor role in a paradigm.
Vasopressin, also named antidiuretic hormone, is a hormone that contributes to the regulation of water and electrolyte homeostasis and constricts arterioles, increasing vascular resistance and blood pressure. The blockage of vasopressin using conivaptan has been recently tested in an RCT and showed no beneficial effects on PP. 34 Indeed, previous data support the concept that vaptans may not only be ineffective but also have negative secondary effects in terms of morbidity and mortality in patients with cirrhosis.35,36 Partial vasopressin agonists in the mesentery may have better projection. Indeed, a recent study demonstrated beneficial effects of FE 204038 reducing PP in cirrhotic rats, 37 and a recently started RCT evaluates the compound FE 204205 in cirrhotic patients with PH. 38
Promotion of vasodilation
Nitric oxide (NO) is a potent vasodilator synthesized by the NO synthase (NOS) that regulates vascular tone and inflammation; however, it becomes markedly deregulated in the cirrhotic liver. Several therapeutic approaches focused at increasing its levels, either by enhancing NO production (NOS-dependent strategies), or by providing NO as an exogenous source or decreasing NO degradation (NOS-independent strategies).
Delivering NO to precise cellular locations, like the liver sinusoid, represents a challenge. However, nanoparticle delivery has lately become an important and emerging field. In fact, recent studies using NO-releasing nanoparticles coated with vitamin A, which is mainly stored in HSCs in the liver, reduced PP in a preclinical cirrhotic model. 39 Similar effects were observed when NO synthesis was enhanced: the NOS cofactor tetrahydrobiopterin (sapropterin) and the NOS transcription enhancer (AVE 9488) decreased PP in cirrhotic animal models,40,41 nevertheless no benefits were observed in an RCT evaluating the first. 42 In this same strategy, a currently ongoing RCT will describe the effectiveness of serelaxin, a drug that has been used for the treatment of acute heart failure due its capability to activate the relaxin receptor and NOS. 43
The soluble guanylate cyclase (sGC) is the only known receptor for NO and represents its downstream mediator. When sGC is activated by NO, it induces the formation of the intermediate cyclic guanosine monophosphate (cGMP), which confers potent vasodilation. In this regard, the stimulation of the nonoxidized form of sGC or the oxidized and free-sGC form may represent novel approaches for activating cGMP production and subsequently ameliorate the vasoconstrictor environment. In fact, a recent study using the nonoxidized and free-sGC stimulator riociguat showed improved liver fibrosis and PP in cirrhotic animals. 44 However, no studies have evaluated the potential of activators of the oxidized form of sGC for the treatment of PH. Considering that oxidative stress plays a key role in cirrhosis and PH, and that sGC activators are able to generate cGMP even in these detrimental conditions, 45 their usefulness as a therapeutic option for PH deserves further investigation.
In addition to sGC strategies, there are other approaches aimed at potentiating the effects of cGMP by preventing its degradation by phosphodiesterase-5 (PDE-5). Indeed, different PDE-5 inhibitors showed lowering effects on PP in cirrhotic patients.46–48
Transcriptional modulators
In addition to specific modulators of vasoactive molecules, other strategies rely on the modulation of transcription factors that regulate the expression of many of these proteins. For example, farnesoid X receptor (FXR) is a bile-acid-responsive transcription factor highly expressed in the liver that controls the expression of many genes involved in metabolic regulation, hepatic fibrosis, vascular homeostasis and inflammation, and thus it has been considered a promising therapeutic target in CLD. Obeticholic acid (OCA), a semisynthetic FXR agonist, has been tested in preclinical models of cirrhosis showing beneficial effects on PH by reducing the IHVR. 49 Indeed, different ongoing clinical trials are investigating its effects in advanced CLD,50–52 including patients with NASH cirrhosis.53,54 This drug, however, has raised some cases of overdose-related toxicity in patients with severe or even mild liver dysfunction, so its dosage needs to be carefully adjusted. 55 In addition to OCA, new nonsteroidal FXR agonists have been developed to evade enterohepatic recirculation. These drugs (e.g. PX201606, GS-9674), have been tested in animals with CLD showing increased endothelial NOS expression, reduced inflammatory response and improved liver injury.56,57 The effects of these newly formulated compounds on advanced CLD and PH require further investigation.
On the other hand, statins are lipid-lowering drugs that have shown strong hepatosinusoidal protective effects in preclinical models of CLD, ultimately leading to PP decrease.58–60 Underlying mechanisms of statins mostly rely on their capability to induce the expression of the transcription factor Kruppel-like factor 2 that leads to liver sinusoidal endothelial cell (LSEC) phenotype amelioration, HSC deactivation, and global improvement in hepatic injury.61–66 Encouraged by the preclinical results, several clinical studies have tested statins in PH-cirrhotic patients. Recent analyses have shown that statins are associated with decreased risk of decompensation, death and hepatocellular carcinoma development.67,68 Moreover, simvastatin has shown beneficial effects when administered alone 69 or combined with beta blockers. 70 Future and ongoing RCTs will extend our knowledge regarding the use of statins for the treatment of PH. 71
Anticoagulants
It is widely accepted that cirrhotic patients have an imbalance in anti- and procoagulant factors, leading to both increased risk of bleedings and microthrombosis in the liver, which may in turn increase liver hypoxia and inflammation. 72 Therefore, different anticoagulants have been lately tested in the setting of CLD with success in the prevention of hepatic fibrosis. 73 Indeed, the direct antithrombin enoxaparin has been shown to reduce PP and liver fibrosis in experimental CLD, 74 confirming at the bench side its clinical benefits on cirrhosis decompensation and survival. 75 A different anticoagulant strategy is the direct inhibitor of factor Xa rivaroxaban, which has been shown to reduce liver microthrombosis, HSC activation and PP in experimental models of cirrhosis. 76 Moreover, it is currently being tested in RCTs on patients with cirrhosis 77 or with portal-vein thrombosis. 78 Additional, ongoing RCTs will provide more data on the use of anticoagulants for CLD-patients. 79
Antiangiogenics
Angiogenesis is an important feature in the development of PH, being triggered by hypoxia and inflammation which are present during hepatic fibrogenesis. 80
Vascular endothelial growth factor (VEGF) is the main proangiogenic factor promoting neovascularization in the mesentery and contributes to the development of increased splanchnic vasodilation. Therefore, different studies have focused on blocking VEGF receptors (VEGFr) to decrease splanchnic circulation and improve PP. Initially, monoclonal antibodies and specific inhibitors of VEGFr2 were used in PH animals showing improvement in portosystemic collateral vessel formation but showing no effect on PP.81,82 Subsequent preclinical studies investigated oral tyrosine kinase inhibitors that block VEGFr2, such as sorafenib,83,84 brivanib84,85 and regorafenib 86 and showed improved PP and systemic shunting in cirrhotic and noncirrhotic rats with PH. Indeed, tyrosine kinase inhibitors have also been shown to decrease liver fibrosis. 87 These beneficial preclinical results were confirmed in a small clinical trial where sorafenib significantly improved PH in patients with liver cirrhosis and hepatocellular carcinoma. 88
Apart from VEGF, other molecules such as pigment epithelium-derived factor (PEDF) or vasohibin-1 have been shown as potent endogenous inhibitors of angiogenesis. In fact, overexpression by adenovirus of PEDF or vashoihin-1 resulted in decreased mesenteric angiogenesis, portosystemic shunting, PH and liver fibrosis.89,90 Nevertheless, no current RCTs are being performed following this approach. It is important to highlight that angiogenesis is not only deleterious but also required for repairmen of hepatic tissue during liver fibrosis resolution, therefore total angiogenesis blockade may not be a realistic therapeutic approach.
Cell death, oxidative stress and inflammation
Prevention of cell death
The main factor in the progression of cirrhosis is chronic liver injury. This is a scenario of persistent cell death (due to different etiologies), which leads to excessive inflammation and associated cytotoxicity, and oxidative stress. This sequence of events has led to the hypothesis that pharmacological inhibition of cell death may represent a therapeutic option for the treatment of PH and CLD (Figure 2).

Schematic representation of different therapeutic approaches for portal hypertension based on their effect on apoptosis, oxidative stress or inflammation as main features of chronic liver damage.
Emricasan is an orally active pan-caspase inhibitor that has proven promising potential in the setting of CLD. Indeed, a preclinical study presented as a meeting abstract describes that CCl4-cirrhotic rats receiving emricasan displayed improved PP and IHVR compared with vehicle-treated rats, in addition to improved liver function and microcirculation, and these effects were accompanied by improved LSEC and HSC phenotype and reduced inflammation. 91 More recently, another study in bile-duct-ligated (BDL) mice reported similar effects of this caspase inhibitor. 92 At the bedside, emricasan did not show positive effects in acute-on-chronic liver failure. 93 However, a proof-of-concept clinical study suggests that emricasan ameliorates PP in patients with compensated cirrhosis and severe PH. 94 The completion of a bigger ongoing clinical trial 95 may confirm these promising results in the near future.
These findings are in agreement with other studies assessing the use of caspase inhibitors in chronic liver diseases, such as NASH (reviewed elsewhere 96 ). Of note, inhibition of the apoptosis signal-regulating kinase 1 is another potential candidate strategy in CLD, as it has shown significant results in reducing the fibrosis stage of NASH patients. 97
Despite the exciting results, there is still controversy on the role of cell-death modulation in CLD. Indeed, a recent study suggests that the use of caspase inhibitors would not avoid cell death, as hepatocytes may still die from necrosis. 98 On the other hand, some vasoprotective drugs modulate hepatic fibrosis at least in part due to selective apoptosis of HSCs, 65 while other studies suggest that treatment with low doses of an apoptotic agent (gliotoxin) may achieve similar effects without affecting hepatocyte viability. 99
Future studies will clarify the usefulness of cell-death inhibition in a context where the injury is not present anymore, such as the regression of CLD after the removal of the hepatic injury.
Antioxidants
During liver damage, increased inflammation, mitochondrial damage and enzymatic alterations [i.e. xanthine and nicotinamide adenine dinucleotide phosphate (NADPH) oxidases] induce oxidative stress. Antioxidants are molecules that compensate this oxidative stress by promoting the conversion of reactive oxygen species (ROS) into less reactive molecules, either by direct enzymatic activity or by transcriptional/post-transcriptional modulation of other enzymes. However, cirrhotic livers are known to produce reduced levels of antioxidants compared with control livers, thus further aggravating the increased oxidative stress in this setting.100,101 For this reason, several studies aimed to develop pharmacological antioxidant-enhancing strategies for CLD. One of these is mitoquinone, a mitochondria-targeted antioxidant with oral administration approved for human use. This drug has already proved positive effects in preventing oxidative damage in models of liver diseases such as NASH or ischemia/reperfusion.102,103 In a recent study, treatment with mitoquinone reduced PP and IHVR in two different preclinical models of CLD [CCl4 and thioacetamide (TAA)] by reducing oxidative stress and fibrosis. 101 Importantly, these antifibrotic effects were also validated in precision-cut liver slices from human tissue, suggesting potential applicability in clinical practice. Moreover, a recent abstract reports similar effects of this drug in the BDL rat model. 104
Apocynin is a natural antioxidant of vegetal origin which has a specific inhibitory effect on NADPH oxidase and thus has been studied for its antioxidant properties.105–107 In this regard, a recent study assessed the effects of apocynin in a model of mild fibrosis. 108 Although authors described amelioration of markers of liver damage, oxidative stress, inflammation and fibrosis, these effects were not observed in a previous study using animals with advanced CLD probably due to a progressive reduction in NADPH oxidase activity during cirrhosis development. 109 This advocates for the use of NADPH oxidase inhibitors during progression but not at advanced stages of CLD. Indeed, a study using a new generation of NADPH oxidase inhibitor demonstrated anti-inflammatory and hepatoprotective effects in a preclinical model of NASH without significant fibrosis, 110 altogether encouraging future studies to ascertain which would be the appropriate therapeutic window for this strategy in cirrhosis.
In addition to their protective effects against oxidative stress, antioxidants may also play an important role in ameliorating microvascular dysfunction, as they prevent the scavenging of NO by the superoxide anion (O2−). 100 In this direction, back in 2006, Hernández-Guerra and collegues assessed whether acute administration of ascorbic acid would prevent the increase in HVPG associated with the defective ability of the cirrhotic endothelium to accommodate the postprandial increase in portal blood flow. Indeed, patients receiving vitamin C displayed lower HVPG after a meal compared with placebo, suggesting improved microvascular function. 111 Interestingly, a more recent publication demonstrated that consumption of dark chocolate had similar effects on the postprandial increase of HVPG. 112 Since then, several studies have addressed the role of antioxidant therapy in the improvement of NO bioavailability. In this regard, the modulation of the activity of the antioxidant enzyme superoxide dismutase (SOD) has proven promising effects in animal models of CLD.113,114 In a study from 2011, administration of tempol, a SOD analog, improved the IHVR of cirrhotic rats in an NO-dependent manner. 114 Similarly, a more recent study assessed the administration of a recombinant form of human manganese SOD in two different animal models of CLD resulting in improved liver hemodynamics, as well as reduced oxidative stress and liver fibrosis. 115 Resveratrol is an antioxidant found in the skin of grapes that activates the histone deacetylase Sirt1, which regulates several vasoprotective pathways leading to NO overexpression. 116 Recent studies have reported hepatic antifibrotic effects of resveratrol in models of mild liver injury linked to its antioxidant properties117–119 while its administration to rats with severe PH reduced PP and hepatic fibrosis, and ameliorated hepatic microvascular dysfunction. 120
Strategies targeting inflammation
Anti-inflammatory strategies
Rapamycin is an immunosuppressor that negatively regulates the mammalian target of rapamycin (mTOR) complexes, which play a central role in the regulation of many of the cellular pathways including proliferation, inflammation, apoptosis and autophagy. In the field of CLD, rapamycin has shown the ability to reduce the PP in rats with PH due to its intrahepatic121,122 and extrahepatic effects.123,124
The Janus kinase 2 protein (JAK2) is involved in the toll-like receptor (TLR) signaling in response to lipopolysaccharide, and upregulates profibrotic and inflammatory genes. As commented above, AG490 is a specific inhibitor of JAK2 but it can also regulate the TLR signaling pathway. Recently, its potent antifibrotic and anti-inflammatory effects were proven in livers of BDL-cirrhotic rats, partially normalizing PH. Furthermore, the same study reported additional extrahepatic hemodynamic amelioration in the partial portal-vein ligation model, 26 while combination with NSBB achieved additive effects. 27
Epoxyeicosatrienoic acids (EETs) are products of the arachidonic acid metabolism with anti-inflammatory properties. 125 These molecules are further metabolized by the soluble epoxide hydrolase (sEH), thus inhibition of this enzyme represents a good strategy in maintaining or increasing EET levels. Indeed, the anti-inflammatory effects of the sEH inhibitor t-TUCB have been recently validated in steatotic livers. 126 In addition to these effects, EETs have also been described to show vasoactive properties, 127 thus suggesting additional potential in liver vascular diseases. Indeed, recent studies reported that t-TUCB treatment ameliorates PH in CCl4-cirrhotic rats, altogether improving fibrogenesis and endothelial dysfunction128,129 while additionally ameliorating extrahepatic circulation. 130 Although inflammatory markers were also downregulated by this inhibitor, whether the hemodynamic effects were additive or directly mediated by inflammation was not assessed. Despite these promising reports, there is some controversy regarding the role of EETs in cirrhosis, as another study reported opposite effects in the splanchnic territory. 21
In addition to conventional anti-inflammatory pharmacological strategies, engineered nanoparticles represent an alternative therapeutic option with targetable delivery. Cerium oxide nanoparticles have been shown to behave as anti-inflammatory agents, with no exception in the liver. Indeed, these nanoparticles prevented macrophage infiltration and overexpression of hepatic inflammatory genes, causing a reduction in oxidative stress and improving PP in rats with mild PH. 131
Antidiabetic drugs
As expected from drugs aimed at normalizing glucose levels, antidiabetic treatments usually target glucose and lipid transport, as well as metabolism. However, these drugs have also been reported to have vasoprotective effects associated with NO bioavailability, inflammation and oxidative stress.132–134 In consequence, these beneficial effects have been assessed in CLD. One such pharmacological strategy is use of liraglutide. This glucagon-like peptide 1 (GLP-1) analog has shown the ability to ameliorate PH and liver microvascular function in cirrhotic animals by reducing HSC activation and the levels of inflammatory markers independently of the GLP-1 receptor. 135 In this same study, the effects of liraglutide were validated in different human models in vitro, thus shortening the gap between the preclinical study and clinical trials. Indeed, liraglutide has shown antifibrotic effects also in NASH patients, 136 thus representing a promising drug with high probabilities of success as a treatment for PH and CLD. Similarly, the antidiabetic drug metformin has shown improvement in liver hemodynamic and fibrosis, which were accompanied by a reduction in inflammation and oxidative stress. 137 Interestingly, the observed hemodynamic effects were complementary to the effects of NSBB. Lastly, fenofibrate, another antidiabetic drug with described positive vascular effects, also improved PP in cirrhotic rats in addition to hepatic fibrosis and microvascular function. 138
An additional strategy to these drugs is the use of antibodies that specifically block the interaction between metabolic hormones and their ligands. Leptin, the satiety hormone, is overexpressed in obese people, who may suffer from leptin resistance. Furthermore, this hormone has been implicated in fibrosis and is highly expressed in cirrhotic patients, probably due to inflammation.139,140 Interestingly, the use of a competitive antibody against leptin receptor proved to be effective in ameliorating PP and IHVR in cirrhotic rats, suggesting that leptin is not only a marker in CLD but is also a targetable effector. 141
Microbiota modulators
Intestinal bacteria are known to modulate splanchnic flow and thus participate in the PH-derived hyperdynamic syndrome. In addition, during cirrhosis, increased gut permeability allows translocation of bacteria or their products into the bloodstream, causing general and hepatic inflammation.142,143 For this reason, different clinical trials have assessed the use of antibiotics (rifaximin,144–146 norfloxacin 147 ) as prophylactic intervention against general infections in decompensated cirrhotic patients, which overall improved their outcome.
However, recent research suggests that the composition of the microbiota population may also play a role in gut permeability and, interestingly, in liver hemodynamics. In this regard, oral administration of the probiotic VSL#3 (a mix of four Lactobacillus species) prevented the increase in the systemic levels of inflammatory cytokines and vasodilators in a rat model of BDL cirrhosis 148 and ameliorated liver function and pro-inflammatory markers in cirrhotic patients compared with the placebo group. 149 Bifidobacterium pseudocatenulatum CECT7765 supplementation had similar anti-inflammatory features. On one hand, liver or blood macrophages isolated from cirrhotic animals and patients (respectively) switched to an anti-inflammatory M2 phenotype when treated in vitro with B. pseudocatenulatum 150 while on the other hand, oral administration of the bifidobacteria to BDL-cirrhotic rats improved their hepatic hemodynamic parameters, liver damage and markers of inflammation in vivo. 151
These findings suggest that not only direct administration of bacteria but also diets that modify the gut microbiota could have therapeutic value in CLD and PH. Indeed, a recent clinical trial reported that cohorts of healthy controls and patients with cirrhosis have different microbiota populations, that microbiota diversity negatively correlates with the degree of cirrhosis, and that cirrhotic patients with diets that ensure higher microbiota diversity had lower risk of hospitalization. 152 Dietary goods associated with microbiota diversity included fermented milk products, coffee, tea, dark chocolate and vegetables, while consumption of carbonated drinks (including caffeinated ones) was predictor of poor diversity. Interestingly, most of the microbiota-beneficial products are rich in polyphenols (antioxidants), thus holding a potential combined effect to that.
Strategies targeting fibrogenesis
As stated above, it is well known that chronic liver damage in combination with paracrine signaling from the activated endothelium lead to the activation of HSCs, promoting fibrogenesis.63,153
Although most of the vasoprotective strategies described above ultimately lead to significant amelioration in fibrosis, there are a few pharmacological approaches that directly target the process of fiber formation/stabilization. One of these is simtuzumab, a monoclonal antibody against lysyl oxidase homologue 2 (LOXL2). LOXL2 is a protein that participates in the cross-linking of collagens and elastin. This protein is highly expressed in fibrotic livers (unlike control livers) and its inhibition with antibodies has shown positive results in preventing and reducing liver fibrosis in preclinical models. 154 Despite these promising results, simtuzumab proved to be ineffective in a recent phase II RCT in NASH-cirrhotic patients, 155 highlighting the controversy that is often found in the translation of some therapies from the laboratory to the clinic.
Cell therapy
The lack of pharmacologic therapeutic options to ameliorate PH and cirrhosis has prompted the use of regenerative therapies or cell-derived interventions. In 2006, Terai and colleagues performed the first clinical trial studying the autologous bone marrow cell infusion (ABMi) in decompensated cirrhotic patients, showing a decrease in Child-Pugh score as well as better liver function. 156 Latterly, infusion of mesenchymal stem cells (MSCs) from bone marrow or from umbilical cord to patients with liver cirrhosis demonstrated amelioration in hepatic fibrosis and function with increased serum albumin levels and with a good safety profile. 157 However, the last investigations in the field are focused on less invasive therapies using induced pluripotent stem cells (iPS). 158 iPS have been proposed as a new therapeutic option to replace damaged hepatocytes by healthy hepatocyte-like cells in CLD. 159 Nevertheless, hepatocyte-like cells induced by reprogramming methods remain immature without the specific hepatocyte characteristics such as detoxification or production of albumin and urea. 160 Along this theme, transplantation of human fetal hepatocytes 161 was evaluated in patients with end-stage CLD; although the small population size of the study was an important limitation for obtaining a robust conclusion, model for end-stage liver disease (MELD) score and portosystemic encephalopathy episodes were ameliorated in transplanted patients. Even though several clinical trials evaluating stem cell therapies in CLD are still ongoing,162–164 none of them specifically performs HVPG measurements; therefore, these RCTs will not provide new results about the effect of stem cells in PH.
On the other hand, preclinical studies have evaluated the effectiveness of cell-derived therapy ameliorating hepatic fibrosis, inflammation and liver hemodynamics using different sources and potency of stem cells in fibrotic and cirrhotic rodents. Specifically, human placenta-derived stem cells (PDSCs) are considered a promising therapeutic option for their proliferation and differentiation capacities, as well as their low immunogenicity potential. 165 In this sense, the anti-inflammatory and regenerative effects of chorionic plate-derived MSC transplantation in CCl4-injured rats 166 and the amelioration in fibrosis and cirrhosis progression in fibrotic rodents receiving human amniotic membrane-derived mesenchymal 167 or epithelial stem cells 168 has been reported. In addition, transplantation of human amniotic MSCs in TAA-induced cirrhotic mice was able to restore the hepatic function and improve liver fibrosis; 169 importantly, this study suggested the PDSCs transplantation as a better therapeutic option in comparison with adipose tissue-derived MSCs.
Regarding the effects of stem cells on PH and hepatic microcirculation, much less is known. Brückner and colleagues suggested that transplantation of hepatocyte-like cells in vitro differentiated from rat adipose tissue MSCs into CCl4-cirrhotic rats led to an amelioration in PH after 3 weeks of treatment without changes in liver dysfunction or fibrosis. 170 However, an ongoing preclinical study presented as a meeting abstract has shown the beneficial effects of transplantation of human amniotic stem cells (mesenchymal or epithelial) in CCl4-induced cirrhotic rats with PH, improving HSC and LSEC phenotype, inflammation and liver function, resulting in an amelioration in PH and microvascular dysfunction. 171
The promising results of cell therapy in preclinical models of CLD and PH support future and ongoing clinical trials using this strategy.
Lifestyle and dietary interventions
The basis of most of the abovementioned strategies is to improve deregulated processes in CLD (vasoconstriction, oxidative stress, etc.) with drugs or other molecules that specifically target these molecular pathways. Alternatively, wider effects on these pathological processes can also be achieved by modification in lifestyle; mainly diet and exercise.
Dietary approaches
The effects of certain diet components (dark chocolate, coffee/tea, fermented milk) on gut microbiota and its implications in CLD have been described above.112,152,172 In summary, consumption of these products ensures a proper diversity in the gut microbiota while some of them also possess antioxidant properties, thus ameliorating liver damage and hemodynamics in CLD. In addition to these, the following dietary supplementations may have the potential to ameliorate PH.
Caffeine intake is extended worldwide, either in the form of coffee, tea or carbonated drinks. Independently of its effects on the gut microbiota, 173 caffeine consumption (in the form of coffee) has been associated with reduced liver fibrosis in precirrhotic patients. 174 Although there are no reports of its effects in PH in humans, prophylactic but also therapeutic treatment of BDL-cirrhotic rats with caffeine or caffeinated coffee ameliorated PP, liver inflammation and fibrosis, while also improving the extrahepatic vasculature.175,176
Taurine is an amino-sulfonic acid ubiquitously expressed in mammals with many pleiotropic effects 177 and is also a component of the so-labeled ‘energy drinks’. It has been reported that taurine deficiency leads to CLD in preclinical models, 178 suggesting a protective role in cirrhosis. Indeed, oral taurine administration has been proven to ameliorate PP due to reduction in fibrosis and systemic vasodilation in a rat model of mild cirrhosis 179 while more recently, a clinical trial in a small cohort of patients with clinically significant PH (HVPG >12 mmHg) also reported reduction in PP. 180 Thus, consumption of low carbonated ‘energy drinks’ (which are rich in taurine, and also contain caffeine) may have a positive impact in PH.
Finally, curcumin is a dietary product most common in Asian countries but extended worldwide that possesses anti-inflammatory, antiangiogenic and antiproliferative properties. 181 Indeed, its administration to BDL-cirrhotic rats decreased hepatic fibrosis and ameliorated liver endothelial phenotype while inducing splanchnic vasoconstriction, which led to a reduction in PP. 182
Lifestyle interventions
In addition to nutritional strategies, nonsedentary lifestyle exerts beneficial effects on CLD complications such as PH. ‘The SportDiet study’ demonstrated that moderate exercise in combination with a controlled diet reduced PP and body weight in overweight patients with compensated cirrhosis and PH. 183 Moreover, another RCT evaluating the effects of exercise and diet intervention in patients with cirrhosis and PH showed an improvement in HVPG determined before and postintervention. 184 Moreover, the impact of exercise therapy on cirrhosis and its complications has been extensively studied in patients185,186 as well as in preclinical models. 187
Conclusion
The knowledge about the pathophysiology of CLD has improved dramatically in the last few years. Consequently, many preclinical studies reported novel treatments with promising improvements in PH and its complications that, importantly, act at different levels of the disease (either improving the microcirculation or the liver’s response to damage). Indeed, some of these proposed treatments are currently being tested in RCTs and could become new options for the treatment of CLD in the near future.
However, it seems obvious that the rate of success of the preclinical strategies at the bedside is still far from optimal. In this regard, new studies on the mechanisms of the disease (still to be fully understood) will provide new insights on novel targetable molecular pathways and metabolites. Nevertheless, we believe that in addition to the important research on drug development and efficacy, it is time to start wondering why these therapies fail in translation. What are the differences between preclinical models and current patients that make these therapies ineffective? Should the preclinical models integrate important variables present in humans, like comorbidities, epigenetics, aging, etc.? Are therapeutic windows different in humans? If so, could we ‘desensitize’ the human liver to these therapeutic windows? In our opinion, this second line of research could widen the current bottleneck in the translation of pharmacological strategies and reopen the box of failed therapeutic options in search of second chances.
Footnotes
Acknowledgements
MV and SG-M contributed equally to this article.
Funding
SG-M has a postdoctoral fellowship from the Stiftung für Leberkrankheiten and the Asociación Española Para el Estudio del Hígado. AF-I has a Sara Borrell contract from the Instituto de Salud Carlos III (CD15/00050). JG-S has received continued funding from the Instituto de Salud Carlos III (currently, FIS PI17/00012), the Spanish Ministry of Science, Innovation and Universities, Centro de Investigación Biomédica en Red en Enfermedades Hepáticas y Digestivas (CIBEREHD), the European Union Funds FEDER ‘una manera de hacer Europa’ and the Stiftung für Leberkrankheiten.
Conflict of interest statement
The authors declare that there is no conflict of interest.
