Abstract
Objectives:
Large-volume paracentesis (LVP) can be time and labor intensive depending on the amount of ascites removed and the method of drainage. Wall suction has been adopted as the preferred method of drainage at many centers, though the safety and benefits of this technique have not been formally evaluated. The primary objective of this study was to define the cost and time savings of wall suction over the traditional glass vacuum bottle method for ascites drainage. The secondary objective was to compare the safety profile and patient satisfaction using these two techniques.
Methods:
We conducted a randomized, controlled pilot study of the wall suction versus vacuum bottle methods for LVP in hospitalized patients. All LVPs were performed under ultrasound guidance by a single proceduralist. Patients with at least 4 liters removed received 25% intravenous albumin, 8 g/liter fluid removed. Demographic, clinical characteristics, and procedure details were recorded. Laboratory and hemodynamic data were recorded for 24 h prior to and 24–48 h post LVP. An electronic chart review was conducted to evaluate procedure-related complications. Data were compared using Fisher’s exact test, t test, or Mann–Whitney U test.
Results:
Thirty-four patients were randomized to wall suction at 200 mmHg (n = 17) or glass vacuum bottle drainage (n = 17). Wall suction was significantly faster and less costly than vacuum bottle drainage (7 versus 15 min, p = 0.002; $4.59 versus $12.73, p < 0.001). There were no differences in outcomes at 24 and 48 h post LVP, or at 60-day follow up.
Conclusion:
Performing LVP using wall suction resulted in significantly shorter procedure time and supply cost savings. There were no differences in outcomes between the groups, suggesting equivalent safety, though larger studies powered to detect small differences are needed. Given its efficiency, convenience, and cost effectiveness, wall suction may be a superior method of ascites drainage for LVP.
Introduction
Ascites is the most common major complication of cirrhosis, with approximately 50% of patients with compensated cirrhosis developing ascites over 10 years [Gines et al. 1987]. In addition to cirrhosis, ascites may result from other diseases such as congestive heart failure and malignancy. As a result, paracentesis is a routinely performed diagnostic and therapeutic procedure. While diagnostic paracentesis is a quick and simple procedure, therapeutic large-volume paracentesis (LVP) can be time and labor intensive depending on the amount of fluid removed and the method of drainage.
Historically the method of drainage used during LVP is a glass vacuum container bottle. In this method, the intra-abdominal drainage catheter is connected to a glass vacuum bottle by means of a puncturing device/needle. Once punctured, the glass vacuum bottle will fill until it reaches its limit (typically 1 liter) at which point the proceduralist will manually disconnect the catheter from that bottle and puncture the next empty glass vacuum container. This process continues until the desired amount of ascites is removed. This traditional method has several limitations. These include rate of ascites drainage, need for multiple containers, and requirement to manually switch between containers after each is filled. Additionally, these bottles are cumbersome to utilize and dispose of, and are costly to supply. Consequently, many hospitals and outpatient practices have transitioned to wall suction as an alternative method of ascites fluid removal. This method is thought to offer multiple benefits compared with the traditional glass vacuum bottle system, though these improvements have not previously been clearly defined. While a single study in the literature describes this method as ‘fast, convenient, and safe’, the complication profile of this method has also not been formally evaluated [Banimahd and Spinello, 2009]. There remains concern that rapid drainage of ascites as used in wall suction may increase the risk for paracentesis-induced circulatory dysfunction (PICD) with resultant renal injury, hemodynamic changes, and hyponatremia.
Despite its widespread use, prospective, randomized trials are lacking to validate wall suction as a superior technique for ascites drainage during LVP. We conducted a randomized, controlled pilot study of the wall suction versus vacuum bottle methods for LVP. Our primary objective was to define the time and cost savings of wall suction over the traditional glass vacuum bottle method for ascites drainage. The secondary objective was to compare the safety profile of LVP and patient satisfaction using these two techniques.
Methods
Patients
We evaluated 34 hospitalized patients who received medical care at the Johns Hopkins Hospital in Baltimore, MD, USA from August 2010 to June 2012. Individuals with ascites (regardless of etiology) who were identified as requiring therapeutic paracentesis by their primary inpatient medical team were recruited for the study. Hospitalized patients who were 18 years or older were eligible. Patients who were unable to provide written informed consent, were pregnant, in the intensive care unit, or had a creatinine of 3 mg/dl or higher or on hemodialysis were excluded. Patients with loculated ascites, peritoneal thickening, or who were previously enrolled in the study were also excluded.
Study design
After written informed consent was obtained, patients were randomized to either the traditional glass vacuum bottle method or the wall suction method for ascites drainage. Randomization was performed using a computer-generated randomization table.
Procedures
All LVPs were performed by a single hospital proceduralist (DL) to eliminate operator variability. The proceduralist remained with the patient for the entire duration of the procedure. Ultrasound guidance was used in all patients to identify the optimal procedure site. Paracentesis was performed using the standard sterile technique that has been previously detailed by Grabau and colleagues, save the final alteration to the method of ascites drainage [Grabau et al. 2004]. A single cannula with multiple side holes was used to drain the ascites for all procedures. Patients who were randomized to the wall suction at 200 mmHg arm had the sterile drainage tubing attached to plastic wall suction canisters that were connected in series instead of directly connecting to a single glass vacuum bottle (Figure 1). Each wall suction device was attached to a pressure gauge that could be set to a specific pressure setting. This gauge maintained the wall suction at that specific setting unless manually changed which allowed providers to monitor the level of suction pressure throughout the entire procedure. As such, calibration was not necessary. Each wall suction canister held up to 2 liters of fluid, and once filled, automatically drained into the adjacent empty canister. These current wall suction canisters are designed to prevent content from leaving the canister in the event of loss of vacuum, thus maintaining sterility.

Wall suction device demonstrating suction canisters connected in series.
All patients who had at least 4 liters of ascites removed were administered 25% intravenous albumin, 8 g/liter of fluid removed in accordance with current standard of care for LVP [Runyon, 2009]. Baseline demographic and clinical data were abstracted from electronic patient charts by a research team member prior to the procedure. Procedure details including anatomic procedure site, total volume of ascites removed, total procedure and drainage time, and number of drainage containers used were documented by a research team member on a standard data collection form during each procedure. Each patient also completed a post-procedure satisfaction survey at the end of the paracentesis. This included assessment of discomfort during the procedure, procedure length, and comparison to prior experience with LVP if applicable. Responses were based on a scale of 1–10, with 10 being very satisfied/comfortable and 1 being very unsatisfied/uncomfortable.
Baseline and post-procedural evaluations
All study patients had a baseline comprehensive metabolic panel (CMP), complete blood count (CBC), and an international normalized ratio (INR) drawn within 24 h prior to their procedure. Follow-up CMP, CBC, and INR were checked approximately 24 h following the procedure. The majority of patients had these labs checked again at approximately 48 h post procedure. Ascites fluid was sent for total protein, albumin, cell count and differential, gram stain and culture from each LVP.
Hemodynamic measures including heart rate, systolic blood pressure, and diastolic blood pressure were documented for the 24 h prior to and 24–48 h following the procedure. These measures were taken on average every 4–8 h and the range (documented as maximum to minimum values) for each variable was recorded for each 24 h period. An electronic chart review was conducted to evaluate for procedure-related complications, including bleeding and LVP-related organ injury. Development of acute kidney injury was defined using the Acute Kidney Injury Network (AKIN) parameters: an abrupt reduction in kidney function (<48 h) defined as an absolute increase in serum creatinine of at least 0.3 mg/dl, or a percentage increase in serum creatinine of at least 50% [Mehta et al. 2007]. Given that urine output was inconsistently documented for our patients, this parameter of the AKIN criteria was not applied. In addition, initiation of hemodialysis, time to repeat LVP and 60-day mortality were also documented for each patient based on follow-up electronic chart review.
Statistical analysis
Data were compared using Fisher’s exact test for categorical variables, Student’s t tests for parametric continuous variables, or Mann–Whitney U test for nonparametric continuous variables. A p value up to 0.05 was considered statistically significant. Statistical calculations were performed using StataIC 11 (StataCorp, College Station, TX, USA).
Approval
This study was approved by the Johns Hopkins Medicine Institutional Review Boards and written informed consent was obtained for all participants.
Results
Thirty-four patients were enrolled in the study and were randomized to either wall suction at 200 mmHg (n = 17) or glass vacuum bottle drainage (n = 17). There were no differences in baseline characteristics between the two groups (Table 1). The majority of patients were white and male. The etiology of ascites was diverse, including viral hepatitis, alcoholic liver disease, and malignancy, but the overall distribution was similar between the two groups. The majority (62%) of patients had undergone prior LVP. The median pre-LVP Model of End Stage Liver Disease score was the same between the two groups, 15. Baseline median pre-LVP creatinine was also not significantly different (0.9 versus 1.2 mg/dl in vacuum bottle and wall suction groups, respectively). There were no significant differences in other baseline labs, including pre-LVP hemoglobin, platelets, INR, sodium, and albumin. The majority of patients were on diuretics at the time of their procedure (64.7% and 58.8%, respectively). Their baseline hemodynamic parameters were also similar.
Baseline patient characteristics.
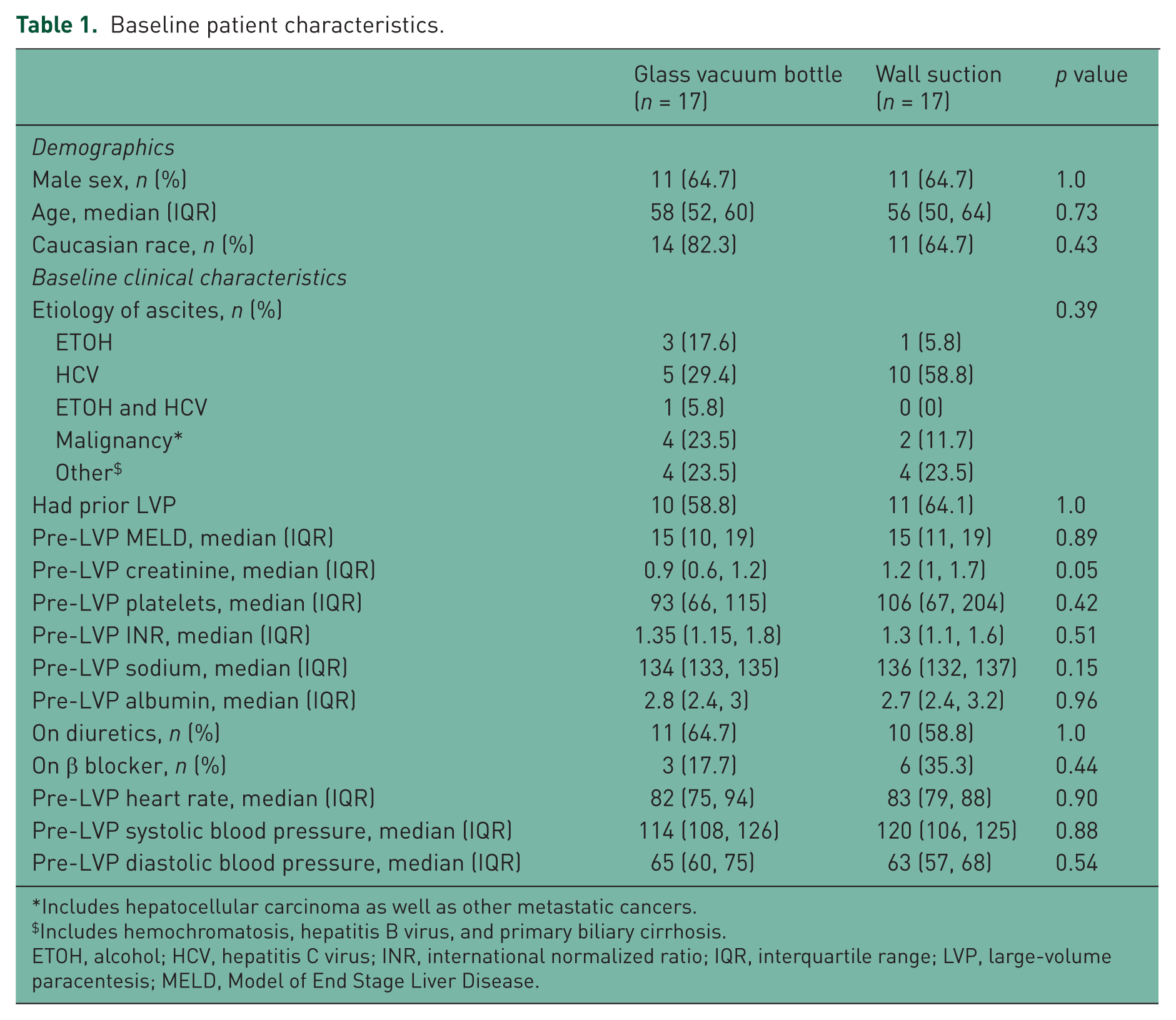
Includes hepatocellular carcinoma as well as other metastatic cancers.
Includes hemochromatosis, hepatitis B virus, and primary biliary cirrhosis.
ETOH, alcohol; HCV, hepatitis C virus; INR, international normalized ratio; IQR, interquartile range; LVP, large-volume paracentesis; MELD, Model of End Stage Liver Disease.
The total median volume of ascites removed was similar regardless of method used (3 liters in vacuum bottle and 3.7 liters in wall suction) (Table 2). There were no instances of premature termination of ascites drainage or instances when the paracentesis needle needed to be replaced. Drainage time was significantly faster in the wall suction group, with a median drainage time of 7 min (range 3.5–28 min) versus a median drainage time of 15 min for the vacuum bottle group (range 9–36 min) (p = 0.002). The supply cost for the wall suction group was also lower. The total mean supply cost was calculated as the cost of one basic paracentesis supply kit per procedure ($25.73), plus the cost of the total number of glass vacuum bottles needed ($3.23 each) or the cost of the total number of wall suction canisters needed ($1.347 each) plus the sterile connection tubing to connect to wall suction ($0.40 each) for each procedure. The mean total cost for supplies was $38.46 in the glass vacuum bottle group and $30.32 in the wall suction group. Given that we were interested in change in cost specifically related to method of ascites drainage, we also calculated the mean supply cost based only on drainage supplies (subtracting the cost of the basic kit which was a baseline cost per procedure). The mean cost for drainage supplies in the glass vacuum bottle group was $12.73 whereas the mean cost in the wall suction group was $4.59 (p < 0.001).
Procedure details and post-procedure events.
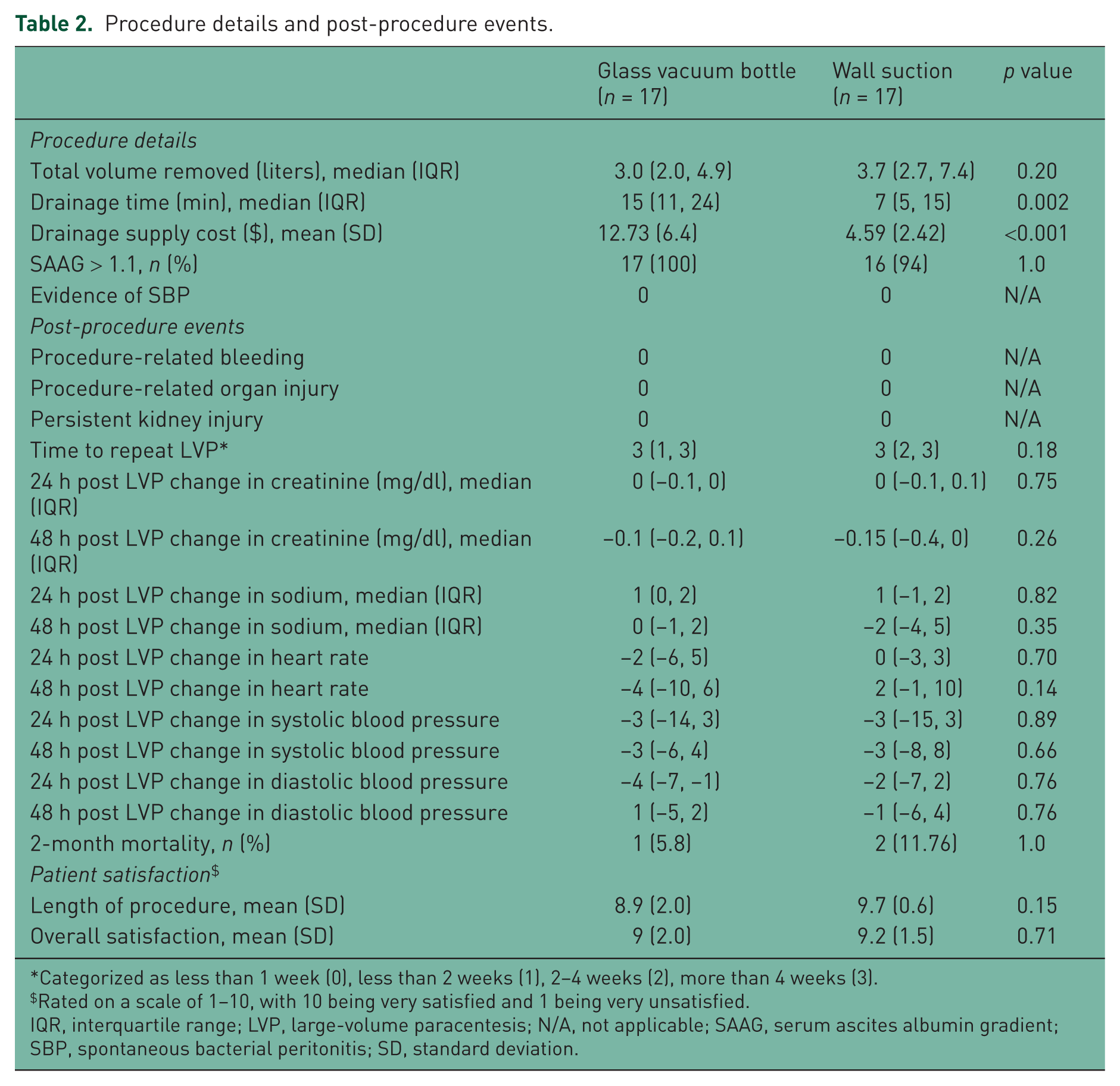
Categorized as less than 1 week (0), less than 2 weeks (1), 2–4 weeks (2), more than 4 weeks (3).
Rated on a scale of 1–10, with 10 being very satisfied and 1 being very unsatisfied.
IQR, interquartile range; LVP, large-volume paracentesis; N/A, not applicable; SAAG, serum ascites albumin gradient; SBP, spontaneous bacterial peritonitis; SD, standard deviation.
The ascites fluid analysis characteristics were similar between the groups. There were no procedure-related complications, including procedure-related bleeding, organ injury, or kidney injury in either group. Additionally, there were no differences in changes in mean heart rate, blood pressure, hemoglobin, creatinine, or sodium at 24 and 48 h post LVP between the groups. An additional sensitivity analysis was performed to assess the impact of restricting analysis to only patients with cirrhosis, and the results were not significantly changed. Follow-up measures including time to repeat LVP and 60-day mortality were also not significantly different between the two methods. Lastly, patient satisfaction in terms of level of discomfort, length of procedure and overall satisfaction were excellent for both methods. Ratings for overall level of discomfort were not affected by faster drainage time with wall suction. Patients with larger volumes of fluid removed overall had very high ratings for satisfaction with overall procedure time and overall experience with the LVP, but similarly high ratings were given by patients with small volumes of ascites drained.
Discussion
LVP is a frequently performed procedure in patients with ascites. Many patients require this procedure serially as symptomatic relief is usually transient, leading to LVP between every 7 to 21 days [Hou and Sanyal, 2009]. When performed using the traditional glass vacuum bottle method, LVP can be a lengthy and technically cumbersome procedure. In response to the drawbacks of this method, many institutions have transitioned to wall suction as the method for ascites drainage. The persistent nationwide shortage of glass vacuum bottles has also caused providers to change to wall suction for ascites drainage. However, case control analyses comparing these two methods are lacking to support this transition. In this context, our data related to the efficiency, cost effectiveness, and safety profile of wall suction versus glass vacuum bottle method of ascites drainage for LVP provide several insights into the benefits of this alternative method.
One of the most striking differences between the two methods was the overall time savings resulting from using the wall suction method. This includes both savings in overall time required to set up and perform the procedure, and time savings directly related to ascites drainage rate. By using a single piece of equipment consisting of multiple suction canisters connected in series, our technique reduced the time required for collection of procedural supplies and also eliminated the need to manually switch between containers as each filled. These methodological changes streamlined the steps required to perform each LVP and cut down on overall procedure time. In addition to cutting down on overall procedure time, there was a significant difference in rate of ascites drainage using the two methods. When using wall suction at 200 mmHg, ascites was drained in less than half the time required when using the glass vacuum bottles. This translates to a median and mean drainage rate of approximately 2 min/liter when using wall suction (range 1.1–3), but 5.3 min/liter when using the glass vacuum bottles (range 4.3–7.5). This improvement in efficiency is highly attractive given that many patients require significant volumes of fluid removal with each LVP. Decreased total procedure time may result in higher clinical productivity. While there is a hypothetical concern that the faster rate of drainage from wall suction may lead to premature termination of the paracentesis due to blockage of the suction catheter, the total volume removed was not different between the two groups and no instances of premature termination were encountered in our study. Lastly, although the total procedure time for outpatients may still be prolonged due to the time required to complete administration of plasma expanders, the time saved on the part of the proceduralist persists regardless of the procedure setting and thus this change in paracentesis method is still beneficial.
These time savings were not only beneficial from a provider standpoint, but also from a patient standpoint. As demonstrated by the patient satisfaction surveys, overall satisfaction with level of discomfort, procedure length, and overall experience was very high in our cohort. Given that ratings were excellent in both groups, we were unable to show a statistically significant improvement in patient satisfaction in this small patient population. Larger studies, especially those evaluating patients with significant volume of ascites removed (>8 liters), may be better able to demonstrate improved patient ratings as a result of decreased procedure time.
We also identified a significant 64% reduction in cost of drainage supplies associated with using wall suction rather than glass vacuum bottles. While the cost reduction in absolute dollars for each individual procedure is not large, when taken in aggregate at a systems level for centers that perform this procedure at a high frequency, this cost savings could be meaningful. Although not formally studied, cost of disposal of supplies from the wall suction method may also be lower given that many institutions are charged for disposal of hazardous materials by weight. Additionally, the waste from wall suction is less hazardous to handle given they are made from plastic as opposed to glass bottles that are prone to shattering. Overall, these significant cost savings are another appealing aspect of this alteration to LVP procedural methods, especially in centers that perform this procedure at high volumes.
Notably, in this pilot study the two methods of ascites drainage appeared to have equivalent safety profiles. For our study, we focused on both short-term and delayed complications related to method of ascites drainage for LVP. Cited complications of paracentesis include infection (peritonitis), bleeding, intestinal perforation/other organ injury, and PICD. A prospective study of 171 patients who underwent 515 paracenteses showed an overall complication rate of 10.5%, with 1.6% of these classified as ‘major complications’ [De Gottardi et al. 2009]. There were no procedure-related complications such as bleeding, organ injury, or peritonitis in either group (Table 2), which is not unexpected in this small sample size. In addition to these overt procedure-related complications, we were specifically interested in evaluating for clinical outcomes associated with the development of PICD. We were specifically interested in these outcomes given that prior studies had raised the concern that development of PICD may be related to faster rates of ascites fluid extraction [Coll et al. 2004; Cabrera et al. 2001; Sola-Vera and Such, 2004].
PICD was first described by Gines and colleagues and is defined as an increase in plasma renin activity (PRA) on the sixth day after LVP of more than 50% pretreatment value to a level of more than 4 ng/ml/h [Gines et al. 1988]. While the incidence of this complication is significantly reduced with the administration of intravenous albumin, PICD is thought to occur in 15% of patients following LVP with administration of intravenous albumin. PICD is characterized by the activation of the renin–angiotensin–aldosterone system with accentuation of arteriolar vasodilatation. This vasodilatation is hypothesized as being multifactorial in nature, including increased synthesis of nitric oxide, changes in intra-abdominal pressure and possibly related to other dynamics of the LVP, including rates of ascites extraction [Sola-Vera and Such, 2004]. Clinically, the manifestations of PICD are often silent in the short term, though some studies have noted faster reaccumulation of ascites, higher incidence of renal impairment and the development of hyponatremia in patients who develop PICD. In the long term, several studies have also noted reduced overall survival rates in patients who develop PICD [Sola-Vera and Such 2004; Ruiz-del-Arbol et al. 1997; Gines et al. 1996].
Several studies have investigated the relationship between rate of ascites extraction and development of PICD, though most had very small sample sizes and did not demonstrate clinically significant outcomes. The study by Coll and colleagues is most frequently cited [Coll et al. 2004]. This study noted that patients with decreased systemic vascular resistance (SVR) 3 h following LVP had higher flow rates of ascites extraction (1.18 versus 0.81 liters/min) and concluded that hemodynamic changes following LVP could be prevented by reducing the flow rate of ascites extraction. Of note, only 22 patients were evaluated and no clinically evident side effects were observed during or after LVP for any patient. Cabrera and colleagues raised concern that rapid changes in intra-abdominal pressure lead to hemodynamic changes, specifically changes in SVR, following LVP. This study similarly only evaluated a very small cohort (11 patients) and no clinically evident side effects were observed [Cabrera et al. 2001]. Studies by Shaheen and Grimm, and Wilcox and colleagues specifically evaluated the effects of more rapid drainage of ascites and did not find differences in clinical outcomes. Shaheen and Grimm evaluated 40 patients who underwent LVP with one group having fluid extraction rates 2.2–3.8 times faster than the other, and did not note differences in incidence of bleeding, hypotension, or ascites fluid leak [Shaheen and Grimm, 1996]. Wilcox and colleagues evaluated 52 patients who underwent LVP using a peritoneal dialysis catheter which resulted in a more rapid drainage of ascites and noted rates of peritonitis, ascites fluid leak, intestinal perforation, and change in creatinine similar to those seen using traditional methods [Wilcox et al. 1992].
To our knowledge, this study is the first randomized, prospective study evaluating both immediate and delayed complications of LVP based on method of ascites extraction. In an attempt to improve upon the prior evaluations, hemodynamic, laboratory, and clinical changes at 24 h, 48 h, and 60 days post LVP were measured. These time intervals were chosen to mirror time points used in prior studies on this topic to facilitate similar comparison of outcomes [Gines et al. 1996; Peltekian et al. 1997]. Based on the concerns raised from prior studies regarding possible adverse short-term outcomes related to rapid rate of ascites extraction, we focused on the development of hemodynamic changes, electrolyte changes, and renal insufficiency following LVP. We did not find any differences in these outcomes between the two groups. Given that patients with cirrhosis are presumed to be at higher risk of developing these complications compared with patients with other etiologies of ascites, we performed a sensitivity analysis to determine this subgroup’s specific level of risk and still did not find a difference between the two methods. On longitudinal follow up, we were interested in the time to repeat LVP as well as overall mortality given findings in prior small studies, but we again did not detect any differences based on method of ascites drainage. In the context of the limited small studies previously performed on this topic, our study offers novel data regarding both the immediate and delayed complication profile following LVP, depending exclusively on the method of ascites extraction.
The major strength of our study is the prospective assessment of a new method of ascites drainage in a sample of hospitalized patients in need of LVP. The longitudinal follow up builds upon the descriptive papers previously published which only focused on immediate complications of LVP following more rapid ascites extraction. Additional strengths include the randomization of participants as well as the elimination of operator variability by having all LVPs performed by the same skilled, experienced hospital proceduralist. There are several limitations to our findings. Given the lack of prospective studies on the topic, we conducted a pilot study with a small sample size which limits our ability to detect small differences in safety profiles between the two methods. Despite the small sample size, we demonstrated significant time and cost savings with the wall suction technique, although it would be reasonable to confirm in a larger prospective study. Additionally, our cohort of patients had a relatively low median volume of ascites removed (<4 liters), and at baseline did not have low systolic blood pressure (114–120). Patients with cirrhosis who have high volumes of ascites removed (>5 liters) and who have baseline hypotension may be at higher risk for developing post-LVP complications and future studies that target these patients who are at high risk may be better able to generalize the risk of using faster rates of ascites extraction to this patient population. Future studies may also be able to evaluate clinical and hemodynamic parameters for longer periods to detect delayed postprocedural changes, especially given that changes to PRA levels to define PICD are measured on the sixth day following LVP. It is encouraging, however, that 60-day post-LVP electronic chart review demonstrated no persistent post-LVP effects in our cohort. Lastly, future evaluations that include outpatients as well as other patients who were ineligible for enrollment in our study but who frequently require LVP (such as patients on hemodialysis) would improve the generalizability of our findings.
In conclusion, in this randomized, controlled pilot study, performing LVP using wall suction at 200 mmHg in hospitalized patients resulted in significantly shorter procedure time and supply cost savings without an increase in procedure-related complications. Given its efficiency, convenience and cost effectiveness, wall suction appears to be a superior method of ascites drainage for LVP. With the current nationwide vacuum bottle shortage, wall suction appears to be the optimal alternative method of ascites drainage for paracentesis. Given the widespread use of wall suction for ascites drainage, it would be important for our findings to be confirmed in a larger prospective study that includes more diverse patient populations and also focuses on patients at high risk of complications.
Footnotes
Acknowledgements
We are most grateful to the hospital proceduralist David Lichtman who performed each large volume paracentesis for this study.
Funding
This work was supported by the National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases (grant R01DK075990).
Conflict of interest statement
The authors declare that there is no conflict of interest.
