Abstract
Irritable bowel syndrome is a functional bowel disorder with gastrointestinal symptoms (e.g. abdominal pain, straining, urgency, incomplete evacuation, nausea, and bloating) that occur alongside bowel function alterations (i.e. constipation, diarrhea, or both). Patients with irritable bowel syndrome may also experience comorbid anxiety and depression. Irritable bowel syndrome is common, with a prevalence estimated between 3% and 28%, affecting patient health and quality of life. Patients with moderate or severe irritable bowel syndrome generally seek medical care, whereas those with milder symptoms may choose self-management. Most patients with irritable bowel syndrome receive outpatient care, but irritable bowel syndrome-related hospitalizations do occur. The pathophysiology of irritable bowel syndrome is multifactorial (i.e. genetics, immune components, changes in the gut microbiota, disturbances in physiologic stress response systems, and psychosocial factors). Management of irritable bowel syndrome can include lifestyle changes, dietary interventions, counseling, psychologic medication, and agents that affect gastrointestinal motility. A number of therapies have emerged in recent years with clinical trial data demonstrating efficacy and safety for patients with irritable bowel syndrome, including agents that target gastrointestinal motility (i.e. linaclotide), gastrointestinal opioid receptors (i.e. asimadoline, eluxadoline), and gut microbiota (i.e. rifaximin). Linaclotide has been shown to significantly improve stool frequency and abdominal pain compared with placebo in constipation-predominant irritable bowel syndrome (number needed to treat, 5.1). Asimadoline shows efficacy in patients with moderate-to-severe irritable bowel syndrome-related pain. Rifaximin provided adequate relief of global irritable bowel syndrome symptoms versus placebo for a significantly greater percentage of patients with diarrhea-predominant irritable bowel syndrome (p < 0.001). Management that encompasses all aspects of irritable bowel syndrome (gastrointestinal symptoms) and comorbid psychologic symptoms (e.g. anxiety or depression) is important for improving overall patient health and well-being.
Introduction
Irritable bowel syndrome (IBS) is a functional bowel disorder characterized by symptoms of abdominal pain or discomfort that occur with changes in bowel function for at least 3 days/month for at least 3 months (Rome III criteria) [Drossman, 2006; Longstreth et al. 2006]. Patients with mild IBS have few symptoms, report good health-related quality of life (HRQOL), and generally seek medical care for symptoms approximately once per year, whereas patients with moderate or severe IBS have a greater number of symptoms (e.g. abdominal pain, bloating, dietary restrictions), report fair to poor HRQOL, and typically seek medical care approximately two to seven times per year [Drossman et al. 2009b, 2011]. IBS is further classified based on stool consistency, including constipation-predominant IBS (IBS-C), diarrhea-predominant IBS (IBS-D), IBS alternating between constipation and diarrhea (IBS-M), or unsubtyped IBS [Longstreth et al. 2006]. Patients with IBS often experience a number of additional gastrointestinal (GI)-related symptoms, including straining, urgency, incomplete evacuation, nausea, and bloating [Hungin et al. 2005, 2014; Longstreth et al. 2006; Ringel et al. 2009; Su et al. 2014]. Bloating has been considered to be one of the most bothersome IBS symptoms by patients; bloating led patients to seek medical care with significantly greater frequency than when bloating did not occur (p = 0.01) [Ringel et al. 2009]. Bloating adversely affected energy level, food intake, and the physical function subdomains of the IBS-quality of life (IBS-QOL) scale [Ringel et al. 2009]. Some symptoms tend to occur more often in a specific subtype of IBS. For example, a significantly greater percentage of patients with IBS-D and IBS-M reported experiencing urgency compared with patients with IBS-C (p < 0.001), while nausea was more commonly reported in patients with IBS-M than in patients with IBS-D or IBS-C (p = 0.01) [Su et al. 2014].
Epidemiology of IBS
The prevalence of IBS has been estimated between 3% and 28%, with IBS-M more prevalent than IBS-C and IBS-D [Brummond et al. 2015; Lin et al. 2014; Locke et al. 2000; Patel et al. 2015; Rasmussen et al. 2015; Rey de Castro et al. 2015; Ringel et al. 2009; Saito et al. 2002; Su et al. 2014]. IBS is also more prevalent in women compared with men [Lovell and Ford, 2012]. Diagnosis and management of patients with IBS occurs primarily in an outpatient setting; however, IBS accounted for 0.03% of US hospital discharges in 2010, with a mean duration of hospitalization of 3.7 days, accounting for a mean of US$21,153 in hospital costs [Mitchell and Drossman, 1987; Sethi et al. 2013]. A 2014 narrative review of studies that examined the cost of IBS reported an annual US cost of US$742–7547 per patient [depending on study year (1992 to 2004), relationship (direct versus indirect) to IBS care, and source of cost data] and a total projected annual cost of US$1.35 billion [Canavan et al. 2014]. Extraintestinal physical symptoms of IBS include fatigue, sleep problems, and back pain, reported by 69.3%, 47.5%, and 37.3% of patients, respectively [Choung et al. 2009; Patel et al. 2015]. In one study, patients reported the intensity of 12 different symptoms on a scale from 0 to 2 using the Patient Health Questionnaire (PHQ), with a score of 0 indicating ‘no bother at all,’ a score of 1 for ‘bothered a little,’ and a score of 2 for ‘bothered a lot,’ for a total score ranging between 0 and 24 [Patel et al. 2015]. Patients with IBS had greater odds of a medium or high total PHQ score (total score ⩾8) compared with patients of a GI clinic without IBS [odds ratio (OR) 1.7; 95% confidence interval (CI) 1.2–2.4]. A high total PHQ score (i.e. ⩾13), indicative of more severe physical symptoms, occurred in 31.7%, 22.5%, and 20.8% of patients with IBS-M, IBS-C, or IBS-D [Patel et al. 2015]. Low back pain is more common in patients with IBS-C than IBS-D (p < 0.01) [Schmulson et al. 1999]. Patients with IBS often experience improvement in pain symptoms after a bowel movement, particularly patients with pain-predominant IBS (i.e. abdominal pain >6 times/year), compared with patients with abdominal pain <6 times/year (50% versus 13%, respectively) [Talley et al. 1990]. Approximately one-quarter of patients in each of these groups had IBS-C. Patients with IBS who develop pelvic floor dyssynergy may experience worsening of IBS symptoms; interestingly, biofeedback therapy for dyssynergia has been shown to resolve IBS symptoms in some patients [Patcharatrakul and Gonlachanvit, 2011; Prott et al. 2010; Wong et al. 2010]. Thus, a number of physical symptoms appear to play a role in the overall well-being of patients with IBS and should not be overlooked or marginalized.
Pathophysiology of IBS
The pathophysiology of IBS has not been fully elucidated, but is multifactorial, and may include, but is not limited to, genetic factors, immune components, alterations in the gut microbiota, disturbances in physiologic stress response systems (e.g. hypothalamic–pituitary–adrenal), and psychosocial factors (Figure 1) [Camilleri, 2012]. A genetic component appears to play a role in IBS, as demonstrated by familial association studies and genome-wide association studies [Ek et al. 2015; Holliday et al. 2014; Levy et al. 2001; Locke et al. 2000] Genome-wide association studies of IBS have identified putative genetic risk loci for IBS [Ek et al. 2015; Holliday et al. 2014]. Further, additional genetic polymorphisms have been associated with IBS and specific disease characteristics (e.g. colonic transit) [Camilleri et al. 2014; Grasberger et al. 2013; Wouters et al. 2014].

Pathophysiology of IBS.
Alterations in the immune system play a role in IBS, including cytokine imbalances, immune cell activation, inflammation, and increased GI membrane permeability. Results of a meta-analysis indicated that levels of the proinflammatory cytokine tumor necrosis factor (TNF)-α were significantly increased in patients with IBS-C, IBS-D, and IBS-M, compared with controls (p ⩽ 0.03 versus controls for all), while levels of the anti-inflammatory cytokine interleukin (IL)-10 did not differ between patients with either of these three subtypes and controls [Bashashati et al. 2014]. Further, serum levels of the proinflammatory cytokine IL-6 were significantly increased in patients with IBS-D compared with controls (32.2 versus 7.5 pg/ml, respectively; p < 0.001) [Rana et al. 2012]. Patients with IBS had increased levels of activated intestinal mucosa immune cells compared with patients of a gastroenterology clinic without IBS, or healthy individuals [Ahn et al. 2014; Chadwick et al. 2002; Coeffier et al. 2010; Guilarte et al. 2007; Martínez et al. 2012; Vivinus-Nébot et al. 2012, 2014]. In patients with IBS-D, the number of daily bowel movements and stool consistency were positively correlated with mucosal levels of activated B-cells and plasma cells [Vicario et al. 2014]. Mast cells, which play an important role in innate immunity, release granules containing histamine, serotonin, proteases, lipid mediators, and cytokines upon activation by stress [Barbara et al. 2006, 2011]. Indeed, patients with IBS-C and IBS-D had an increased number of duodenal mast cells compared with healthy individuals [Walker et al. 2009], and disease severity and abdominal pain were significantly correlated with mast cell counts [Vivinus-Nébot et al. 2012].
Vitamin D, which plays a role in inflammatory processes, may also be of relevance in the pathophysiology of IBS [Yin and Agrawal, 2014]. Vitamin D deficiency was significantly associated with depression, a condition not uncommon in patients with IBS [Fond et al. 2014; Hoang et al. 2011]. Although the role of vitamin D in the pathogenesis of IBS remains to be elucidated, vitamin D supplementation may improve symptoms of IBS, as well as anxiety and depression [Karaahmet et al. 2013; Sprake et al. 2012]. However, data to support this intriguing hypothesis are sparse and limited to case reports [Sprake et al. 2012].
Increased GI membrane permeability is thought to be an important factor in IBS pathogenesis [Martínez et al. 2012]. Patients with IBS appear to have increased GI membrane permeability compared with healthy individuals, with increased severity of disease and abdominal pain significantly associated with membrane permeability (p = 0.002 and p = 0.006, respectively) [Piche et al. 2009; Vivinus-Nébot et al. 2014]. Expression of genes related to membrane permeability and mast cell function was altered in patients with IBS-D compared with healthy individuals [Martínez et al. 2012]. Expression of the tight junction protein occludin was decreased in patients with IBS compared with healthy individuals [Coeffier et al. 2010; Martínez et al. 2012; Vivinus-Nébot et al. 2014]. Thus, impaired GI membrane permeability appears to play a role in IBS.
The gut microbiota of patients with IBS differs qualitatively and quantitatively compared with that of healthy individuals [Carroll et al. 2011; Codling et al. 2010; Durbán et al. 2012, 2013]. Symptoms of IBS have been associated with specific gut microbiota profiles [Jeffery et al. 2012]. Bloating was associated with an increase in GI Cyanobacteria in patients with IBS, whereas increased colonic transit time and constipation were associated with 17 different taxa [Jeffery et al. 2012]. The GI tract of patients with IBS with depression has a lower concentration of bacteria belonging to the family Actinomycetaceae compared with patients with IBS without depression. However, healthy individuals have significantly less Actinomycetaceae than patients with IBS without depression (p = 0.002); the contribution of this bacterial family to the IBS pathology remains to be elucidated. Although specific symptoms have been correlated with expression of microbiota species in patients with IBS, symptom severity has been associated with instability in the gut microbiota, particularly for patients with IBS-D [Durbán et al. 2013]. However, instability of the microbiota was associated with recurrence and remission, with changes to the microbiota profile occurring rapidly (i.e. within days). Further, the microbiota profile differed by symptom severity for a patient with severe IBS-D, with differences between days with mild-to-moderate disease and severe disease. It is still unknown whether alterations in gut microbiota result in the development of IBS symptoms, or occur as a result of IBS.
The bidirectional brain–gut communication axis and brain processing of noxious stimuli appears to play an important role in the pathophysiology of IBS. Patients with IBS may have abnormal colonic transit [Tornblom et al. 2012] and visceral hypersensitivity [Camilleri et al. 2008; Larsson et al. 2012; Posserud et al. 2007] potentially related to altered processing of neuronal signals from the GI tract. A meta-analysis of 18 studies that examined brain region activation after rectal distension demonstrated that patients with IBS displayed differences in brain region activity compared with healthy individuals, particularly in brain regions engaged in emotional arousal [Tillisch et al. 2011]. Patients with IBS also display alterations in autonomic function [Salvioli et al. 2015] and basal levels of stress hormones [Chang et al. 2009], suggesting altered function of the hypothalamic–pituitary–adrenal axis.
Psychosocial factors (e.g. physical, sexual, or psychologic abuse; psychiatric conditions) may play a role in the pathophysiology of IBS; psychosocial problems tend to be reported more commonly in patients with more severe forms of IBS [Afari et al. 2014; Bradford et al. 2012; Chitkara et al. 2008; Drossman et al. 2011; Halland et al. 2014; Knight et al. 2015]. Although the exact role of anxiety and depression in the pathophysiology of IBS is currently unknown, both have been associated with increased risk for IBS. Indeed, anxiety and depression occurred in a significantly greater number of patients with IBS compared with individuals without IBS (p < 0.0001 for both anxiety and depression) [Koloski et al. 2012; Ladabaum et al. 2012]. Further, 38% of patients with IBS have reported thoughts of suicide and, although rare, some patients with IBS have also attempted suicide (5%) [Miller et al. 2004].
The severity of IBS and the intensity of abdominal pain have been positively correlated with anxiety and depression [Rey de Castro et al. 2015]. Patients with IBS-C and IBS-D, but not IBS-M, had significantly greater anxiety compared with healthy individuals (p = 0.04, p = 0.01, and p = 0.06, respectively); only patients with IBS-D had significantly greater incidence of depression than healthy individuals (p = 0.03) [Fond et al. 2014]. A greater percentage of patients with IBS with anxiety or depression had extraintestinal physical symptoms compared with patients without anxiety (44.8% versus 16.8%, respectively; p < 0.001) or depression (57.0% versus 21.5%, respectively; p < 0.001) [Patel et al. 2015]. A greater percentage of patients with IBS received anxiolytics and antidepressants compared with healthy controls (p < 0.0001 for both comparisons); notably, 62% of patients received these agents prior to receiving a diagnosis of IBS [Ladabaum et al. 2012]. Further, patients with IBS were more likely to have psychiatric conditions than individuals without IBS [Gulewitsch et al. 2011; Singh et al. 2012]. A significantly greater percentage of patients with severe IBS had at least one psychiatric disorder compared with patients with mild or moderate IBS (94.4% versus 35.7% and 76.1%, respectively; p = 0.003 and p = 0.02) [Singh et al. 2012]. Thus, the pathophysiology of IBS is multifactorial in nature.
Management of IBS: established approaches
Patients with IBS may benefit from lifestyle changes (e.g. exercise, dietary modification) [Fukudo et al. 2015]. In a study of patients with IBS undergoing military training, 62.9% of patients had improvement from baseline in bowel habits after 9 weeks of lifestyle modification imposed by the training (i.e. no smoking or alcohol consumption, regular meals, physical activity) [Kang et al. 2011]. After 9 weeks of training, mean stool frequency decreased from baseline, and the percentage of patients with normal stools (defined as Bristol stool scale score of 3, 4, or 5) increased from baseline (p = 0.05). In a ran-domized, controlled study of patients with IBS, patients in the group with increased physical activity experienced significant improvement from baseline in IBS severity compared with patients who had no change in physical activity after 12 weeks (p = 0.003) [Johannesson et al. 2011]. Further, patients who increased physical activity had significant improvement from baseline in the emotional (p = 0.002), sleep (p = 0.03), energy (p = 0.006), physical function (p = 0.001), social role (p = 0.001), and physical role (p = 0.008) dimensions of the IBS-QOL instrument after 12 weeks. Thus, increased exercise is a lifestyle change that appears to benefit patients with IBS.
Dietary modification is a therapeutic option preferred by patients with IBS, but may be limited by issues of long-term patient adherence and potential risk of nutritional deficiencies [Gibson et al. 2015]. A meta-analysis of four randomized, controlled clinical studies supported the efficacy of a low fermentable oligo-, di-, and monosaccharides and polyols (FODMAP) diet in patients with IBS, with an estimated number needed to treat (NNT) of 2.2 (95% CI 1.9–2.5) [Khan et al. 2015]. However, long-term maintenance of a low-FODMAP diet may not be practical [Muir and Gibson, 2013] and its efficacy beyond that of traditionally recommended IBS diet (i.e. regular ingestion of meals and snacks, reduced intake of certain foods such as onions, avoidance of carbonated beverages and artificial sweeteners, and ingestion of fiber) is uncertain [Böhn et al. 2015]. Gluten-free diets may be recommended for patients with IBS, although it is unclear how gluten affects IBS symptoms [Vazquez-Roque et al. 2013]. In a randomized, controlled study of patients with IBS-D (based on Rome II criteria), those receiving a gluten-free diet for 4 weeks achieved a significant reduction in daily stool frequency compared with patients receiving diets containing gluten (p = 0.04). The National Institute for Health and Care Excellence recommends avoidance and exclusion diets (e.g. FODMAP) in patients who have persistent IBS symptoms; however, such patients should be under the care of a dietary management expert. Finally, the efficacy and safety of probiotics, which are one of the most common dietary supplements used for the treatment of GI conditions, are limited by the number of quality clinical studies (e.g. adequate sample size, variability in outcomes examined) supporting their use [Didari et al. 2015; Dossett et al. 2014; Whelan, 2014] It is currently unclear which probiotic strain might be appropriate for IBS symptoms, and at what daily therapeutic dose [Whelan, 2014]. Probiotics, if used, should be administered in combination with conventional treatments for IBS for best effect [Ringel and Ringel-Kulka, 2011].
Carbohydrate malabsorption is associated with symptoms of IBS (i.e. abdominal pain, constipation, diarrhea), although affected individuals may not have IBS [Goebel-Stengel et al. 2014]. In a retrospective study of patients with IBS-like abdominal symptoms, 36% and 64% of patients had symptomatic lactose and fructose malabsorption, respectively, as determined by breath testing. Of patients with diagnosed IBS (by Rome III criteria), 22% had fructose malabsorption as determined by breath testing [Melchior et al. 2014]. In these patients, carbohydrate malabsorption can be managed by an elimination diet [Goebel-Stengel et al. 2014].
Given the number of patients with IBS with anxiety or depression, patients with IBS may benefit from psychological (psychotropic) therapies [American College of Gastroenterology Task Force on IBS, 2009; Fukudo et al. 2015]. Indeed, findings of a systematic review and meta-analysis that evaluated 32 randomized, controlled trials of psychological therapies found that a greater percentage of patients receiving control therapy (i.e. monitoring of symptoms, physician’s ‘usual management’, supportive therapy, or placebo) reported that their symptoms of IBS did not improve (76.1%), compared with the percentage of patients that did not experience improvement with psychological therapy (i.e. cognitive behavioral therapy, multicomponent psychological therapy, stress management, and relaxation therapy; 51.9%) [Ford et al. 2014b]. Thus, results of this meta-analysis suggest that psychological therapies are an effective management option in patients with IBS.
Serotonin is a ubiquitous signaling molecule in the body, primarily produced and stored by enterochromaffin cells in the GI tract [Gershon, 2004; Gershon and Tack, 2007]. Serotonin is important for normal GI motility, secretion, and visceral sensitivity [Bennett and Whitney, 1966; Delvaux et al. 1998; Gershon, 1999]. Inactivation of serotonin is mediated by the serotonin reuptake transporter (SERT), which is expressed on GI enterocytes [Gershon, 2004; Wade et al. 1996]. Colonic serotonin and SERT levels were decreased in patients with IBS compared with healthy individuals [Coates et al. 2004]. Selective serotonin reuptake inhibitors (SSRIs) can increase serotonin levels in patients with IBS by inhibiting the reuptake of this neurotransmitter [Chang et al. 2014]. An analysis of pooled data from 7 or 11 randomized, controlled studies of SSRIs or tricyclic antidepressants, respectively, found active treatment was associated with decreased symptoms of IBS compared with placebo (NNT = 4 for both) [Ford et al. 2014a]. Although efficacious for patients with IBS, psychotropic medications can be associated with adverse events (AEs) including drowsiness, dry mouth, and lower libido, with a number needed to harm of 9 [Clayton and Montejo, 2006; Ford et al. 2014a]. The American College of Gastroenterology weakly recommends the use of SSRIs and tricyclic antidepressants for relief of symptoms and pain in patients with IBS, and the National Institute for Health and Care Excellence notes one could consider them as a second-line therapy [Ford et al. 2014a].
Whereas the majority of patients with IBS-C or IBS-D have normal colonic transit times, 12% of patients with IBS-C have slow, and 27% of patients with IBS-D have fast colonic transit times [Tornblom et al. 2012]. A number of therapeutic agents that influence colonic transit are available for the treatment of patients with IBS. Lubiprostone, a selective chloride channel activator indicated for the treatment of women with IBS-C, had exhibited a greater degree of efficacy than placebo for relief of global IBS-C symptoms [Takeda Pharmaceuticals America, Inc., 2013; Drossman et al. 2009a; Ford et al. 2014a]. Results of two phase III clinical studies demonstrated that a significantly larger percentage of patients with IBS-C receiving lubiprostone for 12 weeks achieved overall response (i.e. moderate or significant relief of global IBS symptoms for ⩾2 of 3 months by patients self-report) compared with placebo (17.9% versus 10.1%, respectively; p = 0.001) [Drossman et al. 2009a]. However, lubiprostone has been associated with mild-to-moderate nausea, which may negatively impact use for some patients [Drossman et al. 2009a]. Loperamide, an effective antidiarrheal agent, is not currently recommended for the treatment of patients with IBS, based on insufficient evidence of global IBS symptom relief [Ford et al. 2014a]. Alosetron is a selective 5-hydroxytryptamine (5-HT3) receptor antagonist indicated for the treatment of women with severe, chronic IBS-D refractory to other therapies [Prometheus Laboratories, Inc., 2014]. Early clinical studies of alosetron in patients with IBS included mostly women and failed to demonstrate improved efficacy compared with placebo in men [Bardhan et al. 2000; Camilleri et al. 1999]. Alosetron provided adequate relief of IBS pain and discomfort, and improvement of stool consistency, in men with IBS-D, but did not improve stool frequency, urgency, bloating, or number of pain-free days compared with placebo [Chang et al. 2005]. Because of the risk of severe constipation and ischemic colitis in some patients, prescribing of alosetron is limited to healthcare providers enrolled in a special prescribing program, and patients must sign an acknowledgment form before initiating treatment [Prometheus Laboratories, Inc., 2014; Chang et al. 2010; Schiller and Johnson, 2008]. Analysis of postmarketing data acquired after implementation of the risk management program demonstrated that the incidence of ischemic colitis and constipation remained rare and decreased between 2002 and 2011 [Tong et al. 2013]. Another 5-HT3 receptor antagonist, ondansetron, has also been shown to improve symptoms in patients with IBS-D. In a randomized, placebo-controlled crossover trial, patients with IBS-D who received ondansetron 12–24 mg/day for 5 weeks experienced significantly greater improvement from baseline in mean stool form than placebo (p < 0.001) [Garsed et al. 2014]. In addition, patients who received ondansetron had reduced mean numbers of days per week with urgency (p < 0.001) and mean numbers of days per week with bloating (p = 0.002) compared with placebo [Garsed et al. 2014]. The most frequent AE with ondansetron was constipation, which occurred in a greater percentage of patients who received ondansetron (9%) than placebo (2%); however, most patients responded to dose reduction and continued with the trial [Garsed et al. 2014].
Abnormal contraction of smooth muscle within the GI tract may underlie some IBS symptoms, especially pain; therefore, agents that relax smooth muscles such as antispasmodics and peppermint oil have been evaluated in patients with IBS. A 2014 systematic review of available randomized, placebo- or ‘no treatment’-controlled trials for various antispasmodics (23 trials; 2154 patients) demonstrated that antispasmodics significantly improved IBS symptoms to a greater extent than placebo [NNT for antispasmodics 5 (95% CI 4–9)]. However, the effectiveness of individual antispasmodic agents varied, with only otilonium, hyoscine bromide, cimetropium bromide, pinaverium bromide, and dicyclomine hydrochloride showing benefits above placebo. AEs with antispasmodics were more common than with placebo [relative risk (RR) 1.6; 95% CI 1.1–2.4; number needed to harm 20 (95% CI 9.5–333)]; nonserious AEs of dry mouth, dizziness, and blurred vision were commonly reported [Ford et al. 2014a]. Two systematic reviews of ran-domized, placebo-controlled trials of enteric-coated peppermint oil in patients with IBS (5 trials, 482 patients; 9 trials, 726 patients) showed that peppermint oil was more efficacious than placebo for improving IBS symptoms (RR 2.2; 95% CI 1.8-2.8; NNT 3) [Ford et al. 2014a; Khanna et al. 2014]. Data are somewhat conflicting regarding the incidence of AEs. A pooled analysis study that included 7 randomized, placebo-controlled trials (474 patients) demonstrated a significantly greater risk of experiencing an AE than placebo (RR 1.7; 95% CI 1.3–2.4), whereas a separate study that pooled data from 5 randomized, placebo-controlled trials (482 patients) showed no significant difference from placebo (RR 1.3; 95% CI 0.8–2.1). This dissimilarity may reflect differences in the inclusion of individual studies for the pooled analysis rather than an actual clinical difference in AE occurrence.
The therapies that have been discussed within have been available for patients with IBS for a number of years and, in many cases, much longer. The purpose of this review is to provide gastroenterologists with an update on more recent therapies that have become available (i.e. since 2012), or are currently in development, for the treatment of patients with IBS.
Methods
A PubMed search of English-language articles available through 8 May 2015 was conducted using the following keywords to identify ran-domized, controlled studies performed in humans: ‘irritable bowel syndrome guidelines’, ‘rifaximin and irritable bowel syndrome’, ‘linaclotide and irritable bowel syndrome’, ‘alosetron and irritable bowel syndrome’, ‘ramosetron and irritable bowel syndrome’, ‘asimadoline and irritable bowel syndrome’, and ‘eluxadoline and irritable bowel syndrome’. Reference lists from review articles were used to identify additional studies for inclusion. Further, a ClinicalTrials.gov search of phase III or IV randomized, controlled studies of IBS (primary condition) was conducted on 15 February 2015 to identify additional agents for inclusion in this review.
Newer agents for the treatment of IBS
Targeting GI motility
The prosecretory agent linaclotide is a guanylate cyclase-C agonist indicated for the treatment of patients with moderate-to-severe IBS-C [Forest Laboratories, Inc., 2014; Yu and Rao, 2014]. Linaclotide binding activates guanylate cyclase-C, which leads to phosphorylation of the cystic fibrosis transmembrane conductance regulator (CFTR). Chloride and bicarbonate ions are secreted through the CFTR, while sodium absorption is decreased, leading to an increase of intestinal fluid into the GI lumen, and a subsequent increase in GI transit time [Layer and Stanghellini, 2014].
Linaclotide was shown to be well tolerated and efficacious for the treatment of patients with IBS-C in 2 randomized, double-blind, placebo-controlled studies, as well as additional post-hoc analyses (Table 1) [Castro et al. 2013; Chey et al. 2012; Macdougall et al. 2013; Quigley et al. 2013; Rao et al. 2012, 2014]. In one randomized, controlled study, a significantly greater percentage of patients receiving linaclotide had improvement in IBS symptoms as evaluated using the US Food and Drug Administration (FDA) efficacy endpoint [i.e. change from baseline ⩾30% in mean daily worst abdominal pain scores and an increase from baseline ⩾1 complete spontaneous bowel movement (CSBM) for ⩾6 of the first 12 weeks of treatment] compared with placebo (6 weeks, 33.7% versus 13.9%, respectively; p < 0.0001) [Chey et al. 2012]. A greater percentage of patients who received linaclotide versus placebo met criteria for the individual components of the combined endpoint [i.e. ⩾30% improvement in abdominal pain (48.9% versus 34.5%, respectively) and an increase of ⩾1 in the number of weekly CSBMs from baseline for ⩾6 of 12 weeks (47.6% versus 22.6%, respectively)]. Improvement in the FDA efficacy endpoint was sustained for 13 weeks, with 32.4% and 13.2% of patients receiving linaclotide or placebo, respectively, achieving the FDA efficacy endpoint (p < 0.0001) [Chey et al. 2012]. The percentages of patients who had ⩾30% reduction in average daily worst abdominal pain (36.9 versus 17.4% with linaclotide and placebo, respectively; p < 0.0001) and ⩾3 CSBMs with an increase of ⩾1 CSBM from baseline (15.7 versus 3.5%, respectively; p < 0.0001) also remained significantly greater with linaclotide than placebo during an additional 13 weeks of treatment [Chey et al. 2012]. These findings were supported by a second randomized, controlled study, which found that 33.6% and 21.0% of patients receiving linaclotide or placebo, respectively, achieved response as defined by the FDA efficacy endpoint (p < 0.0001) [Rao et al. 2012]. However, in this study, when patients who received linaclotide for 12 weeks were reassigned to receive placebo for 4 weeks, they experienced an increase in mean daily worst abdominal pain and a decrease in CSBMs comparable with that of patients who had received placebo for 12 weeks.
Summary of randomized, controlled clinical studies of linaclotide in patients with IBS a .

Studies presented are limited to the previous 4 years, due to the large number of clinical studies of linaclotide.
Defined as meeting both an improvement from baseline ⩾30% in mean of daily worst abdominal pain scores and an increase from baseline ⩾1 CSBM for ⩾6 weeks of first 12 weeks of treatment.
Abdominal pain responder defined as patient with improvement from baseline ⩾30% in mean weekly worst abdominal pain score or mean weekly abdominal discomfort score for ⩾6 weeks of first 12 weeks of treatment, with neither score worsening from baseline. Scoring of abdominal pain or discomfort ranged from 0 (none) to 10 (very severe).
Degree-of-relief responder defined as patient with ‘considerable’ or ‘complete’ relief of IBS symptoms (i.e. score ⩽2) for ⩾6 of first 12 weeks of treatment. Symptoms were rated weekly on a scale of 1 to 7, with rating of 1 for ‘completely relieved’, 4 for ‘unchanged’, and 7 for ‘as bad as I can imagine’.
AE, adverse event; CSBM, complete spontaneous bowel movement; DB, double-blind; EMA, European Medicines Agency; EQ-5D, European Quality of Life - 5 Dimensions; EQ-5D VAS, European Quality of Life - 5 Dimensions Visual Analogue Scale; IBS-C, constipation-predominant irritable bowel syndrome; IBS-QOL, Irritable Bowel Syndrome - Quality Of Life questionnaire; LS, least squares; PBO, placebo; PBO-C, placebo-controlled; qd, once daily; R, randomized; tx, treatment.
Post hoc analysis of the two randomized, controlled studies [Chey et al. 2012; Rao et al. 2012] showed that linaclotide significantly improved IBS-QOL from baseline to 12 weeks compared with placebo (p = 0.004 and p < 0.0001 for the two studies) [Quigley et al. 2013]. Indeed, all subscales of the IBS-QOL were significantly improved with linaclotide compared with placebo (p < 0.05 for all comparisons), with the exception of the activity interference subscale for patients in the Rao and colleagues study (p = 0.6). Although linaclotide improved symptoms of IBS and patient quality of life, this agent was associated with diarrhea [Chang et al. 2014; Chey et al. 2012; Quigley et al. 2013; Rao et al. 2012].
Targeting opioid receptors
The µ-, δ-, and κ-opioid receptors expressed in the GI tract are important for the regulation of gut motility and secretion [Bagnol et al. 1997; Holzer, 2009; Lamki and Sullivan, 1983]. Expression of the κ-opioid receptor is increased during inflammation and chronic visceral hypersensitivity [Hughes et al. 2014a]. In mice, expression of δ-opioid receptors increased under conditions of stress compared with no stress [Wade et al. 2012]. Patients with IBS had decreased blood and colonic levels of the endogenous opioid β-endorphin compared with healthy individuals [Hughes et al. 2014b].
The mixed µ-opioid receptor agonist and δ-opioid receptor antagonist eluxadoline was approved in May 2015 for the treatment of IBS-D. Patients with IBS-D receiving eluxadoline in a phase II dose-ranging study had greater efficacy compared with patients receiving placebo after 12 weeks [Dove et al. 2013]. A significantly greater percentage of patients receiving eluxadoline 25 mg or 200 mg twice daily achieved clinical response (i.e. decrease from baseline in mean worst abdominal pain ⩾30% and ⩾2 points, with a daily Bristol Stool Scale score of 3 or 4 on ⩾66% of daily diary entries within a week) after 4 weeks of treatment compared with placebo (12% or 13.8% versus 5.7%, respectively; p = 0.04 for 25 mg and p = 0.02 for 200 mg versus placebo). Clinical response was also significantly greater in patients receiving eluxadoline 100 mg twice daily after 12 weeks of treatment compared with placebo (20.2% versus 11.3%, respectively; p < 0.05). Eluxadoline improved the number of daily bowel movements and decreased the episodes of urgency and incontinence experienced by patients during the 3-month treatment period. Eluxadoline had an overall favorable safety profile, with nausea, abdominal pain, vomiting, and constipation the most commonly reported AEs. Constipation was most common in the eluxadoline 100 mg group (6%); however, no patients in this group discontinued from the study or rated the intensity of constipation as severe. Three serious incidences of pancreatitis were observed (two within the first two doses of eluxadoline 200 mg twice daily and one within 18 days of 25 mg twice-daily dosing), but all incidences quickly resolved without sequelae. Furthermore, US prescribing information warns of an increased risk of sphincter of Oddi spasm, resulting in pancreatitis, as well as an increased risk of pancreatitis not associated with sphincter of Oddi spasm [Patheon Pharmaceuticals, Inc., 2015].
Asimadoline is a κ-opioid receptor agonist currently in development for the management of patients with IBS-D with moderate-to-severe pain (Table 2) [Delvaux et al. 2004; Mangel et al. 2008; Mangel and Hicks, 2012; Szarka et al. 2007]. In a phase II, dose-ranging study of patients with IBS with a mean abdominal pain score of 1.5 (on a scale of 0 to 3, with 0 = no pain, 1 = mild pain, 2 = moderate pain, and 3 = severe pain), treatment with asimadoline 0.15, 0.5, or 1 mg twice daily for 12 weeks did not improve response (i.e. number of months with adequate relief of IBS symptoms) compared with placebo [Mangel et al. 2008]. However, response was significantly improved in patients with moderate or severe pain (pain score ⩾2) receiving asimadoline 0.5 and 1 mg, compared with placebo (p = 0.006 and p = 0.005, respectively). Further, subgroup analysis by IBS type showed that 46.7% and 20% of patients with IBS-D receiving asimadoline 0.5 mg or placebo, respectively, had adequate relief of pain symptoms for ⩾3 of 4 weeks (p = 0.01). A significantly greater percentage of patients with IBS-D receiving asimadoline 0.5 mg achieved a 25% increase in the number of pain-free days during the 12 weeks of treatment compared with placebo (42.9% versus 18%, respectively; p = 0.001). Bowel function improved in patients with IBS-D after treatment with asimadoline 0.5 mg: the number of daily bowel movements decreased significantly from baseline during month 3 with asimadoline compared with placebo (2.3 versus 0.3, respectively; p < 0.05) and the number of days with urgency was decreased during the 3 months of treatment at 0.5 or 1.0 mg. Thus, asimadoline may be a potential future therapy for the treatment of patients with IBS-D.
Summary of randomized, controlled studies of asimadoline in patients with IBS.
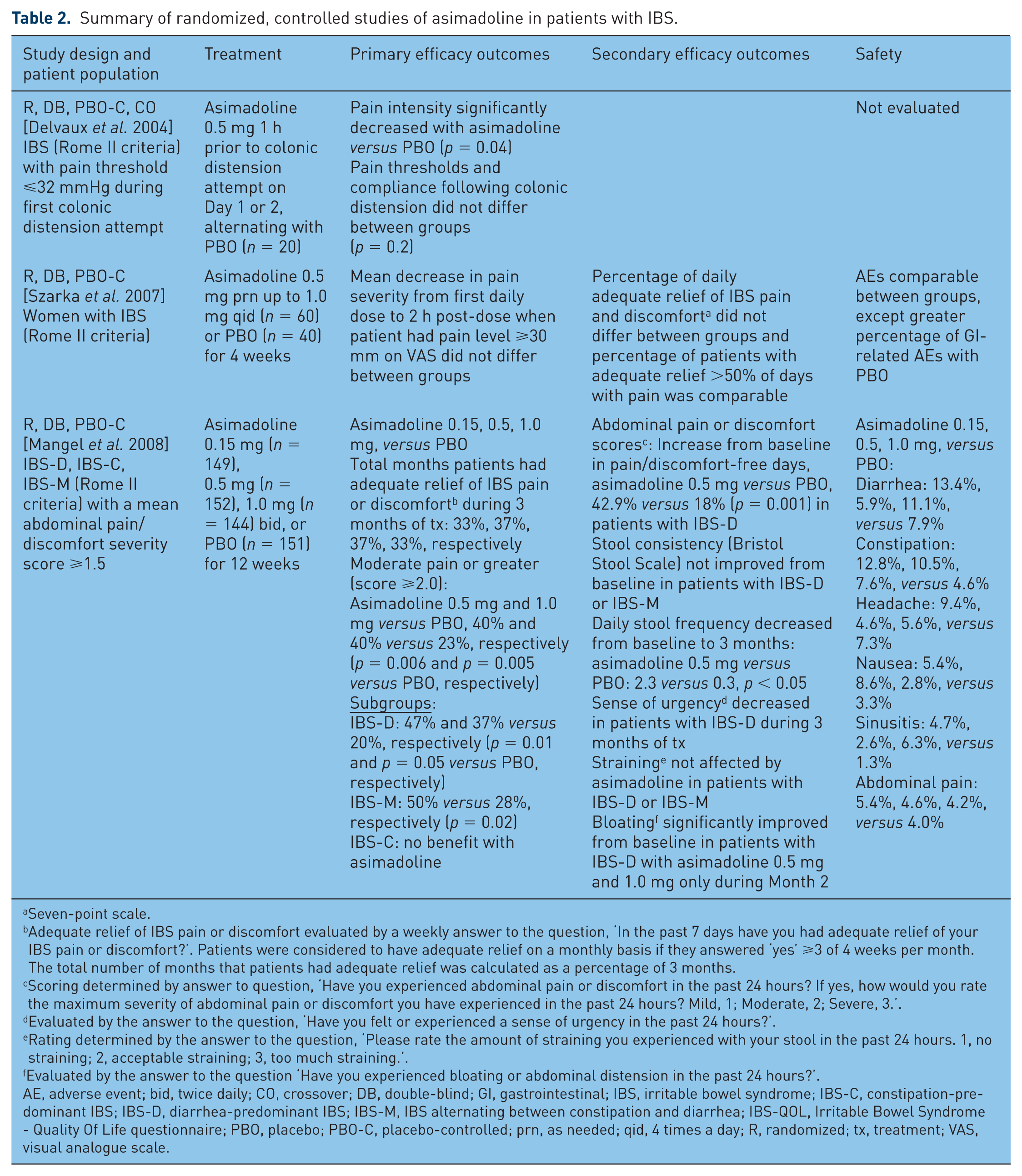
Seven-point scale.
Adequate relief of IBS pain or discomfort evaluated by a weekly answer to the question, ‘In the past 7 days have you had adequate relief of your IBS pain or discomfort?’. Patients were considered to have adequate relief on a monthly basis if they answered ‘yes’ ⩾3 of 4 weeks per month. The total number of months that patients had adequate relief was calculated as a percentage of 3 months.
Scoring determined by answer to question, ‘Have you experienced abdominal pain or discomfort in the past 24 hours? If yes, how would you rate the maximum severity of abdominal pain or discomfort you have experienced in the past 24 hours? Mild, 1; Moderate, 2; Severe, 3.’.
Evaluated by the answer to the question, ‘Have you felt or experienced a sense of urgency in the past 24 hours?’.
Rating determined by the answer to the question, ‘Please rate the amount of straining you experienced with your stool in the past 24 hours. 1, no straining; 2, acceptable straining; 3, too much straining.’.
Evaluated by the answer to the question ‘Have you experienced bloating or abdominal distension in the past 24 hours?’.
AE, adverse event; bid, twice daily; CO, crossover; DB, double-blind; GI, gastrointestinal; IBS, irritable bowel syndrome; IBS-C, constipation-predominant IBS; IBS-D, diarrhea-predominant IBS; IBS-M, IBS alternating between constipation and diarrhea; IBS-QOL, Irritable Bowel Syndrome - Quality Of Life questionnaire; PBO, placebo; PBO-C, placebo-controlled; prn, as needed; qid, 4 times a day; R, randomized; tx, treatment; VAS, visual analogue scale.
Targeting gut microbiota
The nonsystemic antibiotic rifaximin appears to have anti-inflammatory, host-response, and gut microbiota modulatory activities [Bajaj et al. 2013; Brown et al. 2010; Cheng et al. 2010; Debbia et al. 2008; DuPont and Jiang, 2004; Hopkins et al. 2014; Jiang et al. 2010a, 2010b; Maccaferri et al. 2010; Mencarelli et al. 2010, 2011; Schrodt et al. 2013; Terc et al. 2014; Xu et al. 2014]. Rifaximin received regulatory approval for the treatment of IBS-D in May 2015; several studies indicated a favorable efficacy and safety profile for rifaximin in IBS-D (Table 3) [Di Stefano et al. 2011; Pimentel et al. 2006, 2011, 2014]. The identically designed, randomized, placebo-controlled, phase III TARGET 1 and 2 studies examined the safety and efficacy of rifaximin 550 mg 3 times daily for 2 weeks in patients with IBS-D [Pimentel et al. 2011]. The primary efficacy endpoint (i.e. percentage of patients with adequate relief of global IBS symptoms for ⩾2 of the first 4 weeks after the 2-week treatment ended) was achieved by 40.7% and 31.7% of patients receiving rifaximin or placebo, respectively (p < 0.001). Further, a significantly greater percentage of patients receiving rifaximin had adequate relief of IBS-related bloating during the first 4 weeks after the 2-week treatment phase compared with placebo (40.2% versus 30.3%, respectively; p < 0.001). The results of these studies support that rifaximin has a sustained effect in patients with IBS-D, with a 2-week treatment course providing patients with IBS-D benefit for at least 3 months. In addition, three meta-analyses of five randomized, controlled trials of rifaximin for IBS (any subtype) demonstrated significant improvement in overall IBS symptoms with rifaximin compared with placebo (NNT 9–10.6) [Ford et al. 2014a; Menees et al. 2012; Shah et al. 2012]. The safety of rifaximin was favorable and generally comparable with that of placebo; no Clostridium difficile–associated diarrhea was reported in the TARGET 1 and 2 studies. Regarding its safety profile, meta-analyses of rifaximin trials showed no difference in the overall incidence of AEs compared with placebo [Ford et al. 2014a; Menees et al. 2012; Shah et al. 2012]. Furthermore, data indicated that 846 patients would benefit from rifaximin treatment for every 1 patient harmed (number needed to harm 8971; NNT 10.6) [Shah et al. 2012].
Randomized, controlled studies of rifaximin in patients with IBS.

Patients included in modified intention-to-treat analysis.
Defined as relief of symptoms for ⩾2 of first 4 weeks of treatment by self-report.
AE, adverse event; bid, twice daily; CO, crossover; DB, double-blind; CSBM, complete and spontaneous bowel movement; IBS, irritable bowel syndrome; IBS-C, constipation-predominant irritable bowel syndrome; NS, not significant; PBO, placebo; PBO-C, placebo-controlled; ppm, parts per million; R, randomized; tid, 3 times daily; tx, treatment; VAS, visual analogue scale.
An additional phase III study (TARGET 3) indicated that repeat treatment (up to three 2-week cycles of rifaximin 550 mg three times daily) with rifaximin in patients with IBS-D was significantly more efficacious than placebo in improving IBS symptoms (both abdominal pain and stool consistency) and treatment was well tolerated in patients with IBS-D [Lembo et al. 2014]. Rifaximin was not associated with clinically meaningful adverse effects on pathogen emergence or bacterial susceptibility to common antibiotic classes and no sustained disturbance of the overall gut (stool) microbiota was observed. Although not indicated for IBS-C, rifaximin has also been examined in patients with IBS-C, specifically in combination with neomycin [Pimentel et al. 2014]. Rifaximin plus neomycin significantly improved severity of constipation, and symptoms of bloating, straining, and abdominal pain for up to 4 weeks following 2-week treatment, compared with placebo [Pimentel et al. 2014].
Bloating and flatulence in patients with IBS may, in part, be due to bacterial overgrowth, may be diagnosed by breath testing, and is a distinct medical condition from IBS [Saadi and McCallum, 2013; Shah et al. 2013]. Antibiotics (e.g. ciprofloxacin, metronidazole) are generally used to treat small intestinal bacterial overgrowth (SIBO), which is thought to affect up to half of patients with IBS [Saadi and McCallum, 2013; Shah et al. 2013]. In the rifaximin TARGET 1, 2, and 3 studies, SIBO was not tested as part of the inclusion criteria, and also not included in the diagnosis of IBS. Rifaximin, with its favorable safety profile and demonstrated efficacy, appears to be a viable therapeutic option for patients with IBS with or without comorbid SIBO [Scarpellini et al. 2007].
Additional agents for treatment of IBS
The neurokinin-2 receptor antagonist ibodutant is currently in clinical development; however, data from randomized, controlled studies in patients with IBS have not yet been published [Trinkley and Nahata, 2014]. Plecanatide, an analog of the natural peptide uroguanylin, which regulates digestive activity, is currently in phase III trials in patients with IBS-C. Data from a phase II, randomized, controlled trial in patients with IBS-C (n = 424) indicated that various doses of plecanatide (including 1.0, 3.0, and 9.0 mg) provided significant improvement in the number of weekly CSBMs versus placebo (p ⩽ 0.05 for all comparisons) [Miner et al. 2014]. The most common AE with plecanatide was diarrhea [Miner et al. 2014]. Ramosetron, a selective serotonin receptor antagonist, is currently indicated for the treatment of men with IBS-D in Japan, although efficacy (i.e. improvement in global relief of IBS symptoms by patient report) was also demonstrated in women in one study [Matsueda et al. 2008a]. Efficacy was demonstrated in both men and women with IBS-D in a second, phase III study [Matsueda et al. 2008b]. In this study, overall response (i.e. global relief of IBS symptoms by patient report) was significantly greater with ramosetron compared with placebo after 1, 2, 3, and 4 months of treatment (p < 0.001) [Matsueda et al. 2008b], However, when stratified by sex, women reported significant relief of IBS symptoms at month 2 only compared with placebo, whereas men reported significant relief of IBS symptoms versus placebo at all time points. Further, the incidence of AEs, including abdominal distension, constipation, hard stool, and decreased white blood cell count, was ⩾3% higher for women than men. Thus, there are apparent differences in response in men versus women, but the reasons for these differences are currently unknown. Another treatment being examined for patients with IBS-C is chenodeoxycholic acid (CDC), a bile acid traditionally used for gallstone dissolution [Rao et al. 2010]. In a double-blind RCT, patients with IBS-C received placebo, CDC 500 mg, or CDC 1000 mg for 4 days [Rao et al. 2010]. CDC significantly accelerated overall colonic transit within 24 hours compared with placebo (p = 0.005) [Rao et al. 2010]. In addition, among females, CDC significantly improved stool consistency (p = 0.003), increased stool frequency (p = 0.18), and improved ease of passage (p = 0.24) versus placebo [Rao et al. 2010]. Lower abdominal cramping/pain was the most common AE with CDC and was significantly more prevalent in the CDC groups (42% to 45%) than in the placebo group (0%; p = 0.01). Diarrhea (17%-18% with CDC versus 0% with placebo) and nausea (9% to 25% versus 0%) were also common with CDC and were more prevalent with CDC compared with placebo [Rao et al. 2010]. Herbal preparations, such as STW 5 (Iberogast®; Bayer Corporation, Morrisville, NC) may improve IBS symptoms [Madisch et al. 2004]; however, clinical data are limited. There is also preliminary evidence that ketotifen, a mast cell stabilizer used in the treatment of asthma, may improve IBS symptoms [Klooker et al. 2010], but additional adequately powered studies are needed. Agents that have shown efficacy in other GI conditions [e.g. chronic constipation (elobixibat, or A3309 [Chey et al. 2011]), inflammatory bowel disease (AZD9056 [Eser et al. 2015] and 5-aminosalicylic acid [Feagan and Macdonald, 2012; Ford et al. 2011])] may also be beneficial for patients with IBS, but further research is needed.
Conclusions
IBS is a common condition managed primarily in an outpatient setting. Although patients with moderate-to-severe IBS seek medical care with greater frequency compared with patients with mild IBS, it is important to improve the health and overall well-being of all patients with IBS. Treatment should therefore include identification and management of psychologic comorbidities, such as anxiety and depression, as appropriate. The pathophysiology of IBS is unclear, but is thought to include genetic, immunologic, microbial, physiologic stress response, and psychosocial components. Management of patients with IBS includes lifestyle changes, dietary modification, use of psychotropic medications, psychological therapies, and over-the-counter agents targeting GI motility. Newer therapies targeting GI motility (i.e. linaclotide for IBS-C), GI opioid receptors (i.e. eluxadoline for IBS-D), and gut microbiota (i.e. rifaximin for IBS-D) have demonstrated efficacy and safety in patients with IBS. Further, additional therapies currently in phase II and III of development appear to show promise for the treatment of patients with IBS.
Footnotes
Acknowledgements
Technical editorial and medical writing assistance was provided under the direction of A.E.F.-O. by Sophie Bolick, PhD, Synchrony Medical Communications, LLC, West Chester, PA. Funding for this support was provided by Salix, a Division of Valeant Pharmaceuticals North America LLC, Bridgewater, NJ, USA.
Funding
This work was supported by Salix.
Conflict of interest statement
A.E.F.-O. has served on the speakers’ bureau for Salix, and has received payments for lectures.
