Abstract
Background:
Endoscopic management of pelvic abscesses not amenable to percutaneous drainage has been described. The technique employs endoscopic ultrasound (EUS)-guided placement of stents or drains, which may require multiple procedures, is cumbersome and uncomfortable for the patient. We describe the successful management of these abscesses in a single step involving EUS-guided lavage and instillation of antibiotics.
Methods:
Six consecutive patients with seven symptomatic pelvic abscesses not amenable to percutaneous drainage were referred for EUS-guided drainage. The abscesses were aspirated with a 19-gauge needle under EUS guidance and serially lavaged with an equal aspirate to instillation volume of sterile saline until cleared of pus. The residual cavity was then instilled with gentamicin 40 mg/ml. Patients were followed clinically and radiographically with repeat computed tomography or magnetic resonance imaging.
Results:
All patients had rapid resolution of symptoms. The abscesses disappeared completely in four patients. One patient with recurrent diverticulitis and abscess had marked decrease in abscess size and inflammation to permit planned sigmoid resection. One patient with Crohn’s disease had clinical improvement and marked decrease in abscess size, permitting outpatient management of Crohn’s disease.
Conclusions:
EUS-guided lavage and instillation of antibiotics is a simple, one-step approach in the management of pelvic abscesses and may obviate the need for prolonged drain management and repeat procedures in select cases.
Introduction
Pelvic abscesses have traditionally been managed with systemic antibiotics and drainage. In select cases, however, the location of an abscess does not permit percutaneous drainage. Recently, endoscopic drainage with placement of transcolorectal stents or drains has been described as an alternative to drain pelvic abscesses.1–5 This technique entails an initial procedure to place one or more stents or an indwelling drain for lavage, and a follow-up procedure to remove the stent. Of necessity, this technique also creates a tract between the abscess and the lower gastrointestinal (GI) tract. In the largest study, Varadarajulu and colleagues 4 described abscess resolution and no recurrence within 90 days in 24/25 patients using this technique. There were a total of six patients in the studies by Puri and colleagues 5 and Giovannini and colleagues 1 in which the abscess was aspirated only. Of these, three had recurrence and required surgical treatment. The techniques involved here are of course imported from the traditional approach by interventional radiology utilizing drains for the management of abscesses. We present six patients with seven pelvic abscesses managed successfully in a single endoscopic session without placing stents or drains, whereby endoscopic ultrasound (EUS)-guided lavage was followed by antibiotic instillation.
Methods
Six patients presented with seven symptomatic pelvic abscesses between June 2016 to March 2018. All patients provided a Health Insurance Portability and Accountability Act (HIPAA) compliant consent to be included in this study. The etiologies are as follows:
Patient 1: developed an abscess postoperatively following appendectomy (Figure 1).
Patients 2 and 3: developed abscesses (three abscesses) as a consequence of sigmoid diverticulitis.
Patient 4: this patient suffered from liver cirrhosis and ascites, and presented with abdominal pain and neutrocytic peritonitis. On computed tomography (CT) scan a pelvic abscess was identified. There was no history of diverticulitis or appendicitis.
Patient 5: had recurrent and smoldering sigmoid diverticulitis despite prolonged antibiotics and a diverticular abscess that increased in size on antibiotics, not allowing a window for planned sigmoid resection.
Patient 6: has fibrostenotic Crohn’s disease and presented with multiple anal sinus tracts, a gluteal abscess for which a seton was placed and a supra-levator abscess not amenable to trans-anal drainage.

Computed tomography scan of patient 1 revealing a fluid density abscess in the pelvis.
All patients continued to have symptoms while on intravenous antibiotics and the abscesses were deemed inappropriate for percutaneous drainage due to their location.
Endoscopic technique
All procedures were performed by one endoscopist (AK) after institution of intravenous antibiotics. In each case, a curvilinear echoendoscope was used to puncture the abscess with a 19-gauge needle (EchoTip, Cook Medical Inc. Bloomington, IN, USA) and pus aspirated. Serial lavage was then performed through the needle with an instillation volume of sterile saline equal to each aspiration volume to avoid overinjecting the abscess cavity. Initial pus aspiration was slow through the 19-gauge needle, often taking a few minutes for a pus column to even appear in the vacuum syringe, but became easier and quicker with sterile saline injection and dilution. Equal volume lavage was performed until the return became clear or serosanguinous and nonpurulent. The volume of lavage varied markedly; for example, the largest diverticular abscess (~7 cm) required more than 30 min of lavage. The abscess cavity was then filled or injected with gentamicin (40 mg/ml; not to exceed 500 mg total) equaling the final return volume. The needle was withdrawn and the procedure completed. Gentamicin was selected for instillation into the abscess due to its Gram-negative antibacterial coverage, and the concern for introducing fecal flora into the abscess during passage of the EUS needle transrectally; and the availability of gentamicin in concentrated form (40 mg/ml) permitting the instillation of an adequate dose in a small abscess cavity.
Patient assessment
Intravenous antibiotics were initiated for all patients upon presentation and continued in oral form upon discharge for five patients for a total of at least 2 weeks. One patient (patient 5) was discharged on intravenous antibiotics. Each patient was followed clinically and radiographically with a follow-up CT scan or pelvic magnetic resonance imaging scan (MRI) in 2–4 weeks.
Results
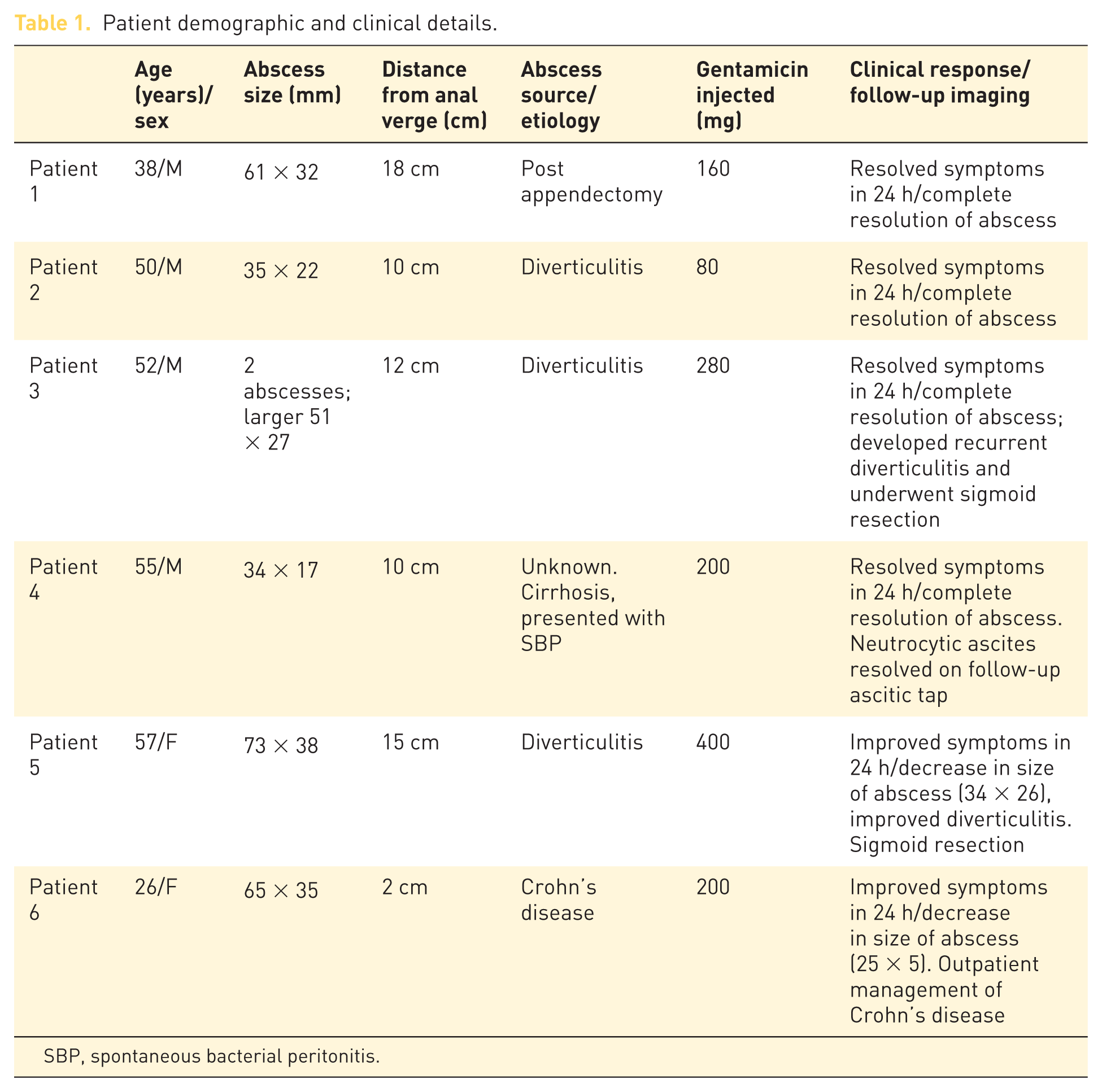
Patient clinical data are summarized in Table 1. The pelvic collections ranged in size from 3.5 to 7.3 cm in greater diameter. All patients were treated as described in the methods section with EUS-guided aspiration, equal volume sterile saline lavage and gentamicin instillation. Subsequent CT imaging for four patients (patients 1–4) revealed complete resolution of abscess (Figure 2 for patient 1).
Patient demographic and clinical details.
SBP, spontaneous bacterial peritonitis.

Follow-up computed tomography scan of patient 1 without evidence of residual or recurrent abscess.
One patient (patient 3) with a pelvic abscess secondary to diverticulitis developed recurrent diverticulitis 3 months later, but without abscess recurrence, and ultimately underwent surgery.
Patient 5 had marked decrease in abscess size from 7.3 to 3.4 cm and improvement in diverticular inflammation permitting planned surgery.
Patient 6 with Crohn’s disease continues to have sinus tracts and anal canal narrowing but has marked improvement in the abscesses; the supralevator abscess decreased in size from 6.5 by 3.5 cm to 2.5 by 0.5 cm.
Discussion
The proximity of pelvic abscesses to nearby GI lumen has led to the development of endoscopic drainage techniques. Typically these include the placement of stents or drains that are then removed in a second procedure. These techniques seem to be effective in their primary aim of abscess drainage, but may require repeated interventions on account of drain occlusion by viscus abscess contents and dislodgment or kinking of the stent. To prevent clogging, the simultaneous placement of an irrigation catheter has been advocated. While effective, these drains typically emerge from the anus and can be uncomfortable for the patient. Furthermore, these drains require serial irrigation over several days, which necessitates involvement of a healthcare worker providing in-home visits or even in-hospital care. Lastly, after resolution of the abscess, a second procedure becomes necessary to remove the stents or catheters. The purpose of this process is to ensure complete removal of pus and infected material over the duration of the indwelling stents or drain. What remains unclear is if the same result of complete evacuation of pus can be achieved in a single treatment session, obviating the need for stents or drains. As presented in our limited experience here, our approach of abscess aspiration followed by irrigation until a clear return has been achieved, and instillation of antibiotics, appears to be successful clinically, yet much simpler to execute, have cost-saving benefits and minimize patient discomfort. A prior case series of ultrasound-guided transcutaneous aspiration and lavage alone to treat pelvic abscesses (2–11 cm in size), without drains, lends support to this approach. 6 While some of the abscesses treated in this series are relatively small and not amenable to stent or drain placement, EUS-guided simple aspiration alone of the abscess may have significant risk of recurrence as seen in the case series by Giovannini and Puri and colleagues. We suggest the key aspects to our approach are first the equal volume serial lavage until clear return, and second the instillation of antibiotics. Intra-abscess injection of antibiotics following lavage has a precedent in other organs, for example, breast. 7 While we chose to inject these abscesses with gentamicin due to the concern of contaminating a closed space with the needle being passed transrectally, there may be the added benefit of antimicrobial activity in the abscess which may not otherwise have adequate antibiotic penetration. Finally, whether this approach will be successful in larger abscesses is unknown and stent or drain placement for lavage may ultimately be required for adequate treatment. For example, patient 5 with smoldering diverticulitis and a 7.3 cm abscess had clinical improvement within 24 h and marked decrease in abscess size in 2 weeks on CT scan; it is unclear if this abscess would resolve completely over time as the clinical time course was interrupted by surgery. Patient 6 presents a unique challenge. While it is unclear if any drainage intervention would resolve the abscess in the setting of Crohn’s disease and multiple sinus tracts, an enteral stent or drain in the setting of Crohn’s disease puts the patient at risk of a permanent fistula. As such, we propose that aspiration, lavage and antibiotic injection is the preferable approach in this situation.
Important limitations of this case series include its small size and the absence of comparative groups. Larger series and a prospective study are necessary to validate the efficacy and safety of this procedure.
Footnotes
Acknowledgements
All authors give final approval for this manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Author contributions: Zachary Zator: data acquisition and analysis, drafting of manuscript, final approval of manuscript. Matthew Klinge: data acquisition and analysis, drafting of manuscript, final approval of manuscript. Wolfgang Schraut: data acquisition and analysis, revision of manuscript for critical content, final approval of manuscript. Allan Tsung data acquisition and analysis, revision of manuscript for critical content, final approval of manuscript. Asif Khalid: conception, design, data acquisition and analysis, drafting of manuscript, final approval of manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
