Abstract
Colonic diverticular disease is common in Western countries and its prevalence increases with age. The large majority of patients (80–85%) will remain entirely asymptomatic throughout their life. In symptomatic cases, most patients will have diverticulosis without inflammation while the remainder will have diverticulitis with or without complications. About 1–2% will require hospitalization and 0.5% will require surgery. Factors predicting the development of symptoms remain to be identified. However, it is generally recognized that diverticular disease is probably related to complex interactions between colon structure, intestinal motility, diet, and genetic features. Epidemiologic studies have demonstrated an association between diverticulosis and diets that are low in fiber and high in refined carbohydrates. Although the causes of symptom development are still unclear, it is thought that previous episodes of intestinal inflammation may play a role. Changes in intestinal microflora could be one of the putative mechanisms responsible for low-grade inflammation. In patients with uncomplicated diverticulosis, a diet abundant in fruit and vegetables is recommended. The current therapeutic approaches in preventing recurrence of symptoms are based on nonabsorbable antibiotics, mesalazine, and/or probiotics. Cyclic rifaximin administration seems to be an adequate approach to relieving symptoms and preventing acute diverticulitis in patients with symptomatic diverticulosis.
The spectrum and biological progression of colonic diverticular disease
Diverticular disease of the colon represents the most common disease affecting the large bowel in the Western world, where it represents one of the most important gastrointestinal diseases in terms of direct and indirect health costs, accounting for approximately 130,000 hospitalizations yearly in the United States [Jacobs, 2007].
Diverticular disease is uncommon and prevalence is largely age dependent, with a rate of less than 5% in people under 40 years of age, increasing up to 65% in people aged 65 years or over; 80% of patients who present with diverticulitis are 50 or older. Hospital admissions of people with diverticular disease have increased over the past two decades. In England, the admission rate increased from 0.56 to 1.20 per 100,000 people/years between 1996 and 2006. The 30-day mortality rate was 5.1%, the 1-year mortality rate was 14.5% and the 28-day readmission rate was 9.6% [Jeyarajah et al. 2009].
Studies on the natural history of the disease have indicated that a large majority (80–85%) of patients with diverticular disease will remain entirely asymptomatic throughout their life. Of the 15–20% of patients presenting with abdominal pain, approximately three quarters will have painful diverticular disease without inflammation while the remainder will have diverticulitis with or without complications. Furthermore, around 1–2% will require hospitalization and 0.5% will require surgery [Stollman and Raskin, 2004].
The terms ‘diverticulosis’ and ‘diverticular disease’ are used to describe the presence of uninflamed diverticula. Diverticulitis is an inflammatory complication of the disease which is commonly accompanied by gross or microscopical perforation, ranging in severity from a single, mild, acute attack of diverticulitis to more severe attacks characterized by perforation and abscess formation, occasionally resulting in chronic complications such as obstruction and fistula formation. Free perforation into the peritoneum causing frank peritonitis can be life threatening but is rare. Diverticular perforation is associated with a twofold increase in mortality compared with the general population [Salzman and Lillie, 2005].
Unfortunately, because of the lack of prospective studies, factors predicting the development of symptoms remain to be identified. However, it has been suggested that evaluation of the colonic motility index (pressure amplitude exceeding 120 mmHg), together with a brief history of left lower quadrant pain, a short segment of involved colon and a relatively young age (about 50 years), may be useful in recognizing patients at risk of developing symptoms [Colecchia et al. 2003] (Figure 1).
The majority of patients harboring colonic diverticula remain asymptomatic throughout their life (asymptomatic diverticular disease); only 20% will develop symptoms and signs of illness. Symptomatic diverticular disease is further subdivided into painful diverticular disease (symptomatic diverticular disease with no inflammation) and diverticulitis (symptomatic diverticular disease with inflammation). Diverticulitis is further subdivided into uncomplicated and complicated diverticulitis. DD, diverticular disease.
Physiopathology
The pathogenetic mechanisms of diverticular disease are still poorly understood, however it is generally recognized that these are probably related to complex interactions between colon structure, intestinal motility, diet, and genetic features [Simpson et al. 2002].
Increased intracolonic pressures have been recorded in patients with diverticulosis. Segmentation of the colon could generate excessively high pressures favoring herniation. In response to increased intraluminal pressure, outpouchings consisting of only mucosa and submucosa may develop and protrude at areas of potential weakness, such as where the bowel wall is penetrated by its vasculature. Stasis or obstruction in the narrow necked diverticulum may lead to bacterial overgrowth and local tissue ischemia. Anaerobes (including bacteroides, peptostreptococcus, clostridium, and fusobacterium species) are the most commonly isolated organisms. Gram-negative aerobes, especially Escherichia coli, and facultative Gram-positive bacteria, such as streptococci, are often cultured as well [Sheth et al. 2008].
Although the cause of colonic diverticular disease has not yet been conclusively established, epidemiologic studies have demonstrated an association between diverticulosis and diets that are low in dietary fiber and high in refined carbohydrates [Eastwood, 2009]. Low intake of dietary fiber results in less bulky stools that retain less water and may alter gastrointestinal transit time; these factors can increase intracolonic pressure and make evacuation of the colonic contents more difficult. In a large prospective study of 43,881 US male healthcare professionals, an inverse relationship was noted between the consumption of insoluble dietary fiber and the development of symptomatic diverticular disease [Aldoori et al. 1998].
Other factors that have been associated with an increased risk of diverticular disease include physical inactivity, constipation, obesity, smoking, and treatment with some drugs. Some investigations have suggested that lack of physical activity is independently associated with an increased risk of symptomatic diverticular disease, while smoking, caffeine and alcohol intake are not associated with a substantially increased risk of asymptomatic disease [Aldoori et al. 1995].
Case–control studies have demonstrated an association between current medication use and diverticular perforation. There are biologically plausible explanations for the development of perforation with exposure to nonsteroidal anti-inflammatory drugs (NSAIDs) with a decrease in prostaglandin synthesis and alterations in large intestine permeability, which serve to decrease colonic wall integrity [Lanas and Scarpignato, 2006]. Similarly, corticosteroid use is associated with a threefold increase in the risk of diverticular perforation [Humes et al. 2011].
Because of increased constipation associated with opiate analgesics, the resultant high-pressure colonic contractions may predispose patients to diverticular perforation. Opiate analgesics use is associated with a twofold increase in the risk of diverticular perforation. Current use of a calcium antagonist, however, is not associated with an increased risk of perforated colonic diverticular disease. Calcium channel blockers may produce a beneficial reduction in the strength and duration of colonic contractions, minimizing episodes of high intracolonic pressure while maintaining basal activity and colonic transit [Humes et al. 2011; Morris et al. 2003].
Since few data exist on risk factors, preventive measures for the development of diverticula are only speculative, and can be aimed only at preventing development of symptoms. Despite controversial data, fiber supplementation is recommended as standard therapy [Marlett et al. 2002; Murphy et al. 2007].
Symptom development
The causes of symptom development in some patients are still unclear. Because patients with diverticulosis who have a history of diverticulitis have more episodes of recurrent abdominal pain and impaired bowel function, it has been suggested that previous episodes of intestinal inflammation may play a role. This finding is in line with recent observations in patients with other gastrointestinal diseases such as infectious enteritis and inflammatory bowel disease (IBD), and as also speculated in irritable bowel syndrome [Colecchia et al. 2003]. Luminal mucosal inflammation (segmental colitis), which shares histological features with IBD, may coexist with colonic diverticula. In patients with these conditions, the presence of a chronic, low-grade intestinal inflammation would induce a sensory-motor dysfunction, leading to symptom development and/or persistence. Patients with symptomatic diverticular disease exhibit increased neuropeptides in mucosal biopsies, which may reflect resolved prior inflammation because it parallels the changes seen in acute and chronic diverticulitis [Simpson et al. 2009].
Changes in intestinal microflora could be one of the putative mechanisms responsible for low-grade inflammation. In patients with diverticular disease, bacterial overgrowth may be present. This bacterial overgrowth, aided by the fecal stasis inside the diverticula, could contribute to chronic low-grade inflammation that sensitizes both intrinsic primary efferent and extrinsic primary afferent neurons. These alterations could lead to smooth muscle hypertrophy, and increased sensitivity to abdominal distension, and finally, to symptom development [Simpson et al. 2003].
Symptomatic but not asymptomatic uncomplicated diverticular disease has been reported to be associated with heightened perception of distension, not only in the diverticula-bearing sigmoid, but also in the unaffected rectum. This hyperperception is not due to altered wall compliance [Clemens et al. 2004].
Patients affected by recurrent attacks of uncomplicated diverticulosis are at high risk of recurrence. If diverticulosis is considered to be a step-up chronic IBD, it has been suggested that patients affected by recurrent attacks of diverticulosis harbor more severe inflammation of the colonic mucosa and then require prolonged treatment [Floch and Bina, 2004].
Treatment of symptomatic diverticular disease
Because the large majority of patients with diverticulosis will remain entirely asymptomatic, there is no need for treatment and no data support any therapeutic recommendations. However, based on the pathogenesis of diverticular disease, a diet rich in vegetables, fruit and fiber is recommended in order to accelerate colonic transit and reduce intraluminal pressure [Murphy et al. 2007].
According to available guidelines, treatment of symptomatic, but uncomplicated, diverticular disease aims to reduce the frequency and severity of diverticular-related symptoms (abdominal pain, bloating, alterations in bowel habit) and to prevent complications. Different agents have been proposed, such as bulking agents, antispasmodics, and topical antibiotics, on the basis of different potential pathophysiological mechanisms; that is, abnormal colonic motility, inadequate intake of dietary fibers, intestinal bacterial overgrowth, and mucosal inflammation [Sheth et al. 2008].
In patients with uncomplicated diverticulosis, a diet with abundant fruit and vegetables is recommended because it seems that this protective effect reduces symptom development and prevents major complications, as demonstrated in uncontrolled studies. A high-fiber diet may prevent formation of additional diverticula, lower the pressure in the lumen, and reduce the chances that one of the existing diverticula will burst or become inflamed. The efficacy of fiber supplementation in the treatment of symptomatic diverticular disease remains controversial, although it is considered a mainstay of treatment [Marlett et al. 2002].
The observed hypermotility of the sigmoid colon in many patients with symptomatic disease provides the rationale for using anticholinergic and spasmolytic agents, which may be effective in some cases of uncomplicated diverticular disease. However, their use remains to be confirmed by controlled studies [Morris et al. 2003].
Prevention of diverticulitis and recurrence of symptoms
Symptomatic uncomplicated diverticular disease usually improves with effective therapy. However, about 61% of patients who have had a previous attack of symptomatic diverticular disease and are not taking any medication to prevent recurrence of symptoms become symptomatic within 1 year and about 4% eventually develop complications. Recurrent attacks of symptomatic uncomplicated diverticular disease may be an important problem in clinical practice. Current therapeutic approaches to prevent recurrence of symptoms of uncomplicated diverticulosis are based on nonabsorbable antibiotics, mesalazine, and/or probiotics [Papi et al. 2005].
Rationale for antibiotic use in symptomatic diverticular disease
In symptomatic but uncomplicated diverticular disease, the rationale for the use of antibiotics has not been clearly established. However, recent studies have suggested the presence, at least in a subgroup of patients, of an intestinal bacterial overgrowth, a condition that may allow an excessive production of bowel gas (such as hydrogen, carbon dioxide, and methane) through carbohydrate fermentation, with secondary development of abdominal pain, bloating and tenderness. Excessive gas production in the bowel has been suggested to play a role in determining abdominal symptoms such as bloating, pain, and discomfort. Antimicrobial drugs have been shown to reduce hydrogen production and gas-related symptoms, and antibiotic therapy raises mean stool weight in patients taking fiber, most likely due to reduced fiber degradation.
Poorly absorbed antimicrobials, unlike systemically available antibiotics, allow localized targeting of enteric pathogens and are associated with minimal risk of systemic toxicity or side effects. The restricted use of nonabsorbable oral antibiotics only for enteric infections should also reduce the development of widespread resistance, a major limitation of current antibiotics for enteric infections [Zullo et al. 2010; Pistoia et al. 2004].
Rifaximin
Rifaximin is an oral, nonsystemic rifamycin analogue with a broad spectrum of activity similar to that of rifampicin. Rifaximin is poorly absorbed in the gastrointestinal tract, therefore it has a high safety profile. Owing to these properties, rifaximin has been tested in different gastrointestinal diseases, including diverticular disease. Rifaximin has broad-spectrum in vitro activity against Gram-positive and Gram-negative aerobic and anaerobic enteric bacteria, and has a low risk of inducing bacterial resistance [Scarpignato and Pelosini, 2005]. Furthermore, with minimal systemic bioavailability, rifaximin may be more conducive to long-term use than other, more bioavailable antibiotics with detrimental side effects [Papi et al. 2005].
Plasma levels of rifaximin are negligible; therefore, bacteria outside the gastrointestinal tract are not exposed to appreciable selective pressure. The risk of bacterial resistance appears to be lower with rifaximin than with systemic antibiotics and is mediated through reversible genomic change. For chromosomally mediated mutation and selection to result in clinically relevant resistance, the mutation cannot be lethal and cannot significantly decrease virulence; otherwise, the resistant trait will not be transmitted. Both in vitro and in vivo studies of the effects of rifaximin on commensal flora suggest that rifaximin-resistant organisms have low viability [Colecchia et al. 2007].
The mechanism by which rifaximin improves symptoms in uncomplicated diverticular disease is not fully understood. A synergistic effect of rifaximin and a high-fiber diet have been suggested to reduce proliferation of gut microflora, with a consequent decrease in bacterial hydrogen and methane production, and/or to expand fecal mass due to a decrease in bacterial degradation of fibers. Furthermore, rifaximin may improve symptoms and lower the frequency of disease complications, reducing intestinal bacterial overgrowth. Rifaximin may also enhance fecal bulking and fecal weight, as shown for other antibiotics, and decrease the intraluminal colonic pressure, one of the pathogenetic mechanisms for diverticula development [D’Inca et al. 2007].
Adverse events (AEs) associated with rifaximin administration. (Adapted from [Bass et al. 2010; Pimentel et al. 2011.]

Rifaximin plus dietary fiber supplementation versus dietary supplementation alone
Although the role of antibiotics in uncomplicated diverticular disease is still debated, recent clinical studies have demonstrated that cyclic administration of rifaximin is more effective in reducing symptoms than fiber supplementation alone. Furthermore, treatment with nonabsorbable antibiotics was shown to reduce symptom frequency and intensity in these patients. The mechanism of rifaximin in reducing the frequency of symptoms and the rate of complications of diverticular disease is only speculative. Rifaximin has been suggested to reduce the metabolic activity of intestinal bacterial flora, the degradation of dietary fibers, and the production of gas. Moreover, eradication of bacterial overgrowth seems to be related to a reduction in intestinal symptoms. The latter effects are important because an increased production of intestinal gas and of gas-related symptoms such as pain and bloating have been documented in these patients. It is important at this stage to identify and characterize patients with diverticular disease who have intestinal bacterial overgrowth, and then to perform controlled clinical trials to evaluate the effects of antibiotic administration on symptom and complication frequency, that is, on the natural history of this disease [Papi et al. 2005] (Figure 3).
Diverticular disease: putative role of intestinal bacterial overgrowth in symptom development. Altered intestinal microflora could contribute to chronic low-grade inflammation which abnormally sensitized both intrinsic primary efferent and extrinsic primary afferent neurons. This condition could lead to neural and muscle dysfunction and, finally, to symptom development. (Adapted from Colecchia et al. [2003].) Antibiotics decrease intraluminal pressure in patients with diverticular disease. SIBO, small intestinal bacterial overgrowth. (Adapted from Frieri et al. [2006].)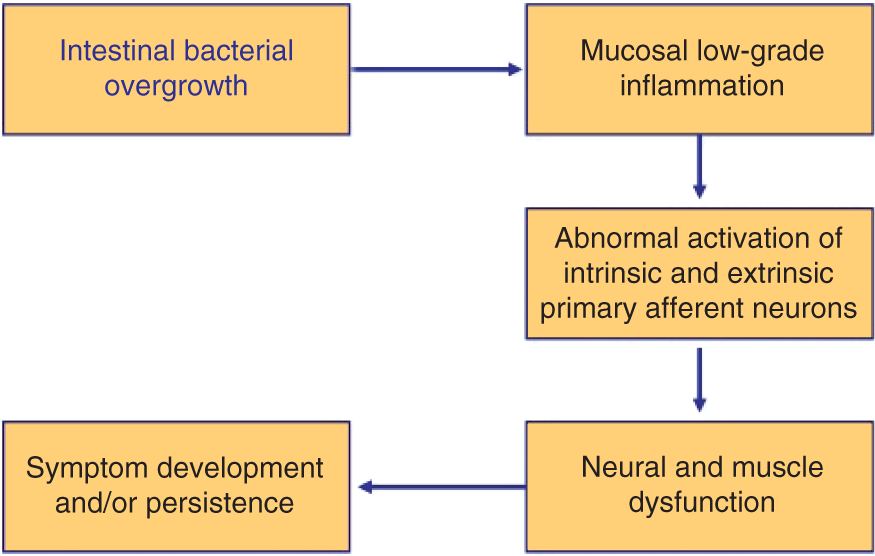

Two randomized controlled trials (RCTs) that use rifaximin in the treatment of symptomatic diverticular disease have been reported. The first RCT (168 people with uncomplicated diverticular disease) compared dietary fiber supplementation (glucomannan 2 g/day) plus oral rifaximin (400 mg twice daily) with dietary fiber supplementation (glucomannan 2 g/day) plus placebo. Both treatments were given for 7 days each month for 1 year. The RCT found that dietary fiber supplementation plus rifaximin significantly increased the proportion of people with no symptoms or only mild symptoms after 1 year of treatment compared with dietary fiber supplementation alone (69% with rifaximin versus 39% with placebo, p = 0.001). The RCT found no significant difference between treatments in the severity of diarrhea, tenesmus, or upper abdominal pain [Papi et al. 1995].
The second open RCT, which included 968 people with diverticular disease, also compared dietary fiber supplementation (glucomannan 4 g/day) plus oral rifaximin (400 mg twice daily for 7 days every month) with dietary fiber supplementation alone (glucomannan 4 g/day). The RCT found that, at 1 year, dietary fiber supplementation plus rifaximin significantly improved global symptom score compared with dietary fiber supplementation alone [Latella et al. 2003].
The efficacy of long-term cyclic administration of rifaximin in symptomatic uncomplicated diverticular disease has been assessed. A RCT enrolling 307 patients over 24 months found that long-term administration of rifaximin 400 mg twice daily for 7 days every month plus dietary fiber supplementation of at least 20 g/day was more effective than dietary fiber supplementation alone in reducing the clinical manifestations of patients with symptomatic, uncomplicated diverticular disease. Furthermore, rifaximin treatment was more effective than fiber administration in influencing the clinical course of the disease because both probability of symptom recurrence and development of disease complications were significantly reduced [Colecchia et al. 2007].
The administration of rifaximin 1200 mg/day improves the benefits of dietary fiber in uncomplicated diverticular disease by preventing its bacterial degradation. This effect was achieved through the prevention of the increase in hydrogen production by bacterial fermentation of the dietary fiber and the reduction in the oroanal transit time [D’Inca et al. 2007].
Fiber supplementation is also indicated in symptomatic diverticulosis in order to relieve symptoms and prevent acute diverticulitis. The best results have been obtained using a combination of soluble fiber, such as glucomannan, and poorly absorbed antibiotics, such as rifaximin, given 7–10 days every month [Papi et al. 2005].
Mesalazine
Mesalazine is an anti-inflammatory drug that inhibits factors of the inflammatory cascade (such as cyclooxygenase) and free radicals, and has an intrinsic antioxidant effect. Some inflammatory complications of diverticulitis, such as edema, are generated by a heightened production of proinflammatory cytokines, reduced anti-inflammatory cytokines, and enhanced intramucosal synthesis of nitric oxide. Thus, salicylates could be effectively used in the treatment of diverticulitis both for the acute phase and recurrence prevention. Tursi and colleagues in Italy confirmed the effectiveness of mesalazine in treating symptomatic uncomplicated diverticular disease of the colon. This study suggested that continuous mesalazine treatment is better than cyclic mesalazine treatment in maintaining remission of recurrent uncomplicated diverticulosis. In fact, about 70% of patients treated with mesalazine every day remained symptom free at the end of the study, whereas only about 50% of patients treated with mesalazine for 10 days every month were symptom free at the end of the study. Continuous mesalazine treatment was able to maintain the remission in patients at high risk of recurrent, symptomatic disease, reducing recurrence and risk of complications. For this reason, continuous treatment with mesalazine may be a good choice as maintenance treatment of symptomatic uncomplicated recurrent diverticulosis [Tursi et al. 2006].
In addition to antibiotics, mesalazine inhibits reactive oxygen metabolites, with which intestinal bacteria interact, thus potentially modifying bacterial activity. The positive effects of these mechanisms on diverticular disease were shown by Trespi and colleagues in a study of 166 patients with acute, noncomplicated diverticular disease reporting mild to moderate symptoms. Patients reported significant improvement in abdominal symptoms during an 8-week course of mesalazine that followed antibiotic therapy. These positive effects even persisted over a follow-up period of 4 years [Trespi et al. 1997].
In another report, with a follow-up period of 12 months and a larger population (244 patients), cycles of mesalazine at a dosage of 400 mg or 800 mg twice daily, lower than normally used for the treatment of IBD, for 10 days every month showed promising results in reducing symptom frequency and symptom severity. This therapy was significantly more effective than rifaximin in 10-day monthly cycles at a dosage of 200 mg or 400 mg twice daily in lowering the mean general symptom score [Comparato et al. 2007].
Data from a recent systematic review reflect the expanding use of mesalazine [Gatta et al. 2010]. Mesalazine given at 800 mg twice daily for 10 days a month has a similarly effect to rifaximin (400 mg twice daily) [Di Mario et al. 2005]. It must however be emphasized that these data came from a single-center, randomized, open trial. Despite these reports, the use of mesalazine following an episode of acute diverticulitis is not widespread and deserves further studies.
Probiotics
Probiotics could be a good option for treating uncomplicated acute diverticulitis. Probiotic action includes production of antimicrobials, competitive metabolic interactions with proinflammatory organisms, and inhibition of adherence and translocation of pathogens. Likewise, probiotics can determine mucosal defense by changing immune and epithelial function, for example, by diminishing tumor necrosis factor, interleukin-1, and interferon gamma. In a recent prospective, randomized, open-label study, 46 consecutive patients previously affected by symptomatic uncomplicated diverticular disease of the colon were enrolled for 6-month follow up. A total of 68% of patients who received treatment with a symbiotic mixture of Lactobacillus acidophilus and Bifidobacterium spp. for 6 months were still symptom free at the end of the follow-up period [Lamiki et al. 2010]. These studies show that antibiotics, mesalazine, and probiotics could be new choices in resolving signs and symptoms of uncomplicated symptomatic diverticular disease of the colon. Further studies are needed to show the effectiveness of these drugs in maintaining remission and preventing diverticulitis throughout a long-term follow-up period [Festi and Colecchia, 2009].
Treatment of diverticulitis
There is a general consensus that conservative treatment is indicated for newly onset uncomplicated diverticulitis because about 70% of patients treated for a first episode recover and have no further problems. However, a 60% risk of developing complications has been reported in patients with recurrent attacks. Conservative treatment aims to relieve symptoms and prevent future complication [Sheth et al. 2008].
For uncomplicated diverticulitis the standard therapy is liquid diet and oral antimicrobials, usually ciprofloxacin and metronidazole. Hospitalization, bowel rest, and intravenous antibacterial agents are mandatory for complicated diverticulitis. Hemorrhage is usually a self-limited event but may require endoscopic or surgical treatment [Kohler et al. 1999]. Once in remission, continuous fiber intake and probably an intermittent course of rifaximin may improve symptoms and reduce diverticulitis recurrence. These preventive strategies will likely improve patients’ quality of life and reduce management costs. A surgical approach to diverticular disease is needed in 15–30% of patients and consists of removing the intestinal segment affected by diverticula. Surgery is indicated in diffuse peritonitis, abscesses, fistulas, stenosis, and after the second to fourth attack of uncomplicated diverticulitis, although rather than the number of episodes it is the severity and the interval between them which is more important [Collins and Winter, 2007]. Young people and patients who are immunocompromised are more likely to undergo surgery [Murphy et al. 2007].
Summary and conclusions
Diverticular disease must be understood as an entity with biological progression from a normal colon to a wide spectrum of clinical scenarios and disease severities. In diverticular disease, as in functional intestinal disease, bacterial flora and intestinal motility seem to play a key role in determining the clinical manifestations of the disease [Colecchia et al. 2003].
Different degrees of mucosal inflammation may be an underlying pathogenetic mechanism in diverticular disease as in other colonic diseases.
The use of nonabsorbable antibiotics in diverticular disease has two aims: relief of symptoms, and prevention of diverticulitis recurrence. Current evidence based on studies with the negligible absorbed antibiotic, rifaximin, administered in long-term cyclic treatments supports this approach. In symptomatic uncomplicated diverticular disease, treatment with rifaximin plus fiber supplementation is effective in relieving symptoms and preventing complications at 1 year [Bianchi et al. 2011]. Rifaximin administration may improve the clinical benefits of a high-fiber diet by interfering with the intestinal microflora, preventing fiber degradation, and by reducing the mucosal damage induced by bacterial overgrowth [D’Inca et al. 2007].
Footnotes
Acknowledgements
Dr Angel Lanas was an invited speaker to a symposium sponsored by Alfa-Wasserman at the United European Gastroenterological week in Barcelona in 2010.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Dr Federico Sopeña declares no conflict of interest in preparing this article.
