Abstract
Autoimmune hepatitis (AIH) is characterized by a T-cell rich infiltrate associated with lobular and interface hepatitis, hypergammaglobulinemia and production of autoantibodies. Genetic risk is linked to the HLA particularly DRB1*0301 and DRB1*0401 alleles in North American and European Caucasian populations. It has recently been suggested that functional deficiencies in CD4+CD25+CD127lowFOXP3+ regulatory T cells contribute to the breakdown of immune tolerance that results in AIH. Most patients respond to immunosuppressive therapy with corticosteroids and can be maintained in remission by low-dose corticosteroid treatment and/or azathioprine. For those who progress to end-stage disease liver transplantation is an effective treatment although it is associated with recurrence. In the future it is likely that biological therapies will allow more targeted therapy designed to switch the balance to immune regulation and thereby restore immune homeostasis. Although treatment for many cases is relatively straightforward and successful problems are encountered in those who fail to respond to standard treatment, are unable to tolerate it or relapse. In such cases alternative therapies should be considered. In addition treatment is complicated in some patients by comorbidity and special care is required during and after pregnancy. We will discuss the current and future therapeutic options for patients with difficult to treat AIH.
Introduction
Autoimmune hepatitis (AIH) is a condition of unknown aetiology, causing acute and chronic liver disease. It is characterized by a T-cell rich interface hepatitis that also contains B cells and plasma cells associated with hypergammaglobulinemia and autoantibody production. There is strong evidence that AIH is driven by antigen-specific T cells that expand as a consequence of a failure of immune tolerance, particularly in type 2 AIH in which the T- and B-cell autoantigen has been defined as a hepatic cytochrome CYP2D6. AIH is often initially asymptomatic and leads to chronic hepatitis if untreated. Most cases either progress rapidly to liver failure or become chronic with progression through cirrhosis to liver failure. The prognosis for most patients is good as a consequence of successful treatment with corticosteroids and immunosuppressive drugs but around 20% fail to respond fully to standard therapy and these patients are at risk of progressive disease. Restoration of immune tolerance either physiologically or pharmacologically would be an ideal option for the management of AIH.
This review discusses treatment of difficult AIH, including current and future therapeutic options, focusing on AIH in asymptomatic patients; AIH that does not respond to first-line treatment; fulminant AIH; AIH in special circumstances including pregnancy, the adolescent patient, the older patient and in the presence of coexisting liver disease; AIH which overlaps with primary biliary cirrhosis (PBC) or primary sclerosing cholangitis (PSC); post liver transplant AIH recurrence.
Prevalence/incidence
AIH is an uncommon condition with a prevalence of 16.9 per 100,000 and annual incidence of 1.9–3 per 100,000 [Boberg et al. 1998; Hodges et al. 1982]. It is characterized by female preponderance – women are affected three times more commonly than men [Boberg et al. 1998; Gregorio et al. 1997] – and affects all ages and races [Czaja and Freese, 2002; Zolfino et al. 2002], although black patients may have worse outcomes [Verma et al. 2007].
Immunohistopathology
Three types of AIH are identified on the basis of circulating autoantibodies [Oo et al. 2010]: type 1 AIH (most common): anti-smooth muscle antibodies (SMAs) or antinuclear antibodies (ANAs); type 2 AIH: anti-liver–kidney microsomal (LKM) antibodies; type 3 AIH: antibodies to soluble liver or pancreas antigens, although these antibodies react with the same antigen and may be referred to as anti-SLA/LP.
Findings on liver biopsy are typically of a plasma cell rich T-cell infiltrate in portal and periportal regions with interface hepatitis associated with ballooning and rosetting of periportal hepatocytes. Fibrosis initially develops in periportal areas, leading to bridging fibrosis and eventually cirrhosis [Baggenstoss et al. 1972].
Symptoms and diagnosis
Diagnosis may not always be straightforward and there is no one definitive diagnostic test. Typical findings at presentation are elevated transaminases, positive serology and hypergammaglobulinemia [Czaja and Freese, 2002] [Feld et al. 2005]. Other metabolic and infectious conditions must be excluded [Czaja and Freese, 2002; Manns et al. 2010a]. Liver biopsy confirms the diagnosis if typical findings described above are seen but in itself does not discriminate between other causes of chronic hepatitis. A scoring system proposed for diagnostic use in studies was subsequently simplified by the International Autoimmune Hepatitis Group for practicing gastroenterologists and hepatologists [Alvarez et al. 1999a; Hennes et al. 2008b] (Table 1).
Simplified scoring system for autoimmune hepatitis (AIH) by the International AIH Group.

ALT, alanine aminotransferase; ANA, antinuclear antibody; AST, aspartate aminotransferase; ERCP, endoscopic retrograde cholangiopancreatography; IgG, immunoglobulin G; LKM, liver–kidney microsomal; SLA, soluble liver antigen; SMA, smooth muscle antibody.
Although most patients with AIH will respond to immunosuppressive therapy, some will progress to fibrosis and cirrhosis, and presentation with established cirrhosis or liver failure is not uncommon. Patients with cirrhosis due to AIH are at risk of hepatocellular carcinoma (HCC) and other malignancies. Screening with standard measures of liver ultrasound and α-fetoprotein (AFP) measurement should be undertaken in patients with cirrhosis who are at risk of HCC. A prospective study of patients with AIH in the UK found HCC occurring at a rate of 1.1% per year, with all tumours occurring in patients with cirrhosis [Yeoman et al. 2008]. There is a higher risk of malignancy after onset of AIH, mainly due to hepatobiliary cancers, skin, colorectal cancers, and lymphoma [Werner et al. 2009]. In a large retrospective review of Swedish patients with AIH there was no association between immunosuppressive agents and lymphoma risk [Werner et al. 2009]. A US cohort study identified male gender, immunosuppression for greater than 3 years as predictive of HCC [Montano-Loza et al. 2009].
Treatment of autoimmune hepatitis
The aim of treatment is to induce and maintain remission and prevent subsequent complications of chronic hepatitis, such as cirrhosis and HCC. American Association for the Study of Liver Disease (AASLD) practice guidelines [Manns et al. 2010a] define remission clinically, biochemically and histologically: disappearance of symptoms; normalization of transaminases, bilirubin and γ globulin; normal hepatic tissue/inactive cirrhosis. The various therapies described in this paper have diverse but related sites of action, as shown in Table 2. An algorithm for treatment of AIH is shown in Figure 1.
Mechanism of action of different immunosuppressive agents.

GI, gastrointestinal; IL-2, interleukin 2; MAPK, mitogen-activated protein kinase; mTOR, mammalian target of rapamycin; NFAT, nuclear factor of activated T cells; NFκB, nuclear factor κB; TNF, tumor necrosis factor.
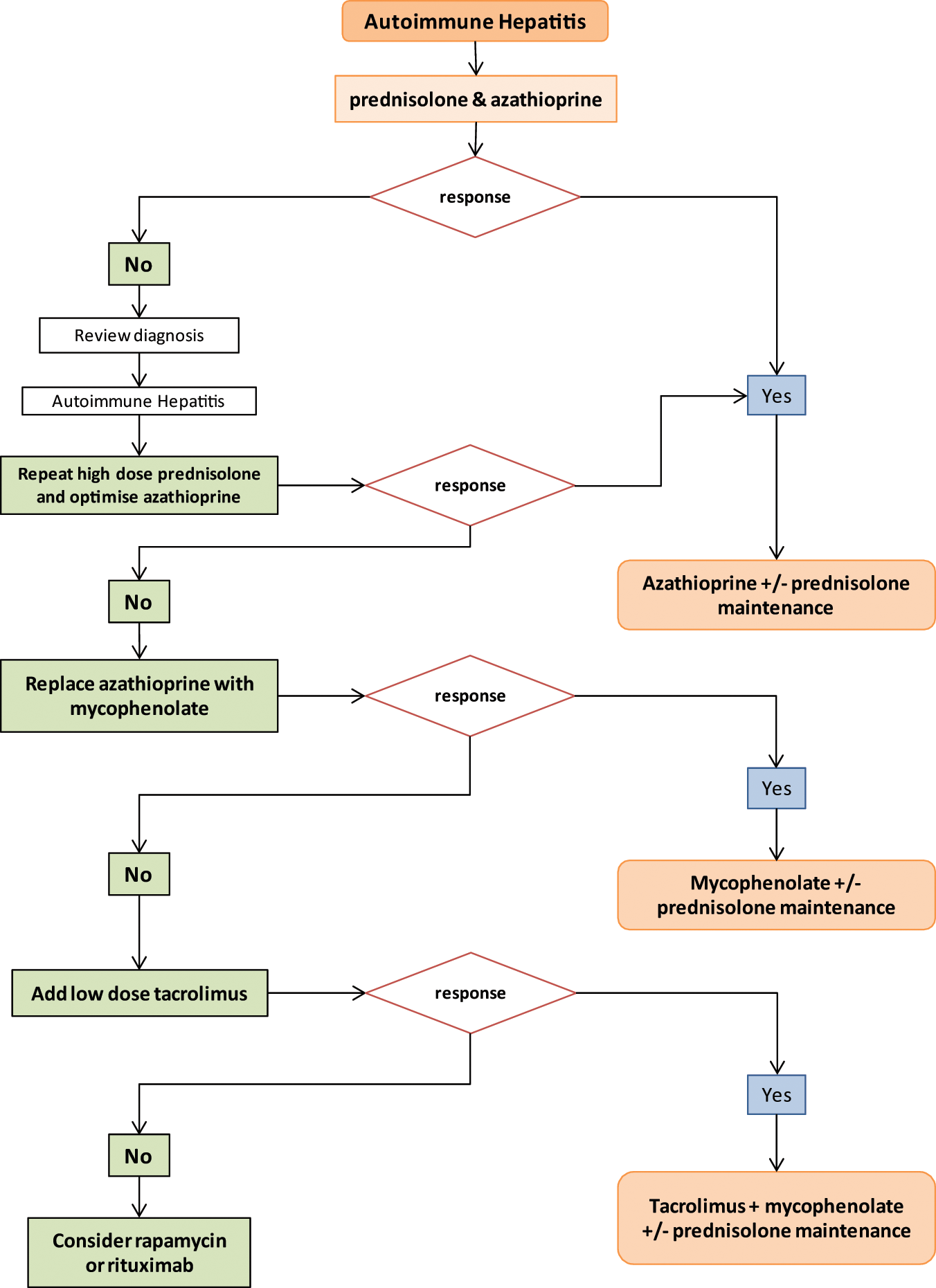
A practical algorithm to guide treatment in autoimmune hepatitis. The algorithm describes our current practice and as outlined above there is little evidence to support some of our choices. We accept that others may disagree with some of the steps but we believe that in the absence of good clinical trial data this represents a logical and reasonable approach to treating autoimmune hepatitis (AIH). The use of prednisolone and azathioprine is accepted practice and there is strong evidence that mycophenolate can be used as second-line therapy in those whose condition fails to respond to azathioprine. There is some evidence for efficacy of calcineurin inhibitors, including tacrolimus, but we concede that the evidence is not strong. We use tacrolimus rather than cyclosporine because it is associated with fewer cosmetic side effects, which is of particular benefit in adolescent patients. There are no clinical trial data to support the use of rapamycin or rituximab in AIH. However, the modes of action of both agents suggest they ought to be effective in AIH and they have proved to be effective in other immune-mediated diseases, including graft rejection (rapamycin) and rheumatoid arthritis and vasculitis (rituximab). Our own limited experience and other anecdotal evidence suggest their use should be formerly tested in AIH.
Corticosteroids have been central to treatment of AIH for 40 years after several early trials demonstrated efficacy [Cook and Mulligan, 1971; Kirk et al. 1980; Soloway et al. 1972]. Initial conflicting results regarding azathioprine [Mackay, 1968; Murray-Lyon et al. 1973] were resolved with data demonstrating clear efficacy and a lower side-effect profile compared with corticosteroid monotherapy when azathioprine is used to reduce or replace corticosteroids [Summerskill et al. 1975]. After induction of remission, monotherapy with azathioprine is often effective [Stellon et al. 1988]. Standard therapy, as recommended by guidelines, is high-dose corticosteroids (40–60 mg or 1 mg/kg) to induce remission, tapered over time with the addition of azathioprine. Some authors suggest that azathioprine should only be introduced once a corticosteroid response has been seen because of the small but real risk of azathioprine hepatitis [Manns et al. 2010a].
With treatment, 65% of patients will achieve remission within 18 months, and 80% within 3 years [Czaja and Freese, 2002; Feld et al. 2005]. Higher transaminase levels at the time of diagnosis predict successful treatment. Remission may allow for cautious withdrawal of medication; however the majority of patients will relapse after cessation of immunosuppression [Feld et al. 2005; Hegarty et al. 1983], particularly those with type 2 AIH in whom immunosuppression should probably never be stopped. If patients do relapse, retreatment with corticosteroids as per de novo disease is indicated in the first instance [Manns et al. 2010a] followed by lifelong maintenance therapy.
Difficult-to-treat autoimmune hepatitis
Treatment failure with steroids and azathioprine is seen in 9% of patients, 13% will improve but not to the point of achieving remission (incomplete response), and a further 13% do not tolerate standard therapy [Manns et al. 2010a]. Patient compliance with medication is crucial before making the diagnosis of treatment failure with conventional therapy. Revisiting the initial diagnosis to exclude alternatives to AIH is necessary. Alternative immunosuppressive regimens may be required to achieve remission in these cases.
It is not uncommon that patients with AIH present with fulminant hepatic failure. The presence of cirrhosis at the time of starting treatment confers higher risk of treatment failure [Feld et al. 2005]. AIH presenting in pregnancy is also challenging to manage. Sometimes, AIH can coexist with other liver diseases or overlap with or progress to PSC, which does not respond well to immunosuppression. Here we review all of these scenarios of difficult to treat AIH in clinical practice.
Asymptomatic patients
We have included this group because it can be difficult to decide when, or if, to treat the asymptomatic patient. Patients with asymptomatic disease are more commonly men [Kogan et al. 2002] but there are no differences in clinical features compared with symptomatic disease. Biochemically, those with asymptomatic disease had lower bilirubin, aspartate transaminase (AST), alanine transaminase (ALT) and immunoglobulin levels. Importantly, however, on histology, moderate or severe lobular hepatitis and periportal fibrosis was seen with similar frequencies in both asymptomatic and symptomatic groups, although symptomatic patients were more commonly found to have cirrhosis. The majority of asymptomatic patients in Kogan’s series became symptomatic during follow up. In addition, a number of patients who were asymptomatic at the onset of AIH developed cirrhosis, end-stage liver failure and required liver transplantation, albeit at a lower rate than those who were symptomatic at the onset of disease.
In a larger study of 126 patients by Feld and colleagues, 25% were asymptomatic at the time of diagnosis [Feld et al. 2005]. Similar to the findings of the Kogan study, asymptomatic patients had lower levels of bilirubin, AST and ALT. Asymptomatic patients had greater lobular inflammation scores on histology. With treatment, higher complete response rates were seen in asymptomatic patients, although outcomes (transplant or liver-related death) at 10 years did not differ. In this study fewer – 25% of asymptomatic patients – became symptomatic. Approximately half of the asymptomatic patients were treated, allowing comparison with those who were treated; however, the numbers of patients reaching endpoints (liver-related death, transplant or all deaths) were too small to reach statistical significance. There was a trend for greater mortality in the group who were not treated. The presence of cirrhosis at baseline had the greatest impact on survival, those patients without cirrhosis having 94% 10-year survival.
Thus, patients with asymptomatic disease may have marked histological disease despite less derangement of biochemical tests. In addition, these patients can progress to advanced liver disease. For these reasons, we recommend that treatment should be considered in patients with asymptomatic disease after liver biopsy to access the necroinflammatory activity. Patients without cirrhosis have an excellent prognosis and side effects of immunosuppressive medications should be taken into consideration. Older patients may not tolerate immunosuppressive drugs. Asymptomatic patients need to be followed to monitor for changes in their disease with serial liver biopsies or elastography. It should be noted that the AASLD guidelines suggest treating young patients who fall into this category [Manns et al. 2010a].
Autoimmune hepatitis that does not respond to conventional therapy
Some patients may fail to respond to conventional therapy or may be intolerant of standard treatment regimes. A complete failure to respond to initial high-dose corticosteroids is unusual and should result in a careful review of the initial AIH diagnosis. Treatment failure more often takes the form of partial responses or relapses. It is important to make sure that patients are adherent, particularly adolescents and young adults who are less likely to be compliant with therapy. If the patient’s condition has failed to respond to first-line treatment then other therapies must be considered. In the case of relapse the patient needs to be retreated with high-dose corticosteroids again.
Budesonide as an alternative to prednisolone
Side effects of corticosteroid therapy may preclude effective treatment and adolescent patients in particular may not accept the cosmetic effects of corticosteroids. Standard protocols use prednisolone but there is now evidence that the synthetic corticosteroid budesonide may be more effective with fewer side effects as a consequence of high rates of first pass metabolism resulting in increased local levels in the liver with relatively lower systemic exposure. Initially suggested to be effective in an open-label trial of 13 patients [Danielsson and Prytz, 1994], a large randomized controlled trial in 207 patients without cirrhosis and with new AIH or a flare of AIH demonstrated superiority of budesonide to prednisolone (both used in combination with azathioprine) with regard to normalization of transaminases [Manns et al. 2010b]. It should be noted that the prednisolone regimen was not standard [Manns et al. 2010a], but used a rapidly decreasing dose. Given the need for first pass metabolism in the liver to generate high local levels, budesonide should not be used in patients with cirrhosis in whom portal hypertension and shunting will reduce first pass metabolism. Budesonide also has the disadvantage of requiring 8 hourly dosing compared with prednisolone, which can be given once daily.
Alternatives to azathioprine for maintenance therapy
Azathioprine is a safe, effective and cheap drug that is the first choice for maintenance therapy once corticosteroids have induced remission. A small minority of patients develop toxicity in the form of bone marrow suppression or gastrointestinal intolerance and in some patients their condition fails to respond to maintenance therapy with azathioprine. There are no controlled clinical trials to inform choices for second-line therapy, but several drugs have been reported to be safe and effective in patients with AIH whose condition fails to respond to azathioprine in open-label and retrospective studies [Richardson et al. 2000; Devlin et al. 2004; Inductivo-Yu et al. 2007].
Mycophenolate mofetil
Mycophenolate mofetil (MMF) arrests DNA replication in T and B lymphocytes. MMF can cause marrow suppression but this is less frequently seen than with azathioprine because of the presence of a salvage pathway in lymphocytes. MMF is frequently associated with nausea and diarrhoea but these can be reduced by using divided doses and tend to improve with time. Response to MMF is associated with response to first-line immunosuppression, that is, azathioprine [Hennes et al. 2008a] in a retrospective review of 36 patients whose condition had failed to respond to standard therapy. A total of 43% achieved remission with MMF if they had been intolerant of azathioprine, whereas only 25% achieved remission with MMF if they had failed to achieve remission with azathioprine. Mycophenolate should not be used in young women of reproductive age, or those who are attempting in vitro fertilization therapy due to its potential teratogenicity.
Cyclosporine
Cyclosporine is a calcineurin inhibitor (CNI), which inhibits effector T-cell function. Therefore, there is a rationale for using cyclosporine in AIH, a T-cell-mediated disease. A small nonrandomized trial in 32 children without previous treatment demonstrated success in inducing remission with cyclosporine monotherapy [Alvarez et al. 1999b]. In a case series of adult patients whose condition had not responded to standard therapy, four of five patients showed response to cyclosporine therapy [Fernandes et al. 1999]. Adverse effects include neurotoxicity, hypertension and hyperlipidaemia, but the major drawback with this agent is the high risk of nephrotoxicity and its use should be confined to units with extensive experience of its administration, for example, transplant units.
Tacrolimus
Tacrolimus is also a CNI, which inhibits T-cell activation and cytokine release via FK-binding proteins. A retrospective study examining the use of tacrolimus in 11 patients with steroid-refractory AIH found that, in combination with azathioprine, tacrolimus improved transaminases and necroinflammatory activity and allowed cessation of steroids in the majority of patients [Aqel et al. 2004]. Another retrospective study of nine patients with steroid-refractory AIH described improvement in histological inflammation, fibrosis and serum ALT in patients treated with tacrolimus in combination with prednisolone [Larsen et al. 2007]. The side-effect profile of tacrolimus is similar to cyclosporine and again the risk of nephrotoxicity means this drug should only be administered by those experienced in using it. As with cyclosporine, drug levels must be monitored to reduce the risk of toxicity.
Cyclophosphamide
Cyclophosphamide is a cytotoxic agent, which is immunosuppressive when used at lower doses. It has been used to treat autoimmune diseases particularly vasculitis. A case series of three patients treated with cyclophosphamide and corticosteroids reported induction and long-term maintenance of remission [Kanzler et al. 1997]. Remission was induced with 1–1.5 mg/kg of cyclophosphamide in combination with a tapering dose of corticosteroids, initiated at 1 mg/kg, then, low doses of corticosteroids (2.5–10 g/day) together with 50 mg/day of cyclophosphamide were used for maintenance. Bladder toxicity is associated with cumulative dosing of cyclophosphamide and again it should probably only be given by clinicians experienced in its use. However, recent data show that cyclophosphamide can activate immune responses by suppressing regulatory T cells, an effect which, in theory, would not be beneficial in AIH. Until its mode of action is better understood, cyclophosphamide should probably be avoided in AIH [Greten et al. 2010].
Methotrexate
Methotrexate is immunosuppressive in low doses. It is widely used to treat rheumatoid arthritis and psoriasis. There is anecdotal evidence of improvement in liver enzymes and improved liver histology in patients with AIH resistant to steroid monotherapy and azathioprine, when treated with moderate dose of methotrexate [Burak et al. 1998]. However, it is teratogenic in women of childbearing age and has been associated with liver fibrosis.
Rapamycin
Rapamycin prevents activation of lymphocytes. It does this by binding a cytosolic protein called FK-binding protein, which inhibits the mammalian target of rapamycin pathway. Rapamycin also has antiproliferative and antiangiogenic effects and has recently been shown to stabilize regulatory T cells in vitro and in vivo [Coenen et al. 2006]. Rapamycin has been developed primarily as an immunosuppressive agent for transplantation where it has proved effective, but its mode of action suggests a potential use in AIH. There is very little published evidence to support its use in AIH, although anecdotal reports suggest efficacy in resistant disease.
Biological therapies for autoimmune hepatitis
Biological therapies might have two potential roles in the treatment of AIH; either as first-line agents to induce remission without the need for corticosteroids or as second-line therapy for patients whose condition fails to respond to conventional treatment. Until efficacy and safety are proven, it is likely that their use will largely be restricted to the latter indication. A plethora of biological drugs are currently coming onto the market, including anticytokine therapies; antibodies and fusion proteins aimed at blocking specific costimulatory receptors involved in T-cell activation; and antibodies and small molecule inhibitors of adhesion molecules and chemokines involved in effector cell recruitment to sites of inflammation. Many of these agents are being developed for other autoimmune diseases, including rheumatoid arthritis, inflammatory bowel disease (IBD), psoriasis and multiple sclerosis, and relatively little emphasis has been put on their potential role in liver disease. Their exciting efficacy in other autoimmune conditions and anecdotal reports of their use in liver disease make formal testing of some of these agents in autoimmune liver disease a priority.
Rituximab
Rituximab is a monoclonal antibody to CD20, which is expressed on B lymphocytes, from early pre-B cells to memory B cells, but which is absent from terminally differentiated plasma cells. Rituximab has been shown to be effective in several autoimmune diseases, particularly rheumatoid arthritis, systemic lupus erythematosus and vasculitides, suggesting it might be effective in AIH. Recent evidence implicating B cells in liver fibrosis further supports its use [Novobrantseva et al. 2005]. Reactivation of dormant hepatitis B virus (HBV) has been associated with rituximab therapy, and HBsAg along with HBV core antigen must be checked before starting treatment. Nucleos(t)ides analogues can be used in patients with positive HBV serology, but the additional risks need to be balanced against the lack of objective evidence of efficacy. A case report of a patient with AIH and idiopathic thrombocytopenic purpura resistant to corticosteroid treatment described normalization of serum transaminases after 4 weeks’ treatment with rituximab [Santos et al. 2006]. A later report of a patient with B-cell lymphoma and PBC/AIH overlap syndrome, resistant to steroid treatment, described improvement in symptoms, liver enzymes and histology after a total of 12 weeks’ treatment with rituximab [Barth and Clawson, 2010]. Our own experience is limited to three patients who were resistant to conventional immunosuppressive therapy. We used one or two intravenous injections to induce remission as determined by transaminases and normalization of gammaglobulin and then maintained with conventional therapy; in one case subsequent biochemical relapses were preceded by the reappearance of CD20 B cells in the blood and responded to repeated rituximab injections at intervals of 3–6 months. The results are promising and toxicity is low but controlled clinical trials are required to evaluate these anecdotal reports and to decide how best to use rituximab.
Antitumour necrosis factor therapy
Tumour necrosis factor α (TNFα) is a proinflammatory cytokine that is implicated in the pathogenesis of several autoimmune disorders, including rheumatoid arthritis, IBD and psoriasis. Therapeutic inhibition can be achieved using monoclonal antibodies such as infliximab or adalimumab or with a recombinant receptor fusion protein such as etanercept [Silva et al. 2010]. In addition, several commonly used drugs have anti-TNF activity, including the xanthine derivative pentoxifylline. The use of biological TNF blockers has been associated with lymphoma development and opportunistic infections. Anecdotal reports suggest that some patients with difficult AIH may respond to anti-TNF therapy and some patients with AIH and IBD have been reported as showing improvement in hepatitis when the IBD is treated with anti-TNFα therapy. However, despite these encouraging anecdotes, other reports have recorded AIH as a side effect of anti-TNF therapies [Coffin et al. 2011; Jansen, 2010; Umekita et al. 2011]. Thus further evidence of efficacy and toxicity is required before such therapies can be recommended.
Antichemokine therapy
Chemokines are chemotactic cytokines that regulate cell migration and positioning and that are implicated in the recruitment of leukocytes from blood into tissues. Several chemokines have been implicated in the recruitment of effector cells to the inflamed liver and these chemokines and their target receptors on effector cells are thus potential therapeutic targets. One such chemokine is CXCL10, previously known as IP-10, which is induced by proinflammatory cytokines in the inflamed liver and which has been associated with effector cell recruitment in immune-mediated and fibrotic liver diseases. A humanized anti-CXCL10 antibody developed by Novimmune (Switzerland) is currently undergoing a phase II clinical trial in the treatment of PBC (http://www.novimmune.com/products/ni-0801.html) and if this shows promise AIH would be a potential indication for future trials with this agent. However, both effector and regulatory subsets of lymphocytes express CXCR3 chemokine receptor for CXCL-10, thus preferential recruitment of lymphocyte subsets may determine the outcome of inflammation. Other chemokine–chemokine receptors that may be targets in AIH include CXCR6 and its receptor CXCL16, which is expressed on inflamed hepatocytes and bile ducts and CCR5 and its ligands [Oo and Adams, 2010].
Modulation of T-cell activation
The mechanisms through which effector T-cell immune responses to autoantigens such as cytochrome P450IID6 (CYP2D6) in AIH type 2 are permitted are being elucidated. An imbalance of effector and regulatory networks can unmask autoimmune responses and it has been reported that AIH is associated with a numerical and functional impairment of regulatory T cells [Longhi et al. 2004, 2006]. This suggests that experimental therapies can be directed at dampening immune cell activation by blocking costimulatory molecules to prevent effective antigen presentation or by giving immunosuppressive cytokines such as interleukin 10. These strategies work very well in animal models but are more difficult to apply to human autoimmune disease and the danger of interfering with powerful immune regulatory molecules in humans has recently been highlighted [Waibler et al. 2008]. One promising experimental approach is to adoptively transfer autologous regulatory T cells with activity against T effector cells to suppress autoimmune responses. Several groups are developing this approach and type 2 AIH, for which the antigen and immune epitopes are well described, is an obvious starting point. However, such approaches remain experimental at present [Longhi et al. 2011].
Management of fulminant autoimmune hepatitis
Fulminant liver failure may rarely be caused by AIH. A review of 115 patients that compared patients with acute presentations to chronic disease suggested that a minority (9%) presented with acute disease, including some with fulminant hepatic failure [Kessler et al. 2004]. The outcome of 11 cases of fulminant AIH in which five patients died suggested that serum total bilirubin levels worsen during days 8–15 and should be considered as a factor indicating liver transplantation [Miyake et al. 2006]. There were no differences in autoimmune markers.
Phelan-McDermid syndrome (caused by a deletion in chromosome 22q13) can lead to fulminant AIH requiring orthotopic liver transplantation in children [Tufano et al. 2009; Bartsch et al. 2010]. Interferon therapy in hepatitis C virus (HCV) has also been associated with fulminant AIH [Kogure et al. 2007]. Fulminant AIH presentation is more common in older patients [Redondo-Cerezo et al. 2002]. In a series of patients (n = 16) with fulminant AIH (encephalopathy, n = 10), only 1 of 12 patients who were treated with corticosteroids showed improvement. Thirteen patients required liver transplantation. Three patients experienced severe disseminated sepsis with corticosteroids [Ichai et al. 2007]. Corticosteroid therapy may not be effective in severe, fulminant forms of AIH and liver transplant assessment should be not be delayed unduly while waiting for a therapeutic response in this group of patients.
Treating autoimmune hepatitis in the presence of liver comorbidity
Fatty liver disease and autoimmune hepatitis
Nonalcoholic fatty liver disease (NAFLD) is the hepatic manifestation of the metabolic syndrome and in developed countries is now the commonest chronic liver disease with a prevalence of up to 46% [Browning et al. 2004; Bedogni et al. 2005; Fan and Farrell, 2009; Williams et al. 2011]. Other elements of this syndrome, including obesity and insulin resistance, are known to exacerbate other liver diseases [Ortiz et al. 2002; Fartoux et al. 2005; Charlton et al. 2006; Hart et al. 2010] and increase the risk of progression to cirrhosis [Liu et al. 2010]. Autoantibodies can be detected in some patients with NAFLD as they can in alcoholic liver disease and patients may have elevated immunoglobulins particularly immunoglobulin A [Cotler et al. 2004]. There are no data regarding the prevalence of features of the metabolic syndrome in AIH, or the impact on outcome. Features of the metabolic syndrome, including diabetes, hypertension and obesity, are exacerbated by corticosteroid treatment. Therefore, it is important to have a clear picture of what is being treated in patients with AIH who also have features of the metabolic syndrome. Both diseases cause persistently elevated transaminases and liver biopsy is often required to distinguish between interface activity associated with AIH and steatohepatitis. It is reasonable to presume that the presence of steatohepatitis in patients with AIH will increase the risk of progressive disease associated with liver inflammation and fibrosis and NAFLD should thus be treated with lifestyle interventions and pharmacological measures if appropriate [Ratziu et al. 2010].
Autoimmune hepatitis in patients with viral hepatitis
Chronic viral hepatitis can present with similar symptoms and biochemical changes to AIH and may be associated with autoantibodies (see below). It is thus important to be sure that one is dealing with two diseases rather than one. However, AIH can develop in patients with either HBV or HCV and patients with AIH can contract viral hepatitis.
Hepatitis B
Reactivation of hepatitis B has been reported in patients during treatment of AIH with immunosuppressant drugs, such as corticosteroids or azathioprine. Clinical manifestations can range from asymptomatic to acute hepatitis and even liver failure. Baseline HBV serology with viral load is recommended for all patients receiving immunosuppressive drugs. Several randomized, placebo-controlled trials have shown that reactivation can be prevented by antiviral prophylaxis, thus HBsAg-positive patients should receive viral load suppression with oral antivirals in the form of nucleos(t)ide analogues before and during immunosuppressive therapy for AIH [Hoofnagle, 2009].
Hepatitis C
Anti-HCV antibodies are prevalent in patients with type 2 AIH [Lenzi et al. 1990] and ANA, SMA, and liver/kidney microsomal type 1 (anti-LKM1) may also occur in chronic hepatitis C infection. The reasons for this association may be a consequence of immune dysfunction resulting in the unmasking of immune responses against autoantigens released during liver injury. Distinct subspecificities, including ANA with the homogeneous pattern and SMA with anti-actin specificity, are found in AIH [Cassani et al. 1997]. Interferon treatment for hepatitis C can provoke AIH [García-Buey et al. 1995]. A small series of seven patients with HCV and AIH found response to corticosteroid treatment in 71% with regard to normalization of transaminases, immunoglobulin G levels, and histological improvement [Schiano et al. 2001].
Autoimmune hepatitis in patients with HIV infection
De novo AIH as an immune reconstitution phenomenon has been described in patients with preexisting autoimmune disease treated for HIV [O’Leary et al. 2008]. In this setting liver biopsy is critical in establishing the diagnosis of AIH and to discriminate other causes of abnormal liver biochemistry including drug toxicity and opportunistic infections [Puius et al. 2008]. Treatment of AIH in patients with AIDS has been associated with life-threatening infections and if possible it may be best to leave treatment until highly active antiretroviral therapy has suppressed HIV levels. Having said that, in some cases severe or progressive AIH may not allow for this and each patient must be treated on their merits [Wan et al. 2009].
Autoimmune hepatitis and pregnancy
Pregnancy is generally safe in patients with well controlled noncirrhotic AIH. However, pregnant patients with cirrhosis face unique risks. These include higher rates of spontaneous abortion, prematurity and a potential for life-threatening variceal haemorrhage, hepatic decompensation, splenic artery aneurysm rupture, and postpartum haemorrhage. A review of 31 women treated for AIH suggested AIH in pregnancy is associated with both maternal and fetal mortality [Heneghan et al. 2001]. There were two maternal deaths (6%): one due to thromboembolic disease during pregnancy and another due to variceal bleeding 6 months after delivery. There were five instances of fetal loss (16%). An Italian cohort of 73 patients reported a miscarriage rate of 15.4% in pregnancies [Floreani et al. 2006] whereas Terrabuio and colleagues reported a 30% rate of fetal loss [Terrabuio et al. 2009]. Steroids and azathioprine are generally safe for mother and baby and need to be continued during pregnancy. Mycophenolate is contraindicated due to its possible teratogenic effect. AIH may present during pregnancy and should be considered in any pregnant woman with deranged liver tests.
Pregnancy is associated with natural immune tolerance, required to prevent the fetus being rejected by the mother [Aluvihare et al. 2004; Malhotra et al. 2002]. This can result in disease remission and better control during pregnancy and in some patients a reduced requirement for immunosuppression. However, after delivery there is a real risk of immune rebound once the tolerogenic effects of pregnancy are lost and patients should be monitored closely for this in the first few weeks following delivery [Elsing et al. 2007]. Thus AIH requires careful monitoring jointly by obstetricians and hepatologists not only during pregnancy but also into the post-partum period [Aggarwal et al. 1999]. Endoscopic surveillance of patients with cirrhosis for oesophageal varices and prophylaxis with β blockers or banding should eliminate bleeding risk.
Nonadherence to therapy in adolescents
AIH can affect children and young adults. The most common form is type 1 but LKM+ type 2 also occurs predominantly in children and adolescents. Adolescents frequently display poor compliance with medical advice and poor adherence to therapy. This may be exacerbated by the cosmetic effects of corticosteroids. Disease relapse or failure to obtain satisfactory control in adolescents should always bring up the possibility of poor adherence to therapy even if the patient tells you they are taking their medication. The risks can be reduced by the involvement of an active multidisciplinary team, including psychologists, youth workers and dedicated nurses who can provide support and education during this difficult period [Kerkar et al. 2006] and regular monitoring of immunosuppressant drugs.
When and how to treat autoimmune hepatitis in older patients
AIH is increasingly diagnosed in older patients in whom it may have an indolent course. Older patients with AIH differ from younger patients with regard to clinical and serological parameters: older patients are more likely to have cirrhosis at diagnosis, which was suggested by previous studies [Czaja and Carpenter, 2006; Floreani et al. 2006; Miyake et al. 2007]. In addition, older patients with AIH are more likely to have other autoimmune diseases, particularly thyroid disease [Czaja and Carpenter, 2006; Granito et al. 2005]. HLA DR4 is more frequently associated with the older patient whereas a higher proportion of younger patients are HLA DR3. Of note, a small UK series examining AIH in older patients found none of the patients died from complications of liver disease [Newton et al. 1997]. This may have been a consequence of shorter follow up in older patients, although a similar observation was made by Granito and colleagues in an Italian study [Granito et al. 2005].
With regard to treatment of AIH in older patients, Czaja and Carpenter found less frequent failure of corticosteroid treatment in older patients compared with younger patients [Czaja and Carpenter, 2006]. However, other data show similar responses to standard therapy in older patients [Granito et al. 2005; Miyake et al. 2007]. Given the often indolent nature of the disease in older patients, studies are required to determine how aggressively immunosuppressive therapy is required in this population.
Overlap syndromes with other autoimmune liver diseases
AIH may present with biochemical, immunological, clinical or histological features and other autoimmune liver diseases or may subsequently develop features of biliary disease [Buuren et al. 2000; Manns et al. 2010a]. Sequential syndromes involving PBC and AIH typically present with features of PBC first, whereas in those involving PSC and AIH, features of AIH usually occur initially and progress to biliary disease [Talwalkar et al. 2002; Abdo et al. 2002; Buuren et al. 2000; Floreani et al. 2005].
In the context of PBC, features more commonly associated with AIH including interface hepatitis and elevated transaminases probably represent one end of a spectrum of immune-mediated injury in which damage to hepatocytes is more prominent [Longhi et al. 2011; Waibler et al. 2008]. Patients with coexisting features of AIH and PBC have a worse outcome compared with those with AIH alone [Silveira et al. 2007]. There is now good evidence that patients with PBC associated with florid interface hepatitis respond to immunosuppressive therapy whereas those in whom the biliary feature predominates failed to respond to immunosuppression and should be treated with ursodeoxycholic acid alone [Chazouillères et al. 1998; Al-Chalabi et al. 2008; Joshi et al. 2002].
Both AIH and PSC can be associated with IBD [Perdigoto et al. 1992; Broomé and Bergquist, 2006] and there is evidence that they can occur sequentially in patients who present with typical AIH subsequently developing PSC [Abdo et al. 2002], particularly in children and adolescents. Some patients may have biliary disease from the start along with AIH, which are not detected if liver biopsy is not performed. The King’s Liver Unit reported the presence of histological and radiological biliary features compatible with PSC in 50% of children who otherwise fulfilled criteria for AIH [Gregorio et al. 2001]. Czaja and Carpenter reported that 20 of 84 adult patients with AIH had biliary changes on liver biopsy and responded to immunosuppressive therapy [Czaja and Carpenter, 2001]. The paediatric patients usually respond to corticosteroids, unlike patients with classical PSC. However, as the biliary features progress, response to immunosuppression may be lost [Gregorio et al. 2001]. Thus AIH/PSC overlap is more common than previously suspected and biliary features should be sought carefully in patients with AIH both histologically and if indicated by magnetic resonance cholangiography. This is particularly true if an elevated alkaline phosphatase is noted [Gohlke et al. 1996]. Addition of ursodeoxycholic acid rather than an increase in immunosuppression may improve biochemistry in such patients.
Liver transplantation and autoimmune hepatitis
Transplantation is indicated in patients with fulminant liver failure due to acute AIH, or more commonly, decompensated cirrhosis secondary to AIH. Swedish cohort data showed that liver transplantation is more common in patients experiencing relapses when off treatment, those who have higher international normalized ratio and are younger at the time of diagnosis of AIH [Werner et al. 2010]. Analysis of the European Transplant Registry [Schramm et al. 2010] for patients who underwent liver transplantation for AIH suggested 5-year survival was 0.73 compared with 0.83 for patients transplanted for PBC and incidence of fatal episodes of infection after transplantation was higher in patients transplanted for AIH. The overall success of liver transplantation is good but a proportion of patients, up to 25% in some series [Ratziu et al. 1999; Duclos-Vallee et al. 2003], are at risk of developing recurrent disease despite maintenance immunosuppression to prevent graft rejection. Recurrent AIH is characterized by the persistence of autoantibodies, elevated immunoglobulins and typical features on liver biopsy, although whether such changes truly represent recurrent disease is unclear because autoantibodies and interface hepatitis are detected in some patients transplanted for conditions other than AIH [Czaja, 2009]. The long-term use of low-dose steroids post transplant has been claimed to reduce the incidence of recurrent AIH [Montano-Loza et al. 2009], but recurrent disease may progress to cirrhosis or graft failure [Ratziu et al. 1999], warranting protocol biopsies to make the diagnosis and allow for treatment [Duclos-Vallee et al. 2003].
Conclusion
AIH is a complex autoimmune disease with unknown aetiology that occurs as a consequence of a breakdown in immune tolerance. The majority of patients respond to conventional treatment with corticosteroids and azathioprine and have a good prognosis. These patients are relatively straightforward to manage. However, in those whose condition fails to respond adequately to conventional treatment or who have side effects, careful consideration should be given to the choice of alternative treatment. There are limited data from randomized controlled trials but increasing experience from case reports and case series in the use of a number of alternative immunosuppressive agents including novel biological agents. It is imperative that in the future these approaches are subjected to rigorous clinical trials to allow future treatment decisions to be evidence based rather than empirical choices based on anecdote. In some situations, such as asymptomatic patients, pregnancy, old age and when there are concomitant liver diseases, treatment may not be straightforward and will need to be tailored to the individual patient. In addition, the option of not treating asymptomatic patients who do not have fibrosis may be considered. All such patients should ideally be managed by experienced hepatologists in specialist clinics with established protocols, supported by clinical nurse specialists with psychological support for adolescent patients. Liver transplantation is the only effective treatment for end-stage, decompensated AIH or patients who develop fulminant liver failure or HCC, but recurrence is seen in up to 25% of patients and needs careful assessment to discriminate between true recurrent autoimmune disease and other causes of graft hepatitis, including allograft rejection. We are entering an exciting phase during which many novel immune modulatory therapies are being introduced into clinical practice. These new drugs should revolutionize the way we treat AIH but require systematic evaluation and will probably be applied to specific subgroups of patients using a stratified/personalized medicine approach.
Footnotes
Funding
Richard Parker and Ye Htun Oo are both funded by the Medical Research Council.
Conflict of interest statement
All authors do not have any financial arrangements to disclose regarding this manuscript.
