Abstract
Background:
Portal vein pressure (PVP) is crucial in the diagnosis, treatment monitoring, and prognosis of portal hypertension (PHT). At present, the hepatic venous pressure gradient (HVPG) and the portal vein pressure gradient (PPG) are the main approaches for portal pressure measurement. However, HVPG and PPG are limited in application due to invasive operations and high requirements for medical conditions. A more convenient way to measure portal pressure is demanded.
Objectives:
To detect whether the endoscopic variceal pressure (EVP) could predict the PVP.
Design:
This is a single-center retrospective study that detects whether the EVP could predict the PVP.
Methods:
Thirty-six patients were considered eligible for pre-emptive transjugular intrahepatic portosystemic shunt (TIPS) and were willing to accept the endoscopy-TIPS hybrid surgery (continuous operation within 24 h) from April 2023 to October 2023 retrospectively. EVP was measured in the procedure of the modified sandwich method. HVPG, PPG, and PVP were measured following TIPS operation. EVP was compared with HVPG, PPG, and PVP in groups divided by different sarin classifications and whether portal vein thrombosis (PVT) or not.
Results:
EVP was successfully measured and recorded in all. Totally, HVPG was lower than PPG, PPG was lower than PVP, and EVP manifested between PPG and PVP. In the PVT group, HVPG was lower than PPG, PPG was lower than EVP, and EVP manifested between PPG and PVP. In the non-PVT group, HVPG was lower than PPG, PPG was lower than PVP, and EVP manifested between PPG and PVP. In the gastroesophageal varices group, HVPG was lower than PPG, and PPG was lower than EVP.
Conclusion:
As a novel way for prediction, EVP measurement might be useful and convenient in PHT. Moreover, it might be closer to the PVP.
Keywords
Introduction
PHT is a common complication with increased pressure of the portal vein system caused by multiple causes. Approximately 90% of PHT is caused by cirrhosis.1,2 A series of complications such as ascites, splenomegaly, gastroesophageal varices (GOV), hepatorenal syndrome, and so on could be caused by PHT. 3 GOV is one of the several complications of PHT, and its rupture is the most common cause of death in patients with cirrhosis. Esophageal and gastric variceal bleeding (EGVB) occurs in 30%–50% of patients with liver cirrhosis, and 15%–30% of patients die of it. 4
Portal vein system pressure is of great significance in the diagnosis, predicting complications of PHT, prognosis, and evaluation of the drug efficacy. 5 Portal vein system pressure measurements include invasive pressure measurement methods: portal vein pressure gradient (PPG) measurement, hepatic venous pressure gradient (HVPG) measurement, and direct puncture measurement. Noninvasive pressure measurement methods include laboratory examination, imaging examination, noninvasive prediction models, endoscopic ultrasound (EUS) pressure measurement, etc.6–9 At present, PPG and HVPG are relatively accurate methods for measuring portal vein pressure (PVP). It can be obtained by direct surgical pressure measurement, transjugular intrahepatic portosystemic shunt (TIPS), intraoperative pressure measurement, and percutaneous liver puncture pressure measurement. However, it is not suitable for most patients due to invasive operations and limited measurement methods. In recent years, EUS-guided PPG measurement has become a new research trend due to its safety and simplicity of operation. However, further research is still needed, and EUS-PPG is limited to the centers with relevant technologies. 10 HVPG is the “gold standard” for the diagnosis of portal hypertension (PHT). HVPG plays an important role in diagnosis, risk stratification, treatment selection, evaluation of treatment response, and prognosis.11,12 At the same time, the accuracy of HVPG is affected by factors such as anesthesia status, body position, presence or absence of portal vein thrombosis (PVT), balloon position, collateral circulation, etc., and has high requirements for medical conditions and physician teams, so its reference value is limited.13,14 Some studies report combination of noninvasive methods can partially replace HVPG to predict portal vein system pressure and provide some guidance for diagnosis, prognosis, and complication prediction.15,16 However, due to a large number of data sources, the accuracy is easily affected, and the data are easily missing, it has not yet been applied to clinical practice.
Endoscopy is a timely hemostatic treatment commonly recognized by guidelines at home and abroad, including ligation, sclerotherapy, or combined therapy.17,18 The evaluation of the risk of rebleeding after endoscopic treatment and the main reference for the secondary prevention indications of TIPS are derived from the appearance of endoscopic varices during endoscopic treatment and the prediction of the effect after treatment.19,20 There is a lack of accurate PVP data reference, which can only be preliminarily predicted by the diameter of varices under endoscopy and whether there is a red sign. The study is designed to measure the endoscopic variceal pressure (EVP) in the procedure of endoscopic treatment, and then compare EVP with HVPG, PPG, and PVP in the following TIPS process. The results are expected to be the available measurement for PVP prediction.
Methods
Study population
The study retrospectively enrolled 36 patients diagnosed with EGVB caused by decompensated hepatocirrhosis in Suining Central Hospital from April 2023 to October 2023 (all patients who were willing to undergo the hybrid operation and met the inclusion and exclusion criteria during the period). Based on both indications of the modified sandwich method and the pre-emptive TIPS according to BAVENO VII, the endoscopy-TIPS hybrid operation was performed after the written informed consent was signed. 21 The general information was collected before the operation, and the EVP, HVPG, PPG, and PVP were collected and recorded. All the data were analyzed according to Sarin’s varices classification and PVT or not.
Inclusion and exclusion criteria
Inclusion criteria: (1) Age 18–75 years; (2) liver cirrhosis PHT: PHT is diagnosed by one or more of clinical features, imaging features, endoscopic features, liver biopsies, or HVPG; (3) the diagnosis of EGVB was confirmed, which met the indications of endoscopic treatment; (4) indicators of TIPS: pre-emptive TIPS according to BAVENO VIII: pre-emptive TIPS with PTFE-covered stents within 72 h (ideally 24 h) is indicated in patients bleeding from EV, GOV1, and GOV2 who meet any of the following criteria: Child–Pugh class C <14 points or Child class B >7 with active bleeding at initial endoscopy or HVPG >20 mmHg at the time of hemorrhage 20 ; and (5) written informed consent with the endoscopy-TIPS hybrid operation.
Exclusion criteria: (1) endoscopic examination and treatment contraindications, (2) contraindications of TIPS operation, (3) minors, (4) pregnancy, (5) regional PHT, (6) unsuccessful endoscopy operation, (7) unsuccessful TIPS operation, (8) unsuccessful pressure measurement during TIPS operation, and (9) non-variceal bleeding.
Study design
The study was a retrospective, self-controlled design and conformed to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 22 A total of 36 patients consistent with the inclusion and exclusion criteria were enrolled. After the written informed consent was signed, the general information, including gender, age, causes of cirrhosis, and PVT or not, was recorded. Then all patients underwent the endoscopic-TIPS hybrid operation. The operation procedure was as follows.
Under general anesthesia, a varix greater than 0.5 cm in diameter was found under endoscopy (Figure 1(a)). After puncturing the varix with blood returning in the transparent needle sheath (Figure 1(b)), the injection needle (Boston Scientific Interject™; 23 ga (0.6 mm 240 cm)) was connected to a pressure sensor (SJ-DOI-100, Zhejiang Sujia medical equipment Co., LTD; Jiaxing, Zhejiang Province, China) and multi-function monitor (BeneVision N15, Shenzhen Mindray Biomedical Electronics Co., LTD; Shenzhen, Guangdong, China). The pressure values were read and recorded (supplemental material). Then the contrast agent (Ioversol Injection; Jiangsu Hengrui Pharmaceutical Co., Ltd; 100 ml, 35 g; Lianyungang, Jiangsu Province, China) was injected through the needle (Figure 1(c)). The blood flow direction was observed under digital subtraction angiography (DSA) monitoring (SIEMENS) to make sure the needle tip was in the varix vessel (Figure 1(d)). Then, the modified sandwich method was performed (Figure 1(e)).

(a) Varices were found in gastroscopy. (b) After the successful puncture, the needle sheath was seen to return blood and then connected to the monitor to measure the pressure and read the data. (c) The contrast agent was injected by the injection needle. (d) The contrast agent and flow direction were observed under DSA monitoring. (e) Sclerosing agent injection. (f) Cyanoacrylate injection.
The TIPS operation followed after the endoscopic operation. After the neck was sterilized and covered with a drape, the right internal jugular venous approach was acquired, and then the catheter (Torcon NB® Advantage Catheter; HNB5.0-38-100-P-10S-0; Cook Incorporated; Bloomington, Indiana, USA) was inserted to measure the inferior vena cava pressure. Then the balloon catheter (CHESS558012) was advanced into the right hepatic vein under DSA to measure the HVPG. The vascular puncture kit (RUPS-100; Cook Incorporated) was then guided anteriorly and punctured from the right hepatic vein into the right/left portal vein. 23 Next, the catheter was induced into the splenic vein. To complete portal vein system angiography and then PPG and PVP were measured. After a balloon dilatation catheter (Cook Incorporated; PTA-35-80-8-6.0) was introduced to dilate the tract, the covered stent (GORE.VIATORR; PTB8107275W) was placed to connect the right hepatic vein to the portal vein. 24 After the catheter was removed, the postoperative PPG was measured, and the portal venography was performed again to confirm sufficient drainage. Finally, the catheter and RUPS-100 were removed, and gauze was used to cover the incision in the neck. The EVP, HVPG before the TIPS operation, preoperative PPG, postoperative PPG, and PVP were collected for analysis.
Statistical analyses
The statistical data were analyzed using SPSS (25.0 version, IBM, Armonk, NY, USA). The quantitative data were expressed as mean ± standard deviation or quartile (the 25th and 75th percentiles). The categorical data were expressed as frequency (percentage). Student’s t-test was used when the quantitative data were compared between two groups. When there were multiple groups of quantitative data, the one-way test was used to analyze whether there were differences, and Dunnett’s test or Tukey’s test was used to compare with each other. Categorical data were analyzed by the Chi-square test or Fisher’s exact test (when there was any frequency less than 5). Statistical significance was defined as a two-sided p-value of <0.05.
Results
Baseline characteristics
The baseline characteristics are as follows according to PVT or not (Table 1) and the types of Sarin classification (Table 2).
Baseline characteristics of the study population according to PVT or not.
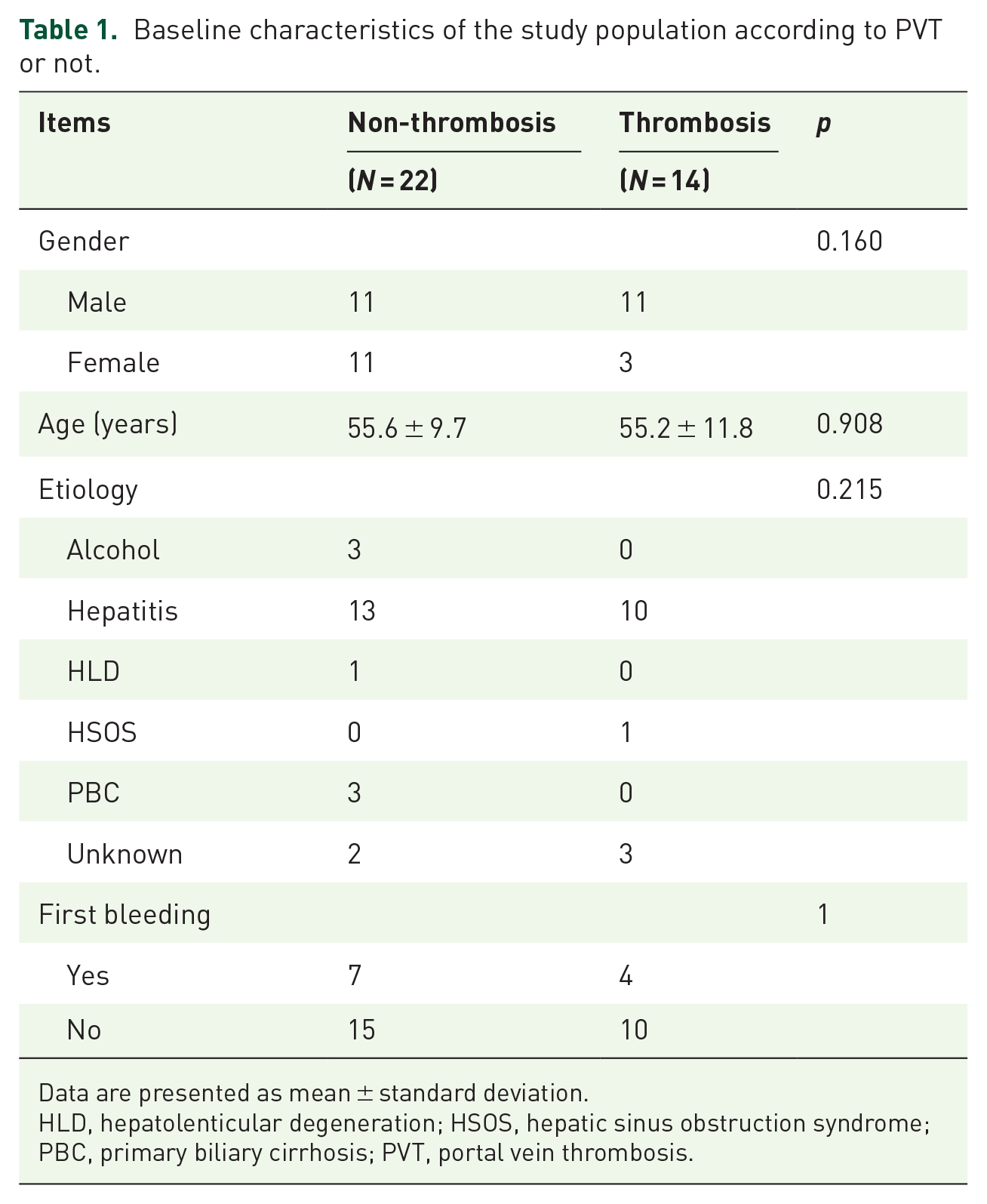
Data are presented as mean ± standard deviation.
HLD, hepatolenticular degeneration; HSOS, hepatic sinus obstruction syndrome; PBC, primary biliary cirrhosis; PVT, portal vein thrombosis.
Baseline characteristics of the study population according to the Sarin typing.
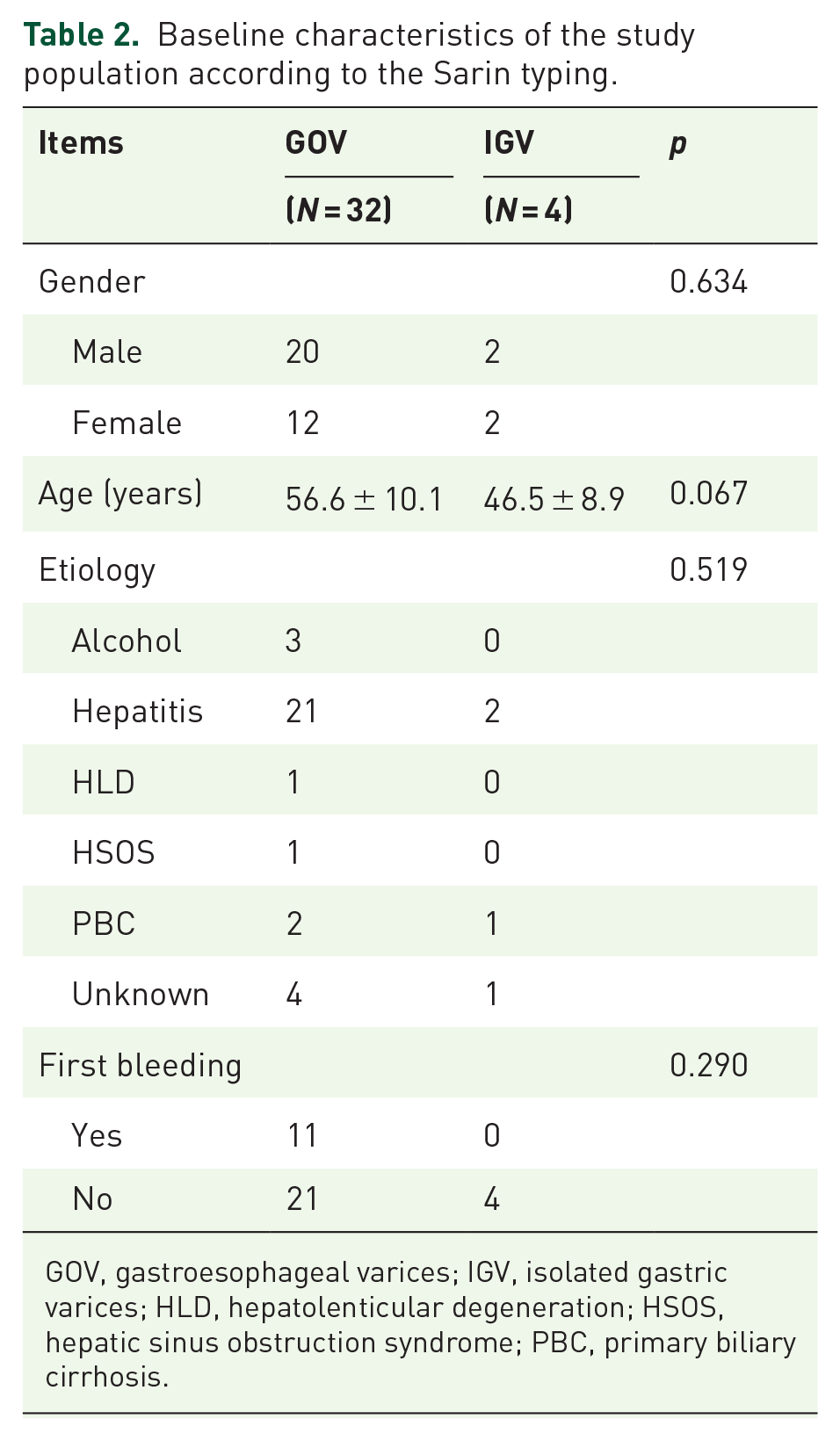
GOV, gastroesophageal varices; IGV, isolated gastric varices; HLD, hepatolenticular degeneration; HSOS, hepatic sinus obstruction syndrome; PBC, primary biliary cirrhosis.
Comparisons of HVPG, preoperative PPG, EVP, and preoperative PVP totally
Totally, HVPG (15.49 ± 7.06) was lower than the PPG (25.17 ± 5.83) (p < 0.0001), EVP (30.06 ± 11.13) (p < 0.0001), and PVP (37.44 ± 7.16) (p < 0.0001). There was no statistical difference between PPG and the EVP (p = 0.0529). The PPG was lower than the PVP (p < 0.0001). The EVP was lower than the PVP (p = 0.0009) (Figure 2).

Comparisons of HVPG, PPG, EVP, and PVP in total group. The median and quartiles are marked in the figure.
Comparisons of HVPG, preoperative PPG, EVP, and preoperative PVP in the PVT group
In the patients with PVT, the HVPG (15.89 ± 7.33) was lower than the PPG (25.64 ± 6.63) (p = 0.0061), EVP (29.86 ± 10.32) (p = 0.0024), and PVP (37.57 ± 7.40) (p < 0.0001). There was no statistical difference between PPG and EVP (p = 0.7343). The PPG was lower than the PVP (p = 0.0008). There was no statistical difference between EVP and PVP (p = 0.1702) (Figure 3).

Comparisons of HVPG, PPG, EVP, and PVP in the PVT group. The median and quartiles are marked in the figure.
Comparisons of HVPG, preoperative PPG, EVP, and preoperative PVP in the non-PVT group
In the patients with non-PVT, the HVPG (15.23 ± 7.05) was lower than the PPG (24.86 ± 5.41) (p = 0.0011), EVP (30.18 ±11.85) (p < 0.0001), and PVP (37.36 ± 7.15) (p < 0.0001). There was no statistical difference between PPG and EVP (p = 0.1479). The PPG was lower than the PVP (p < 0.0001). The EVP was lower than the PVP (p = 0.0245) (Figure 4).

Comparisons of HVPG, PPG, EVP, and PVP in the non-PVT group. The median and quartiles are marked in the figure.
Comparisons of HVPG, preoperative PPG, EVP, and preoperative PVP in the GOV group
According to the Sarin classification of type GOV, the HVPG (16.02 ± 7.19) was lower than the PPG (26.28 ± 4.95) (p < 0.0001), EVP (32.00 ± 10.18) (p < 0.0001), and PVP (38.94 ± 5.65) (p < 0.0001). The PPG was lower than the EVP (p = 0.0111) and PVP (p < 0.0001). The EVP was lower than the PVP (p = 0.0012) (Figure 5).
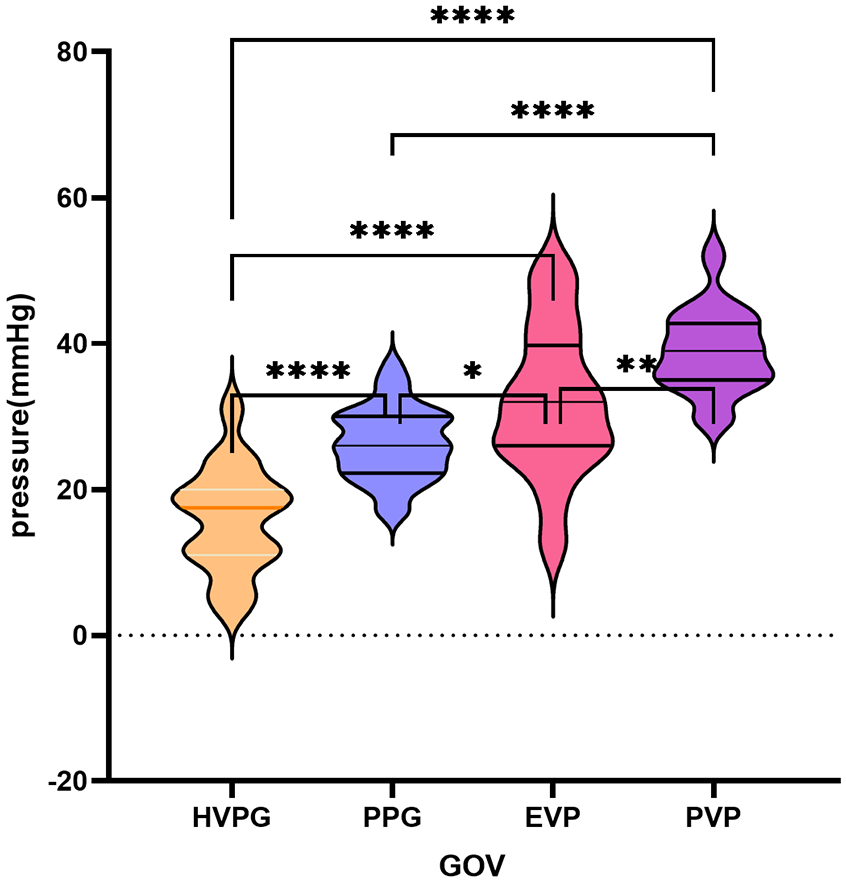
Comparisons of HVPG, PPG, EVP, and PVP in the GOV group. The median and quartiles are marked in the figure.
Comparisons of HVPG, preoperative PPG, EVP, and preoperative PVP in the IGV group
According to the Sarin classification of type IGV, there was no statistical difference in the IGV (Figure 6).

Comparisons of HVPG, PPG, EVP, and PVP in the IGV group. The median and quartiles are marked in the figure.
Adverse events
There were no severe adverse events, such as ectopic embolism and spray-like bleeding after needle withdrawal. Three patients had minor oozing blood after EVP measurement and modified sandwich method treatment, which stopped within 10 s.
Discussion
Endoscopic examination and treatment are considered first-line treatment for EGVB, which is timely, intuitive, and accurate. 25 However, in the treatment of PHT, endoscopic treatment is often considered to have certain limitations in the choice of second-line treatment due to the absence of varices pressure-related data as a guide. Nevertheless, current methods of portal vein system pressure measurement are invasive and limited by medical conditions. In this study, the variceal pressure was measured by endoscopy, which was directly associated with the bleeding varices. All of the varices with certain diameters were punctured by an endoscopic needle, which was a common step of the modified sandwich method, and it was easy to perform for endoscopists without additional invasion. The modified sandwich method is suitable for gastric variceal hemorrhage, and esophageal variceal hemorrhage which is not suitable for endoscopic variceal ligation (EVL). 17 The steps before sclerosing agent and cyanoacrylate injections are similar to other vascular pressure measurements, but not used in EGVB treatment. The key technical point is to keep the needle in the varix vessel for a while, but this is not difficult for experienced operators. The other innovative point of EVP measurement is the use of contrast and DSA in the process, the contrast in the varix could show the flow direction to prevent the potential risk of ectopic embolism and to make sure the needle is in the varix. If there is no condition for DSA or TIPS operation in some hospitals, the needle can be verified in the varix by repeated intrathecal refluxes to ensure accurate pressure measurements.
In this study, HVPG was found to be lower than PPG, and PPG was lower than PVP, which was due to hepatic collaterals, PVT, hepatic vein locations, and other reasons reported before, which is consistent with Berzigotti’s study.26–29 EVP was found to be higher than PPG and lower than PVP, which suggested that EVP might be closer to PVP than PPG. The reasons might be as follows: first, the measured varices of EVP were directly connected to the portal vein; second, EVP measurement is unnecessary to subtract the hepatic vein free pressure or right atrial pressure; thus, it was not affected by right heart function. In addition, according to Laplace’s law, the wall tension of the varices determines the risk of bleeding. 30 In PVT and non-PVT groups, EVP was found between PPG and PVP, which indicated that the measurement of EVP might not be affected by PVT. In the GOV group, EVP was also found between PPG and PVP. Most GOVs are derived from the gastric coronary vein, and some are derived from the posterior gastric or short gastric veins from the splenic vein. Therefore, EVP was lower than PVP. In the IGV group, the varices in the stomach are directly connected to the inferior vena cava through the gastric-renal shunt or the spleen–kidney shunt. Therefore, it tended to be lower than PPG in our study. Due to the small sample size, although the result in the IGV group was not statistically different, meaningful results might be obtained by expanding the sample size and multicenter studies.
In the 1950s, Palmer 31 first proposed the direct puncture of blood vessels to measure the variceal pressure, which was regarded as the gold standard for measuring the variceal pressure. However, it may cause complications such as bleeding and infection and cannot be measured repeatedly. After the emergence of flexible endoscopy, new variceal pressure measurement techniques have emerged, such as needle puncture, endoscopic balloon technique, and endoscopic gauge.30,32,33 However, needle puncture has the risk of invasion and bleeding, and the endoscopic balloon technique is not reliable for blood vessels with a diameter of less than 3 mm and needs special equipment, endoscopic gauge is affected by artifacts such as esophageal movement. According to LaPlace’s law, Jackson et al. 34 calculated variceal wall tension by direct sonographic and manometric measurements to predict bleeding risks. However, its application is limited because of the invasion. In this study, EVP measurement did not increase invasion because it was performed during modified sandwich therapy. The shortcomings of EVP measurement are not yet applicable in varices without bleeding and esophageal varices without EVL contradictions.
There are several limitations in our study. First, due to the small sample size, stratified analysis of cirrhosis etiology and statistical analysis of the IGV1 group could not be performed. A large sample multicenter study should be performed to improve the standard operating procedure and threshold interval calculation of EVP. Second, it is a retrospective single-center study, which needs to verify the accuracy of EVP during TIPS and be included in a multi-center cohort for further verification. If the verification is successful, the clinical significance of EVP could be further explored.
Conclusion
In conclusion, EVP might provide a new reference for the selection of EGVB secondary prevention with no ray and less invasion than HVPG and PPG. Moreover, EVP measurement could become a useful and convenient method in PHT prediction. However, more samples including a variety of liver disease populations, small varices, IGV, and ectopic varices are needed to establish an EVP cutoff valve for predicting Clinically Significant Portal Hypertension (CSPH) and standardizing EVP measurement.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
