Abstract
Background:
Clonidine is considered an alternative treatment for refractory diarrhoea. The evidence in the literature is scarce and not conclusive. The present paper’s purpose is to gather available evidence and provide a systematic answer regarding the effectiveness of clonidine for diarrhoea.
Method:
We performed a systematic review of clonidine and its effect on diarrhoea. Meta-analysis was performed with a random effects model of the standardized mean difference (SMD) or the weighted mean difference and heterogeneity was quantified with I2 and publication bias was assessed with Egger’s and Begg’s test. Subgroup analyses and meta-regression were performed to investigate sources of heterogeneity. Any empirical study describing use of clonidine for diarrhoea in humans independent of age was included. For the meta-analysis, papers had to provide sufficient data to produce an effect measure, while case reports were not included in the meta-analysis and are discussed narratively only.
Results:
A total of 24 trials and seven case reports were identified. Clonidine (median dose 300 μg/day) has been used for treatment of diarrhoea in irritable bowel syndrome, faecal incontinence, diabetes, withdrawal-associated diarrhoea, intestinal failure, neuroendocrine tumours and cholera; studies were also performed on healthy volunteers. Results indicate a strong effect of clonidine on diarrhoea (SMD = −1.02, 95% confidence interval [CI] −1.46 to −0.58) with a decrease of stool volume by 0.97 l/day, stool frequency by 0.4 times/day and increase in transit time by 31 minutes. In a sensitivity analysis of studies with functional diarrhoea and sample size over 10 subjects, the effect was similar −0.99 (95% CI −1.54 to −0.43). There is however significant heterogeneity and publication bias. Heterogeneity decreased in subgroup analyses by condition but not with other factors examined. A limitation of the present study includes small study effects.
Conclusion:
Clonidine is effective for treatment of diarrhoea and should be considered as an alternative when all other medications have failed.
Introduction
Diarrhoea means different things to different patients: loose stool consistency and frequent bowel movements are prime characteristics. Diarrhoea is said to be chronic when it lasts more than 4 weeks. Chronic diarrhoea is an important medical condition that troubles patients [Schiller, 2005]. Treating diarrhoea is a difficult issue for the gastroenterologist and the general physician who needs effective medications [Vohmann and Hoffmann, 2013]. The differential diagnosis is broad, and diagnostic evaluation may be complex. In spite of the large number of causes of chronic diarrhoea, and in spite of extensive investigations, no cause for the symptoms is found in a large group of patients, who are referred to as having functional diarrhoea. The precise prevalence of chronic diarrhoea is unknown [Tack, 2012]. Diarrhoea kills an estimated 2.5 million people each year, with about 60–70% of them being children under 5 years of age [Guarino et al. 2012].
The main causes of chronic diarrhoea seem to depend on the socioeconomic status of the population surveyed [Li and Vaziri, 2012; Schiller, 2012]. In developed countries, the most frequent diagnoses made in patients with chronic diarrhoea are irritable bowel syndrome, idiopathic inflammatory bowel disease, malabsorption syndrome, chronic infections and idiopathic secretory diarrhoea. In less-developed countries, chronic bacterial, mycobacterial and parasitic infections are the most common causes of chronic diarrhoea; functional disorders, inflammatory bowel disease and malabsorption (from a variety of unspecified causes) are also common in this setting. Treatment of diarrhoea includes oral rehydration therapy, dietary modifications, antibiotics, absorbents, probiotics, anticholinergics, opiates and opiate-like medications, bile acid binders, budesonide, somatostatin analogues, alpha adrenergic agonists, proton pump inhibitors and antihistamines and calcium channel blockers [Halland and Talley, 2012; Kaiser and Surawicz, 2012; Li and Vaziri, 2012; Randall et al. 2012; Tack, 2012].
An important treatment of chronic diarrhoea which has proven resistant to standard treatments is clonidine, an alpha 2-adrenergic receptor agonist [Dharmsathaphorn, 1986; Fedorak and Field, 1987; Tack, 2012]. Activation of alpha 2-adrenergic receptors on enterocytes increases fluid and electrolyte absorption and inhibits secretion while at the same time decreasing bowel transit time [Schiller et al. 1985; Gregersen et al. 1989; Rubinoff et al. 1989]. In particular, clonidine can inhibit gastrointestinal motor activity by presynaptically inhibiting acetylcholine release from nerves in the myenteric plexus and at the neuromuscular junction, while it also has antinociceptive effects mediated by alpha-2 receptors in the spinal cord, brainstem, and forebrain [Unnerstall et al. 1984; Bharucha et al. 1997]. Clonidine reduced colonic and rectal tone, increased colonic and rectal compliance, and reduced colonic and rectal perception of distention in healthy subjects [Bharucha et al. 1997; Malcolm et al. 2000]. Secondary to the potential adverse effects including hypotension, this category should be cautiously used in treating diarrhoea especially in dehydrated patients. Due to the less-central hypotensive effect, clonidine is the medication of choice in this category. Clonidine has been used in treatment of chronic secretory diarrhoea of unknown aetiology, diarrhoea associated with narcotics withdrawal and diabetic diarrhoea, diarrhoea caused by chemotherapy or graft versus host disease, among many others [Li and Vaziri, 2012].
Until now, there has been no systematic assessment of the available literature on the effectiveness of clonidine for diarrhoea. This makes it difficult for clinicians to suggest this treatment as an option, since the literature is quite sparse and fragmented. A systematic review of this topic will provide answers for the clinician regarding clinical effectiveness and potential limitations.
Methods
Study eligibility criteria
The inclusion criteria were: any empirical study (abstract and full paper) describing treatment of diarrhoea with clonidine in humans independent of age. Diarrhoea was considered in a broad manner and anything that could be considered a proxy for diarrhoea was considered (e.g. stool volume changes, intestinal transit changes, etc.). Any studies with clonidine analogues (e.g. lidamidine) [Heredia Diaz et al. 1979, 1983; Heredia Diaz and Kajeyama Escobar, 1981; Goff, 1984; Edwards et al. 1986; Edwards and Read, 1986; Gasbarrini et al. 1986; Sninsky et al. 1986; Vicencio Tovar et al. 1986; Masukawa et al. 1987; Allison et al. 1988; Prior et al. 1988; Rodriguez Magallan et al. 1997; Awad et al. 2000] or with primary focus gastric emptying [Rosa-E-Silva et al. 1995; Asai et al. 1997; Thumshirn et al. 1999; Huilgol et al. 2002; Kwiatek et al. 2009] were excluded. There was no restriction to language of papers and the types of interventions included observational studies, randomized controlled trials, case series and case reports. For the meta-analysis papers had to provide sufficient data to produce an effect measure, while case reports were not included in the meta-analysis and are going to be discussed narratively only.
Search strategy and terms
PRISMA guidelines and MOOSE checklist for systematic reviews and meta-analyses were used [Stroup et al. 2000; Moher et al. 2009]. Electronic database searches were conducted in Google Scholar, PubMed/Medline, Scopus, EMBASE and Cochrane Library with no year limits. Publisher databases were also searched (Sciencedirect.com, link.springer.com, Wiley Library Online, Highwire press and Nature.com). The keywords for search were: clonidine, bowel, intestine, diarrhoea, diabetes, jejunostomy, ileostomy, irritable bowel syndrome, transit, stool, inflammatory bowel disease, cholera, infection, withdrawal. The date of search was 1 September 2015. The bibliographies from all included manuscripts and hand searching of relevant neurogastroenterology journals were used to identify further references.
Study selection, data extraction and quality assessment
The resulting studies (in abstract form) were assessed against the inclusion criteria. When there was insufficient information available in the abstract, the full text was reviewed. The investigators then extracted data from the selected studies including: author, year of publication, aim of the study, country, continent, sample size, mean age, male percentage, study design and results. The quality of studies (risk of bias) was assessed with Cochrane Collaboration’s tool [Higgins et al. 2008]. For the meta-analysis, studies were examined for p-values, means and standard deviations, correlation coefficients or other metrics depicting the effect of clonidine in diarrhoea. Metrics were converted to the standardized mean difference (SMD) [Lipsey and Wilson, 2001; Cooper et al. 2009] or weighted mean difference (WMD) where similar outcomes were identified [Borenstein et al. 2008]. Examples of outcomes included changes in stool volume, intestinal transit, and number of spontaneous bowel movements or any other relevant outcome depicting directly or indirectly improvement in diarrhoea.
Statistical analysis
Quantitative analysis was performed with Stata 12.0 (StataCorp LP, Texas) and Review Manager 5.3 (Cochrane Collaboration, Copenhagen). SMDs/WMDs were extracted from studies when available. The strength of association was categorized as following: small, SMD = 0.2; medium, SMD = 0.5; and large, SMD = 0.8 [Cohen, 1988; Faraone, 2008]; WMD has units and reflects the units of the outcome under description (e.g. litres per day for stool volume). A random effects model was used to produce a pooled estimate of the SMDs/WMDs. Statistical heterogeneity was assessed using Cochran’s Q test, which examines the null hypothesis that all studies are evaluating the same effect [Higgins et al. 2003]. Statistical significance for heterogeneity was set as p ⩽ 0.10. Heterogeneity was quantified using the I2 statistic, indicating the percentage of total variation across studies that is due to heterogeneity rather than chance [Gillum et al. 2000; Higgins et al. 2003]. An I2 value of 0% was considered to indicate no observed heterogeneity whilst a value >50% was considered to indicate substantial heterogeneity [Higgins and Thompson, 2002; Huedo-Medina et al. 2006; Bowden et al. 2011; Fragkos et al. 2014]. Heterogeneity was further investigated with subgroup analysis and meta-regression. Publication bias was assessed using funnel plots and Egger’s and Begg’s test [Begg and Mazumdar, 1994; Egger et al. 1997; Fragkos et al. 2016]. A funnel plot was created for the clinical measures with more than 10 studies [Sterne et al. 2011]. This is a scatter plot of the effect estimates from individual studies against a measurement of the study’s sample size or precision. Resemblance of a symmetrical inverted funnel supports that findings are due to sampling variation alone; thus, absence of bias [Sterne et al. 2011]. Sensitivity analyses were performed to quantify the effect of clonidine on diarrhoea when restricted to trials/case series with patient groups and sample size over 10 subjects. We carried out a cumulative meta-analysis of the effect of clonidine on diarrhoea based on the date of publication and condition.
Results
Systematic review and meta-analysis of trials
Studies and quality assessment
The systematic review of the literature resulted in 24 eligible trials/case series and seven eligible case reports (Figure 1 and Table 1). The case reports [Mcarthur et al. 1982; Sacerdote, 1986; Zimmermann et al. 1986; Roof, 1987; Migliore et al. 1988; Scholz et al. 1991; Schworer et al. 1995] are discussed narratively in a separate section below because of possible high placebo rates and are not included in the meta-analysis. The remaining 24 trials/case series included 10 studies with healthy volunteers (total sample size = 150 participants) [Schiller et al. 1985; Baxter et al. 1987; Pressman et al. 1987; Baumer et al. 1989; Gregersen et al. 1989; Rubinoff et al. 1989; Thollander et al. 1997, 1999; Mann and Shinkle, 1998; Viramontes et al. 2001] and 14 studies (total sample size = 493 patients) with faecal incontinence, irritable bowel syndrome, diabetes, alcohol withdrawal, heroin withdrawal, ultra-rapid opioid detoxification, short bowel syndrome (high output ileostomy or high rectal output), intestinal transplant and cholera patients [Fedorak et al. 1985; Bretzke, 1987; Gupta and Jha, 1988; Rabbani et al. 1989; Baumgartner and Rowen, 1991; Morali et al. 1991; Rovera et al. 1997; Camilleri et al. 2003, 2009; Mcdoniel et al. 2004; Buchman et al. 2006; Farzam and Najafi, 2009; Bharucha et al. 2010, 2014]. Results regarding statistical analysis of these studies are described in the following.
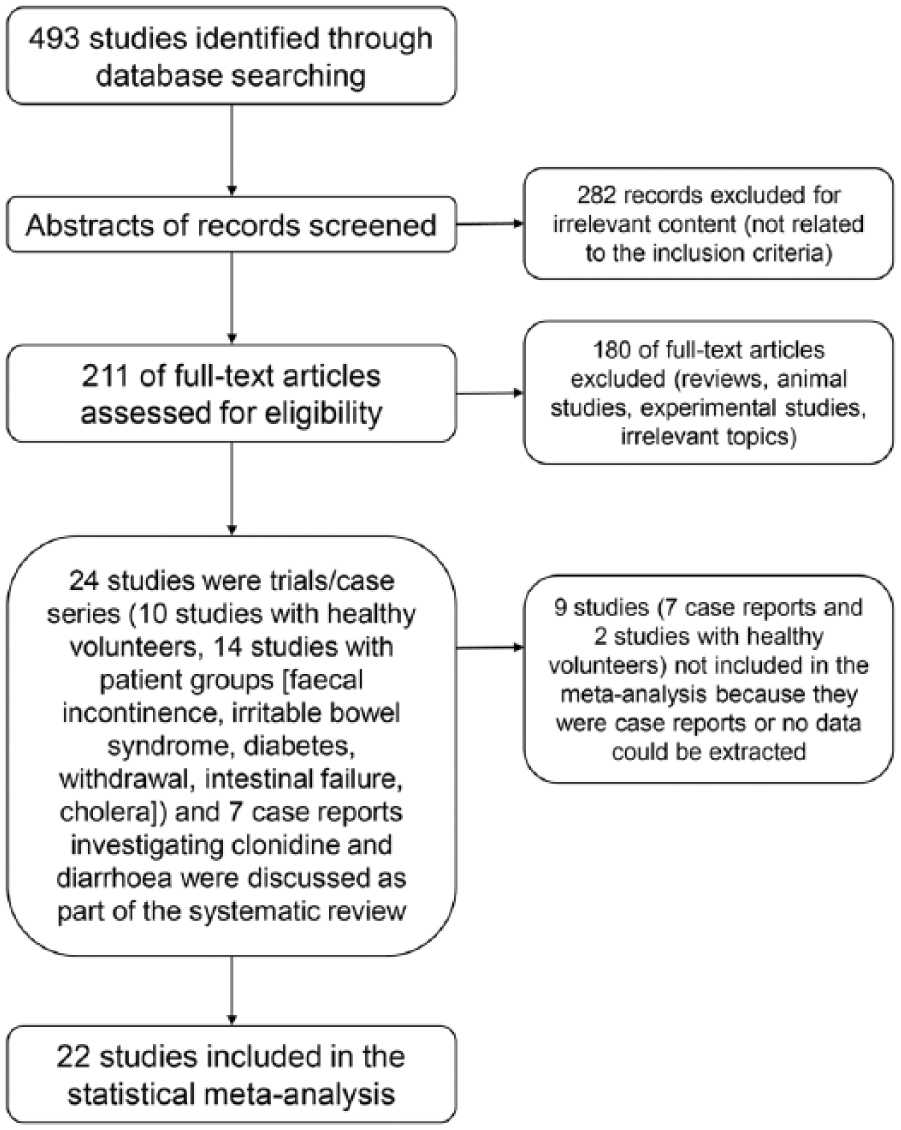
Flow diagram of the systematic review and meta-analysis.
Empirical studies for clonidine and diarrhoea.

Study not included in the meta-analysis because no data could be extracted from paper.
The total number of participants was 643 (range 2–120, median 13, mean 28, males 291). The mean age was 36 years (standard deviation [SD] 6). Clonidine was used as an oral or transdermal preparation with mean daily doses ranging between 110 and 900 μg/day (mean dose 344 μg/day, median dose 300 μg/day). Regarding quality assessment of the included papers, there is a high risk of selection bias due to lack of randomization and performance and detection bias due to lack of blinding; however the presentation of results seems adequate (see Figure 2 and Supplementary Figure 1).
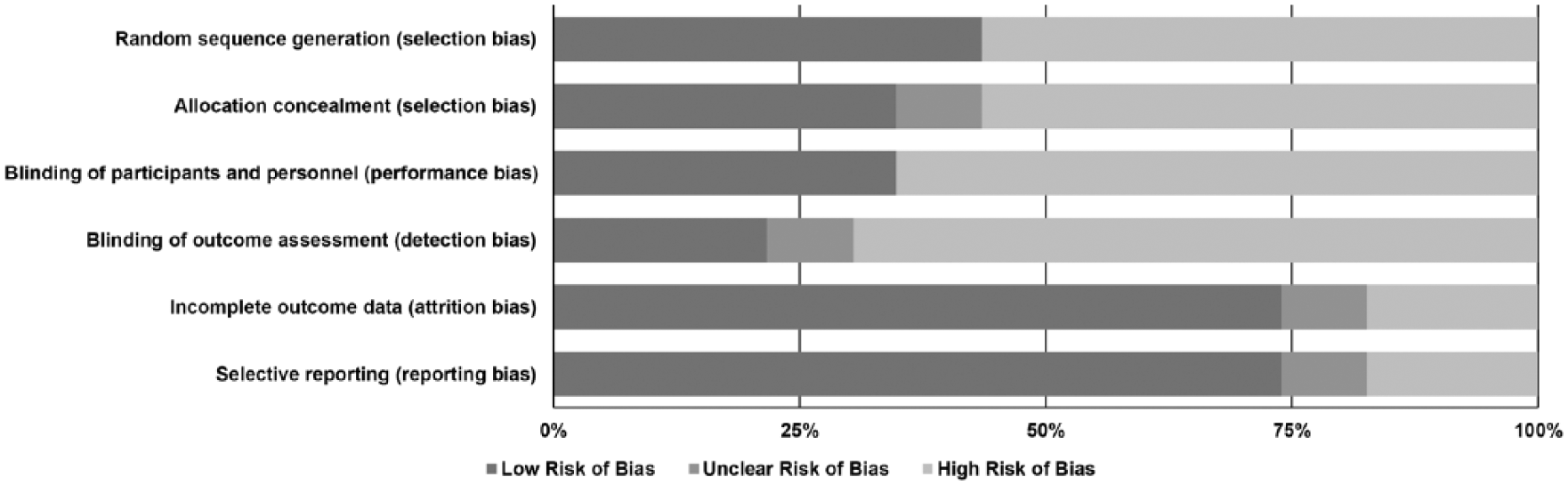
Risk of bias graph: reviewers’ judgements about each risk of bias item presented as percentages across all included studies.
Meta-analysis of the main outcome
Main effect
Meta-analysis was initially performed on studies involving healthy volunteers and patient groups, which included 22 studies that data could be extracted from. The studies by Thollander and colleagues [Thollander et al. 1997, 1999] did not allow for numerical data extraction. Overall, clonidine had a significant effect on diarrhoea with a mean SMD of −1.02 (95% confidence interval [CI] −1.46 to −0.58; see Figure 3A), which is strong according to Cohen’s criteria [Cohen, 1988]. However, there was significant heterogeneity (I2 = 82%). The funnel plot for publication bias indicates asymmetry (Figure 4) and Begg’s test was significant (p = 0.012) but not Egger’s test (p = 0.151). Hence, publication bias is possibly present due to small study effects.

Forest plots of the standardized mean difference (SMD) regarding the efficacy of clonidine for diarrhoea. (A) Forest plot of all studies (n = 22). (B) Forest plot with all studies (n = 14) except healthy volunteers. (C) Forest plot with all studies (n = 10) regarding functional diarrhoea (diabetes, irritable bowel syndrome, faecal incontinence and withdrawal). (D) Forest plot with all studies (n = 8) regarding functional diarrhoea and sample size over 10 subjects.
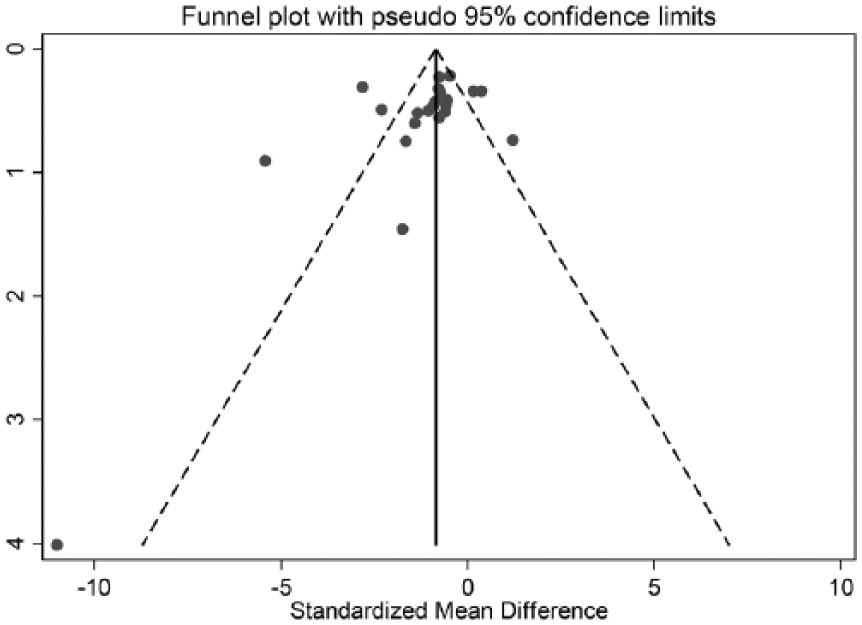
Funnel plot indicating probable publication bias in the reporting of results for clonidine’s efficacy for diarrhoea.
Sensitivity analysis and cumulative meta-analysis
The first step of sensitivity analysis was to include only the 14 studies with patient groups and not healthy volunteers [Fedorak et al. 1985; Bretzke, 1987; Gupta and Jha, 1988; Rabbani et al. 1989; Baumgartner and Rowen, 1991; Morali et al. 1991; Rovera et al. 1997; Camilleri et al. 2003, 2009; Mcdoniel et al. 2004; Buchman et al. 2006; Farzam and Najafi, 2009; Bharucha et al. 2010, 2014]. Clonidine still had a strong significant effect on diarrhoea with a mean SMD of −1.17 (95% CI −1.74 to −0.60; see Figure 3B), very similar to the previous overall SMD of −1.02. Heterogeneity was still present in a similar magnitude (I2 = 86%) as well as publication bias. When we included only the 10 studies with diagnoses that essentially constitute functional diarrhoea (diabetes, irritable bowel syndrome, faecal incontinence and withdrawal) [Fedorak et al. 1985; Bretzke, 1987; Gupta and Jha, 1988; Baumgartner and Rowen, 1991; Morali et al. 1991; Camilleri et al. 2003, 2009; Farzam and Najafi, 2009; Bharucha et al. 2010, 2014], clonidine still had a significant strong effect on diarrhoea with a mean SMD of −1.04 (95% CI −1.54 to −0.53; see Figure 3C). Furthermore, when we included only the studies with sample size over 10 patients [Gupta and Jha, 1988; Baumgartner and Rowen, 1991; Morali et al. 1991; Camilleri et al. 2003, 2009; Farzam and Najafi, 2009; Bharucha et al. 2010, 2014], the mean SMD did not change much: −0.99 (95% CI −1.54 to −0.43; see Figure 3D).
Further sensitivity meta-analysis was performed to examine the influence of individual studies in the overall mean SMD. No individual study lowers the effect of clonidine significantly since the overall mean always remains less than −0.87, indicating a constant strong effect. Two studies that lower the effect of clonidine to −0.87 are those of Rovera and colleagues [Rovera et al. 1997] and Camilleri and coworkers [Camilleri et al. 2009] (Figure 5A). Cumulative meta-analysis shows that a constant strong statistically significant effect of clonidine on diarrhoea is observed after publication of the ninth trial in 1989 (SMD ⩽ −0.74), while a significant strong effect appears as early as 1985 with the second trial (SMD = −1.45); see Figure 5B. Although subsequent trials have increased the precision of the point estimate, no substantive change has occurred in the direction or magnitude of the treatment effect. Cumulative meta-analysis by condition shows that after inclusion of studies with healthy volunteers the SMD does not change substantially, ranging between −1.02 and −1.18 with more precise confidence intervals (Figure 5C).

(A) Sensitivity analysis for each individual study. (B) Cumulative meta-analysis by year. (C) Cumulative meta-analysis by condition.
Heterogeneity analysis
Next, we examined the potential sources of heterogeneity. We performed subgroup analyses with respect to geographical information (country and continent of study), study criteria (randomized or not, controlled or not), the condition under which the effect of clonidine was examined and the effect type [clonidine effect (pre- and post-treatment); clonidine versus placebo; clonidine effect (condition versus healthy controls); clonidine dose effect; clonidine versus codeine; and clonidine versus chlordiazepoxide). Results are shown in Table 2 and funnel plots for subgroup analyses are shown in Supplementary Figures 2–7. With respect to study criteria and geographical information, heterogeneity remained in the subgroups (I2 > 50%), indicating that they were not potential factors. Interestingly though, in certain subgroups of the effect type and condition heterogeneity was low (I2 < 50%). With respect to condition, heterogeneity was not present anymore in the subgroups of diabetes, faecal incontinence and withdrawal; and regarding effect type, I2 was still high when effect was compared pre- and post-clonidine (80%), against placebo (59%) and healthy controls (94%) but was 0% when clonidine was compared against chlordiazepoxide in withdrawal.
Subgroup analyses of the efficacy of clonidine.
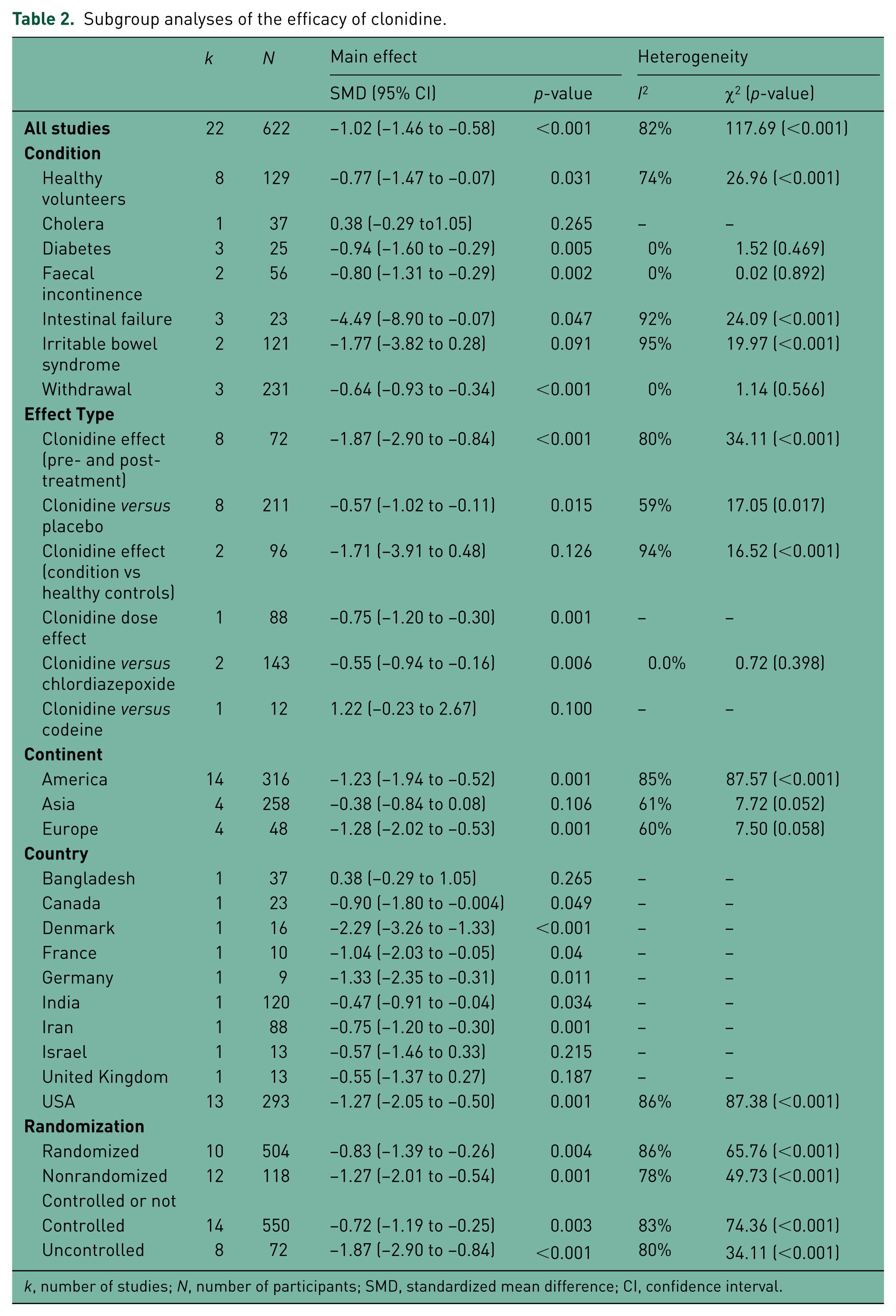
k, number of studies; N, number of participants; SMD, standardized mean difference; CI, confidence interval.
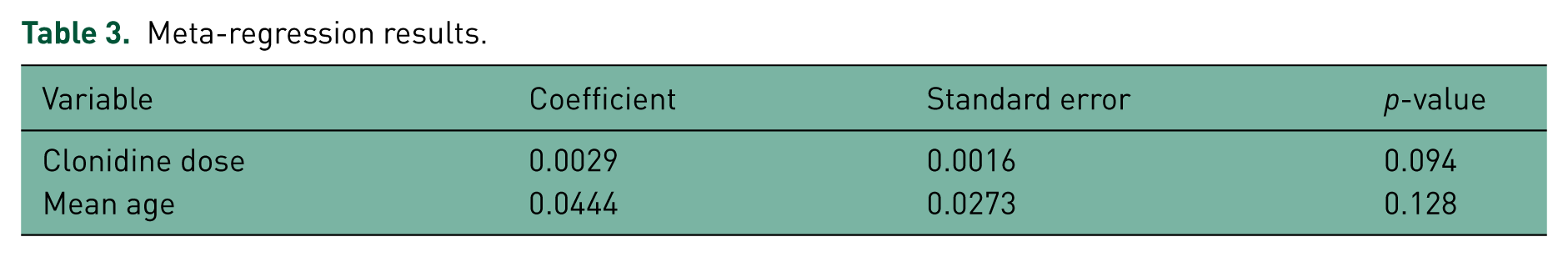
We finally examined continuous variables that could be sources of heterogeneity. The results of meta-regression are shown in Table 3. Clonidine dose and mean age do not predict heterogeneity in the SMD (coefficient p-values >0.05).
Meta-regression results.
Other outcomes
Stool volume was described in six papers [Fedorak et al. 1985; Schiller et al. 1985; Rabbani et al. 1989; Rovera et al. 1997; Mcdoniel et al. 2004; Buchman et al. 2006]. The WMD was calculated: clonidine reduced stool volume by −0.97 l/day (95% CI −2.27 to 0.33; I2 = 97%). The SMD indicated a strong significant decrease in stool volume (SMD = −2.08, 95% CI −3.88 to −0.27; I2 = 89%; see the Forest plots shown in Supplementary Figures 8 and 9).
Stool frequency was described in five papers [Fedorak et al. 1985; Bretzke, 1987; Camilleri et al. 2003; Bharucha et al. 2010, 2014]. The WMD was calculated: clonidine decreased stool frequency by −0.4 times/day (95% CI −1.0 to 0.2; I2 = 27%) [Bretzke, 1987; Camilleri et al. 2003; Bharucha et al. 2010, 2014]; when clonidine was compared pre- and post-treatment it decreased stool frequency by −1.5 times/day (95% CI −4.2 to 1.2; I2 = 64%) [Bretzke, 1987; Bharucha et al. 2010] and when clonidine was compared with placebo it decreased stool frequency by −0.3 times/day (95% CI −0.7 to 0.3; I2 = 0%) [Camilleri et al. 2003; Bharucha et al. 2014]. Finally, the overall SMD indicated decrease in stool frequency (SMD = −0.24, 95% CI −0.64 to 0.15; I2 = 14%) [Fedorak et al. 1985; Bretzke, 1987; Camilleri et al. 2003; Bharucha et al. 2010, 2014] (Supplementary Figures 10 and 11). The decrease of stool frequency does not appear to be clinically significant and this result is consistent with the mode of action of clonidine.
Stool consistency was described in four papers [Fedorak et al. 1985; Camilleri et al. 2003; Bharucha et al. 2010, 2014]. The SMD indicated that there was a weak non-significant improvement in stool consistency after clonidine treatment (SMD = −0.30, 95% CI −0.77 to 0.18; I2 = 29%; see Supplementary Figure 12). Nocturnal incontinence was described in two papers [Fedorak et al. 1985; Bretzke, 1987]. The SMD indicated that there was an important reduction in the presence of this symptom after clonidine treatment (SMD = −2.20, 95% CI −3.53 to -0.87; I2 = 0%; see Supplementary Figure 13).
Gastrointestinal transit time was described in eight papers [Baxter et al. 1987; Pressman et al. 1987; Baumer et al. 1989; Rubinoff et al. 1989; Morali et al. 1991; Mann and Shinkle, 1998; Viramontes et al. 2001; Camilleri et al. 2003]. The SMD indicated an increase in transit time, however this was nonsignificant (SMD = 0.31, 95% CI −0.17 to 0.79; I2 = 53%; see Supplementary Figure 14). The WMD was also calculated: clonidine increased significantly orocaecal transit time by 0.52 hours (31.2 minutes; 95% CI 0.06−0.98 h; I2 = 61%) [Baxter et al. 1987; Pressman et al. 1987; Baumer et al. 1989; Rubinoff et al. 1989; Morali et al. 1991; Mann and Shinkle, 1998; Camilleri et al. 2003] (Supplementary Figure 15).
Furthermore, clonidine decreased rectal losses of sodium (SMD = −0.81, 95% CI −1.67 to 0.05; I2 = 59%; see Supplementary Figure 16) [Schiller et al. 1985; Rabbani et al. 1989; Buchman et al. 2006] and chloride (SMD = −0.75, 95% CI −1.34 to 0.16; I2 = 0%) (Supplementary Figure 17) [Schiller et al. 1985; Rabbani et al. 1989], but did not affect bicarbonate (SMD = 0.29, 95% CI −0.63 to 1.21; I2 = 53%; see Supplementary Figure 18) and potassium losses (SMD = 0.11, 95% CI −0.46 to 0.67; I2 = 0%; see Supplementary Figure 19) [Schiller et al. 1985; Rabbani et al. 1989] while it did not affect absorption of sodium (SMD = 0.31, 95% CI −0.44 to 1.06; I2 = 0%; see Supplementary Figure 20) and potassium (SMD = 0.20, 95% CI −0.55 to 0.94; I2 = 0%; see Supplementary Figure 21) [Schiller et al. 1985; Buchman et al. 2006]. Absorption of fat, nitrogen, magnesium, calcium [Buchman et al. 2006], chloride, bicarbonate and water [Schiller et al. 1985] were not affected.
Several other outcomes were described in papers which are discussed narratively. Bharucha and colleagues [Bharucha et al. 2010, 2014] performed an uncontrolled study and then a placebo-controlled study of the effect of clonidine in faecal incontinence. In the uncontrolled study there was a more prominent effect of clonidine reducing the days of faecal incontinence, faecal incontinence episodes and proportion of bowel movements being incontinent or with staining. There was greater satisfaction with therapy in the uncontrolled study than in the placebo controlled study but neither study seemed to have an effect on lifestyle, coping or depression or on loperamide use. Camilleri and colleagues [Camilleri et al. 2009] also did not find any effect of the use of clonidine for irritable syndrome on depression and anxiety as measured by the hospital anxiety depression scale.
Moreover, most studies did not find any effect of clonidine on anal pressures, rectal compliance or sensation [Camilleri et al. 2003; Bharucha et al. 2010, 2014]. However, Camilleri and colleagues [Camilleri et al. 2009] reported that clonidine reduced sensation threshold for pain and increased rectal compliance in patients with irritable bowel syndrome and Viramontes and colleagues [Viramontes et al. 2001] reported that clonidine increased colonic compliance and reduced fasting tone without altering colonic response to a meal, aggregate sensation to distensions and sensation of pain in healthy volunteers.
Finally, two studies examined the influence of certain gene variations on the effect clonidine. Camilleri and colleagues [Camilleri et al. 2009] reported that α2A, GNβ3 and SLC6A4 genotypes significantly modify responses to clonidine on sensory and motor gastrointestinal functions (rectal sensation of gas, urgency, pain) in health and irritable bowel syndrome but a later study by the group in patients with faecal incontinence [Bharucha et al. 2014] reported that the α2A-C1291G polymorphism did not predict effects of clonidine with respect to faecal incontinence severity score, Bristol stool score or quality of life.
Narrative discussion of case reports
There have been seven case reports in the literature describing the use of clonidine to treat severe refractory diarrhoea (Table 1). Their description is interesting, albeit should be treated with caution due to the lack of control and the possibility of placebo effect. The indications have been diabetes, tumours with neuroendocrine features (carcinoid syndrome, lung cancer with VIP secretion, VIPoma) and high output ileostomy. Dose range was 100–1200 μg/day and route of administration could be oral or transdermal. The highest dose of clonidine was used in a 46-year-old patient with high-output ileostomy (up to 10 l/day) who was treated with clonidine, after loperamide, tinctura opii, cholestyramine and somatostatin had failed to reduce stool volume to less than 6 l/day. Under combined treatment with clonidine 1200 μg/day and somatostatin 6 mg/day, which was well tolerated, stool weights were normalized within 24 hours. Neuroendocrine tumours seemed to require higher doses of clonidine and lower doses were used in diabetic patients.
Side effects
The side effects for each study are describe in Table 1. The most prevalent side effect was dry mouth with a mean risk ratio of 12.3 (95% CI 2.5–61.2; I2 = 0%) for clonidine versus placebo [Camilleri et al. 2003; Bharucha et al. 2014], appearing up to 100% of patients who had received clonidine [Bretzke, 1987; Rubinoff et al. 1989; Baumgartner and Rowen, 1991; Thollander et al. 1997, 1999; Bharucha et al. 2010, 2014]. Drowsiness, light-headedness and fatigue had mean risk ratios over 1 for clonidine versus placebo but with nonsignificant 95% CIs (2.37, 95% CI 0.80–7.00; 3.32, 95% CI 0.89–12.42; and 1.50, 95% CI 0.63–3.61, respectively; I2 = 0% for all analyses) [Camilleri et al. 2003; Bharucha et al. 2014]. Hypotension varied from between 0% [Fedorak et al. 1985; Mcdoniel et al. 2004; Buchman et al. 2006] and 45% [Gupta and Jha, 1988] in patients who received clonidine but there was a general conception that this was a drop in systolic blood pressure up to 32.9 units with no symptomatology [Rubinoff et al. 1989; Morali et al. 1991]; however, a few cases of severe orthostatic hypotension [Rovera et al. 1997] were described (Table 1). Finally, other side effects were cold/cough/fever, headache, constipation, bradycardia and tachycardia: all general autonomic symptoms.
Discussion
We performed a systematic review of studies examining the effect of clonidine against diarrhoea. This is the first such review to the best of our knowledge. As a general comment, we can observe that the literature regarding clonidine starts as far back as the 1980s, however its use and reporting comes in a more fragmented manner than one would expect: only 31 studies (mainly observational nonrandomized) spanning a period of almost 40 years.
This is not something to disregard. Although this study shows an objective decrease of diarrhoea by clonidine, the quality of studies is compromised by selection bias and the results might be limited by publication bias and heterogeneity. Interestingly, diabetes, faecal incontinence and withdrawal were three group subgroups without heterogeneity; no group however included many studies. Small studies effect was a pattern observed in the present review and we dare say it almost reflects some sort of caution regarding the use of clonidine. This may be due to its potential side effects, most serious being hypotension and rebound hypertension when stopped abruptly. This does not come as a surprise since it is something observed in our current practice at University College Hospital, London. Clonidine prescription requires pharmacy approval for unlicensed use and frequently patients need inpatient trials of this medication before commencing outpatient treatment. Moreover, its use by anaesthetists during anaesthesia could equally trouble the general practitioner or gastroenterologist who might not be willing to try this drug as an outpatient.
Nevertheless, the present study appears to dispel some of practitioners’ and specialists’ cautions. Most studies report an asymptomatic drop in systolic blood pressure, which is acceptable considering the severity of their patient’s diarrheal symptoms. Moreover, the use of clonidine appears to treat effectively their refractory diarrhoea, having failed multiple other treatments: the benefits outweighing the risk. The most reported side effect was dry mouth increasing the risk just over 12 times compared with placebo. The WMDs calculated from the study were only a subset of the whole group of studies because not all studies reported on raw values to allow for such calculations. This decreases the power of these calculations and should be kept in mind.
On another topic, this study seems to corroborate previous findings regarding clonidine’s acting mechanism. Clonidine seems to decrease on average one litre of stool per day, improving consistency and decreasing frequency by one movement every 2 days approximately while additionally increasing gastrointestinal transit up to 1 hour. It also reduces sodium and chloride rectal losses. The effect of clonidine seems to be stronger when clonidine was compared pre- and post-treatment.
Within its limitations, the present study suggests that clonidine is a considerable option for refractory diarrhoea. However, one must take into account the limitations which were the presence of heterogeneity, small sample size of individual studies which in its turn was amplified by design biases.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
