Abstract
Background:
Temporary stent placement is widely performed for pancreatic duct stenosis due to chronic pancreatitis. A fully covered self-expandable metal stent (FCSEMS) has a larger diameter, and therefore longer stent patency, and the effect of expansion of the main pancreatic duct stricture may be obtained. However, if stent migration upstream occurs, stent removal is extremely difficult. In addition, because of the diameter gap between the FCSEMS and the main pancreatic duct, stent-induced ductal change may occur. To prevent these adverse events, the technical feasibility, safety and efficacy of the placement of a novel 6 mm diameter FCSEMS with a long suture, to facilitate its removal in cases of stent migration upstream, were evaluated in a pilot study.
Methods:
Between December 2014 and August 2015, symptomatic chronic pancreatitis patients with abdominal pain and a main pancreatic head duct stricture were enrolled. Stent placement for main pancreatic duct stricture was performed under endoscopic retrograde cholangiopancreatography (ERCP) guidance and stent removal was performed within 6 months.
Results:
A total of 13 patients were retrospectively enrolled in this study. Metal stent insertion was successfully performed in all patients and clinical success was high (12/13, 92%). As adverse events, stent migration upstream was seen in two patients. Another 11 patients successfully underwent stent removal without any adverse events. During follow up (median 258 days), 2 patients still underwent pancreatic duct stenting because of continuing main pancreatic duct stricture.
Conclusion:
In conclusion, this novel FCSEMS is acceptable for stent placement in cases of chronic pancreatitis with a main pancreatic duct stricture.
Introduction
Pancreatic stenting using a plastic stent is widely performed for pancreatic stricture due to chronic pancreatitis [Levy et al. 2000; Godil and Chen, 2000; Smits et al. 1995; Binmoeller et al. 1998; Neuhaus, 2002; Cahen et al. 2007; Costamagna et al. 2006]. After stent removal, recurrence of symptoms may occur due to recurrent or persistent stricture of the main pancreatic stricture. To treat main pancreatic duct stricture, stent placement is needed using large diameter stents. However, a metallic stent has a larger diameter than a plastic stent. Therefore, longer stent patency and the effect of expansion of the main pancreatic duct stricture may be obtained compared with a plastic stent. Unsatisfactory results for patients with a dominant stricture in the main pancreatic duct using an uncovered self-expandable metal stent have been reported [Eisendrath and Deviere, 1991; Shin et al. 2006; Kahaleh et al. 2008]. However, a fully covered self-expandable metal stent (FCSEMS) prevents tissue ingrowth and can be removed, though a high frequency of stent migration is one of the disadvantages. If stent migration upstream beyond the main pancreatic duct stricture occurs, stent removal is extremely difficult. In addition, because of the diameter gap between the FCSEMS and the main pancreatic duct, stent-induced ductal change may occur more easily than with a plastic stent [Kozarek, 1990].
To prevent these adverse events, the technical feasibility, safety and efficacy of placement of a novel 6 mm diameter FCSEMS with a long suture to facilitate its removal in cases of stent migration upstream were evaluated in a pilot study.
Patients and methods
Patients and study design
In this retrospective study between December 2014 and August 2015, symptomatic chronic pancreatitis patients with abdominal pain and a main pancreatic head duct stricture were consecutively enrolled. Chronic pancreatitis was diagnosed according to the American Pancreatic Association practice guideline in chronic pancreatitis [Conwell et al. 2014]. All patients were native of pancreatic stenting.
After cephalic stone clearance had been performed, stent placement for main pancreatic duct stricture was performed under endoscopic retrograde cholangiopancreatography (ERCP) guidance and stent removal was performed within 6 months.
All patients provided written informed consent before the procedure. This study was approved by the institutional review board of our hospital.
The novel fully covered self-expandable metal stent
Figure 1 shows the novel fully covered self-expandable metal stent (Niti-S Biliary S-Type Stent Long Suture; TaeWoong Medical, Seoul, Korea; Century Medical Inc., Tokyo, Japan). The diameter of this stent is 6 mm and the delivery system is 8Fr. This diameter may prevent stent-induced ductal change because it is smaller than that of the stents used in previous reports (8 or 10 mm) [Moon et al. 2010]. This stent also has a long suture (9 cm).

Novel fully covered self-expandable metal stent.
Pancreatic stent placement and removal procedures (video)
First, the duodenoscope was advanced into the duodenum and, using an ERCP catheter, the contrast medium was injected into the main pancreatic duct (Figure 2a). After pancreatography was performed, a 0.025 inch guidewire was also placed across the stricture site. After that, stent delivery system was inserted into the main pancreatic duct (Figure 2b) and stent placement was performed (Figure 2c, d). If it was difficult to insert the stent delivery system, 4 mm balloon dilation using a balloon catheter (REN biliary dilation catheter; KANEKA, Osaka, Japan) was performed. Within 6 months, stent removal was performed inside the operative channel of the endoscope. If the stricture had disappeared, no further stent placement was performed (Figure 3).

Technical tips of placement of pancreatic metal stent.

Case of disappeared pancreatic duct stricture.
Statistical analysis
Technical success was defined as stent placement into the main pancreatic duct across the stricture site. Clinical success was also defined as the resolution of symptoms and disappearance of the dilatation of the main pancreatic duct after stent placement. Adverse events were judged according to the American Society for Gastrointestinal Endoscopy (ASGE) lexicon [Conwell et al. 2014]. In addition, early adverse events were defined as those seen within 14 days after stent placement, and late adverse events were defined as those seen after 14 days.
Results
A total of 13 patients (6 men, 7 women; median age: 54 years; range: 36–81 years) were retrospectively enrolled in this study. Table 1 shows the characteristics of these patients. The etiologies of chronic pancreatitis were alcoholic (n = 8) and idiopathic (n = 5). The median length of the main pancreatic duct stricture was 8.7 (range: 5.5–22.7) mm and the median diameter of the main pancreatic duct was 6.9 (range: 5.0–9.2) mm. All patients had experienced abdominal pain due to chronic pancreatitis or acute pancreatitis several times.
Patient’s characteristics.
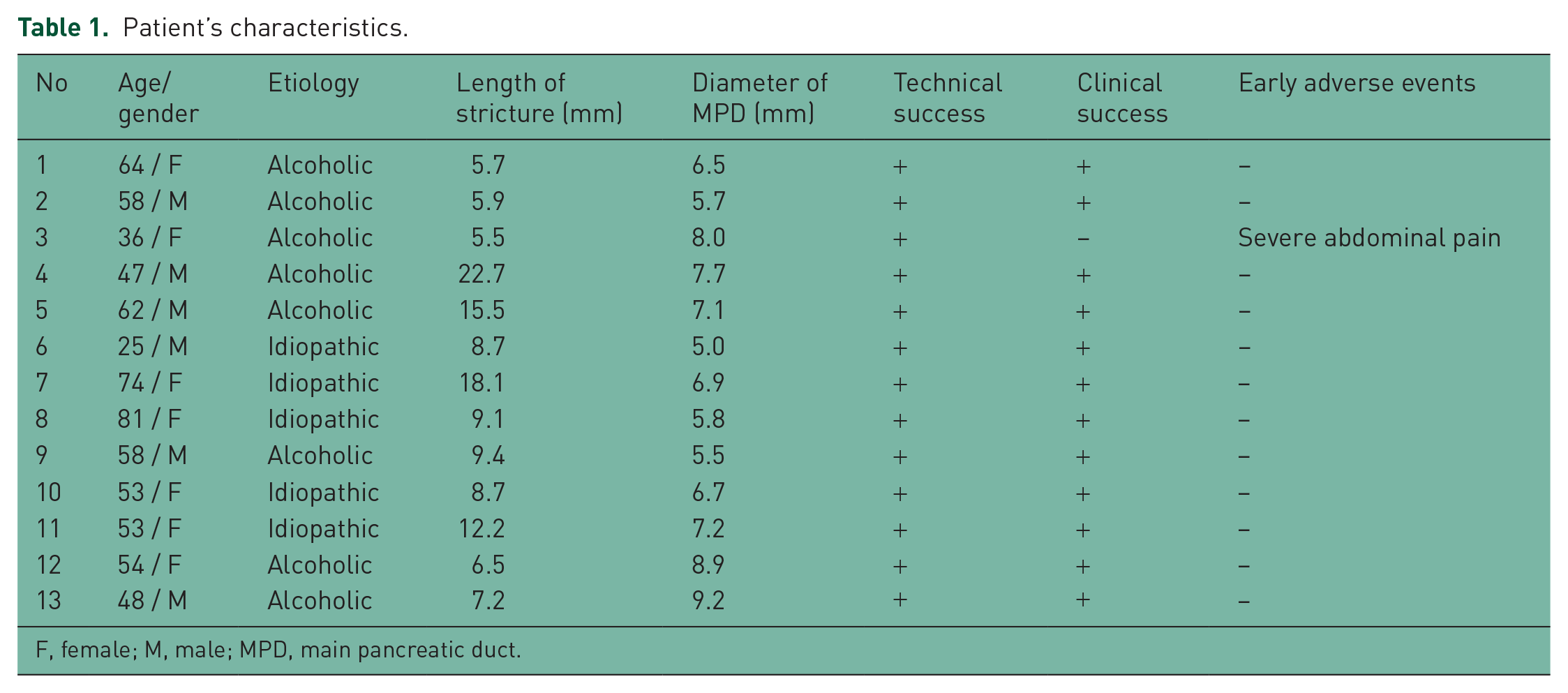
F, female; M, male; MPD, main pancreatic duct.
Metal stent insertion was successfully performed in all patients (technical success 100%). Clinical success was high (12/13, 92%). However, severe abdominal pain due to stent expansion occurred in 1 patient and stent removal was performed after 2 days.
Table 2 shows the patients’ outcomes in this study. The length of the metal stent was mostly 6 cm (6 cm n = 10, 8 cm n = 3). Stent placement was performed trans-minor papilla in one patient; the others were trans-major papilla. As adverse events, stent migration downstream was seen in two patients. Stricture of the main pancreatic duct was resolved on pancreatography under ERCP guidance and therefore re-intervention, such as stent placement, was not performed. Another 11 patients successfully underwent stent removal without any adverse events. The median period of stent placement was 172 (range: 153–185) days.
Clinical outcomes.
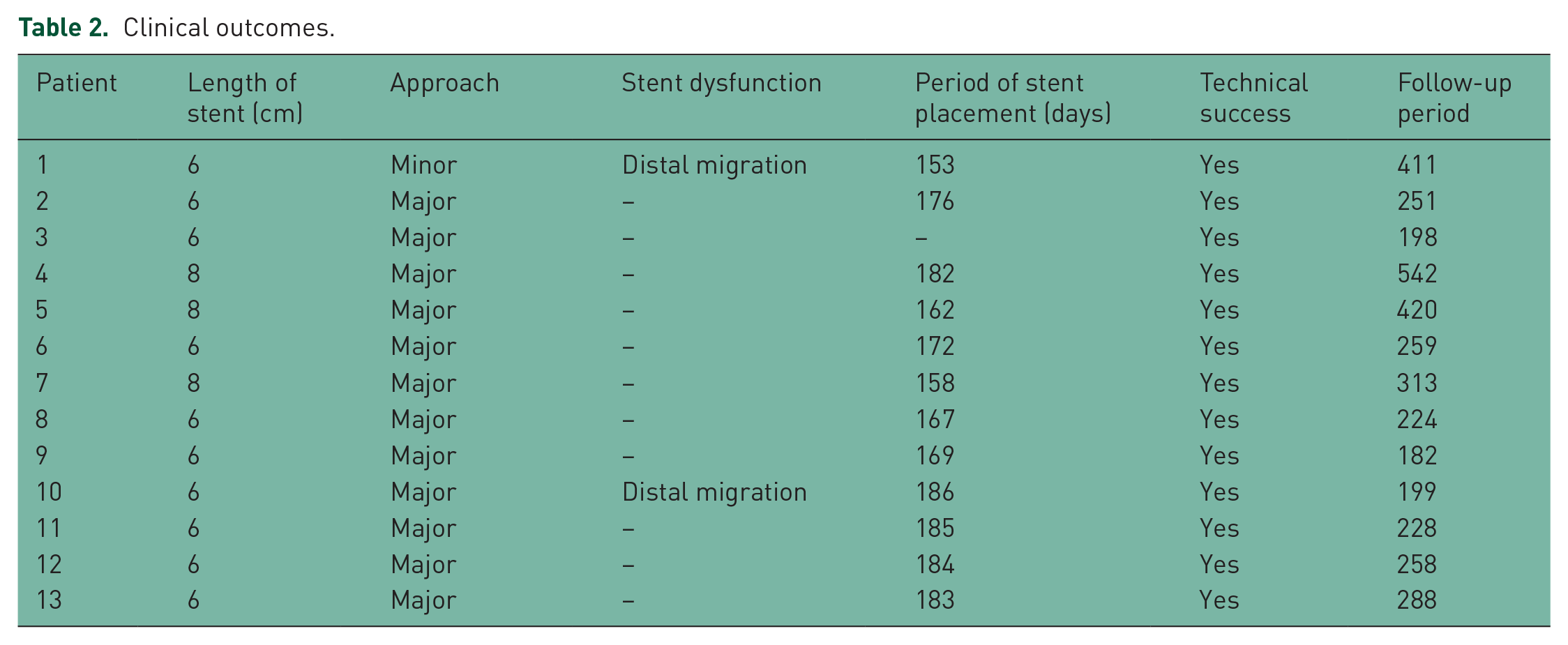
During follow up (median: 258 days; range: 198–542 days), 2 patients (numbers 3 and 7) still underwent pancreatic duct stenting by a plastic stent (patient no. 3) and a metal stent (patient no. 7) because of continuing main pancreatic duct stricture.
Discussion
Temporary stent placement is widely performed for pancreatic duct stenosis due to chronic pancreatitis. In this procedure, plastic or metal stent placement has been reported. To date, several reports on plastic stent placement have been published. Binmoeller and colleagues reported 87% sustained clinical improvement over a mean follow up of 4.9 years [Binmoeller et al. 1995]. Cremer and colleagues reported pain relief in 94% of patients during a mean follow up of 3 years [Cremer et al. 1991]. Therefore, plastic stent placement has several impacts on the clinical course. However, long-term follow up and frequent stent exchange are needed and thus a patient’s quality of life may decrease.
Recently, self-expandable metallic stent (SEMS) placement has been reported. Table 3 shows recently published clinical trials of FCSEMS placement for pancreatic duct stricture due to chronic pancreatitis [Sauer et al. 2008; Park et al. 2008; Moon et al. 2010; Giacino et al. 2012; Akbar and Baron, 2012].
Recent published literature for pancreatic duct stenting for chronic pancreatitis using a fully covered metal stent.

FCSEMS, fully covered self-expandable metal stent.
Park and colleagues reported that FCSEMS placement without a flare end for main pancreatic duct stricture caused by chronic pancreatitis was performed in 13 patients. In their study, stent migration downstream occurred frequently [Park et al. 2008]. Fortunately, although stent migration upstream occurred in one patient, they successfully performed re-intervention, because the distal end of the migrated stent upstream was placed in the pancreatic orifice. If complete stent migration upstream occurs into the main pancreatic duct through the stricture site, stent removal may be challenging.
Moon and colleagues, however, reported a similar study using a modified FCSEMS. Their stents had flared ends and irregular cell size showing different magnitudes of segmental radial force depending on the size of the cells. In their study, stent migration was not seen in any patients. However, stent-induced ductal changes were relatively frequent (5 patients, 16%). Stent-induced ductal change may be caused by ischemic injuries related to excessive outward radial pressure of a proximal flared stent end [Moon et al. 2010]. In the present study, therefore, we selected a 6 mm diameter stent that was non-flared, although the risk of stent migration was present. Indeed, in this study, there were no counterpart on side ducts or development of pseudocysts after covered stent insertion and there was no secondary hyperplasia in the duct at stent removal. Stent migration downstream occurred in two patients. If migration upstream occurs, stent removal may be possible because of the long suture. Therefore, this novel FCSEMS having an anti-migration system in the proximal site, such as wide flare, may be better.
Conclusion
Although the present study has several limitations, such as a small sample size and no long-term follow up, this novel FCSEMS is acceptable for stent placement in cases of chronic pancreatitis with a main pancreatic duct stricture.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
